Abstract
Slash-and-mulch agroforestry systems can reduce greenhouse gas emissions by mulching the vegetation instead of burning it. This mulch layer then contains greater stocks of organic material than after burning, making it a potential source of N2O and CH4 efflux during decomposition. We examined N2O and CH4 efflux from slash-and-mulch AFS using a two-way factorial design: with and without P + K fertilization, and with and without a nitrogen-fixing tree (Inga edulis). We hypothesized that inclusion of N-fixing trees would increase N2O efflux and that CH4 efflux would increase due to increased soil moisture with mulching. We measured trace gas fluxes prior to the end of Rotation 1, and after mulching to begin Rotation 2. N2O efflux increased with I. edulis during the year prior to, but not after, mulching. No differences by treatment were detected for CH4 efflux before or after mulching. Site conversion from secondary forest to Rotation 2 resulted in a 130% increase in N2O efflux and a 430% decrease in CH4 efflux. The CO2e increase of 2,400 kg ha−1 was an order of magnitude less than estimated releases of trace gases from burning (38,400 kg ha−1). For both N2O and CH4, land disturbance during mulching led to larger changes in trace gas fluxes than either P + K fertilization or inclusion of the N-fixer. The order-of-magnitude estimates of trace gas release as CO2e from mulching and the addition of N-fixers appears to be less than that from burning alone.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Agriculture and deforestation for agriculture are responsible for nearly 25% of greenhouse gas (GHG) emissions globally (Smith et al. 2014; Tubiello et al. 2015). Agroforestry systems (AFS) have been proposed to reduce the GHG emissions of agricultural practices (Campos & Nepstad 2006). Forest loss, partially for conversion to agriculture, in Brazilian Amazonia has generated at least 5% of global GHG emissions (Nepstad et al. 2009). Smallholdings (< 100 ha) in Brazilian Amazonia occupy ~ 38% of the total land area under cultivation (Brondizio et al. 2009) and are typically prepared for cultivation using slash-and-burn AFS, in which the standing vegetation is cut, left to dry, and then burned. The initial pulse of available nutrients via ash increases crop production. Leaching, erosion, and crop export cause fertility to decline, leading to abandonment after 2–3 years (Denich et al. 2004). Clearing forest via fire leads to pulses of GHG emissions at clearing and, if followed by conventional agriculture, continuously increased fluxes of CO2, CH4, and N2O (Davidson et al. 2001).
AFS vary in their potential to mitigate emissions of CO2, methane (CH4), and nitrous oxide (N2O). Slash-and-mulch systems have been adopted in some areas to replace the use of fire in slash-and-burn systems (Kato et al. 1999). In one study comparing slash-and-burn to slash-and-mulch, the global warming potential through the first two years of the cycle was estimated at 21 and 3.6 Mg CO2e ha−1, respectively (Davidson et al. 2008). This difference was largely due to the avoided release of 12 kg N2O–N ha−1 and 630 kg-CH4 ha−1 during the burn. During cropping, however, slash-and-mulch treatments resulted in CH4 efflux (16 kg CH4 ha−1) compared to slash-and-burn that resulted in consumption (−5 kg CH4 ha−1). Similarly, soil N2O efflux during cropping was greater in the slash-and-mulch treatment (4.2 vs 2.9 kg N2O–-N ha−1). This previous study applied 90 kg-N ha−1 as urea in addition to including the N-fixer Inga edulis; therefore the role of the N-fixer could not be separated from the N fertilization effect on N2O efflux during the cropping phase. N-fixing trees have been implicated as a source of increased N2O efflux (Rosenstock et al 2014). A recent meta-analysis synthesized global agroforestry and reforestation research using N-fixing trees and found 39 papers fitting their search criteria (Kou-Giesbrecht & Menge 2021), although it included only one site from the Amazon basin, and it was from natural forest. The later work concluded that N2O efflux might double from planting N-fixing trees for reforestation, which could offset ~ 4% of the benefits of atmospheric CO2 uptake derived from tree growth.
In the same locale as the Davidson et al (2008) research (Igarapé Açu, Pará, Brazil), an improved fallow study with N-fixing tree species, including I. edulis, but without slash-and-mulch preparation, reported low rates of N2O emissions (2 kg N2O–N ha−1) that did not differ from control treatments that lacked N fixing trees (Verchot et al. 2008). In addition, CH4 was consumed in both the wet and dry season, with estimated annual consumption of −4 kg CH4 ha−1, which did not differ between treatment and control. A similar result was found in the Peruvian Amazon where, after burning, a multistrata AFS with I. edulis showed no increase in N2O efflux or decrease in CH4 consumption compared to unburned 13-yr-old secondary forest (Palm et al. 2002).
Fertilization with inorganic N can increase production of N2O in many sites via nitrification and denitrification (Castro et al. 1994 and 1995; Basiliko et al. 2009) and can suppress CH4 emissions in upland sites due to stimulation of methanotrophic bacteria (Banger et al. 2012). The use of N-fixing trees in AFS is a common substitution for mineral N fertilizer (Rosenstock et al 2014) and can increase aboveground biomass and N content (Brienza Jr, 1999; Joslin et al. 2011). This increased N content can be used to fertilize subsequent crops as a green manure after burning or mulching. N-fixers can also increase soil-N content, through root turnover or litter decomposition (Danso et al. 1992), yet increases in N2O efflux are inconsistent (Palm et al. 2002; Verchot et al. 2008).
In an AFS in the Kenyan Highlands, rates of N2O efflux were correlated with the N content of residues (i.e., green manure) applied prior to cropping (Millar et al. 2004). Rates of N2O efflux during the first 84-days were ~ 2 vs. 0.2 kg-N2O–N ha−1, respectively, when residues from improved fallows with an N-fixing tree were compared to natural fallow residues. Mulching may also enhance soil-moisture retention under the organic layer (Davidson et al. 2008), which can alter rates of N cycling, and rates of denitrification or methanogenesis due to changes in % water filled pore space (WFPS).
It is unclear how N-fixers and mulching may influence CH4 and N2O fluxes but may be important given the high global warming potentials (GWP) of these gases (Davidson et al. 2004a). Upland forest soils of Brazilian Amazonia are usually sinks for atmospheric CH4 (Schlesinger 1997; Verchot et al. 2008), although soils can be sources of CH4 when saturated (Megonigal, 2004). This research addresses the lack of data regarding trace gas effluxes from secondary forest and agroforestry systems in Amazonia.
In the Amazonian study reported here, in the six years of improved secondary forest fallow growth that followed initial mulching and cultivation, planted I. edulis biomass N-content was the primary driver of increased system N-content (Joslin et al. 2016). After these 6 years, I. edulis was the dominant tree species where planted, comprising an estimated 82 and 71% of planted tree biomass with or without P+K fertilization, respectively. In P+K fertilized treatments Inga represented as much as 87.5% of N in the planted trees and held 2.6-fold more N than N in planted trees when Inga was not included (Joslin et al. 2016, 2019). Upland soils of secondary forests in Amazonia are generally N-limited (Jordan 1985), contributing to low N2O fluxes, but P limitation have also been observed, with productivity increase up to 25% with P-fertilization (Davidson, 2004b; Cunha et al 2022). Fertilization with P can also stimulate activity of soil microbial communities that produce CH4 or N2O, such as methanogenic or nitrifying bacteria (Banger et al. 2012), as can potassium (K) for soil CH4 production (Conrad & Klose 2005). As such, we attempt to estimate the influence of an N-fixing tree and P+K fertilization on N2O efflux and total CO2e of our AFS.
