Abstract
Diabetic retinopathy (DR) is a diabetic complication that results in visual impairment and relevant retinal diseases. Current therapeutic strategies on DR primarily focus on antiangiogenic therapies, which particularly target vascular endothelial growth factor and its related signaling transduction. However, these therapies still have limitations due to the intricate pathogenesis of DR. Emerging studies have shown that premature senescence of endothelial cells (ECs) in a hyperglycemic environment is involved in the disease process of DR and plays multiple roles at different stages. Moreover, these surprising discoveries have driven the development of senotherapeutics and strategies targeting senescent endothelial cells (SECs), which present challenging but promising prospects in DR treatment. In this review, we focus on the inducers and mechanisms of EC senescence in the pathogenesis of DR and summarize the current research advances in the development of senotherapeutics and strategies that target SECs for DR treatment. Herein, we highlight the role played by key factors at different stages of EC senescence, which will be critical for facilitating the development of future innovative treatment strategies that target the different stages of senescence in DR.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diabetic retinopathy (DR) is the most common ocular complication of diabetes mellitus (DM) and a leading cause of blindness among working-age people [1]. Research on the pathogenesis of DR aims to reduce the global prevalence of this disease. Microvascular disease is the primary pathogenic hallmark of DR and can develop into different and continuous stages [2]. The process begins with mildly increased vascular permeability and proceeds to nonproliferative diabetic retinopathy (NPDR), which is distinguished by vascular leakage and occlusion. As focal ischemia and hypoxia expand and worsen, new capillary sprouts from pre-existing vessels, mainly driven by increased VEGF levels [3]. With the formation of compensatory retinal angiogenesis, DR progresses to the stage of proliferative diabetic retinopathy (PDR). However, these newly-formed vessels subsequent to angiogenesis are tortuous, leaky and mislocated, leading to the disturbance on retinal structure [3]. In the end stage of PDR, preretinal fibrosis often occurs and exacerbates the disturbance. Throughout the process described above, retinal vascular endothelial cells (ECs) are continuously exposed to hyperglycemia-mediated metabolic disorders, making them essential target cells for vascular diseases.
Current therapeutic strategies for DR focus on microvascular lesions, with targeting endothelial alterations being the most widely utilized therapeutic pathway. In the clinical setting, DR treatment is still dominated by laser therapy and antiangiogenic therapies (AATs) in PDR patients. However, these therapies still lack broader effectiveness and have raised safety concerns [4]: the absence of preventive treatment for patients in the early stage of DR; failure to address the underlying causes of retinal ischemia, which leads to recurrent neovascularization; and the inhibition of normal vascularization and neuron survival. Therefore, developing selective therapeutic targets to inhibit pathological neovascularization while promoting normal vessel regeneration is urgent.
In recent years, many studies have revealed the existence and multiple roles of senescent ECs (SECs) in the progression of DR, especially in the formation and regression of pathological neovascularization. Mechanistically, various types of stress damage induced or aggravated by DM drive the accumulation of SECs, which are sustained and spread via the senescence-associated secretory phenotype (SASP) specific to senescent cells (SCs). Simultaneously, therapies targeting SECs are booming and show considerable promise in DR treatment and prevention. This finding points to an additional sustainable path based on existing laser therapy and AATs because therapies targeting SECs not only target the regression of pathological neovascularization but also promote the regeneration of functional vessels [5, 6]. On this basis, these therapies tend to remain safer. However, the development and application of therapies targeting SECs still face many challenges. In conclusion, this review focuses on the mechanisms of EC senescence in the pathogenesis of DR and summarizes the current research advances in strategies targeting SCs and SECs.
Phenotypic and functional changes in endothelial cells in diabetic retinopathy
Damage to vessels and endothelial cells is one of the most prominent pathological changes in diabetic retinopathy
Although DR is now more accurately defined as a neurovascular disease [7], retinal microangiopathy is still one of the major pathological changes in DR. Under normal conditions, EC monolayers line the vascular network and serve as a selective barrier and blood flow regulator, supplying the neuroretina with enough oxygen and nutrients. However, when blood glucose levels rise, ECs are critical target cells for vascular injury. The breakdown of cell‒cell junctions between adjacent ECs, as well as EC apoptosis, are the main forces for acellular capillary formation and internal blood‒retinal barrier (iBRB) destruction during NPDR. Studies have indicated a significant reduction in the expression of tight junction proteins in the retinas of diabetic rats [8]. In the high glucose environment, significantly increased inflammatory factors and adhesion molecules, including tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), intercellular adhesion molecule-1 (ICAM-1), matrix metalloproteinase-9 (MMP-9), also reduce and destroy cell-cell junctions [8] while promoting EC apoptosis. At the early stage of DR, vascular endothelial growth factor (VEGF) can act as a pro-inflammatory factor modulating inflammatory responses [2, 9]. Given that VEGF as a growth factor directly promotes EC survival and proliferation [10], it is important to emphasize that subsequent excessive inflammation and reactive oxygen species (ROS), rather than VEGF itself, directly leads to EC apoptosis. In this process, ROS induced by high glucose levels can promote the expression of proinflammatory genes while also aggravating EC shedding and apoptosis [11]. In general, EC apoptosis is mainly induced by hyperglycemia [12, 13], inflammation [14] and ROS [15].
During PDR, retinal vascular ECs undergo continuous and excessive proliferation and migration, thus promoting pathological neovascularization [4]. With increased retinal ischemia, VEGF levels increase and promote EC survival, proliferation, permeability, and chemotaxis to VEGF sources, leading to excessive proliferation of ECs and the formation of new vessels. Furthermore, the chronic inflammatory environment induced by high glucose levels also provides a driving force for the direct engagement of ECs in angiogenesis and the indirect involvement of secreted angiogenic mediators in angiogenesis [4].
Endothelial cells participate in the impairment of neurovascular unit function in diabetic retinopathy through intercellular communication
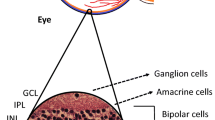
DR is a neurovascular disorder caused by the destruction of the retinal neurovascular unit (NVU) [7, 16]. The structure of the NVU is controlled by the communication among neurons, glial cells and vascular cells. ECs are highly sensitive to signals from surrounding cells and their environment, allowing them to regulate retinal blood flow and vascular permeability through various interactions. In a hyperglycemic environment, EC injury progressively aggravates structural defects and dysfunction in the NVU.
Pericyte-EC interactions are essential to maintain the structural and functional integrity of iBRB. Under normal conditions, pericytes can maintain EC quiescence and survival by secreting angiopoietin-1 (ANG1) [17, 18]; in turn, ECs secrete platelet-derived growth factor-BB (PDGF-BB) to activate PDGFRβ on pericytes, thereby promoting pericyte proliferation, migration, and survival [19]. During the NPDR period, pericyte loss is perceived as an early feature and basis of iBRB disruption [20]. Further, pericytes undergo apoptosis and detachment before ECs and with greater loss [21]. This impairment causes EC damage by disrupting pericyte-EC interactions. Subsequently, increased EC apoptosis and reduced transduction of PDGFRβ signaling in the hyperglycemic environment, in turn exacerbates pericyte apoptosis [22]. Eventually, the progressive loss of ECs and pericytes leads to acellular capillary formation, basement membrane (BM) thickening [23], and progressive disintegration of the iBRB. In addition, the altered protein composition in thickened BM reduces the filtering capacity of the barrier [24] and hinders communication between ECs and other cells within the NVU [23], further deteriorating the physiological structure and function of the NVU. When the iBRB breaks down, immune privilege is also disrupted; thus, circulating immune cells and inflammatory factors infiltrate the NVU, and chronic inflammation persists and contributes to gliosis and neuronal damage [25].
In the end stage of PDR, preretinal fibrosis often occurs for repeated neovascular bleeding and wound healing [26]. Myofibroblasts are the dominant cells of fibrosis, which can arise from activation of fibroblasts, and intercellular transdifferentiation of ECs and pericytes [27, 28]. Specifically, Abu El-Asrar et al. have demonstrated ECs proliferate and undergo endothelial-to-mesenchymal transition (EndoMT) in PDR [27]. Similarly, regulated by TGF-β, PDGF, VEGF, etc., pericytes are activated, dislocated, and transformed into myofibroblasts, which is called pericyte-myofibroblast transition (PMT) [28,29,30]. Ultimately, multiple intercellular communications and transformations (including EndoMT, PMT, and the activation of fibroblasts) mediate the formation of extraretinal fibrovascular membranes, leading to retinal neuronal damage and degeneration.
Along these lines, a cascade of responses promoted by ECs continue to exacerbate structural disruption and dysfunction of the NVU, including iBRB disruption, gliosis, neuronal degeneration, impaired autoregulation of blood flow and control of vascular integrity. These pathological changes ultimately result in impaired visual function.
