Abstract
Dunes provide numerous ecosystem services including habitat for flora and fauna, coastal protection through mitigation of wind and wave energy, and as a barrier to storm flooding. Dune vegetation that inhabits these systems plays a vital role in building dunes and resisting erosion. In the United States, the Virginia coast is a transitional temperature zone for several dominant dune grasses, where the northern species, Ammophila breviligulata (C3) and the southern species, Uniola paniculata and Panicum amarum (both C4) overlap. At Back Bay National Wildlife Refuge (NWR), Virginia, warming temperatures are resulting in native species range shifts altering biotic interactions. Additionally, the invasive sedge, Carex kobomugi (C3) has become more prevalent in the region with unknown effects on the landscape. To understand the impact of Carex on the native plant community, we quantified species distribution and morphological traits of three dominant native species, A. breviligulata, Panicum, and Uniola as well as invasive Carex. We found that Carex was most dominant and exhibited similar individual traits when compared to native species. The suite of plant traits was unique in Carex, with less variation than in native species allowing for rapid nutrient acquisition and growth. These traits, combined with species distribution data suggest that Carex may limit Ammophila abundance. Similarly, presence of Carex had a negative effect on Panicum growth, but no discernible effect on Uniola. Success of Carex at Back Bay NWR may be indicative of the future spread of the species due to above and belowground morphological traits, which may give the invasive an advantage with climate warming.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biotic invasions have been an increasing problem with climate change (McKinney and Lockwood 1999; Dukes and Mooney 1999) affecting growth and persistence of endemic species (Reviewed In: Hughes 2000; Walther et al. 2002). These biotic shifts occur in most ecosystems, including coastal systems (Grabherr et al. 1994; Walther et al. 2002; Wang et al. 2019; Henry and Sorte 2022), where sea-level rise and oceanic storms exacerbate climate change influences. Perhaps most important, is the mitigation of invasive species effects within coastal dune systems. Dunes provide a multitude of ecosystem services, such as habitat for shore birds, water purification, carbon sequestration, and recreation (Reviewed In: Barbier et al. 2011), but also protect economically valuable coastal infrastructure from wind and wave action, as well as rising sea levels (Ruggiero et al. 2001; Hesp 2002; Mullins et al. 2019). An additional concern is that low elevation coastal zones include 10% of the world’s population (McGranahan et al. 2007). Predicting responses of coastal dunes to a range of climate change scenarios and potential biotic invasions is limited by a critical lack of knowledge of morphological plant traits (above and belowground) for native and invasive species that influence resource acquisition and dune dynamics (i.e., dune building, erosion).
Dunes are formed through the feedback of vegetation and sediment deposition (Hesp 2002; van IJzendoorn et al. 2021). Dune vegetation is limited to species that can withstand high abiotic stress such as salt spray, blowing sand, heat, drought, frequent flooding, and nutrient limited soils (Ehrenfeld 1990; Young et al. 1994). Within a dune system, species composition (due to associated morphological traits) can determine the size, shape, and erosion resistance of a particular dune (Feagin et al. 2015; Hacker et al. 2019; Mullins et al. 2019). Many of these traits including height, stem density, burial response, and aboveground plant cover determine dune shape and size (Woodhouse et al. 1977; Hacker et al. 2012; Zarnetske et al. 2012; Reviewed In: Feagin et al. 2015). Knowledge of these aboveground metrics are incorporated into dune management plans; however, belowground characteristics are less understood, especially the influence of species-specific belowground traits on dune building and erosion resistance (Reijers et al. 2020; Walker and Zinnert 2022). Belowground traits influence resource capture for plant growth, propagation throughout the dune system, and dune erosion resistance (Reubens et al. 2007; de Battisti and Griffin 2020). These traits are of equal importance to aboveground characteristics when assessing dune stability, and they may be altered by plant invasions, but belowground traits have not been thoroughly investigated.
Grasses play a vital role in dune building and the introduction of non-natives may have many unintended impacts. An example of these potential impacts can be seen in the introduction of Ammophila arenaria and Ammophila breviligulata along the US Pacific Northwest coast. Expansion of these species has led to significantly taller and wider dunes than those built by native species (Hacker et al. 2012; Zarnetske et al. 2012). Along the US Atlantic coast, Carex kobomugi (Asiatic sand sedge, hereafter referred to by genus) is expanding from Massachusetts to North Carolina, dominating habitat previously occupied by native dune grasses (Charbonneau et al. 2020). Carex proliferates via clonal propagation, which facilitates rapid expansion (Hilton et al. 2006; Hacker et al. 2012). The dense root structure of Carex may enhance dune building and providing protection from storm induced erosion (Wootton et al. 2005; Charbonneau et al. 2016). However, it is important to consider all ecological impacts (positive and negative) when evaluating an invasive species. Due to the dune building function of non-natives along with expanding infrastructure in the North American Pacific Coast, where monitoring the establishment and expansion has been the primary focus (Hacker et al. 2012). In comparison, along the Atlantic Coast efforts are focused on Carex removal (Wootton et al. 2005) as protection of native dune grasses is important for wildlife habitat, including threatened and endangered species.