Here we extend the findings on biomass and N contents to rotation age (9 years) and also test the effects of P+K fertilization and the presence of I. edulis on N2O and CH4 fluxes. These trace gas fluxes were measured during the final year of the first crop-fallow rotation (Year 9) and during Year 1 of the second crop-fallow rotation (Year 10). We hypothesized that: 1) fluxes of N2O and CH4 will be greater in the main-plot P+K fertilized treatment, as well as in sub-plot treatment with the presence of I. edulis during the final year of the first crop-fallow rotation; 2) that during Year 1 of the second crop-fallow rotation these differences would increase due to newly added mulch material; and 3) that N2O and CH4 effluxes would be greater nearer (0.25 m) to I. edulis trees than further away (2 m).
Methods
Site description
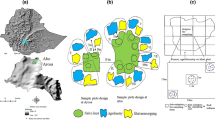
The research was conducted at the Fazenda Experimental de Igarapé Açu (FEIGA) of the Universidade Federal Rural da Amazônia (UFRA) in the Municipality of Igarapé Açu (1°07′41″S 47°47′15″ W), approximately 110 km East of Belém, Pará, Brazil (Fig. S1). One of the oldest continually inhabited agricultural areas in Amazonia, the Bragantina landscape is dominated by urban areas, row-crop farms, plantation forests, cattle ranches, and secondary forests. Primary forests are < 2% of the area (Denich et al. 2004).
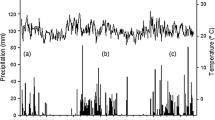
Upland soils in the municipality of Igarapé Açu are predominantly Kandiudults (Rego et al. 1993) with a bulk density (BD) of 1.2 g cm−3 from 0 to 5 cm and 1.4 g cm−3 from 5 to 10 cm (Joslin et al. 2011). Kandiudults are Ultisols in the US Department of Agriculture classification and are typically equated to Acrisols in the Food and Argiculture soil classification. These soils have a kandic horizon, or subsurface accumulation of low activity (< 16 cmolc kg−1 clay) illuvial clays, under a udic (ud) soil moisture regime defined as no periods of 90 days dry down. Igarapé Açu has an average annual temperature of 26°C and annual rainfall of 2500 mm (IBGE, 1996; cited in: Kato et al. 2005); the driest months are August—November and the wettest months are January–May. During the years 2000–2008, the FEIGA weather station recorded mean monthly rainfall over 280 mm from January to May, peaking at 420 mm in March, and minimum rainfall occurring from September–November, reaching its low in November with 30 mm (Fig. S2).
Species descriptions
The five planted tree species are native to forests of the Bragantina region. Inga edulis (Leguminosae) is the only planted known N-fixer, although other N-fixing species may be present due to natural recruitment in secondary succession. Schizolobium amazonicum (Leguminosae) and Ceiba pentandra (Bombacaceae) are rapidly growing pioneers with soft wood. Parkia multijuga (Leguminosae) and Cedrela odorata (Meliaceae) are slower growing tropical hardwoods (See Joslin et al. 2011 for more complete species description).
All five tree species were planted with the food crop manioc (Manihot esculenta), which was harvested after 12 and 20 months during the first crop-fallow rotation. Manioc growth response was reported in Joslin et al. (2011, 2019). After converting secondary forest via mulching, and prior to planting the second rotation, all S. amazonicum were harvested. Inga edulis were cut to stumps with residue placed in the mulch row. Larger individuals of the other species were marked and avoided as best as possible during mulching to allow for their continued growth. Within ten days of mulching via tractor in July 2014, the same tree species were planted along with manioc to begin the second rotation of the crop-fallow cycle.
Plot establishment
In March of 2005, a one-hectare study site was created from a 7-year-old secondary forest within FEIGA by clearing via mulching tractor as described by Joslin et al. (2011). Experimental treatments were applied to the prepared site in June 2005, where four blocks (N = 4) were divided into four 24 × 24 m plots (n = 16). Trees and manioc were planted simultaneously, with trees planted at 4 × 1.8 m spacing, for a total of 78 trees per plot, or 1,354 trees ha−1. The crop species M. esculenta was planted at 1 × 1 m spacing, for a rate of 10,000 stems ha−1.
A factorial combination of fertilization treatment and N-fixing species additions was assigned in a split-plot randomized complete block design. The main-plot fertilizer treatment (Fert) consisted of no fertilization (PK-) and fertilization (PK+) as an application around the base of planted trees of 46 kg P ha−1 as P2O5 (100 kg of 46% Simple-Super Phosphate) and 30 kg K ha−1 applied as KCl (50 kg of 60% KCl). Sub-plot treatments (Nfix) consisted of planting 26 trees each of the native species S. amazonicum, C. odorata and C. pentandra together (I-), or in combination with the N-fixing species I. edulis as well as P. multijuga (I+). In I+ treatment, I. edulis, accounted for half of trees planed, while the remainder was divided evenly among the remaining species.
Gas sampling
Gas sample collections, as well as soil and litter layer sampling, were performed over two years: during the final year of secondary forest growth of the first crop-fallow cycle (Rotation 1) and after conversion via mulching tractor to begin the second crop-fallow cycle (Rotation 2; See Table 1). Samples were collected in May and November of 2013, and March and late May of 2014 to complete Rotation 1. Samples were collected in early July 2014 (one week after site preparation), two weeks later in late July, November 2014, March 2015, and July 2015. To capture seasonality in rainfall samples were collected by: Rainy Season (Jan.–May), Transition Season (June–Aug.) and Dry Season (Sept.–Dec.). No soil moisture measurements or soil samples were collected during the dry seasons of 2013 and 2014.
Gas sampling was conducted using 15-cm diameter PVC ventilated static chambers (Pihlatiea et al. 2013). Eight chambers were installed per plot 2.5 cm into the soil 1-h prior to closing the chamber. After closing the chamber, a sample was drawn in 10 mL syringes and stored in 7 mL pre-evacuated vials (Exetainers, Labco Inc., UK) immediately (T = 0), and again at T = 10, 30, and 50 min. Vials were taken to the University of Georgia, Athens, GA for analysis. Samples were analyzed using a gas chromatograph with electron capture (N2O) and flame ionization (CH4) detectors (Shimadzu Corporation GC-2014, Kyoto, Japan). Gas efflux for N2O and CH4 were calculated following Shrestha et al. (2014). Soil efflux data was not normally distributed for either N2O or CH4 when analyzed as a single data set or when analyzed by Rotation, so all gas efflux data were log-transformed for statistical analysis.
Gas sampling by distance from tree
To assess the effect of the N-fixer on gas efflux by distance from the tree, sampling transects of 25, 50, 100, and 200 cm from each of two randomly selected trees were installed in all plots; in plots with I. edulis (I+) one of the trees sampled was I. edulis (I) paired with one randomly selected non-Inga tree of the remaining species (nI). In plots without I. edulis (I-), pairs of trees were randomly selected as n1 and n2.