Endothelial cell senescence
Cellular senescence
Cellular senescence is considered a cellular state in response to specific physiological processes or various stressful insults [31]. During tissue development and remodeling, cellular senescence often occurs in a physiologically programmed form. However, pathological cellular senescence is triggered by various senescence stressors, including telomere attrition, DNA damage, mitochondrial dysfunction, oxidative stress, and oncogenic activation [31]. Owing to the persistence of these stressors, cellular senescence can occur at any stage of an organism.
Notably, cellular senescence and aging are not synonymous but are closely related. Aging refers to a decline in an organism’s function during adulthood, often accompanied by a significant accumulation of cellular senescence. This is because, with age, there is a general accumulation of stressful insults to cells in the organism, making them more susceptible to entering the senescent state. Moreover, the immune clearance of SCs weakens with age [32]. This imbalance between the production and removal of SCs is an inevitable trend in the natural aging of organisms. On this basis, certain diseases, such as DM, tend to accelerate the generation of this imbalance, resulting in premature cellular senescence [33]. Focal accumulation of SCs can cause specific tissue dysfunction, whereas systemic accumulation of SCs is involved in the overall decline in body function, which is indeed associated with aging.
Phenotypes of cellular senescence
Cellular senescence can be subdivided into many subtypes depending on the cell type and stressor, but these subtypes share several core features. The International Cellular Senescence Association (ICSA) agreed that cellular senescence is characterized by four key features: prolonged and generally irreversible cell cycle arrest, SASP, macromolecular damage (especially DNA damage), and altered metabolism [34]. These characteristics do not exist independently, but are interdependent. The first two characteristics are described here.
Cell cycle arrest
Cell cycle arrest is the most prominent feature of cellular senescence, and this cell cycle withdrawal distinguishes it from cell quiescence and terminal differentiation [35]. Cell cycle arrest can be activated by persistent DNA damage triggered by multiple stressors and ultimately converges on the upregulation of the p53/p21WAF1/CIP1 and/or p16INK4A/RB tumor suppressor pathways [31]. However, the severity, duration and type of damage determine whether the above mechanisms lead to cellular senescence. For example, minor damage to DNA triggers the DNA damage response (DDR), and the cell cycle restarts when the DDR is complete, whereas more extreme DNA damage often leads directly to p53-induced apoptosis [36,37,38].
Senescence-associated secretory phenotype (SASP)
SCs actively secrete various substances collectively termed SASP, including chemokines, cytokines, growth factors, and insoluble factors. Coppé et al. reported that when cells overexpress only cyclin-dependent kinase inhibitors, in the absence of DNA damage, they acquire many senescence phenotypes, including cell cycle arrest, but fail to secrete SASP components [39]. These findings suggest that the SASP is usually activated when cells sense macromolecular damage, especially DNA damage. Cytosolic DNA is usually derived from retrotransposable elements, damaged mitochondrial DNA, and cytosolic chromatin fragments. Cytoplasmic DNA can trigger inflammatory pathways expressing SASP via the cyclic GMP‒AMP synthase (cGAS)-stimulator of interferon genes (STING) pathway [40]. Coopted components of the innate immune machinery in SCs are also sensors in this process, particularly retinoic acid-inducible gene I [41] and inflammasomes [42]. In addition, the SASP is regulated by differential activation and interaction of transcription factors (including nuclear transcription factor-κB [NF-κB], CCAAT/enhancer-binding protein β [C/EBPβ], and GATA4 [43, 44]), mammalian target of rapamycin (mTOR) and p38 mitogen-activated protein kinase (MAPK) signaling pathways [43, 45].
Depending on the stressor, cell type, and duration of senescence, the expression of the above mechanisms varies differentially; thus, the SASP is also heterogeneous [46]. This heterogeneity of substances also determines the variability of SASP function, with beneficial aspects, including the ability to mediate developmental senescence [47, 48], wound healing [49], and tissue remodeling [50], and deleterious aspects, including the ability to promote tissue destruction and persistent chronic inflammation [51]. When the SASP enhances and spreads senescence in both autocrine and paracrine ways, these effects can be propagated through wider peripheral tissues, which is termed bystander senescence [52, 53].
SCs must undergo metabolic reprogramming to maintain a growth arrest state for cell survival and to secrete highly complex, dynamic, and heterogeneous SASPs. This process is frequently accompanied by a series of metabolic shifts, such as increased glycolysis, decreased fatty acid oxidation, and increased ROS production [54]. This mainly stems from the mitochondrial dysfunction and lysosome-associated autophagy imbalance that accompany cellular senescence. The former is usually characterized by alterations in mitochondrial mass, dynamics, composition and the electron transport chain. For example, the activity of complex IV and cytochrome c oxidase, components of the mitochondrial electron transport chain, decreases with age, leading to decreased mitochondrial oxidative phosphorylation and increased ROS [55].
Phenotypes and biomarkers of senescent endothelial cells
Phenotypes of senescent endothelial cells
ECs are arranged in monolayers on the vascular network and participate in vascular maintenance and barriers while being directly exposed to continuous blood flow. Morphologically, SECs become enlarged and flat [56], reducing the local cell density and sites for establishing cell‒cell junctions and decreasing their potential for dynamic remodeling. Second, SECs also exhibit stronger adhesion to the substrate and greater contractility, which is accompanied by extracellular matrix remodeling. Together, these changes endow SECs with a phenotype of enforced stasis and resistance to remodeling, attenuating endothelial repair and adaptation to local blood flow [57,58,59].
SECs often express proinflammatory SASP factors, including IL-6, IL-8, IL-1β, ICAM-1, VEGF, and MMPs [60]. These proinflammatory factors contribute to the disruption of cellular junctions, increased endothelial permeability, and inflammatory cell infiltration accompanying EC senescence. In particular, a unique feature of the endothelial SASP is its reduced nitric oxide (NO) and increased ROS levels, which promote vascular dysfunction [61]. In addition to its protective effects on the vasculature, NO also has anti-cellular senescence effects, such as reducing ROS production through direct inhibition or scavenging of NADPH [62]. Therefore, reduced endothelial-derived NO secretion indirectly enhances, promotes cellular senescence, and contributes to vascular dysfunction, such as impaired NO-dependent dilation. Similar to other types of SCs, increased ROS production by SECs is attributed mainly to mitochondrial and lysosomal dysfunction; however, the resulting alterations in glucose metabolism differ. Quiescent ECs primarily rely on increased fatty acid oxidation and decreased glycolysis, while proliferating ECs depend heavily on glycolysis for proliferation and angiogenesis [63, 64]. Early studies have shown that in oxygen-induced retinopathy (OIR) mice, the accumulation of SCs (including SECs) is strongly consistent with pathological angiogenesis in terms of temporal and spatial localization [6]. Therefore, the effects of mitochondrial dysfunction on the cellular state of proliferating ECs may be more rooted in ROS production rather than energy supply.
Biomarkers of senescent endothelial cells
Based on the molecular level changes behind these phenotypes, in vivo SECs can be detected with various relevant biomarkers. Owing to the lack of specificity of endothelial senescence biomarkers, the biomarkers commonly used to detect SECs are widely established features of different types of cellular senescence [65].
The most widely used assay is the histochemical detection of elevated senescence-associated β-galactosidase (SA-β-gal) activity at pH 6.0, which is based on increased lysosomal content in SCs [66]. However, an increase in SA-β-gal activity can also occur in other biological contexts [67] and is therefore insufficient to serve as evidence for identifying cellular senescence alone. Classical senescence-related biomarkers also involve molecules related to DNA damage and cell cycle arrest, including γH2AX, p16, ARF, p53, p21, p15, p27 and hypophosphorylated RB [31].
In addition, negative biomarkers that should be absent in SCs, such as Ki67 or BrdU incorporation, can be used to exclude SCs [68]. Taken together, a combination of two or more biomarkers, such as SA-β-gal and γH2AX staining, should be used to identify SCs accurately [68, 69]. In addition to traditional immunohistochemical staining of frozen or fixed isolated tissues, the development of liquid biopsies has given rise to the promise of circulating ECs, such as endothelial progenitor cells (EPCs) and vascular endothelial progenitor cells, as potential biomarkers of EC senescence [65]. Recently, a powerful tool called TEMPO (tracing expression of multiple protein origins) has been successfully developed and has yielded surprising new insights into aqueous humor liquid biopsies [70]. TEMPO simultaneously integrates microvolume liquid-biopsy proteomics, single-cell transcriptomics, and artificial intelligence, which provides new opportunities for the molecular assessment of cell type-specific aging in vivo.