Carex invasion has the potential to alter native plant community structure and traits but has not been evaluated in North American dunes. The Virginia, USA coastline represents the northernmost range for southern species and the southernmost range for northern species (Goldstein et al. 2018), with increasing temperatures causing latitudinal range shifts in coastal (Huang et al 2018; Goldstein et al. 2018). Above and belowground traits provide a mechanistic understanding of resource use for the growth of dominant species. Quantifying the interactions of invasive species, such as Carex, with native counterparts is essential for long-term restoration projects and navigating predicting future species range shifts. Our objective is to quantify the influence of invasive Carex kobomugi on the native community for a climatically transitional dune system and determine the plant traits that enhance resource acquisition relative to native species. To achieve this goal, we (1) quantified species distributions and abundance across the dune to identify relationships relative to dune position, (2) evaluated morphological traits of Carex relative to native species, and (3) quantified the effect of Carex on the community through changes in species richness, traits, and species composition.
Methods
Study site
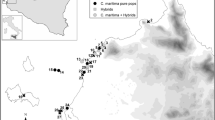
This study was conducted at Back Bay National Wildlife Refuge (NWR), Virginia Beach, VA, USA (36.6880° N, 75.9218° W) along the northernmost portion (1.6 km) of dune ridge (Fig. 1). The area of Back Bay NWR was home to multiple tribes within the Powhatan chiefdom. They were either forcibly removed by the English colonizers or died during the smallpox epidemic by the early 1700s (Rountree 1990). The land this research was completed on was stolen from these Indigenous communities and needs to be respected in accordance with their ideals of land preservation and returned to the remaining modern-day tribes.
The northern section of the site is currently closed to the public and has not been the focus of active management; the Carex population remains intact. Throughout the southern portion, Carex has been removed through yearly targeted spraying of glyphosate along with the planting of native dune grasses including Ammophila breviligulata (American beach grass), Panicum amarum (bitter switchgrass), Uniola paniculata (sea oats), and Spartina patens (saltmeadow cordgrass).
Carex is native to eastern Asia and was introduced in New Jersey, US in the 1920s (Small 1954). The invasion of Carex in the US has primarily been studied along the New Jersey coast, but the species has spread further south causing concern for land managers. The three focal natives here are Ammophila breviligulata, Uniola paniculata, and Panicum amarum (hereafter referred to by genus). Ammophila is a C3 grass (cool-season) that has dominated much of the northern US east coast, with limited distribution in North Carolina and farther south (Goldstein et al. 2018). Ammophila is often used in restoration projects but is sensitive to high temperatures (Woodhouse 1978) that occur with climate warming. Uniola is a C4 grass (warm-season) and dominates the southern Atlantic and Gulf coasts. This species has expanded northward due to warming temperatures (Goldstein et al. 2018). Panicum is a C4 grass that exists along the entire east coast and is typically used in tandem with Ammophila for dune grass planting projects. Panicum is a moderate dune builder that is less studied (Woodhouse 1978; Hodel and Gonzalez 2013). It is unclear how Carex invasion is influencing interactions between native dune grasses as warming induced range shifts occur.
Field sampling
Plant community composition and aboveground traits
To assess dune composition and potential species interactions, transects were established from the dune toe to the back dune (Fig. 1b) and spaced ~ 120 m apart (n = 15) throughout June 2020. Plots (0.25 m2) were positioned 5 m apart and included the toe, face, crest, and back dune (Fig. 1b) with 5 plots per transect. Within each plot, percent cover, number of shoots, and maximum height of each species were recorded. Percent cover of individual species and bare sand were ocularly estimated with a quadrat, rounded to the nearest 5% and totaling 100%. Relative importance was calculated for each species present, using relative cover, relative frequency, and relative stem densities (Eq. 1). This calculation ranges from 0 to 300 with higher numbers being of higher importance at the site.
Equation 1 Calculation for relative importance values by species
Biomass and belowground trait measurements
Based on species composition from community composition plots, monocultures (within a minimum 0.25 m2 plot) of dominant species (Ammophila, Panicum, Uniola, and Carex, Fig. 1) were selected for further above and belowground morphological measurements (n = 10 per species) from the dune face. Additional cores were selected from mixed species plots where Carex was present with native species and absent (mixed native species only, n = 10).
All cores were collected in June 2020, except for monocultures of Panicum which were collected in June 2021. Following modified methods of Charbonneau et al. (2016), cores were collected to 60 cm depth using a slide hammer (AMS, Inc.; American Falls, ID), bagged, and placed in a cooler for further processing. Aboveground biomass within the core was cut at the soil surface and placed in a bag for later processing. Cores were processed by wet-sieving using the #6 (3.35 mm), #18 (1.00 mm), and #35 (500 µm) standard sieves to remove all belowground material. Roots were scanned using Epson Perfection V800 picture scanner (Epson America Inc., Long Beach, California, USA) and WinRHIZO software (Regent Instruments Inc, Québec, Canada) to quantify total root length and surface area for different root diameter classes. After scanning, the total below and aboveground material was dried at 60℃ for 72 h to determine biomass. Root tissue density (RTD) was calculated as the ratio of dried root material to volume of the roots. Dried material (both above and belowground) was analyzed by Cornell Isotope Laboratory for % carbon (C), % nitrogen (N), δ15N, and δ13C. Isotope analysis of δ15N aided in the determination of N source, and to give an indication of nitrogen fixation (Unkovich 2013), while analysis of δ13C is a proxy for water use efficiency (Bacon 2004).