For analysis of the flux data, means were calculated for trees in multiple ways. First, at the plot level, analysis of means for the paired trees, regardless of species, were pooled for a single mean efflux for the plot. A second level of analysis compared the paired trees within treatment. A third level of analysis pooled both trees in the I- plot and compared that mean to the mean of the sampled I. edulis tree (I) and non-Inga tree (nI) in the I+ plot.
Soil moisture and soil sampling
Soil moisture measurements were taken with a Hydrosense II soil moisture probe (Campbell Scientific, Logan, UT) after static chamber caps were removed. During two sampling events in the dry season VWC could not be measured because the probe could not be fully inserted into the soil. Also, the soil moisture probe failed during the July 2015 sampling. Volumetric water content (VWC) data from the available measurement dates were normally distributed across the entire data set.
After gas and VWC measurements were completed, the mulch layer and 0–10 cm soil were sampled from within the static flux chambers in each plot. Soil samples were air-dried and sieved through a 2 mm screen. Subsamples were then ground to powder using a vial roller or ball mill grinder (SPEX, Metuchen, NJ). Soil and litter samples were analyzed for carbon (C) and total nitrogen (TN) concentration using a CN analyzer (CE Elantech, Lakewood, NJ). Soil samples were also extracted for analysis with 1 M KCl to assess available N and analyzed for Total Extractable N (TEN) via chemiluminescence (TNM-L, Shimadzu Corporation, Kyoto, Japan). This measure of total soil N and total extractable N was used to evaluate soil N controls on N2O efflux. Soil samples extracted with 1 M KCl were also analyzed for NH4-N using flow injection analysis (OI Analytical, College Station, TX).
Soil N availability
To capture plot-level N availability over a one-year time frame, we utilized buried resin capsules (UniBest PST-1, Bozeman, MT) at the bottom of 10 cm deep × 2 cm diameter PVC plastic tubes. Rotation 1 sampling spanned the year of pre-mulching gas sampling (May 2013–May 2014) and Rotation 2 sampling took place from site conversion in July 2014 until July 2015. A plastic tube was inserted into the soil, removed with soil, and the resin capsule was placed inside the bottom of the tube and replaced into the soil. Resin capsules were extracted with 2 M KCl solution and analyzed for Resin-captured N (RCN), as was describe above for TEN. Total RCN (mg L−1) in the extracts was compared across treatments.
Statistical analyses
All statistical analyses were performed using SAS (SAS, Cary, NC, USA) using p < 0.1 as significant. T-test analysis indicated that soil gas fluxes differed between Rotations 1 and 2 for both N2O and CH4 (P > t: p < 0.001), so all analyses for soil gas fluxes were conducted within Rotation for each gas. Soil GHG fluxes were analyzed using Proc MIXED. In the factorial model the main-plot treatment was with or without P+K fertilization (Fert), and the split-plot treatment was with or without the presence of the N-fixing tree species I. edulis (Nfix). Fert and Nfix were fixed factors and Block was a random factor. Repeated measures analysis was used to test Date and Treatment*Date effects with plot as the repeated measure subject.
Regression analysis was used to test the effects of environmental variables, including pH, and soil and litter layer C and N concentrations, on N2O and CH4 flux using Proc REG. Since not all variables were present during each sampling date, multiple regression analysis was supplemented by individual regressions of each variable against N2O and CH4 flux to take advantage of all available data. Statistical nomenclature for Trees in the transect sampling is as follows: Inga edulis trees = I, non-Inga trees in the I+ plot = nI trees in the I- plot = nN.
Results
Nitrous oxide
Treatment-level responses
At the end of Rotation 1, 9 years after study initiation, mean soil N2O fluxes from May 2013 to May 2014 ranged from 8.1 to 13.7 µg m−2 h−1, with the lowest flux coming from plots without I. edulis (I-) and the greatest mean flux coming from the full factorial treatment (PK+I+). Soil efflux of N2O differed in the presence of I. edulis (I+), with a mean of 12.5 ± 5.4 compared to 8.2 ± 5.4 µg m−2 h−1 in the I- treatment (Fig. 1). However, N2O efflux did not differ in the main-plot (Fert), with no Fert*Nfix interaction during Rotation 1 (Table S1).
Soil CH4 and N2O efflux in a slash-and-mulch improved-fallow agroforestry system in eastern Amazonia of Brazil. Main plot treatment (Fert) with P+K fertilization (PK+) or without (PK-), and sub-plot treatment (Nfix) planted with I. edulis (I+) or without (I-). Sampling was performed for 1-year at the end of Rotation 1 and during the year following site preparation with a mulching tractor at the beginning of Rotation 2. Capital letters indicate effect of Rotation, lower case letter indicate differences between treatments within a rotation
Soil N2O efflux differed by Date during Rotation 1 (p < 0.001; Table S1) and was greatest in May 2013 and again in May 2014, and lowest during November 2013 (Fig. 2). Neither the Fert*Date nor Fert*Nfix*Date interaction was significant, but the Nfix*Date interaction was significant (p < 0.01; Table S1), with the flux lowest in November 2013 in the I- treatment (2.7 ± 4.4 µg m−2 h−1) and highest during May 2014 in the I+ treatment (17.9 ± 5.0 µg m−2 h−1; Fig. 2).
Soil N2O efflux (mean ± 1SE) in a slash-and-mulch improved-fallow agroforestry system in eastern Amazonia of Brazil. Main plot treatment (N = 4) with P+K fertilization (PK+) or without (PK-), and sub plot treatment planted with I. edulis (I+) or without (I-). Sampling was performed for 1-year at the end of Rotation 1 and during the year following site preparation with a mulching tractor at the beginning of Rotation 2
After slash-and-mulch conversion of the 9-yr-old secondary forest, Rotation 2 measurements (July 2014 to July 2015) of soil N2O fluxes ranged from 20.8 to 26.4 µg m−2 h−1 (Fig. 2), but did not differ in main-plot or sub-plot treatments, or in the Fert*Nfix interaction (Tables 2 and S1). Soil N2O efflux differed by Date during Rotation 2 (p < 0.0001; Table S1) and was greatest during the month after secondary forest conversion (July 7 and 28, 2014; Fig. 2), but other interactions -by Date did not differ.
Tree-level responses
Soil N by distance from sampled trees did not differ across sampled dates (Tables S2 and S2). However, in these transect samples, P+K fertilization increased soil N (i = 0.09), but I. edulis did not. Further, in May 2014, in the final month of secondary succession, soils in transects from Inga trees had higher N than non-Inga trees in I+ plots (p = 0.02), but Inga trees did not differ from trees in I- plots. Soil-N in transect sampled soils did not differ in Rotation 2.
For measurements with distance from tree, soil N2O efflux measured by Tree type ranged from 8.2 to 12.8 µg N2O–N m−2 h−1, producing a trend of increased flux in the presence of I. edulis and I+ trees, compared to trees in the I- treatment (p = 0.10; Table 3). The Tree*Date interaction (p < 0.01) revealed that soil N2O efflux was lowest in May 2013 for trees in I- plots and was highest in March 2014 near non-Inga trees in the I+ plots. Soil N2O efflux did not differ by distance from tree during Rotation 1.