Inducers of endothelial cell senescence
The unique physiological context of ECs explains the multiple triggers for them to undergo cellular senescence. The location of the vascular tree where ECs are located determines their hemodynamic environment. ECs at the sites of branches and curvatures are exposed to disturbed flow, which results in greater cellular turnover and faster replicative senescence [71, 72]. In contrast, circulating factors in the blood that induce senescence have systemic effects, including effects on oxygen, glucose, lipids, amino acid metabolites, immune cells, inflammatory cytokines, and toxic substances [73]. As an organism ages, various physiological stresses accumulate within ECs, which eventually results in EC senescence. Several studies have demonstrated that hyperglycemia promotes the accumulation of stress-induced injury both in vivo and in vitro, culminating in an earlier onset of EC senescence. This will be further discussed below.
Endothelial cell senescence occurs in diabetic retinopathy
Evidence of senescent endothelial cells in diabetic retinopathy
Many studies have shown a strong causal relationship between DR development and EC senescence, particularly through a series of factors dominated by hyperglycemia. Compared with those in control patients with nonvascular ocular pathologies, the expression of VEGF-A, plasminogen activator inhibitor-1 (PAI-1), IL-6, and IL-8, which are involved in paracrine senescence, is significantly elevated in the vitreous fluid of PDR patients (p < 0.01 and p < 0.001) [5]. Crespo-Garcia et al. further confirmed the presence of senescent vascular cells in retinas from postmortem globes of PDR patients at a later stage [6]. Related experimental studies provide further evidence for EC senescence in DR. In vitro, ECs cultured with high glucose exhibit increased SA-β-gal activity [74]. In vivo, premature senescence of retinal ECs has also been demonstrated in streptozotocin (STZ)-induced hyperglycemic mice, as evidenced by increased mRNA levels of senescence markers, including p16INK4A, p21, and p53 [6].
Key factors that induce endothelial cell senescence in diabetic retinopathy
During the development of DR, hyperglycemia, a chronic metabolic disorder, continues to diffuse its harmful effects through the vascular system to retinal tissues, ultimately resulting in neurovascular lesions. During this process, ECs directly exposed to a hyperglycemic environment undergo metabolic reprogramming dominated by altered glucose metabolism (Fig. 1). This metabolic alteration further triggers mechanisms such as advanced glycation end products (AGEs), the hexosamine pathway, the polyol pathway, and the protein kinase C (PKC) pathway [75]. These mechanisms ultimately converge mainly on oxidative stress and chronic inflammation, which directly shape the senescent phenotype of ECs. Oxidative stress and chronic inflammation often have similar origins and can stimulate each other, together promoting EC senescence. For example, the accumulation of AGEs increases the production of ROS and proinflammatory cytokines such as TNF-α [76]. In turn, the prooxidative environment contributes to the activation of the NF-κB signaling pathway, which promotes the expression and recruitment of proinflammatory cytokines [77]. In addition, the hyperglycemia-induced reduction in NO indirectly increases the vulnerability of ECs to stressful insults. The crosstalk of these alterations, in turn, exacerbates the four classical metabolic abnormalities mentioned above [78]. Notably, these alterations also appear in the endothelial SASP, which is proinflammatory and has increased ROS and decreased NO. This finding confirms the relevance and reasonability of the occurrence, maintenance, and spread of EC senescence under hyperglycemic conditions.
Oxidative stress
Increased oxidative stress under hyperglycemic conditions is the most important factor promoting EC senescence. Excess intracellular ROS are derived mainly from oxidative phosphorylation uncoupling and increased NADPH oxidase (Nox) activity [79, 80]. In addition, aberrant activation of multiple metabolic pathways is associated with hyperglycemia-induced increases in oxidative stress, including increased glucose influx through the polyol and hexosamine pathways, activation of the PKC pathway and increased formation of AGEs [81].
Mechanistically, excess ROS likely drive EC senescence through DNA damage and DDR pathway activation. Oxidative DNA damage often occurs at telomeres [82, 83], which can lead to telomere uncapping [84, 85], telomere shortening and length-independent telomere damage [86]. In addition to damage to telomeres themselves, high glucose also induces EC senescence by decreasing telomerase activity within ECs [87, 88] and is associated with increased ROS. Combining the above phenomena and related studies, some studies indicate that telomeres may serve as highly sensitive sensors of stress, with the type of damage depending on the degree of oxidative stress [86]. In addition to nuclear DNA, mitochondrial DNA (mtDNA) is more vulnerable to extensive and persistent oxidative damage (particularly displacement loops) because of the lack of protective histones [89]. In a hyperglycemic environment, increased Nox activity increases the expression of MMPs [90], and then, ROS stimulation facilitates the translocation of MMPs into the mitochondria, which also impairs mtDNA [91]. Damaged mtDNA leads to dysfunctional transcription and protein synthesis, further disrupting electron transport and exacerbating ROS production. Ultimately, mitochondrial dysfunction acts as both a driver and a consequence of EC senescence [92, 93].
In addition to increased oxidative stress production, hyperglycemia also causes a decrease in antioxidant levels within ECs. The levels of antioxidants, including reduced glutathione, superoxide dismutase, and catalase, are lower in the retinas of diabetic rats than in those of nondiabetic rats [94]. Similarly, sirtuins (SIRTs), which act as NAD+-dependent deacetylases and ADP-ribosyl transferases [95], are also important targets for oxidative stress-induced EC senescence under hyperglycemic conditions. SIRT1 and SIRT6 can promote cellular repair and stress resistance in response to elevated cellular NAD+, while SIRT3 localized in mitochondria can protect cells from oxidative stress [95]. However, hyperglycemia suppresses SIRT1 expression, which decreases the expression of mitochondrial antioxidant enzymes (mainly manganese superoxide dismutase) via a histone acetylase- and forkhead box O1 (FOXO1)-mediated pathway, leading to mitochondrial ROS production [33]. In turn, the endothelial expression of SIRT1 and SIRT6 is reduced in response to oxidative stress, which strengthens the p53/p21 pathway [96,97,98]. In conclusion, depletion of mitochondrial SIRTs mediates glucose-induced oxidative stress and EC senescence [99].
Chronic inflammation
Chronic inflammation is a pathophysiological hallmark of DM and a recognized activator of cellular senescence. The cGAS-STING pathway is one of the most important regulators of EC senescence and chronic inflammation. On the one hand, the accumulation of extracellular and cytoplasmic DNA in ECs activates the cGAS-STING pathway, which leads to EC senescence by activating kinase tank-binding kinase 1 and type I IFN secretion [100, 101]; on the other hand, the activated cGAS-STING pathway activates the NF-κB signaling pathway, which stimulates the expression of TNF, IL-1β, IL-6, and IL-8, among others [102], thereby initiating and maintaining EC senescence [103]. Like oxidative stress, chronic inflammation can also drive EC senescence through DNA damage and DDR pathway activation. For example, TNFα-induced EC senescence initiates a positive feedback loop through sustained activation of the Janus kinase/signal transducer and activator of transcription (JAK-STAT) pathway, leading to prolonged DNA damage and cytokine secretion that perpetuates the senescent state [104].
NO reduction
NO is vasculoprotective and antagonistic to EC senescence [62]; thus, the key effectors of hyperglycemia-induced EC senescence are likely related to NO levels. Normally, endothelial nitric oxide synthase (eNOS) utilizes the precursor substance L-arginine to produce NO, which is also a substrate for arginase. In diabetic retinas and high glucose-treated retinal ECs, arginase activity is increased, and less L-arginine is available to eNOS, resulting in decreased NO production [105]. Moreover, several studies have demonstrated that hyperglycemia can lower NO levels by inhibiting eNOS generation, activity and coupling [62, 106,107,108]. Specifically, eNOS coupling is attributed mainly to the depletion of L-arginine and tetrahydrobiopterin (BH4) induced by hyperglycemia [106]; thus, uncoupled eNOS and Nox can produce superoxide using molecular oxygen as a substrate. Next, superoxide binds NO with high affinity to form peroxynitrite (ONOO−), and then ONOO− can oxidize BH4 and disrupt the zinc-thiolate clusters of eNOS, further culminating in eNOS uncoupling [109]. Furthermore, various ROS (such as ONOO− and H2O2) can induce EC senescence by increasing endothelial arginase expression and activity and reducing NO bioavailability [110, 111]. Studies have demonstrated that a DM-induced increase in arginase 1 expression promotes EC senescence through activation of the p16INK4A and p53 signaling pathways [74].