For each species, additional roots were collected (n = 10) for tensile strength measurements. Roots were washed and stored in 15% ethanol until processing. For measurements, roots were rehydrated in water for at least 30 min (Böhm 1979; Bischetti et al. 2003). Clamping of roots followed a modified procedure, similar to De Baets et al. (2008), using sandpaper (3 M, 220 grit) and foam (Walker and Zinnert 2022). Tensile strength was quantified with an MTS Insight 30 Universal Testing Machine (UTM) and MTS Advantage Wedge Action Grips (MTS Systems Corporation; Eden Prairie, MN) with a 50N load cell and connected to the software, Testworks 4. The software calculates peak stress required to break the root (i.e., tensile strength).
Statistical analysis
Plant community composition and aboveground traits
Due to non-normality, cover was log + 1 transformed to account for 0’s in data across the dune profile; plant height and stem density were log transformed. Two-way mixed model analysis of variance (ANOVA) identified differences in cover by location and species with plot as a random variable. Analysis of covariance (ANCOVA) determined species differences in plant height and stem density with species cover as the covariate. Tukey tests were used to examine post-hoc significant differences among groups.
Biomass and belowground trait measurements
All biomass and belowground trait variables were log transformed. Analysis of covariance (ANCOVA) identified species differences in biomass and belowground trait variables (i.e., above and belowground biomass, root:shoot, root diameter, root surface area, fine root surface area, root tissue density) with species cover as the covariate. ANCOVA quantified species differences in peak stress with root diameter as a covariate for the tensile strength. Species differences in aboveground and belowground nutrient and isotope data were determined with ANOVA. Tukey tests were used to examine post-hoc significant differences among groups.
The multiple above and belowground variables (i.e., cover, stem density, height, biomass, root traits, nutrients and isotopes) for each species from coring plots along the face, were analyzed with Multiple Response Permutation Procedure (MRPP) with Euclidean distance to detect significant differences among species. Multivariate results were visualized with Nonmetric Multidimensional Scaling (NMS) using Euclidean distance.
Effects of Carex
Differences in percent cover, height, and stem density for Ammophila, Panicum, and Uniola were assessed in plots with and without Carex (data from full community plots) using multi-factor ANOVA (location, Carex presence/absence). We used a Bonferroni p-value adjustment accounted for multiple tests (α = 0.017). Total above and belowground biomass differences among mixed species coring plots where Carex was present and absent were determined with two-factor ANCOVA (Carex presence/absence, native species identity, and native cover as a covariate). All statistics were completed in R version 4.1.0.
Results
Distribution of species on the dune
Carex had the highest percent cover of all species across the dune profile, decreasing from the back to the dune toe (Fig. 2, Fig. S1). In comparison to the dominant native grasses (Ammophila, Panicum, Uniola), Carex had higher cover at every dune location except the toe where it was rarely found (species × location: F = 2.67, p < 0.01); Fig. 2). Ammophila was dominant at the dune toe and decreased in cover at the crest and back dune. In order of relative importance at the site the species from highest to lowest importance were: Carex, Ammophila, Panicum, Uniola, and Spartina patens (Table S1). The most frequent species in order were Carex, Panicum, Uniola, and Ammophila (Table S1). Other species present on the dunes in low quantities included: Spartina patens, and the forbs Gamochaeta purpurea, Hydrocotyle bonariensis, and Krigia virginica (Table S1).
Species plant traits
Stem density was lowest in Panicum (6 ± 1) compared to the other natives (F = 13.41, p < 0.01). The tallest species were Uniola and Ammophila (~ 70 cm), while Carex and Panicum were shorter (~ 29 cm) (F = 108.7, p < 0.0001; Table 1). Aboveground biomass was highest for Uniola (> 300%, F = 29.92, p < 0.0001, Table 1). Carex belowground biomass was > 2 times more than Panicum (F = 3.98, p = 0.02). Root:shoot was relatively low with Uniola and Panicum significantly lower than Carex (F = 7.65, p < 0.001). Ammophila and Carex had smaller average root diameter (0.33 mm, F = 14.44, p < 0.0001) than Uniola and Panicum, which was reflected in higher tensile strength for both species (F = 16.53, p < 0.0001; Fig. 3). Ammophila had the highest RTD (F = 11.26, p < 0.0001) and highest tensile strength (p < 0.0001). Ammophila tensile strength was > 2 times higher than that of all other species.