After mulching, soil N2O efflux measured with distance from trees ranged from 22.9 – 25.5 µg N2O–N m−2 h−1 but did not differ between Tree types (Table 3). There was a trend of decreased flux after the initial post-mulching sampling in the Tree*Date interaction (p = 0.07; Fig. 2), with N2O flux highest in association with I. edulis in July 2014 and lowest in November 2014 for all Tree types. Distance from planted trees did not affect soil N2O efflux during Rotation 2 (Tables 3 and S3).
Methane
Treatment-level responses
At the end of Rotation 1, 9 years after initial mulching, soil CH4 fluxes ranged from 6.5 – 31.8 µg m−2 h−1 between May 2013 and May 2014 (Fig. 1), with the lowest flux in the control treatment (PK-I-) and highest in the full factorial treatment (PK+I+). Soil efflux of CH4 did not differ in the main-plot or in the sub-plot treatments during Rotation 1. Soil CH4 efflux varied by Date during Rotation 1 (p < 0.01) and was lowest in May 2014 (Fig. 3), but no other interactions -by Date were significant (Table S1).
Soil CH4 efflux (mean ± 1SE) in a slash-and-mulch improved-fallow agroforestry system in eastern Amazonia of Brazil. Main-plot treatment (N = 4) with P+K fertilization (PK+) or without (PK-), and sub-plot treatment planted with I. edulis (I+) or without (I-). Sampling was performed for 1-year at the end of Rotation 1 and during the year following site preparation with a mulching tractor at the beginning of Rotation 2
For Rotation 2 measurements, July 2014–July 2015, soil CH4 efflux ranged from −1.4 to 7.6 µg m−2 h−1 (Fig. 1) but did not differ in the main-plot or the sub-plot treatments (Table S1). The only consumption of CH4 reported here was in the control treatment, −1.4 ± 17.9 µg m−2 h−1. Soil CH4 efflux differed by Date during Rotation 2 (p < 0.01; Table S1) and was greatest in the four weeks after mulching (July 7 and 28, 2014) and lowest during March 2015 (Fig. 3), but no other interactions -by Date were significant (Table S1).
Tree-level responses
Soil-C by distance from sampled trees did not differ (Tables S2 and S3). However, in these transect samples, P+K fertilization increased soil-C (p = 0.07). The Fert*Tree interaction (p = 0.10) revealed that Inga with P+K fertilization had greater soil-C than unfertilized Inga trees (p = 0.06) and P+K fertilized trees in the I- plot had higher soil-C than unfertilized Inga (p = 0.02). In the final measurement of secondary succession, soils in transects from Inga trees had higher C than non-Inga trees in I+ plots (p < 0.01), but Inga trees did were not differ from trees in I- plots. The Tree*Distance interaction was significant in May 2014 (p < 0.001), in which soil-C near I trees was greater at all distances than soil-C at 0.5 m distant from nI trees.
For measurements by distance from tree, soil CH4 efflux ranged from 9.2 to 28.6 µg m−2 h−1 at the individual tree level during Rotation 1 (Table 2), but no differences were detected for other variables or interactions. After mulching to initiate Rotation 2, soil CH4 efflux with distance from planted trees generated between 0.5—10.3 µg m−2 h−1, in which there was a Tree*Distance interaction (p = 0.02; Table 3) as CH4 efflux associated with I. edulis was lower than I- trees at 1.0 m. No differences were detected for other variables.
Environmental responses
Soil attributes
Soil pHH2O and pHCaCl2 (Table S4) did not differ in the main or sub-plot. Neither surface soil (0-10 cm) pHH2O nor pHCaCl2 predicted CH4 nor N2O efflux in Rotations 1, 2, or overall. Mean soil volumetric water content (VWC) was 28.2 ± 1.2% among measured dates, although for three of the driest sampling dates VWC could not be collected. The lowest VWC (%) was in the rainy season (March 2015) sampling of Rotation 2 (23.3% ± 2.3). The highest soil VWC readings (31.3 ± 1.6 and 31.2 ± 1.4%, respectively) were recorded in the final sampling prior to, and the first sampling after, mulching for Rotation 2 (May 2014 and July 7, 2014). VWC was second highest in May 2013 and July 28, 2014 (28.3% ± 2.0 and 28.1% ± 1.3, respectively), while the median VWC value (24.8% ± 1.3) was recorded in March of 2014. VWC did not differ by Fert or Nfix and there was no Fert*Nfix interaction. Rotation 1 and 2 VWC did not differ, but soil VWC differed by Date (p < 0.001) and Fert*Date interaction (p < 0.0001), with highest VWC during the first sampling event after mulching on July 7, 2014 in the PK + treatment, and the lowest in PK+ treatment in March 2014 prior to mulching.
Environmental controls on N2O and CH4 efflux
Stepwise regression for N2O efflux did not yield significant predictors in either Rotation. Regression for CH4 efflux found that VWC and total N recovered with resin capsules (RCN; Table 4) were significant regressors across both Rotations (Table 5). RCN was negatively correlated to CH4 efflux (p.e. = −0.04), and was a weak predictor in the multivariate regression (Partial R2 = 0.08), but logVWC was positively correlated with CH4 efflux (p.e. = 0.8) and was a strong predictor (R2 = 0.7). During Rotation 1, stepwise regression showed that logVWC, TN, and Litter-C concentration were the best predictors of soil CH4 efflux, explaining 58% of the variation (Table 5).
Since all variables were not available during each sampling event for multivariate regression, univariate regression analyses were performed for each variable (Table 6). Despite the larger sample size for the univariate regressions, there were no significant regressors for N2O efflux in either Rotation. In contrast, logVWC and TN concentration were significant during Rotation 1 for CH4 efflux (p = 0.001 and 0.02, respectively) while soil-C:N and logNH4 were significant for CH4 efflux during Rotation 2 (p = 0.05 and 0.04, respectively).
Discussion
The environmental benefits of AFS with N-fixers has been questioned due to the potential for increased GHG efflux (Rosenstock et al 2014; Kou-Giesbrecht & Menge 2019, 2021). In this research we created a novel system for the simultaneous cultivation of the staple crop manioc with native trees for eventual timber sale. We also incorporated the N-fixer I. edulis for improved fallow and green manure. To evaluate the impact of this novel, slash-and-mulch system we measured the seasonal soil effluxes of N2O and CH4 during the year prior to, and the year following, site conversion by mulching tractor. We expected that use of P+K fertilizer (PK+) and the inclusion of the N-fixing tree Inga edulis (I +) would increase soil fluxes of both CH4 and N2O, that the newly deposited mulched biomass layer would increase fluxes of both, and that fluxes of both would be greater nearer to individual I. edulis trees.
Environmental responses
Slash-and-mulch research from the Bragantina region has shown increased soil water retention, by up to 22%, compared to slash-and-burn (Comte et al. 2012). Fluxes of both CH4 and N2O are affected by VWC; anaerobic soils increase flux of both gases, while aerobic soils often result in CH4 consumption (Segers 1998; Butterbach-Bahl et al. 2013). In this research, methane was consumed in control plots during the dry season but consumption was reduced or production increased during the rainy season, which is consistent with previous findings (Vasconcelos et al. 2004). Davidson et al. (2008) reported consumption of CH4 in secondary forests in Eastern Amazonia, and emissions of CH4 immediately after mulching of secondary forests, but did not correlate these findings to VWC.