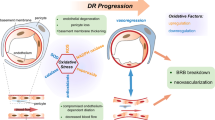
Key factors inducing endothelial cell senescence in diabetic retinopathy. The figure above illustrates a series of mechanisms through which hyperglycemia induces endothelial cell senescence. Among these, increased inflammation, increased ROS and decreased NO are the focus of this section’s discussion, and these changes are also simultaneously reflected in the endothelial SASP. This confirms the relevance and reasonability of the occurrence, maintenance, and spread of EC senescence under hyperglycemic conditions. DDR, DNA damage response; SASP, senescence-associated secretory phenotype; NO, nitric oxide; ROS, reactive oxygen species; AGEs, advanced glycation end products. This figure was created using Figdraw
The role and significance of endothelial cell senescence in diabetic retinopathy
Endothelial cell senescence exacerbates diabetic retinopathy
Endothelial cell senescence exacerbates nonproliferative diabetic retinopathy
The prominent lesions during the NPDR period are acellular capillary formation and iBRB disruption, which eventually progress to microvascular rarefaction (Fig. 2). First, microvascular rarefaction arises directly from vessel regression, a process mediated by massive cell shedding and apoptosis. The continuous proliferation and enhancement of the endothelial SASP significantly affects vascular cell survival, which is strongly attributed to increased ROS, decreased NO, and increased proinflammatory cytokines (e.g., IL-1β, IL-6, TNF-α, chemokines, and adhesion molecules) [62, 112,113,114,115]. This tissue-destructive SASP is a significant modulator of the harmful effects of SECs: it maintains and expands the chronic inflammatory environment by increasing circulating cytokine levels and immune cell infiltration, disrupting cell‒cell junctions and promoting vascular cell apoptosis. Furthermore, these factors can interact to enhance pro-apoptotic effects. For example, IL-1β stimulates ROS production and accelerates EC apoptosis in retinal capillaries by activating the NF-κB signaling pathway, and this effect is intensified under high glucose conditions. In turn, elevated ROS and reduced NO induce inflammatory pathways, particularly the NF-κB signaling pathway, culminating in chronic, sterile and low-grade vascular inflammation and exacerbating cell shedding and apoptosis [116, 117].
In addition to vessel regression, angiogenesis impairment may play a central role in microvascular rarefaction. At the molecular level, ECs initiate and maintain angiogenic processes mainly through VEGF signaling pathways. These findings suggest the potential of EC senescence to promote angiogenic impairment. First, SECs are unable to proliferate due to cell cycle arrest, inhibiting their angiogenic capacity. EPCs, as precursors of ECs, can complement damaged ECs by differentiating into mature ECs, thus maintaining the integrity of the endothelial monolayer. However, senescence can impair the compensatory capacity of EPCs and their potential for vascular regeneration [118].
Another reason for the lack of angiogenic capacity is the resistance of SECs to angiogenic inducers. This is indicated by the impaired angiogenic response of primary cerebromicrovascular ECs to exogenous VEGF in aged rats [119]. In addition, force microscopy suggests that SECs have diminished migratory capacity, as evidenced by the tendency of SECs to be static and firmly adherent to the substrate [56]. In terms of exogenous factors, an obvious contribution is that SECs reduce the production of the angiogenesis inducer VEGF [120], thus impairing the direct promotion of angiogenesis. Since NO is located downstream of VEGF-induced angiogenesis [121], impaired bioavailability of endothelial-derived NO also disrupts the dynamic balance between angiogenesis and vessel regression, thereby promoting microvascular rarefaction [122].
As NPDR lesions progress, retinal microvascular rarefaction and occlusion worsen, accompanied by a decrease in vascular perfusion density [123, 124]. Microaneurysms, which refer to vessel outpouching and leakage, are characteristic tissue lesions during the NPDR period [3]. Early studies suggest that endothelial senescence is involved in the development of microaneurysms [125]. In regions of capillary nonperfusion, pericyte loss and capillary endothelial damage contribute greatly to microaneurysm formation, with the former widely recognized as a central factor [126, 127]. During this process, dysfunction of SECs and spreading of SASP impair pericyte survival and recruitment, resulting in reduced pericyte coverage. Given that pericytes mediate leukocyte extravasation, pericyte loss may contribute to intralumenal accumulation of leukocytes [127]. This, together with pro-inflammatory SASP, exacerbates endothelial damage. At the BM level, the loss of vascular cells reduces BM synthesis, while increased MMP-9 levels in pro-inflammatory SASP disrupt the BM during the later stages of microaneurysm formation [60, 128]. These changes render the vessel wall weak and fragile, making it predispose to dilation and leakage under high capillary hydrostatic pressure [126].
Endothelial cell senescence exacerbates proliferative diabetic retinopathy
During PDR, uncontrolled angiogenesis occurs in areas of sparse retinal microvasculature to compensate for tissue hypoxia and nutrient deprivation. However, these neovessels tend to be fragile, leaky, and misdirected. Accumulating evidence suggests a strong link between pathological angiogenesis and EC senescence (Fig. 2). Crespo-Garcia et al. reported that SCs accumulate in OIR mice (which can be considered an alternative model of PDR) and reach a maximal burden at the peak of pathological neovascularization (P17); at this time, gene set variation analysis (GSVA) of single-cell transcriptomic data revealed that ECs (~ 65–60%) are enriched in senescence-related transcripts [6]. Furthermore, Binet et al. observed robust activation of the RAS pathway (RAS triggers senescence via DDR and p53 activation) in pathological preretinal neovascularization sprouts at P17 and ultimately determined that the SECs overlapped with the neovascular tufts [129]. However, cellular senescence is only involved in pathological angiogenesis rather than healthy vessels [6]. More specifically, as pathological preretinal neovascularization occurs, cells of the vascular unit rapidly engage pathways that activate p16INK4A and upregulate the prosurvival protein BCL-xL, ultimately leading to cellular senescence [6].
This process can be partly explained by a self-protective mechanism triggered by the impaired metabolism of RGCs in the ischemic and hypoxic environment that protects them from hypoxia-induced cell death by entering a senescent state [5]. This process triggers a series of senescence-related effectors that induce cellular senescence, including EC senescence. However, these SCs maintain and spread senescence through autocrine and paracrine secretion of proinflammatory SASPs and thus inhibit long-term vascular repair while promoting pathological neovascularization [5].
This conflicting protection may stem from the dual nature of senescence and the uncontrollable nature of SASP diffusion. In addition, the apoptosis and clearance of SECs reduce pathological neovascularization and induce reparative vascular remodeling [129]. Along these lines, we speculate that SECs may stimulate pathological angiogenesis via the proinflammatory and progrowth SASP and potentially shape neovascularization into a fragile and leaky pathological phenotype as a feature component of pathological neovascularization.
Endothelial cell senescence induces vascular remodeling
Endothelial cell senescence induces physiological vascular remodeling
Although SECs are often viewed as harmful factors that induce or accelerate the course of vascular disease, some studies have revealed their positive role in physiological and pathological vascular remodeling in organisms. During embryonic development, developmentally programmed senescence is widespread and is thought to promote tissue development and patterning through immune clearance and SASP-mediated tissue remodeling [47, 48]. Accordingly, physiological vascular remodeling, as one of the segments of tissue remodeling, may involve the participation of SECs. Notably, p16, p53, and DNA damage are not detected in these structures during developmental senescence [31, 47, 48]. This is because developmental senescence is not mediated by DNA damage but rather by a series of developmental cues that activate signaling pathways, such as phosphatidylinositol-3-kinase (PI3K) and transforming growth factor-β (TGF-β), which ultimately converge on p21 [31]. Moreover, the SASPs of these SCs lack senescence-enhancing mediators such as IL-6 and IL-8 [48, 130, 131], which may help maintain the stability and controllability of tissue development.
Endothelial cell senescence induces pathological vascular remodeling
It has been suggested that cellular senescence may have initially emerged as a developmental mechanism, which was later adapted during evolution to play an adult role. This phenomenon confers rootedness to the beneficial effects of cellular senescence in different stages, including tissue remodeling during development and cancer suppression, tissue regeneration, and damage repair in adulthood. In contrast to physiological tissue remodeling during development, injury repair in adulthood can be viewed, in some ways, as pathological tissue remodeling, which refers to a reparative response in favor of the organism after lesions. SCs appear transiently at the site of tissue injury, promoting wound healing and the elimination of potentially dangerous cells via specific SASP factors with subsequent immune clearance.
Indeed, such pathological tissue remodeling has previously been reported in PDR [132, 133] and other retinopathies, such as age-related macular degeneration (AMD) and retinopathy of prematurity (ROP), manifesting as spontaneous regression of late pathological neovascularization. The involvement of SECs in pathological angiogenesis during PDR has been mentioned previously; however, Binet et al. reported that SECs, which recruit immune cells through the SASP and lead to subsequent self-clearance, can induce pathological neovascularization and further promote vascular repair remodeling [129] (Fig. 2). This process can be viewed as a “self-correcting” program for the retinal vasculature.
The two seemingly contradictory functional outcomes of SECs may stem from the shift in SASP components in a highly context-dependent manner, which is critical for proper resolution of tissue damage. ECs in a persistent senescent state in pathological vessels appear to be highly sensitive to extracellular signals, such as damage-associated molecular patterns, which can exacerbate the proapoptotic and proinflammatory properties of SASPs [134]. Corroborating this, at the tipping point from maximal pathological angiogenesis to normalization of the vasculature at P17 of OIR, transcript enrichment occurs primarily in processes related to inflammation [129]. Since SECs in pathological neovascular sprouts strongly activate RAS signaling, the timely shift in the secretory profile to the inflammatory-type SASP may be related to attenuated NOTCH1 activity in RAS-induced senescence [43, 129]. In the late stage of RAS-induced senescence, the downregulation of the NOTCH1 intracellular domain permits derepression of the proinflammatory transcription factor C/EBPβ, and ultimately, SCs express proinflammatory factors such as IL-6 and IL-8 [43] in concert with NF-κB signaling. Accordingly, in the late stages of pathological neovascularization, RASV12-activated SECs undergo immune clearance through proinflammatory SASP recruitment.