Carex had higher %N in the aboveground biomass than all other species (F = 26.5, p < 0.001) but lower %N than Uniola in belowground material (F = 5.0, p < 0.01, Figure S2). Carex had significantly less aboveground %C than all other species (F = 12.8, p < 0.0001) with no differences found belowground (F = 3.5, p = 0.03, Figure S2). These differences in Carex led to the same patterns for C:N, lower aboveground (F = 19.1, p < 0.0001) and higher belowground (F = 8.7, p < 0.001, Figure S2) compared to the natives. Panicum had lower ẟ15N aboveground (F = 6.5, p = 0.001) whereas Carex had lower belowground ẟ15N (F = 8.7, p < 0.001, Figure S3). Belowground ẟ15N values were close to 0 in Carex, an indication of possible nitrogen fixation. Species separated out by C3 (Carex and Ammophila) and C4 (Panicum and Uniola) for ẟ13C values aboveground (F = 282.4, p < 0.0001) and belowground (F = 224.2, p < 0.0001, Figure S3).
Multivariate analysis based on the aforementioned traits revealed that species differed significantly from each other (MRPP: t = −12.9, p < 0.0001). When visualized using NMS (stress = 5.52; variance explained: Axis 1 = 64.9%, Axis 2 = 21.9%), Carex exhibited lower variability and did not overlap with native species (Fig. 4). Above and belowground %C, belowground C:N, height and aboveground biomass were correlated with axis 1 (Table S2) contributing to the majority of Carex separation from other species occurred.
Effects of Carex on plant community
Across the dune profile at Back Bay NWR, Carex grew predominantly in mixed plots, most frequently with Panicum and Uniola (Table 2). All native species were more commonly found in mixed plots than monoculture. Species richness increased for plots with Carex (2.3 ± 0.14) relative to those without (1.4 ± 0.10), with Carex included in the count when present. Carex did not appear to inhibit growth of Uniola or Ammophila, but a decline in Panicum cover (F = 14.2, p < 0.01) and stem density (F = 5.9. p = 0.013) was observed when Carex was present. (Fig. 5). When comparing biomass from mixed species core plots, aboveground biomass was not affected by the presence of Carex, but belowground biomass was significantly higher with Carex present (F = −10.09, p = 0.02; Fig. 6).
Boxplots of species-specific response to the presence of Carex in mixed community composition plots. Asterisks indicate significant difference results of multi-factor ANOVA (location, Carex presence/absence) with Bonferroni adjustment (α = 0.0125). Points indicate outliers. a Percent Cover and b Stem Counts in 0.25 m2 plots. c Height in each plot
Discussion
Coastal communities are impacted significantly from climate change, especially sea-level rise and increasing storms, both strength and frequency (Suursaar et al. 2015). Climate change also contributes to increased plant invasions and altered native species distributions, leading to novel species interactions and potential loss of native species (Bradley et al. 2010; Dukes and Mooney 1999). Our objective was to identify traits that contribute to the success of the invasive sedge, Carex, on a coastal dune with overlapping distributions of northern and southern native species. We defined success as expanding populations and relative declines in native populations. Community composition of Back Bay NWR dunes differed from northern study sites where Carex has been more closely monitored (Small 1954; Charbonneau et al. 2020). We demonstrated that Carex is the most dominant species at this site, co-occurring with native species. As a C3 species, Carex exhibits overlapping individual traits (e.g., aboveground biomass, root diameter, root tissue density) of both C3 (Ammophila) and C4 species (Panicum). But when considering the entire suite of plant traits, Carex exhibited unique traits that promote resource acquisition and had less variability relative to native species. In mixed plots, there was greater belowground biomass with Carex present than in mixed plots of that were composed only of native species. Carex was found to have a negative impact on the growth of Panicum although no impacts were observed with other species. The limited occurrence of Ammophila with Carex along with reduced cover across the dune relative to Panicum suggests that both species may be impacted by Carex expansion.
Community composition and species interactions
Dunes of the US mid-Atlantic region are heavily dominated by Ammophila, Uniola, Panicum, and Spartina patens (Hacker et al. 2019). These species co-occur and disperse at different elevations and dune positions (Hacker et al. 2019; Woods et al. 2023). The dunes at Back Bay NWR have different levels of management activity. In the northern portion of our study site, Carex was not actively managed and was the most dominant species based on cover and relative importance values. Ammophila, Panicum, and Uniola were the dominant native species, but with less overall abundance and frequency relative to Carex. Carex had highest abundance on the dune face, crest, and back, but was absent at the dune toe where Ammophila was most abundant. Ammophila was less likely to co-occur with Carex and was less abundant on the dune face, differing from dunes at Back Bay NWR that have been actively managed for Carex removal (Woods et al. 2023). Both species are C3 and likely to compete for space and resources due to timing of active growth (Bossuyt et al. 2005; Charbonneau et al. 2020), but extended phenology of Carex may provide a competitive advantage (personal observation; Hess et al. 2019). Based on findings in New Jersey (Charbonneau et al. 2020) and Back Bay NWR (our study and Woods et al. 2023), Ammophila growth and distribution is likely constrained by Carex.