Soil moisture was a significant predictor during both Rotations for CH4, but only marginally important for N2O (p = 0.13). Within each Rotation the influence of VWC was less clear, as it was a good predictor of CH4 and N2O efflux in Rotation 1 but not for either gas during Rotation 2. Nine years of secondary forest growth and mulch decomposition may have stabilized soil N and C concentrations, and soil moisture distribution, reducing soil nutrient and moisture variability. However, the large pulse of woody material put onto the soil surface during site preparation likely caused heterogeneous distribution of N and C resources, and soil moisture conditions leading to inconsistent responses to VWC (Millar et al. 2004; Kravchenko et al. 2017). The lack of dry season soil moisture data, and thus a limited distribution of VWC values, likely also impacted regression relationships. Previous research found better relationships for N2O efflux with %WFPS across broad (20–100%) ranges (Davidson et al. 2001; Reich et al. 1997).
Other environmental variables measured were inconsistent in predicting soil CH4 and N2O efflux. Soil pH, for instance, lowers soil methanogenic and nitrifying bacterial activity at low pH (Oertel et al. 2016), yet we observed no effects of soil pH on efflux of either gas. Production of CH4 and N2O are dependent on C and N resources in the soil on which bacteria act (Hütsch 1998; Bodelier & Laanbroek 2004), yet the data presented here showed variable responses. For example, when analyzed across both Rotations, CH4 was correlated with extractable TN (i.e., TEN) and total soil N (i.e., TN), but not with any C variables. Alternatively, N2O efflux was not related to any N variables during either Rotation, but was correlated to litter layer mass and litter layer C content. It is difficult to draw strong conclusions about environmental variables controlling efflux rates from these data.
Tree level responses
We hypothesized that plots with the N fixing tree I. edulis would have greater N2O efflux and measured rates of N2O efflux would decrease with increasing distance from the stem of I. edulis due to soil N changes from root and leaf litter inputs (Tobita et al 2010). By the end of Rotation 1, the I- treatment generated less N2O than the I+ treatment, but within the I+ treatment tree types did not differ. These findings suggest that the influence of I. edulis on N cycling and N2O efflux was not constrained to the canopy cover or dripline of the tree but had an influence on N2O efflux at the plot level. This may be due to the high density of Inga trees in that treatment (50% of all trees in I+), as well as biotic factors (such as monkeys, coatimundi, and ants, which were all frequently observed during Rotation 1) above and below ground that have the potential to distribute N-rich Inga components (such as fruits and leaves).
Treatment level responses
Methane
We hypothesized that both fertilization with P+K and inclusion of the N-fixing I. edulis would enhance soil CH4 efflux. Despite large CH4 efflux percentage differences these effects were not significant. In a controlled setting, effects of P+K fertilization on CH4 efflux were observed (Conrad & Klose 2005) but the large variance in this field experiment indicates that other factors obscured contributions to higher CH4 efflux.
Mean CH4 efflux was generally greater in Rotation 1 than 2, with the PK+I- treatment as the exception. Enhanced porosity in the surface soil due to the input of mulched material likely played a role in reducing CH4 efflux in Rotation 2 via increased aeration. Soil CH4 efflux increased immediately after mulching and decreased over the following year as the mulch layer decomposed. This pattern is consistent with other slash-and-mulch studies from Eastern Amazonia, in which soil CH4 efflux peaked after mulching and planting, but then declined soon after, and remained relatively stable over the next year (Davidson et al. 2008). Conversion of tropical forests to agriculture has also shown either reduced rates of consumption of CH4 (Keller et al. 1990), or production of CH4 (Keller & Reiners 1994).
Relatively large rates of CH4 production immediately after mulching and sustained consumption as soon as four months afterward indicate that site disturbance itself is a primary influence on CH4 efflux. The most intense agricultural activity in this research took place at and directly after mulching (via tree and crop planting), with virtually no disturbance activities during the following year of gas efflux measurement, which is consistent with research that showed a positive correlation between intensity of agricultural activity in AFS and CH4 efflux (Palm et al. 2002).
Nitrous Oxide—N2O
By the end of Rotation 1 inclusion of the N-fixing tree I. edulis increased N2O efflux by 52%. In other research from Eastern Amazonia, inclusion of the N-fixer I. edulis increased N2O efflux during secondary succession between 0.25 – 0.47 kg N ha−1 yr−1 (38 – 76%), and in improved-fallow increased (N2O + NO)-N by 0.17 kg N ha−1 yr−1 (8.5%; Verchot et al. 2008). However, in the present study, P+K fertilization did not cause an increase in N2O production in either Rotation.
An increase in N2O emissions during the first month after site preparation and deposition of mulched material returned to pre-mulch levels 1 year after mulching, which is consistent with other research showing large pulses of N2O emissions after inorganic N fertilization (Signor et al. 2013) and after green manure application (Millar et al. 2004). Incorporating green manure from N-fixing trees of an improved-fallow AFS increased N2O soil efflux by 620—1,725% over 84 days, and as much as 90% of N2O-N loss occurred during the first 30 days (Millar et al. 2004), where the highest rates of N2O-N loss accompanied the lowest C:N mulch (113—560 g-N ha−1 ton−1 of leguminous mulch vs. 30 g-N ha−1 ton−1 with unimproved fallow mulch; Table S5). Data presented here indicate that 515 g-N ha−1 were lost as N2O over the 28 days from the date of mulching until the end of July, equivalent to 24% of total emissions during the first year after mulching (Table S6), or 0.14% of newly added mulch layer N. Incorporation of organic residue in the Millar et al. (2004) study led to ~ 1.25% of applied N lost as N2O. Inorganic N fertilizers can cause greater losses, as found in Brazil, where 1–12% of N applied as urea and ammonium nitrate was lost as N2O (Signor et al. 2013). Results here indicate that mulched secondary vegetation is more conservative of agroecosystem N than biomass burning or applying mineral-N fertilizers.
Plant residue can create “hot spots” of N2O emissions by absorbing water, creating conditions for nitrification and/or denitrification (Kravchenko et al. 2017). Mulch layer additions at site preparation for Rotation 2 added 106 Mg ha−1 of plant residue and 360 kg N ha−1, contributing to increased N2O flux compared to Rotation 1. Soil N2O efflux increased immediately after mulching, decreasing over time as the forest floor litter layer decomposed. However, the spatial distribution of soil N2O efflux was not due to the proximity of planted N-fixing tree species. These data suggest that N-fixing tree species planted in improved-fallow systems contributed to enhanced soil N2O efflux by enhancing N content of the agroecosystem at the plot scale. Other research from tropical forests indicates that N-fixing trees do not facilitate growth or N acquisition of neighboring trees (Taylor et al. 2017) or increase biomass recovery of secondary forests (Lai et al. 2018). In a meta-analysis (Kou-Giesbrecht & Menge 2021), reporting mostly on temperate forests, increased N2O efflux at the stand level was also evident. Results reported here, however, agree most closely with Verchot et al. (2008) that inclusion of I. edulis in an improved-fallow mix increased soil N2O fluxes.