In particular, neutrophils are recruited mainly by IL-8 [135] and stimulated to release neutrophil extracellular traps (NETs) via IL-1β and C-X-C motif chemokine ligand 1 (CXCL1); subsequently, NETs promote apoptosis (confined to pathological tufts) and the removal of SECs, ultimately leading to the regression of pathological angiogenesis and the regeneration of functional vessels [129]. In addition, studies in OIR models and DR have confirmed an increase in the number of mononuclear phagocytes (MPs) in retinal tissues [136, 137], which may be related to the activation and infiltration of MPs induced by the inflammatory milieu and cellular stress in the ischemic retina [138]. Multiple studies have shown that MPs may mitigate neovascularization and eliminate existing pathological neovascularization by killing (FasL/Fas system) and phagocytosing stressed ECs in pathological neovascular clusters [138,139,140,141].
In terms of the end result, the physiological purpose of this vascular remodeling is positive and self-repairing and is an evolutionarily adaptive mechanism. This raises the issue of whether existing cell senescence can be exploited rationally to treat PDR. However, uncontrolled neutrophils, abundant NET components, and corresponding antibodies derived from organisms may disturb retinal homeostasis, resulting in other lesions, such as microvascular occlusions and small vessel vasculitis [142]. Therefore, the practicality and feasibility of this therapy are still under discussion.
Dual effects of endothelial cell senescence in diabetic retinopathy. Endothelial cell senescence plays a dual role in different stages of diabetic retinopathy. The harmful aspects are characterized by the induction of microvascular rarefaction during NPDR and pathological angiogenesis during PDR; the beneficial aspects are manifested by vascular remodeling in the later stage of pathological angiogenesis, which is a self-correcting process, including the regression of pathological angiogenesis and regeneration of functional vessels. This effect switch is closely related to the shift in components of the endothelial SASP in a highly context-dependent manner. SECs, senescent endothelial cells; SASP, senescence-associated secretory phenotype; iBRB, internal blood–retinal barrier; BM, basement membrane; NETs, neutrophil extracellular traps; IL-8, interleukin-8; IL-1β, interleukin-1β; CXCL1, C-X-C motif chemokine ligand 1. This figure was created using Figdraw
Advances in strategies for targeting senescent endothelial cells
Strategies for targeting senescent cells
Potential clinical implications of senescence have galvanized research into therapeutic approaches targeting SCs. Numerous studies have shown that the application of senotherapeutics might extend a healthy lifespan and ameliorate age-related diseases. Generally, senotherapeutics are classified into two types: (1) senolytics, which selectively eliminate SCs, and (2) senomorphics, which attenuate the destructive SASP instead.
Senolytics
As mentioned above, SCs upregulate senescent cell antiapoptotic pathways (SCAPs) that defend SCs against their own proapoptotic SASP and increase the number of intracellular drivers of apoptosis. Along these lines, the first-generation senolytics ablate SCs by targeting different SCAP network nodes, including HSP90 [143], tyrosine kinase receptors [144], ephrin receptor B1 [145], BCL-2 family members [146, 147], p53 and p38 MAPK. Correspondingly, strategies that have entered clinical trials include fisetin, dasatinib (approved by the FDA in 2006 [144, 148]), quercetin, and the combination of dasatinib and quercetin [147, 149]. Owing to the heterogeneity of SCAPs across different types of SCs, the effects of these drugs on removing SCs are also biased. In addition, preclinical drug candidates that function mainly by inhibiting BCL-2 family members include A1331852, A1155463, and navitoclax. These three drugs selectively induce apoptosis in senescent human umbilical vein endothelial cells (HUVECs) [146, 147]. However, complete inhibition of individual SCAP nodes is compensated by high doses of drugs, where the associated off-target effects lead to a greater risk of toxicity. Therefore, in the future, low-dose combinations and drugs with mild effects on multiple targets should be developed.
Second-generation senolytics arose with the application of methods such as random high-throughput drug library screens. Owing to the increase in lysosomal mass and SA-β-gal activity observed in SCs, one approach involves the development of galacto-oligosaccharide-coated nanoparticles. The nanoparticles carry toxic cargos that can be internalized by SCs and subsequently activated by SA-β-gal [150]. Similarly, β-galactosidase-activated prodrugs are preferentially processed by SCs, resulting in their selective killing [151, 152].
The emergence of senescence immunotherapy contradicts the traditional idea of relying on external drugs to eliminate SCs directly. This method is based on enhancing immune-mediated senescence clearance and advocates reasonable and moderate coordinated intervention in SCs. One idea is to augment senescence surveillance, such as by targeting endothelial pathways with ICOS-activating antibodies [153]. Alternatively, we can synergistically combine former senolytics with senescence clearance, effectively reducing the quantity of SCs below the burden threshold. In recent years, targeting surface proteins heterogeneously expressed by SCs has emerged as a promising immunotherapy strategy, such as the development of chimeric antigen receptor T-cell (CAR-T) adoptive cell transfer [154] and the elimination of protein programmed death-ligand 1 (PD-L1) SCs [155].
Senomorphics
Senomorphics have emerged as alternative therapeutic approaches for senolytics, which alleviate the effects of detrimental senescent functions via SASP inhibition. Generally, senomorphics target certain components of SASP pathways that lead to the production of SASP factors through NF-κB, JAK, SIRT1, mTOR, or other pathways involved in the induction and maintenance of the SASP [156]. Senomorphic agents that have been widely studied include ruxolitinib, metformin, resveratrol, rapamycin, fibroblast growth factor 21 (FGF21), and p38 MAPK inhibitors. Owing to the nature of senomorphics, continuous regular administration is required to maintain efficacy, which may lead to more side effects than intermittent administration of senolytics and may also lead to off-target effects in nonsenescent cells by inhibiting cytokine secretion, which may be essential. In addition to inhibiting senescence diffusion, senomorphics can act as immunotherapies to improve senescence surveillance efficiency, such as through blocking chronic inflammatory factors that suppress immune surveillance [157].
Advances in targeting senescent endothelial cells in diabetic retinopathy and related complications
An increasing number of studies have demonstrated that EC senescence triggers or accelerates the pathological process of multiple ocular vascular diseases (especially those that are age-related), including DR, diabetic macular edema (DME), AMD, ROP, and glaucoma [158]. Owing to the prominent role of EC senescence in pathological angiogenesis, most related senotherapeutics currently target proliferative retinopathies, particularly PDR and DME.
UBX-1967 is a potent senolytic member of the BCL-2 family. In the OIR model, the intravitreal administration of UBX-1967 inhibits pathological angiogenesis while promoting vascular regeneration [6]. In terms of mechanism, UBX1967 selectively depletes a population of SECs associated with retinopathy that expresses Col1a1 [6].
In addition to UBX-1967, UBX-1325 is currently the most promising drug candidate. UBX-1325 is a small molecule inhibitor of BCL-xL and is tailored to treat senescence in age-related eye disorders such as PDR and DME. UBX-1325 has been shown to promote apoptosis in a senescence-associated OIR model [159]. Furthermore, UBX-1325 improved the retinal vasculature in both OIR and STZ mice by effectively inhibiting retinal neovascularization and reducing vascular permeability [159]. In a phase 2b study in patients with DME, a single injection of UBX1325 led to a statistically significant and clinically meaningful improvement in the mean best corrected visual acuity (BCVA) through 48 weeks compared with sham treatment. To date, UBX1325 has favorable safety and tolerability profiles, with no evidence of intraocular inflammation [160].
Approaches focused on increasing NO production reduce SECs from upstream. 2(S)-Amino-6-boronohexanoic acid (ABH) has the potential to alleviate DR via arginase inhibition since it can blunt the diabetes-induced increase in p16INK4A and p38 in retinal ECs [74]. In addition, eNOS activation or the ingestion of NO-boosting substances, including L-arginine, L-citrulline and antioxidants, can delay endothelial senescence under high glucose conditions [161].
In conclusion, future diabetes-related research should focus more on the endothelial lineage. Furthermore, more experimental and clinical studies are needed to develop strategies to target SECs and to determine the impact that other diabetes treatments may have on EC senescence and its downstream effects. Notably, judicious safety monitoring is necessary, as the long-term effects of clearing SCs in humans are not known. If successful, a progressive focus should be placed on exploring ways to improve the ability of antiaging therapies to prevent rather than treat DR.