Phenological timing of growth and response to warming temperatures (both minimum and maximum) may be key determinants of growth for species and provide opportunities for further research. In higher latitudes along the Atlantic coast, Ammophila dominates entire dunes (Burk 1968; Emery and Rudgers 2009; Goldstein et al. 2018). Summer temperature constraints on Ammophila growth and known competitive effects with Uniola may be additional factors limiting the distribution and abundance at our site (Seneca and Cooper 1971; Harris et al 2017; Brown et al. 2018; Woods et al 2023). Uniola was most abundant on the face and crest, frequently co-occurring with Carex. Panicum was dominant on the dune face and back, but less is known about the recent distribution in this region despite use in restoration projects (Seneca et al. 1976; Long et al. 2013). High abundance of Panicum at Back Bay NWR and other Virginia dunes relative to earlier reports (Conn and Day 1993; Sabo 2023) may indicate climatically driven shifts in species composition. Our study did not examine the phenology and growth over time of species, but Ammophila may be most impacted by warming temperatures and Carex invasion in the region. Quantifying temperature limitations to growth across the dune profile will enable future species dominance predictions with climate warming.
Morphological differences in graminoids
Graminoids were the dominant life form of the Back Bay NWR dunes but exhibited distinct differences in morphological traits (Walker and Zinnert 2022). Carex and Panicum were > 50% shorter than Ammophila and Uniola. Carex had high stem density (similar to Ammophila) relative to Panicum and Uniola. The short stature and high stem density of Carex creates unique growth relative to the native species and may impede germination or growth of other species (Silvertown 1980). In addition, it spreads multiple leaves along the soil surface (compared to upright growth of native grasses) which may provide an aboveground competitive advantage by increasing local soil moisture (Wetzel and van der Valk 1998; Deutsch et al. 2010) in the dune system, although not measured in our study.
Carex exhibited individual traits similar to Ammophila (i.e. above and belowground biomass, root:shoot, stem density, and root diameter) and Panicum (i.e. above and belowground biomass, root:shoot, height, and root tissue density). Although Carex and Ammophila had similar root diameters, tensile strength was > 2 times higher in Ammophila, indicating higher erosion resistance (Davidson et al. 2020; Walker and Zinnert 2022; Figlus 2022). Ammophila root traits (smaller diameter and high root tissue density) may contribute to drought tolerance with lower relative growth rates (Wahl and Ryser 2000; Fort et al. 2013). Leaf and root δ13C showed species differences based on photosynthetic pathways (C3, C4), indicating lower water use efficiency in Ammophila and Carex (Ellsworth and Cousins 2016). Most interesting, Carex exhibited several traits that may provide a competitive advantage relative to native species: higher leaf %N, more N allocated per unit C in leaves (lower C:N), thus possibly enhancing photosynthetic rates for faster growth (Leuning et al. 1995). Carex was the only species with evidence suggesting symbiotically fixed N2 (ẟ15N values close to 0, Robinson 2001). Rhizosheath fixation through symbiotic rhizosphere relationships has been suggested in other non-N2 fixing coastal species as a way to acquire N in an N-limited system (Brown and Zinnert 2021). Root C:N was highest in Carex and coupled with low RTD indicates a more rapid root growth strategy compared with native species, further contributing to success as an invasive species (Wahl and Ryser 2000).
Effects of Carex on the landscape
Vegetation along dunes exist more frequently in community with other species than in monoculture as we found in our analysis. Of the native species, Uniola was most frequently found co-occurring with Carex and was unaffected in growth metrics by the presence of Carex. Carex and Uniola differed in most traits, indicating potentially different spatial–temporal niches, and providing opportunities to determine if niche differentiation allows for co-existence. Conversely, Panicum and Carex were similar in aboveground biomass and stature, yet Carex presence was detrimental to Panicum growth (i.e., lower cover and stem density). Carex ability to acquire higher leaf N has the potential to outcompete Panicum due to similar aboveground niche space and faster growth. Although Carex belowground biomass was similar to native species when examined in monoculture, when co-occurring with other species, presence of Carex with native species increased belowground biomass compared to mixed plots with native species only. The mechanism for this increased biomass cannot be determined from our study design but implies an effect of Carex to the plant community.
Belowground biomass, proportion of fine roots, and high tensile strength are indicators of erosion resistance, but it is unclear how these traits are distributed throughout the dune (de Battisti and Griffin 2020; Walker and Zinnert 2022; White 2022). Increased belowground material provides more surface area for sand to bind, but the belowground components are more complex than just gathering biomass (i.e., Feagin et al. 2015). Extensive belowground structures also aid in nutrient and water uptake in these low nutrient soils (Klimešová et al. 2023). Although Ammophila has traits associated with erosion resistance (e.g., small root diameter, high tensile strength), the long, linear dunes formed by Ammophila could be lost due to Carex invasion (Charbonneau et al. 2020) and known temperature constraints on Ammophila growth (Seneca and Cooper 1971; Woods et al. 2023). Changes in species composition could have a major impact on dune building and coastal erosion (Zarnetske et al. 2012). The effect of Carex invasion remains to be determined for locations with higher temperatures where C4 species are expected to thrive.