Annualized trace gas fluxes
The above ANOVA analyses for gas fluxes across time and four replicate blocks are robust for assessing the treatment responses. To assess the potential year-long GHG contribution, however, we annualized the soil CH4 flux rates, which adds uncertainty but follows previously published research (Verchot et al. 1999). We extrapolated the measured flux rate (µg hr−1) by 24 h day−1 by the number of days in each season for the available data to provide an order of magnitude estimate. Annualized soil CH4 flux rates were higher in each treatment during Rotation 1 than for any treatment during Rotation 2 (Table S6), with fluxes ranging from 2.7 to -0.3 kg CH4 ha−1 yr−1 during Rotations 1 and 2, respectively. Annualized soil CH4 flux rates estimated here fall within the range of flux rates reported in other studies from Amazonia (Vasconcelos et al. 2004; Davidson et al. 2004a and 2008; Verchot et al. 2000; Cattânio et al. 2002; Fernandes et al. 2002; Nepstad et al. 2002). However, following mulch treatment the annualized flux rates estimated here for CH4 are one order of magnitude lower (0.3 vs. 10.2 kg CH4 ha−1 yr−1) than reported by Davidson et al. (2008). In contrast, our estimates are very similar during fallow (2.7 vs. 2.4 kg CH4 ha−1 yr−1) to those from a slash-and-mulch study in close proximity (<1 km) to the current research location. In Davidson et al. (2008) mulching increased CH4 efflux, while in the current study, as soon as four months after site conversion via mulching tractor, soils became a sink for CH4, although fertilized plots consumed less CH4 than unfertilized plots.
Estimates of annualized soil N2O flux rates were generated in the same way as for CH4, and, in contrast to patterns of CH4 emissions, decreased in each treatment during Rotation 2 compared to any treatment during Rotation 1 (Table S6), with fluxes ranging from 0.6 to 2.9 kg N2O–N ha−1 year−1. The range of soil N2O-N flux reported here is similar to other research in Amazonia (0.33–6.9 kg N ha−1 year−1; Vasconcelos et al. 2004; Davidson et al. 2001; Verchot et al. 1999, 2000, and 2008; Cattânio et al. 2002; Fernandes et al. 2002; Nepstad et al. 2002; Palm et al. 2002; Table S5). Compared to Davidson et al. (2008) emissions reported here for N2O are lower during fallow and following mulch treatment. At the end of the fallow during Rotation 1, the unfertilized treatment with I. edulis (PK-I+) generated 0.25 kg N ha−1 yr−1 (38%) more N2O than control (PK-I-), while the full factorial treatment of fertilization with I. edulis (PK+I+) produced 0.47 kg N ha−1 yr−1 (76%) more N2O than fertilization without I. edulis (PK+I-). After mulching, total emissions of N2O increased such that during Rotation 2 there were 1.36 kg N ha−1 year−1 (89%) greater emissions from PK+I+ than from PK+I-.
CO2 Equivalents of N2O and CH4 emissions
We used the methodology in Davidson et al. (2008) to estimate the global warming potential (GWP) of the slash-and-mulch treatments when compared to slash-and-burn. Again, the limited number of sampling dates creates uncertainty but the annualized estimates can provide a sense of changes in magnitude. Since we did not perform a burn treatment, we used the estimates from Davidson et al. (2004a) to estimate the N2O and CH4 emissions that would have been created by burning of the mulched biomass at site conversion, relative to the 99.6 Mg ha−1 of biomass from that study, and compared those values to the observed fluxes during the first year after mulching for Rotation 2. In addition to the fluxes of N2O and CH4, we added CO2 generated by the mulching tractor, 780 kg CO2 ha−1, to all mulching treatments, and 12.5 and 11 kg ha−1 CO2 equivalents for P and K fertilizer. Release of CO2 during burning and during mulch decomposition were not considered.
Emissions of N2O and CH4 during burning alone were up to one order of magnitude greater than emissions Year 1 post-mulching, so regardless of whether N-fixers were included in the planting mix, the improved-fallow slash-and-mulch system had far lower estimated emissions than burning (Table 7). Concerns about the release of N2O due to the inclusion of N-fixers in forests or AFS should be balanced against these potentially greater impacts of burning or mineral N fertilization (Rosenstock et al. 2014; Kou-Giesbrecht & Menge 2021). However, burning of the improved-fallow, full-factorial treatment (PK+I+) is estimated to generate nearly twice the CO2 equivalents as the control (PK-I-) largely due to greater biomass. Inclusion of I. edulis in the planting mix had no effect on CH4 efflux, although CH4 efflux declined in all treatments after mulching reducing GWP. For N2O emissions, the incorporation of the N-fixing tree had no effect on GWP after mulching without fertilization but increased GWP by 33% with P + K fertilization.
Conclusions
Inclusion of the N-fixing tree species I. edulis in this improved-fallow slash-and-mulch agroforestry system significantly increased soil flux of N2O but only after site preparation via mulching tractor. I. edulis did not enhance CH4 emissions. Mean values for CH4 and N2O were higher in the presence of the N-fixing tree, but high variance negated the statistical significance of these observations. The effects of the N-fixing tree were detectable at the plot level, but not at the individual tree level or by distance from the tree, suggesting that the mechanism of flux enhancement is not restricted to interactions with individual trees but rather at a broader spatial scale. Fertilization with P+K did not significantly impact soil CH4 or N2O flux. Substitution of fire with mulching for site preparation will benefit agricultural GHG budgets by reducing N2O and CH4 emissions. Incorporation of the N-fixer I. edulis in the planting design of the AFS had a minimal impact on GWP. Use of slash-and-mulch technology reduced total GHG emissions and the inclusion of N-fixing species in improved-fallow practices did not eliminate those benefits.
Data availability
Data is available at: https://docs.google.com/spreadsheets/d/1alM9k1_lUMk3Xo_urTlsXbfF6foZbgyG/edit#gid = 156011827.