Summary and prospects
The dual role of EC senescence in DR vasculopathy has gradually been demonstrated. SECs both exacerbate microvascular rarefaction and pathological angiogenesis and participate in the regression of pathological neovascularization and regeneration of functional vessels in the later stage. These studies further promote the development of strategies for targeting the clearance of SECs, which shows great potential to compensate for the shortcomings of current DR treatments. This is because the strategy differentiates between pathological and physiological vessels at the source and targets the elimination of pathological vessels and remodeling of physiological vessels, resulting in a long-lasting solution to retinal ischemia. Although the multiple anti-senescence therapies under discussion have shown varying degrees of benefit, most of them are still in the initial experimental phase. Therefore, the actual risks of these drugs cannot be fully assessed until more candidates continue or move further into clinical trials. However, the current lack of reported risks may be an encouraging sign for the use of strategies targeting SCs or SECs.
In addition, many questions remain to be explored and further verified in the current research on cellular senescence. As mentioned previously, depending on the cell type, stressor, and duration of senescence, SCs may exist in any given tissue and secrete SASPs with different compositions. In fact, the high heterogeneity of SCs at the single-cell level complicates our understanding of the implications of SCs in tissue and therapeutic targeting. In addition, almost all cells in the retina can act as effectors or donors of proinflammatory factors and ROS and interact through them. Therefore, it is difficult to analyze which cell type plays a role in which phase of DR and to confirm the extent to which SECs contribute, yet this is necessary for the selection of therapeutic strategies.
Moreover, the lack of unique and cell-specific biomarkers for the direct detection of SC subpopulations in specific pathophysiologic contexts is another fundamental challenge. This increases the difficulty in elucidating the similarities and complexities of SCs. For example, since SCs may be beneficial in some contexts, complete elimination of SCs or total inhibition of the SASP may be detrimental. Therefore, future studies should focus on identifying reliable, sensitive, and specific biomarkers of cellular senescence to more precisely target SCs that secrete persistent, tissue-damaging SASP components. Notably, the effects of cellular senescence described thus far seem to be related to duration: acute, transient senescence tends to be beneficial (e.g., tissue development, injury repair), whereas persistent senescence seems to have a negative impact on the restoration of tissue homeostasis (e.g., chronic inflammation, tissue lesions). From this perspective, comparing and selecting biomarkers related to effects that occur during chronic senescence, rather than during the early acute response, may contribute to the above purposes. On these bases, a more generalized approach is to collect both more general and more specialized data and tailor them for different purposes. In addition, recent advances in single-cell transcriptomics approaches, such as spatial transcriptomics, provide a potentially promising tool in the search for new biomarkers of SC subpopulations.
In conclusion, further exploration of the heterogeneity of cellular senescence and the mechanisms of spatiotemporal regulation of the SASP, as well as the molecular mechanisms and precise characterization of EC senescence, is needed in the future. Furthermore, exploring how these mechanisms can be manipulated for therapeutic purposes will profoundly impact the development of therapeutic approaches for vascular diseases.
Data availability
No datasets were generated or analysed during the current study.
References
Bourne RRA, Jonas JB, Bron AM et al (2018) Prevalence and causes of vision loss in high-income countries and in Eastern and Central Europe in 2015: magnitude, temporal trends and projections. Br J Ophthalmol 102:575–585. https://doi.org/10.1136/bjophthalmol-2017-311258
Gui F, You Z, Fu S et al (2020) Endothelial dysfunction in Diabetic Retinopathy. Front Endocrinol (Lausanne) 11:591. https://doi.org/10.3389/fendo.2020.00591
Antonetti DA, Klein R, Gardner TW (2012) Diabetic retinopathy. N Engl J Med 366:1227–1239. https://doi.org/10.1056/NEJMra1005073
Antonetti DA, Silva PS, Stitt AW (2021) Current understanding of the molecular and cellular pathology of diabetic retinopathy. Nat Rev Endocrinol 17:195–206. https://doi.org/10.1038/s41574-020-00451-4
Oubaha M, Miloudi K, Dejda A et al (2016) Senescence-associated secretory phenotype contributes to pathological angiogenesis in retinopathy. Sci Transl Med 8:362ra144. https://doi.org/10.1126/scitranslmed.aaf9440
Crespo-Garcia S, Tsuruda PR, Dejda A et al (2021) Pathological angiogenesis in retinopathy engages cellular senescence and is amenable to therapeutic elimination via BCL-xL inhibition. Cell Metab 33:818–832e7. https://doi.org/10.1016/j.cmet.2021.01.011
Solomon SD, Chew E, Duh EJ et al (2017) Diabetic Retinopathy: A position Statement by the American Diabetes Association. Diabetes Care 40:412–418. https://doi.org/10.2337/dc16-2641
Tang L, Zhang C, Yang Q et al (2021) Melatonin maintains inner blood-retinal barrier via inhibition of p38/TXNIP/NF-κB pathway in diabetic retinopathy. J Cell Physiol 236:5848–5864. https://doi.org/10.1002/jcp.30269
Wang J, Xu X, Elliott MH et al (2010) Müller cell-derived VEGF is essential for diabetes-induced retinal inflammation and vascular leakage. Diabetes 59:2297–2305. https://doi.org/10.2337/db09-1420
Wang J-C, Li G-Y, Wang B et al (2019) Metformin inhibits metastatic breast cancer progression and improves chemosensitivity by inducing vessel normalization via PDGF-B downregulation. J Exp Clin Cancer Res 38:235. https://doi.org/10.1186/s13046-019-1211-2
Morgan MJ, Liu Z (2011) Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res 21:103–115. https://doi.org/10.1038/cr.2010.178
Kageyama S, Yokoo H, Tomita K et al (2011) High glucose-induced apoptosis in human coronary artery endothelial cells involves up-regulation of death receptors. Cardiovasc Diabetol 10:73. https://doi.org/10.1186/1475-2840-10-73
Elshaer SL, Lemtalsi T, El-Remessy AB (2018) High glucose-mediated tyrosine nitration of PI3-Kinase: a Molecular switch of Survival and apoptosis in endothelial cells. Antioxid (Basel) 7:47. https://doi.org/10.3390/antiox7040047
An Y, Geng K, Wang H-Y et al (2023) Hyperglycemia-induced STING signaling activation leads to aortic endothelial injury in diabetes. Cell Commun Signal 21:365. https://doi.org/10.1186/s12964-023-01393-w
Bhatt MP, Lim Y-C, Hwang J et al (2013) C-peptide prevents hyperglycemia-induced endothelial apoptosis through inhibition of reactive oxygen species-mediated transglutaminase 2 activation. Diabetes 62:243–253. https://doi.org/10.2337/db12-0293
Simó R, Stitt AW, Gardner TW (2018) Neurodegeneration in diabetic retinopathy: does it really matter? Diabetologia 61:1902–1912. https://doi.org/10.1007/s00125-018-4692-1
Kim I, Kim HG, So JN et al (2000) Angiopoietin-1 regulates endothelial cell survival through the phosphatidylinositol 3’-Kinase/Akt signal transduction pathway. Circ Res 86:24–29. https://doi.org/10.1161/01.res.86.1.24
Augustin HG, Koh GY, Thurston G, Alitalo K (2009) Control of vascular morphogenesis and homeostasis through the Angiopoietin-Tie system. Nat Rev Mol Cell Biol 10:165–177. https://doi.org/10.1038/nrm2639
Sweeney MD, Ayyadurai S, Zlokovic BV (2016) Pericytes of the neurovascular unit: key functions and signaling pathways. Nat Neurosci 19:771–783. https://doi.org/10.1038/nn.4288
Klaassen I, Van Noorden CJF, Schlingemann RO (2013) Molecular basis of the inner blood-retinal barrier and its breakdown in diabetic macular edema and other pathological conditions. Prog Retin Eye Res 34:19–48. https://doi.org/10.1016/j.preteyeres.2013.02.001