Conclusions
Success of Carex at Back Bay NWR, Virginia is indicative of future spread of the species due to specific morphological traits that create a competitive advantage relative to native species. Carex has previously been documented to displace native grasses (specifically Ammophila) farther north along the Atlantic coast. Our research documents potential competition with Ammophila and Panicum in a climatically transitional dune system. Although there are few studies of Panicum in this region, it was the second most frequent species after Carex, a possible indication of climatically induced range shift by the C4 species. We found that native species more frequently co-occur, which may benefit wildlife habitat and coastal erosion through increased biodiversity. The invasion of Carex may lead to a monoculture dune system over time if left unmanaged. Future research should evaluate the impacts to ecosystem services resulting from monocultural spread of the invasive sedge compared to the functional role of multi-species communities. Regardless, removing invasive species and revegetating dunes with native species provides a suite of traits able to provide broader habitat for multiple native species while providing erosion resistance. Understanding climate change impacts to native species, including effects of non-native species, will enhance predictions of dune composition in future climate scenarios.
References
Bacon MA (2004) Water use efficiency in plant biology. Blackwell, Oxford
Barbier EB, Hacker SD, Kennedy C, Koch EW, Stier AC, Sillman BR (2011) The value of estuarine and coastal ecosystem services. Ecol Monogr 81(2):169–193. https://doi.org/10.1890/10-1510.1
Bischetti GB, Bonfanti F, Greppi M (2003) Misura della resitenza alla trazione delle radice: apparato sperimentale e metodologica d’analisi. Quaderni Di Idronomia Montana 21:349–360 ((in Italian))
Böhm W (1979) Root parameters and their measurement. In: Methods of studying root systems. ecological studies (analysis and synthesis), vol 33. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-67282-8_12
Bossuyt B, Honnay O, Hermy M (2005) Evidence for community assembly constraints during succession in dune slack plant communities. Plant Ecol 178:201–209. https://doi.org/10.1007/s11258-004-3287-8
Bradley BA, Wilcove DS, Oppenheimer M (2010) Climate change increases risk of plant invasion in the Eastern United States. Biol Invasions 12:1855–1872. https://doi.org/10.1007/s10530-009-9597-y
Brown JK, Zinnert JC (2021) Trait-based investigation reveals patterns of community response to nutrient enrichment in coastal mesic grassland. Diversity 13:19. https://doi.org/10.3390/d13010019
Brown JK, Zinnert JC, Young DR (2018) Emergent interactions influence functional traits and success of dune building ecosystem engineers. J Plant Ecol 11(4):524–532. https://doi.org/10.1093/jpe/rtx033
Burk CJ (1968) A floristic comparison of lower Cape Cod, Massachusetts and North Carolina Outer Banks. Rhodora 70(782):215–227. https://www.jstor.org/stable/23308551
Charbonneau BR, Wnek JP, Langley JA, Lee G, Balsamo RA (2016) Above vs. belowground plant biomass along a barrier island: implications for dune stabilization. J Environ Manag 182:126–133. https://doi.org/10.1016/j.jenvman.2016.06.032
Charbonneau BR, Nicoletta R, Wootton LS (2020) A decade of expansion of the invasive plant Carex kobomugi in a coastal foredune system. Biol Invasions 22:2099–2112. https://doi.org/10.1007/s10530-020-02240-6
Conn CE, Day FP (1993) Belowground biomass patterns on a coastal barrier island in Virginia. Bull Torrey Bot Club 120(2):121–127. https://doi.org/10.2307/2996941
Davidson SG, Hesp PA, da Silva GM (2020) Controls on dune scarping. Prog Phys Geogr 44(6):923–947. https://doi.org/10.1177/0309133320932880
De Baets S, Poesen J, Reubens B, Wemans K, De Baerdemaeker J, Muys B (2008) Root tensile strength and root distribution of typical Mediterranean plant species and their contribution to soil shear strength. Plant Soil 305:207–226. https://doi.org/10.1007/s11104-008-9553-0
de Battisti D, Griffin JN (2020) Below-ground biomass of plants, with a key contribution of buried shoots, increases foredune resistance to wave swash. Ann Bot 125:325–333. https://doi.org/10.1093/aob/mcz125
Deutsch ES, Bork EW, Willms WD (2010) Separation of grassland litter and ecosite influences on seasonal soil moisture and plant growth dynamics. Plant Ecol 209:135–145
Dukes JS, Mooney HA (1999) Does global change increase the success of biological invaders? Tree 14(4):135–139. https://doi.org/10.1016/S0169-5347(98)01554-7
Ehrenfeld JG (1990) Dynamics and processes of barrier island vegetation. Rev Aquatic Sei 2:437–480
Ellsworth PZ, Cousins AB (2016) Carbon isotopes and water use efficiency in C4 plants. Curr Opin Plant Bio 31:155–161
Emery SM, Rudgers JA (2009) Ecological Assessment of dune restorations in the Great Lakes Region. Res Ecol 18(S1):184–194. https://doi.org/10.1111/j.1526-100X.2009.00609.x