References
Banger K, Tian H, Lu C (2012) Do nitrogen fertilizers stimulate or inhibit methane emissions from rice fields? Glob Change Biol 18(10):3259–3267
Basiliko N, Khan A, Prescott CE, Roy R, Grayston Sue J (2009) Soil greenhouse gas and nutrient dynamics in fertilized western Canadian plantation forests. Can J for Res 39(6):1220–1235
Bodelier PLE, Laanbroek HJ (2004) Nitrogen as a regulatory factor of methane oxidation in soils and sediments. FEMS Microbiol Ecol 47(3):265–277
Brienza Jr S (1999) Biomass dynamics of fallow vegetation enriched with leguminous trees in the Eastern Amazon of Brazil. PhD Dissertation, Georg-August-Univ, Göttingen, Germany
Brondizio ES, Cak A, Caldas MM, Mena C, Bilsborrow R, Futemma CT, Ludewigs T, Moran EF, Batistella M (2009) Small farmers and deforestation in Amazonia. Amazonia Glob Change Geophys Monogr Ser 186:117–143
Butterbach-Bahl K, Baggs EM, Dannenmann M, Kiese R, Zechmeister-Boltenstern S (2013) Nitrous oxide emissions from soils: How well do we understand the processes and their controls? Philos Trans R Soc B 368:20130122. https://doi.org/10.1098/rstb.2013.0122
Campos MT, Nepstad DC (2006) Smallholders, the Amazon’s new conservationists. Conserv Biol 20(5):1553–1556
Castro MS, Peterjohn WT, Melillo JM, Steudler PA, Gholz HL, Lewis D (1994) Effects of nitrogen fertilization on the fluxes of N2O, CH4, and CO2 from soils in a Florida slash pine plantation. Can J for Res 24(1):9–13
Castro MS, Steudler PA, Melillo JM, Aber JD, Bowden RD (1995) Factors controlling atmospheric methane consumption by temperate forest soils. Global Biogeochem Cycles 9(1):1–10
Cattânio JH, Davidson EA, Nepstad DC, Verchot LV, Ackerman IL, IA, (2002) Unexpected results of a pilot throughfall exclusion experiment on soil emissions of CO2, CH4, N2O, and NO in eastern Amazonia. Biol Fertil Soils 36:102–108
Comte I, Davidson R, Lucotte M, de Carvalho CJR, Oliveira FA, da Silva BP, Rousseau GX (2012) Physicochemical properties of soils in the Brazilian Amazon following fire-free land preparation and slash-and-burn practices. Agr Ecosyst Environ 156(1):108–115
Conrad R, Klose M (2005) Effect of potassium phosphate fertilization on production and emission of methane and its 13C-stable isotope composition in rice microcosms. Soil Biol Biochem 37(11):2099–2108
Cunha HFV et al (2022) Direct evidence for phosphorus limitation on Amazon forest productivity. Nature 608:558–562
Danso SKA, Bowen GD, Sanginga N (1992) Biological nitrogen fixation in agro-ecosystems. Plant Soil 141:177–196
Davidson EA, Bustamente MMC, Pinto AS (2001) Emissions of nitrous oxide from soils of native and exotic ecosystems of the Amazon and Cerrado regions of Brazil. The Scientific World 1(S2):312–319
Davidson EA, Ishida FY, Nepstad DC (2004a) Effects of an experimental drought on soil emissions of carbon dioxide, methane, nitrous oxide, and nitric oxide in a moist tropical forest. Glob Change Biol 10:718–730
Davidson EA, de Carvalho CJR, Vieira ICG, Figueiredo RO, Moutinho P, Ishida FY, dos Santos MTP, Guerrero JB, Kalif K, Sabá RT (2004b) Nitrogen and phosphorus limitation of biomass growth in a tropical secondary forest. Ecol Appl 14(4):S150–S163
Davidson EA, Sá TDAB, Carvalho CJR, Figueiredo RO, Kato MSA, Kato OR, Ishida Y (2008) An integrated greenhouse gas assessment of an alternative to slash-and-burn agriculture in eastern Amazonia. Glob Change Biol 14:998–1007
Denich M, Vielhauer K, Kato MSA, Block A, Kato OR, Sá TDA, Lücke W, Vlek PLG (2004) Mechanized land preparation in forest-based fallow systems: the experience from Eastern Amazonia. Agrofor Syst 61:91–106
Fernandes SAP, Bernoux M, Cerri CC, Feigl BJ, Piccolo MC (2002) Seasonal variation of soil chemical properties and CO2 and CH4 fluxes in unfertilized and P-fertilized pastures in an Ultisol of the Brazilian Amazon. Geoderma 107(3–4):227–241
Hütsch BW (1998) Methane oxidation in arable soil as inhibited by ammonium, nitrite, and organic manure with respect to soil pH. Biol Fertil Soils 28:27–35
Jordan CF (1985) Nutrient cycling in tropical forest ecosystems: principles and their application in management and conservation. Wiley and Sons, New York
Joslin AH, Markewitz D, Morris LA, Oliveira FA, Figueiredo RO, Kato OR (2011) Five native tree species and manioc under slash-and-mulch agroforestry in the eastern Amazon of Brazil: plant growth and soil responses. Agrofor Syst 81(1):1–14
Joslin A, Markewitz D, Morris LA, Oliveira FA, Kato OR (2016) Improved fallow: growth and nitrogen accumulation of five native tree species in Brazil. Nutr Cycl Agroecosyst 106(1):1–15
Joslin AH, Vasconcelos SS, Oliviera FA, Kato OR, Morris LA, Markewitz D (2019). A slash-and-mulch improved-fallow agroforestry system: growth and nutrient budgets over two rotations. Forests 10:1125, Published On-line 10 December, 2019 | https://doi.org/10.3390/f10121125
Kato MSA, Kato OR, Denich M, Vlek PLG (1999) Fire-free alternatives to slash-and-burn for shifting cultivation in the eastern Amazon region: the role of fertilizers. Field Crop Res 62(2–3):225–237
Kato OR, Kato MSA, de Carvalho CR, Figueiredo RO, Sá TDA, Vielhauer K, Denich M (2005) Manejo de vegetação secundária na Amazônia visando ao aumento da sustentabilidade do uso agrícola do solo. XXX Congresso Brasileiro de Ciência do Solo. Recife, Brazil
Kou-Giesbrecht S, Menge D (2019) Nitrogen-fixing trees could exacerbate climate change under elevated nitrogen deposition. Nat Commun 10:1493. https://doi.org/10.1038/s41467-019-09424-2
Kou-Giesbrecht S, Menge DNL (2021) Nitrogen-fixing trees increase soil nitrous oxide emissions: a meta-analysis. Ecology 102(8):e03415. https://doi.org/10.1002/ecy.3415
Kravchenko AN, Toosi ER, Guber AK, Ostrom NE, Yu J, Azeem K, Rivers ML, Robertson GP (2017). Hotspots of soil N2O emission enhanced through water absorption by plant residue. Nat Geosci, Published Online: 5 JUNE 2017 | https://doi.org/10.1038/NGEO2963
Keller M, Mitre ME, Stallard RF (1990) Consumption of atmospheric methane in soils of central Panama: Effects of agricultural development. Global Biogeochem Cycles 4(1):21–27
Keller M, Reiners WA (1994) Soil-atmosphere exchange of nitrous oxide, nitric oxide, and methane under secondary succession of pasture to forest in the Atlantic lowlands of Costa Rica. Global Biogeochem Cycles 8(4):399–409
Lai HR, Hall JS, Batterman SA, Turner BL, van Breugel M (2018) Nitrogen fixer abundance has no effect on biomass recovery during tropical secondary forest succession. J Ecol 2018:1–13. https://doi.org/10.1111/1365-2745.12979
Megonigal JP, Hines ME, Visscher PT (2004) Anaerobic metabolism: linkages to trace gases and aerobic processes. In: Schlesinger WH (ed) Biogeochemistry. Elsevier-Pergamon, Oxford, pp 317–424
Millar N, Ndufa JK, Cadisch G, Baggs EM (2004). Nitrous oxide emissions following incorporation of improved-fallow residues in the humid tropics. Glob Biogeochem Cycles 18:GB1032, https://doi.org/10.1029/2003GB002114
Nepstad D, Moutinho P, Dias-Filho MB, Cardinot DE, G, Markewitz D, Figueiredo R, Vianna N, Chambers J, Ray D, Guerreiros JB, Lefebvre P, Sternberg L, Moreira M, Barros L, Ishida FY, Tohlver I, Belk E, Kalif K, Schwalbe K, (2002) The effects of partial throughfall exclusion on canopy processes, aboveground production, and biogeochemistry of an Amazon forest. J Geophys Res 107(D20):8085. https://doi.org/10.1029/2001JD000360
Nepstad D, Soares-Filho BS, Merry F, Lima A, Moutinho P, Carter J, Bowman M, Cattaneo A, Rodrigues H, Schwartzman S, McGrath DG, Stickler CM, Lubowski R, Piris-Cabezas P, Rivero S, Alencar A, Almeida O, Stella O (2009) The end of deforestation in the Brazilian Amazon. Science 326:1350–1351
Oertel C, Matschullat J, Zurbaa K, Zimmermanna F, Erasmi S (2016). Greenhouse gas emissions from soils—a review. Chemie der Erde—Geochemistry, 76(3):327–352
Palm CA, Alegre JC, Arevalo L, Mutuo PK, Mosier AR, Coe R (2002) Nitrous oxide and methane fluxes in six different land use systems in the Peruvian Amazon. Global Biogeochem Cycles 16(4):1073
Pihlatiea MK, Christiansen JR, Aaltonen H, Korhonen JFJ, Nordbo A, Benanti G, Giebels M, Helmy M, Sheehy J, Jones S, Juszczak R, Klefoth R, Lobo-do-Vale R, Rosa AP, Schreiber P, Serça D, Vicca S, Wolf B, Pumpanen J (2013) Comparison of static chambers to measure CH4 emissions from soils. Agric for Meteorol 171–172:124–136
Rego RS, Silva BNR, Raimundo SO (1993). Detailed soil survey in an area in the municipality of Igarapé Açu, Pará. In: Junk WJ and Bianchi H (Eds.) Summaries of Lectures and Posters Presented at the 1st SHIFT-Workshop in Belém (Brazil), March 8–13. CNPq, IBAMA, BMFT, Geesthact, Germany.