Li W, Yanoff M, Liu X, Ye X (1997) Retinal capillary pericyte apoptosis in early human diabetic retinopathy. Chin Med J (Engl) 110:659–663
Geraldes P, Hiraoka-Yamamoto J, Matsumoto M et al (2009) Activation of PKC-delta and SHP-1 by hyperglycemia causes vascular cell apoptosis and diabetic retinopathy. Nat Med 15:1298–1306. https://doi.org/10.1038/nm.2052
Roy S, Kim D (2021) Retinal capillary basement membrane thickening: role in the pathogenesis of diabetic retinopathy. Prog Retin Eye Res 82:100903. https://doi.org/10.1016/j.preteyeres.2020.100903
Hayden MR, Sowers JR, Tyagi SC (2005) The central role of vascular extracellular matrix and basement membrane remodeling in metabolic syndrome and type 2 diabetes: the matrix preloaded. Cardiovasc Diabetol 4:9. https://doi.org/10.1186/1475-2840-4-9
Cantón A, Martinez-Cáceres EM, Hernández C et al (2004) CD4-CD8 and CD28 expression in T cells infiltrating the vitreous fluid in patients with proliferative diabetic retinopathy: a flow cytometric analysis. Arch Ophthalmol 122:743–749. https://doi.org/10.1001/archopht.122.5.743
Tedeschi T, Lee K, Zhu W, Fawzi AA (2022) Limited hyperoxia-induced proliferative retinopathy: a model of persistent retinal vascular dysfunction, preretinal fibrosis and hyaloidal vascular reprogramming for retinal rescue. PLoS ONE 17:e0267576. https://doi.org/10.1371/journal.pone.0267576
Abu El-Asrar AM, De Hertogh G, van den Eynde K et al (2015) Myofibroblasts in proliferative diabetic retinopathy can originate from infiltrating fibrocytes and through endothelial-to-mesenchymal transition (EndoMT). Exp Eye Res 132:179–189. https://doi.org/10.1016/j.exer.2015.01.023
Xia M, Jiao L, Wang X-H et al (2023) Single-cell RNA sequencing reveals a unique pericyte type associated with capillary dysfunction. Theranostics 13:2515–2530. https://doi.org/10.7150/thno.83532
Chen Y-T, Chang F-C, Wu C-F et al (2011) Platelet-derived growth factor receptor signaling activates pericyte-myofibroblast transition in obstructive and post-ischemic kidney fibrosis. Kidney Int 80:1170–1181. https://doi.org/10.1038/ki.2011.208
Zhao Z, Zhang Y, Zhang C et al (2022) TGF-β promotes pericyte-myofibroblast transition in subretinal fibrosis through the Smad2/3 and Akt/mTOR pathways. Exp Mol Med 54:673–684. https://doi.org/10.1038/s12276-022-00778-0
Muñoz-Espín D, Serrano M (2014) Cellular senescence: from physiology to pathology. Nat Rev Mol Cell Biol 15:482–496. https://doi.org/10.1038/nrm3823
Prata LGPL, Ovsyannikova IG, Tchkonia T, Kirkland JL (2018) Senescent cell clearance by the immune system: emerging therapeutic opportunities. Semin Immunol 40:101275. https://doi.org/10.1016/j.smim.2019.04.003
Mortuza R, Chen S, Feng B et al (2013) High glucose induced alteration of SIRTs in endothelial cells causes rapid aging in a p300 and FOXO regulated pathway. PLoS ONE 8:e54514. https://doi.org/10.1371/journal.pone.0054514
Gorgoulis V, Adams PD, Alimonti A et al (2019) Cellular Senescence: defining a path Forward. Cell 179:813–827. https://doi.org/10.1016/j.cell.2019.10.005
He S, Sharpless NE (2017) Senescence in Health and Disease. Cell 169:1000–1011. https://doi.org/10.1016/j.cell.2017.05.015
d’Adda di Fagagna F (2008) Living on a break: cellular senescence as a DNA-damage response. Nat Rev Cancer 8:512–522. https://doi.org/10.1038/nrc2440
Childs BG, Baker DJ, Kirkland JL et al (2014) Senescence and apoptosis: dueling or complementary cell fates? EMBO Rep 15:1139–1153. https://doi.org/10.15252/embr.201439245
Hsu C-H, Altschuler SJ, Wu LF (2019) Patterns of early p21 Dynamics Determine Proliferation-Senescence Cell Fate after Chemotherapy. Cell 178:361–373e12. https://doi.org/10.1016/j.cell.2019.05.041
Coppé J-P, Desprez P-Y, Krtolica A, Campisi J (2010) The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol 5:99–118. https://doi.org/10.1146/annurev-pathol-121808-102144
Rodier F, Coppé J-P, Patil CK et al (2009) Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nat Cell Biol 11:973–979. https://doi.org/10.1038/ncb1909
Liu F, Wu S, Ren H, Gu J (2011) Klotho suppresses RIG-I-mediated senescence-associated inflammation. Nat Cell Biol 13:254–262. https://doi.org/10.1038/ncb2167
Schroder K, Tschopp J (2010) The inflammasomes. Cell 140:821–832. https://doi.org/10.1016/j.cell.2010.01.040
Ito Y, Hoare M, Narita M (2017) Spatial and temporal control of Senescence. Trends Cell Biol 27:820–832. https://doi.org/10.1016/j.tcb.2017.07.004
Kang C, Xu Q, Martin TD et al (2015) The DNA damage response induces inflammation and senescence by inhibiting autophagy of GATA4. Science 349:aaa5612. https://doi.org/10.1126/science.aaa5612
Freund A, Patil CK, Campisi J (2011) p38MAPK is a novel DNA damage response-independent regulator of the senescence-associated secretory phenotype: p38 regulates the senescence secretory phenotype. EMBO J 30:1536–1548. https://doi.org/10.1038/emboj.2011.69
Childs BG, Durik M, Baker DJ, van Deursen JM (2015) Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nat Med 21:1424–1435. https://doi.org/10.1038/nm.4000
Muñoz-Espín D, Cañamero M, Maraver A et al (2013) Programmed cell senescence during mammalian Embryonic Development. Cell 155:1104–1118. https://doi.org/10.1016/j.cell.2013.10.019
Storer M, Mas A, Robert-Moreno A et al (2013) Senescence is a developmental mechanism that contributes to embryonic growth and patterning. Cell 155:1119–1130. https://doi.org/10.1016/j.cell.2013.10.041
Demaria M, Ohtani N, Youssef SA et al (2014) An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell 31:722–733. https://doi.org/10.1016/j.devcel.2014.11.012
Mosteiro L, Pantoja C, Alcazar N et al (2016) Tissue damage and senescence provide critical signals for cellular reprogramming in vivo. Science 354:aaf4445. https://doi.org/10.1126/science.aaf4445
Franceschi C, Campisi J (2014) Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J Gerontol Biol Sci Med Sci 69(Suppl 1):S4–9. https://doi.org/10.1093/gerona/glu057
Nelson G, Wordsworth J, Wang C et al (2012) A senescent cell bystander effect: senescence-induced senescence. Aging Cell 11:345–349. https://doi.org/10.1111/j.1474-9726.2012.00795.x
Barinda AJ, Ikeda K, Nugroho DB et al (2020) Endothelial progeria induces adipose tissue senescence and impairs insulin sensitivity through senescence associated secretory phenotype. Nat Commun 11:481. https://doi.org/10.1038/s41467-020-14387-w
Kirkland JL, Tchkonia T (2017) Cellular Senescence: a translational perspective. EBioMedicine 21:21–28. https://doi.org/10.1016/j.ebiom.2017.04.013
Ungvari Z, Labinskyy N, Gupte S et al (2008) Dysregulation of mitochondrial biogenesis in vascular endothelial and smooth muscle cells of aged rats. Am J Physiol Heart Circ Physiol 294:H2121–2128. https://doi.org/10.1152/ajpheart.00012.2008
Chala N, Moimas S, Giampietro C et al (2021) Mechanical fingerprint of Senescence in endothelial cells. Nano Lett 21:4911–4920. https://doi.org/10.1021/acs.nanolett.1c00064
Orsenigo F, Giampietro C, Ferrari A et al (2012) Phosphorylation of VE-cadherin is modulated by haemodynamic forces and contributes to the regulation of vascular permeability in vivo. Nat Commun 3:1208. https://doi.org/10.1038/ncomms2199
Franco D, Milde F, Klingauf M et al (2013) Accelerated endothelial wound healing on microstructured substrates under flow. Biomaterials 34:1488–1497. https://doi.org/10.1016/j.biomaterials.2012.10.007
Ting LH, Jahn JR, Jung JI et al (2012) Flow mechanotransduction regulates traction forces, intercellular forces, and adherens junctions. Am J Physiol Heart Circ Physiol 302:H2220–2229. https://doi.org/10.1152/ajpheart.00975.2011
Graves SI, Baker DJ (2020) Implicating endothelial cell senescence to dysfunction in the ageing and diseased brain. Basic Clin Pharmacol Toxicol 127:102–110. https://doi.org/10.1111/bcpt.13403