Feagin R, Figulus J, Zinnert J, Sigren J, Martinez M, Silva R, Smith W, Cox D, Young D, Carter G (2015) Going with the flow or against the grain? The promise of vegetation for protecting beaches, dunes, and barrier islands from erosion. Front Ecol Environ 13(4):203–210. http://www.jstor.org/stable/44000749
Figlus J (2022) Designing and implementing coastal dunes for flood risk reduction. In: Brody S, Lee Y, Kothuis BB (eds) Coastal flood risk reduction. Elsevier, Amsterdam, pp 287–301
Fort F, Jouany C, Cruz P (2013) Root and leaf functional trait relations in Poaceae species: implications of differing resource-acquisition strategies. J Plant Ecol 6(3):211–219. https://doi.org/10.1093/jpe/rts034
Goldstein EB, Mullins EV, Moore LJ, Biel RG, Brown JK, Hacker SD, Jay KR, Mostow RS, Ruggiero P, Zinnert JC (2018) Literature-based latitudinal distribution and possible range shifts of two US east coast dune grass species (Uniola paniculata and Ammophila breviligulata). PeerJ 6:e4932. https://doi.org/10.7717/peerj.4932
Grabherr G, Gottfried M, Pauli H (1994) Climate effects on mountain plants. Nature 369:448. https://doi.org/10.1038/369448a0
Hacker SD, Zarnetske P, Seabloom E, Ruggiero P, Mull J, Gerrity S, Jones C (2012) Subtle differences in two non-native congeneric beach grasses significantly affect their colonization, spread, and impact. Oikos 000:001–011. https://doi.org/10.1111/j.1600-0706.2011.18887.x
Hacker SD, Jay KR, Cohn N, Goldstein EB, Hovenga PA, Itzkin M, Moore LJ, Mostow RS, Mullins EV, Ruggiero P (2019) Species-specific functional morphology of four US Atlantic coast dune grasses: biogeographic implications for dune shape and coastal protection. Diversity 11(5):82. https://doi.org/10.3390/d11050082
Harris A, Zinnert J, Young D (2017) Differential response of barrier island dune grasses to specific interactions and burial. Plant Ecol 218(5):609–619. https://doi.org/10.1007/s11258-017-0715-0
Henry AK, Sorte CJB (2022) Impact assessment of coastal marine range shifts to support proactive management. Front Ecol Environ 20(3):161–169. https://doi.org/10.1002/fee.2447
Hesp P (2002) Foredunes and blowouts: initiation, geomorphology and dynamics. Geomorphol 48:245–268. https://doi.org/10.1016/S0169-555X(02)00184-8
Hess MC, Mesléard F, Buisson E (2019) Priority effects: emerging principles for invasive plant species management. Ecol Eng 127:48–57
Hilton M, Harvey N, Hart A, James K, Arbuckle C (2006) The impact of exotic dune grass species on foredune development in Australia and New Zealand: a case study of Ammophila arenaria and Thinopyrum junceiforme. Aust Geogr 37(3):313–334. https://doi.org/10.1080/00049180600954765
Hodel RG, Gonzalez E (2013) Phylogeography of Sea Oats (Uniola paniculata), a dune-building coastal grass in Southeastern North America. J Hered 104(5):656–665. https://doi.org/10.1093/jhered/est035
Huang H, Zinnert JC, Wood LK, Young DR, D’Odorico P (2018) Non-linear shift from grassland to shrubland in temperate barrier islands. Ecology 99(7):1671–1681. https://doi.org/10.1002/ecy.2383
Hughes L (2000) Biological consequences of global warming: is the signal already. Tree 15(2):56–61. https://doi.org/10.1016/s0169-5347(99)01764-4
Klimešová J, Martínková J, Bartušková A, Ott JP (2023) Belowground plant traits and their ecosystem functions along aridity gradients in grasslands. Plant Soil 487:39–48
Leuning R, Kelliher FM, De Pury DGG, Schulze ED (1995) Leaf nitrogen, photosynthesis, conductance and transpiration: scaling from leaves to canopies. Plant Cell Environ 18(10):1183–1200
Long ZT, Fegley SR, Peterson CH (2013) Fertilization and plant diversity accelerate primary succession and restoration of dune communities. Plant Ecol 214:1419–1429. https://doi.org/10.1007/s11258-013-0263-1
McGranahan G, Balk D, Anderson B (2007) The rising tide: assessing the risks of climate change and human settlements in low elevation coastal zones. Env Urban 19(1):17–37. https://doi.org/10.1177/0956247807076960
Mckinney ML, Lockwood JL (1999) Biotic homogenizationL a few winners replacing many losers in the next mass extinction. Trends Ecol Evol 14(11):450–453. https://doi.org/10.1016/S0169-5347(99)01679-1
Mullins E, Moore LJ, Goldstein EB, Jass T, Bruno J, Durán Vinent O (2019) Investigating dune-building feedback at the plant level: insights from a multispecies field Experiment. Earth Surf Process Landforms 44:1734–1747. https://doi.org/10.1002/esp.4607
Reijers VC, Lammers C, de Rond AJA, Hoetjes SCS, Lamers LPM, van der Heide T (2020) Resilience of beach grasses along a biogeomorphic successive gradient: resource availability vs. clonal integration. Oecologia 192:201–212
Reubens B, Poesen J, Danjon F, Geudens G, Muys B (2007) The role of fine and coarse roots in shallow slope stability and soil erosion control with a focus on root system architecture: a review. Trees 21:385–402. https://doi.org/10.1007/s00468-007-0132-4