Reich PB, Grigal DF, Aber JD, Gower ST (1997) Nitrogen mineralization and productivity in 50 hardwood and conifer stands on diverse soils. Ecology 78(2):335–347
Rosenstock TS, Tully KL, Arias-Navarro C, Neufeldt H, Butterbach-bahl K, Verchot LV (2014) Agroforestry with N2-fixing trees: sustainable development’s friend or foe? Current Opinion in Environmental Sustainability 6:15–21
Schlesinger WH (1997) Biogeochemistry: an analysis of global change. Academic press, San Diego, CA, USA
Segers R (1998) Methane production and methane consumption: a review of processes underlying wetland methane fluxes. Biogeochemistry 41(23–51):1998
Shrestha RK, Strahm BD, Sucre EB, Holub SM, Meehan N (2014) Fertilizer management, parent material, and stand age influence forest soil greenhouse gas fluxes. Soil Sci Soc Am J 78(6):1831–2113. https://doi.org/10.2136/sssaj2014.03.0118
Signor D, Cerri CEP, Conant R (2013). N2O emissions due to nitrogen fertilizer applications in two regions of sugarcane cultivation in Brazil. Environ Res Lett, 8:015013. https://doi.org/10.1088/1748-9326/8/1/015013
Smith, P et al. (2014) Agriculture, forestry and other land use (AFOLU). In: Edenhofer O et al (eds)Climate change 2014: mitigation of climate change. Contribution of working group III to the fifth assessment report of the intergovernmental panel on climate change, pp. 813–922. Cambridge Univ. Press
Taylor BN, Chazdon RL, Bachelot B, Menge DNL (2017) Nitrogen-fixing trees inhibit growth of regenerating Costa Rican rainforests. Proc Natl Acad Sci 114(33):8817–8822. https://doi.org/10.1073/pnas.1707094114
Thomas GW (1996) Soil pH and soil acidity. In: Sparks DL Methods of soil analysis: part 3 chemical methods, soil science society of America book series, Madison, WI, pp 475–490
Tobita H, Hasegawa SF, Tian X, Nanami S, Takeda H (2010) Spatial distribution and biomass of root nodules in a naturally regenerated stand of Alnus hirsuta (Turcz.) var. sibirica. Symbiosis 50:77–86
Tubiello FN, Salvatore M, Ferrara AF, House J, Federici S, Rossi S, Biancalani R, Golec RDC, Jacobs H, Flammini A, Prosperi P, Cardenas-Galindo P, Schmidhuber J, Sanchez MJS, Srivastava N, Smith P (2015) The contribution of agriculture, forestry and other land use activities to global warming, 1990–2012. Glob Change Biol 21:2655–2660
Vasconcelos SS, Zarin DJ, Capanu M, Littell R, Davidson EA, Ishida, FY Santos EB, Araújo MM, Aragão DV, Rangel-Vasconcelos LGT, Oliveira FA, McDowell WH, de Carvalho CJR (2004) Moisture and substrate availability constrain soil trace gas fluxes in an eastern Amazonian regrowth forest. Glob Biogeochem Cycles 18:GB2009, https://doi.org/10.1029/2003GB002210
Verchot LV, Davidson EA, Cattânio JH, Ackerman IA, Erickson HE, Keller M (1999) Land use change and biogeochemical controls of nitrogen oxide emissions from soils in eastern Amazonia. Glob Biogeochem Cycles 13(1):31–46
Verchot L, Davidson EA, Cattânio JH, Ackerman IA (2000) Land-use change and biogeochemical controls of methane fluxes in soils of Eastern Amazonia. Ecosystems 3:41–56
Verchot LV, Brienza S Jr, de Oliveira VC, Mutegi JK, Cattânio JH, Davidson EA (2008) Fluxes of CH4, CO2, NO, and N2O in an improved fallow agroforestry system in eastern Amazonia. Ecosyst Environ 126:113–121
Acknowledgements
We acknowledge the career and legacy of Dr. Lawrence (Larry) Morris (1952—2021). Dr. Morris served as co-advisor to the lead author (AJ) for his MS and served as a member on his PhD committee. AJ offers a special debt of gratitude to Dr. Morris for creating new opportunities for adventure, learning, and serving. The passing of Larry Morris in 2021 was a great loss to the soil science community. His enthusiasm and love for engaging young people in the scientific endeavor and for developing partnerships that spanned continents, brought out the best in other people. Larry’s work at UGA touched the lives of undergraduate and graduate students, faculty, and industry professionals. His friendships developed during graduate school stimulated a love for Brazil, which helped create exchange programs that took US students (such as AJ) to Brazil and brought Brazilian students to the United States. His collaborations led to the creation of a new generation of soil science and research professionals in both countries. His heart was enormous. Thank you Larry, and may the Light of Eärendil forever guide you.
Author information
Authors and Affiliations
Contributions
AJ—Wrote the manuscript text, analyzed the data, assisted in creating the research design, implemented the research study, and conducted the fieldwork. FAO—Assisted in creating the research design, coordinated research activities and facilities, and provided intellectual contributions to the research. OK—Assisted in creating the research design, directed overall research project under which this research was conducted, provided material support to the research, and provided intellecutal contributions to the research. SSV—Provided material support to the research, and provided intellectual contributions to the research. LM—Assisted in creating the research design, provided intellectual contibutions to the research, performed editing of the manuscript, served on the dissertation committee of AJ, and conducted fieldwork. DM—Assisted in creating the research design, provided intellectual contibutions to the research, performed editing of the manuscript, served as dissertation committee chair to AJ, provided material support to the research and analysis, and conducted fieldwork.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Joslin, A.H., de Assis Oliveira, F., Kato, O.R. et al. Ten years of improved-fallow slash-and-mulch agroforestry in Brazilian Amazonia: Do nitrogen-fixing trees affect nitrous oxide and methane efflux?. Agroforest Syst (2024). https://doi.org/10.1007/s10457-024-01053-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10457-024-01053-z