Robinson D (2001) 15N as an integrator of the nitrogen cycle. Trends Ecol Evol 16:153–162
Rountree HC (1990) Pocahontas’s people the Powhatan Indians of Virginia through four centuries. University of Oklahoma Press
Ruggiero P, Komar PD, McDougal WG, Marra JJ, Beach RA (2001) Wave runup, extreme water levels and the erosion of properties backing beaches. J Coast Res 17(2):407–419
Sabo A (2023) Dune building dynamics impact cross-island connectivity and barrier island characteristics. MS Thesis, Virginia Commonwealth University
Seneca ED, Cooper AW (1971) Germination and seedling response to temperature, daylength and salinity by Ammophila breviligulata from Michigan and North Carolina. Bot Gaz 132(3):203–215
Seneca ED, Woodhouse WW, Broome SW (1976) Dune Stabilization with Panicum amarum the North Carolina coast. For: US Army Corps of Engineers Coastal Engineering Research Center. Fort Belvoir, VA
Silvertown J (1980) Leaf-Canopy-Induced seed dormancy in a grassland flora. New Phytol 85(1):109–118
Small JA (1954) Carex kobomugi at Island Beach, New Jersey. Ecology 35:289–291. https://doi.org/10.2307/1931128
Suursaar Ü, Jaagus J, Tõnisson H (2015) How to quantify long-term changes in coastal sea storminess? Estuar Coast Shelf Sci 156:31–41
Unkovich M (2013) Isotope discrimination provides new insight into biological nitrogen fixation. New Phytol 128(3):643–646
van IJzendoorn CO, de Vries S, Hallin C, Hesp PA (2021) Sea level rise outpaced by vertical dune toe translation on prograding coasts. Sci Rep 11:12792. https://doi.org/10.1038/s41598-021-92150-x
Wahl S, Ryser P (2000) Root tissue structure is linked to ecological strategies of grasses. New Phytol 148(3):459–471. https://doi.org/10.1046/j.1469-8137.2000.00775.x
Walker SL, Zinnert J (2022) Whole plant traits of coastal dune vegetation and implications for interactions with dune dynamics. Ecosphere 13:e4065. https://doi.org/10.1002/ecs2.4065
Walther GR, Post E, Convey P, Menzel A, Parmesan C, Beebee TJC, Fromentin JM, Hoegh-Guldberg O, Bairlein F (2002) Ecological responses to recent climate change. Nature 416:389–395. https://doi.org/10.1038/416389a
Wang CJ, Li QF, Wan JZ (2019) Potential invasive plant expansion in global ecoregions under climate change. PeerJ 7:e6479. https://doi.org/10.7717/peerj.6479
Wetzel PR, van der Valk AG (1998) Effects of nutrient and soil moisture on competition between Carex stricta, Phalaris arundinacea, and Typha latifolia. Plant Ecol 138:179–190
White AE 2022. Biotic characteristics of managed and unmanaged coastal dunes in the Outer Banks, North Carolina. MS Thesis. Virginia Commonwealth University
Woodhouse WW, Seneca ED, Broome SW (1977) Effect of species on dune grass growth. Int Biometeor 2(3):256–266. https://doi.org/10.1007/BF01552879
Woodhouse WW (1978) Dune building and stabilization with vegetation. Fort Belvoir, VA
Woods NN, Kirschner A, Zinnert JC (2023) Intraspecific competition in common coastal dune grasses overshadows facilitation on the dune face. Restor Ecol 31:e13870. https://doi.org/10.1111/rec.13870
Wootton LS, Halsey SD, Bevaart K, McGough A, Ondreicka PP (2005) When invasive species have benefits as well as costs: managing Carex kobomugi (Asiatic sand sedge) in New Jersey’s coastal dunes. Biol Invasions 7:1017–1027. https://doi.org/10.1007/s10530-004-3124-y
Young D, Erickson D, Semones S (1994) Salinity and the small-scale distribution of three barrier island shrubs. Can J Bot 72(9):1365–1372. https://doi.org/10.1139/b94-167
Zarnetske PL, Hacker SD, Seabloom EW, Ruggiero P, Killian JR, Maddux TB, Cox D (2012) Biophysical feedback mediates effects of invasive grasses on coastal dune shape. Ecology 93(6):1439–1450. https://doi.org/10.1890/11-1112.1
Acknowledgements
This work was done in collaboration with the biologists at Back Bay NWR, with our main point of contact being Lauren Mowbray. Field and laboratory assistance was provided by: Cameron Clay, Meg Goeke, Audrey Kirschner, Edward A. Long, Allyson Richins, & Shannon Walker. Emotional support was provided by: Past & Present members of the CPEL lab. Thank you to Donald R. Young for comments to an earlier version of the manuscript.
Funding
This project was funded by the Garden Club of Virginia Conservation Fellowship to Emily Riffe and VCU College and Humanities Dean’s funds to Julie Zinnert.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Emily Riffe and Julie Zinnert. The first draft of the manuscript was written by Emily Riffe and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have not disclosed any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Riffe, E.C., Zinnert, J.C. Impact of invasive Carex kobomugi on the native dune community in a US mid-Atlantic coastal system. Biol Invasions 26, 1195–1208 (2024). https://doi.org/10.1007/s10530-023-03237-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-023-03237-7