Donato AJ, Morgan RG, Walker AE, Lesniewski LA (2015) Cellular and molecular biology of aging endothelial cells. J Mol Cell Cardiol 89:122–135. https://doi.org/10.1016/j.yjmcc.2015.01.021
Hayashi T, Yano K, Matsui-Hirai H et al (2008) Nitric oxide and endothelial cellular senescence. Pharmacol Ther 120:333–339. https://doi.org/10.1016/j.pharmthera.2008.09.002
De Bock K, Georgiadou M, Schoors S et al (2013) Role of PFKFB3-driven glycolysis in vessel sprouting. Cell 154:651–663. https://doi.org/10.1016/j.cell.2013.06.037
Kalucka J, Bierhansl L, Conchinha NV et al (2018) Quiescent endothelial cells upregulate fatty acid β-Oxidation for Vasculoprotection via Redox Homeostasis. Cell Metab 28:881–894e13. https://doi.org/10.1016/j.cmet.2018.07.016
Han Y, Kim SY (2023) Endothelial senescence in vascular diseases: current understanding and future opportunities in senotherapeutics. Exp Mol Med 55:1–12. https://doi.org/10.1038/s12276-022-00906-w
Dimri GP, Lee X, Basile G et al (1995) A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci USA 92:9363–9367. https://doi.org/10.1073/pnas.92.20.9363
Yang N-C, Hu M-L (2005) The limitations and validities of senescence associated-beta-galactosidase activity as an aging marker for human foreskin fibroblast Hs68 cells. Exp Gerontol 40:813–819. https://doi.org/10.1016/j.exger.2005.07.011
Biran A, Zada L, Abou Karam P et al (2017) Quantitative identification of senescent cells in aging and disease. Aging Cell 16:661–671. https://doi.org/10.1111/acel.12592
González-Gualda E, Baker AG, Fruk L, Muñoz-Espín D (2021) A guide to assessing cellular senescence in vitro and in vivo. FEBS J 288:56–80. https://doi.org/10.1111/febs.15570
Wolf J, Rasmussen DK, Sun YJ et al (2023) Liquid-biopsy proteomics combined with AI identifies cellular drivers of eye aging and disease in vivo. Cell 186:4868–4884e12. https://doi.org/10.1016/j.cell.2023.09.012
Chiu J-J, Chien S (2011) Effects of disturbed flow on vascular endothelium: pathophysiological basis and clinical perspectives. Physiol Rev 91:327–387. https://doi.org/10.1152/physrev.00047.2009
Warboys CM, de Luca A, Amini N et al (2014) Disturbed flow promotes endothelial senescence via a p53-dependent pathway. Arterioscler Thromb Vasc Biol 34:985–995. https://doi.org/10.1161/ATVBAHA.114.303415
Bloom SI, Islam MT, Lesniewski LA, Donato AJ (2023) Mechanisms and consequences of endothelial cell senescence. Nat Rev Cardiol 20:38–51. https://doi.org/10.1038/s41569-022-00739-0
Shosha E, Xu Z, Narayanan SP et al (2018) Mechanisms of Diabetes-Induced endothelial cell senescence: role of arginase 1. Int J Mol Sci 19:1215. https://doi.org/10.3390/ijms19041215
Stabenow LK, Zibrova D, Ender C et al (2022) Oxidative glucose metabolism promotes senescence in vascular endothelial cells. Cells 11:2213. https://doi.org/10.3390/cells11142213
Rashid G, Benchetrit S, Fishman D, Bernheim J (2004) Effect of advanced glycation end-products on gene expression and synthesis of TNF-alpha and endothelial nitric oxide synthase by endothelial cells. Kidney Int 66:1099–1106. https://doi.org/10.1111/j.1523-1755.2004.00860.x
Birch J, Passos JF (2017) Targeting the SASP to combat ageing: Mitochondria as possible intracellular allies? https://doi.org/10.1002/bies.201600235. Bioessays 39:
Kang Q, Yang C (2020) Oxidative stress and diabetic retinopathy: molecular mechanisms, pathogenetic role and therapeutic implications. Redox Biol 37:101799. https://doi.org/10.1016/j.redox.2020.101799
Korshunov SS, Skulachev VP, Starkov AA (1997) High protonic potential actuates a mechanism of production of reactive oxygen species in mitochondria. Febs Lett 416:15–18. https://doi.org/10.1016/S0014-5793(97)01159-9
Brownlee M (2005) The pathobiology of Diabetic complications. Diabetes 54:1615–1625. https://doi.org/10.2337/diabetes.54.6.1615
Madsen-Bouterse SA, Kowluru RA (2008) Oxidative stress and diabetic retinopathy: pathophysiological mechanisms and treatment perspectives. Rev Endocr Metab Dis 9:315–327. https://doi.org/10.1007/s11154-008-9090-4
Oikawa S, Kawanishi S (1999) Site-specific DNA damage at GGG sequence by oxidative stress may accelerate telomere shortening. FEBS Lett 453:365–368. https://doi.org/10.1016/s0014-5793(99)00748-6
Hewitt G, Jurk D, Marques FDM et al (2012) Telomeres are favoured targets of a persistent DNA damage response in ageing and stress-induced senescence. Nat Commun 3:708. https://doi.org/10.1038/ncomms1708
Anderson R, Lagnado A, Maggiorani D et al (2019) Length-independent telomere damage drives post-mitotic cardiomyocyte senescence. EMBO J 38:e100492. https://doi.org/10.15252/embj.2018100492
Morgan RG, Donato AJ, Walker AE (2018) Telomere uncapping and vascular aging. Am J Physiol Heart Circ Physiol 315:H1–H5. https://doi.org/10.1152/ajpheart.00008.2018
Victorelli S, Passos JF (2017) Telomeres and cell senescence - size matters not. EBioMedicine 21:14–20. https://doi.org/10.1016/j.ebiom.2017.03.027
Zhong W, Zou G, Gu J, Zhang J (2010) L-arginine attenuates high glucose-accelerated senescence in human umbilical vein endothelial cells. Diabetes Res Clin Pract 89:38–45. https://doi.org/10.1016/j.diabres.2010.03.013
Hayashi T, Kotani H, Yamaguchi T et al (2014) Endothelial cellular senescence is inhibited by liver X receptor activation with an additional mechanism for its atheroprotection in diabetes. Proc Natl Acad Sci USA 111:1168–1173. https://doi.org/10.1073/pnas.1322153111
Chen XJ, Butow RA (2005) The organization and inheritance of the mitochondrial genome. Nat Rev Genet 6:815–825. https://doi.org/10.1038/nrg1708
Ago T, Kuroda J, Kamouchi M et al (2011) Pathophysiological roles of NADPH oxidase/nox family proteins in the vascular system. -Review and perspective-. Circ J 75:1791–1800. https://doi.org/10.1253/circj.cj-11-0388
Kowluru RA, Kowluru A, Mishra M, Kumar B (2015) Oxidative stress and epigenetic modifications in the pathogenesis of diabetic retinopathy. Prog Retin Eye Res 48:40–61. https://doi.org/10.1016/j.preteyeres.2015.05.001
Correia-Melo C, Marques FD, Anderson R et al (2016) Mitochondria are required for pro‐ageing features of the senescent phenotype. EMBO J 35:724–742. https://doi.org/10.15252/embj.201592862
Wiley CD, Velarde MC, Lecot P et al (2016) Mitochondrial dysfunction induces senescence with a distinct secretory phenotype. Cell Metab 23:303–314. https://doi.org/10.1016/j.cmet.2015.11.011
Sadikan MZ, Nasir NAA, Iezhitsa I, Agarwal R (2022) Antioxidant and anti-apoptotic effects of tocotrienol-rich fraction against streptozotocin-induced diabetic retinopathy in rats. Biomed Pharmacother 153:113533. https://doi.org/10.1016/j.biopha.2022.113533
Houtkooper RH, Pirinen E, Auwerx J (2012) Sirtuins as regulators of metabolism and healthspan. Nat Rev Mol Cell Biol 13:225–238. https://doi.org/10.1038/nrm3293
Ota H, Akishita M, Eto M et al (2007) Sirt1 modulates premature senescence-like phenotype in human endothelial cells. J Mol Cell Cardiol 43:571–579. https://doi.org/10.1016/j.yjmcc.2007.08.008
Zu Y, Liu L, Lee MYK et al (2010) SIRT1 promotes proliferation and prevents senescence through targeting LKB1 in primary porcine aortic endothelial cells. Circ Res 106:1384–1393. https://doi.org/10.1161/CIRCRESAHA.109.215483
Lee O-H, Woo YM, Moon S et al (2020) Sirtuin 6 deficiency induces endothelial cell senescence via downregulation of forkhead box M1 expression. Aging 12:20946–20967. https://doi.org/10.18632/aging.202176
Liu J, Chen S, Biswas S et al (2020) Glucose-induced oxidative stress and accelerated aging in endothelial cells are mediated by the depletion of mitochondrial SIRTs. Physiol Rep 8:e14331. https://doi.org/10.14814/phy2.14331
Tanaka Y, Chen ZJ (2012) STING specifies IRF3 phosphorylation by TBK1 in the cytosolic DNA signaling pathway. Sci Signal 5:ra20. https://doi.org/10.1126/scisignal.2002521
Frisch SM, MacFawn IP (2020) Type I interferons and related pathways in cell senescence. Aging Cell 19:e13234. https://doi.org/10.1111/acel.13234
Chen Q, Sun L, Chen ZJ (2016) Regulation and function of the cGAS-STING pathway of cytosolic DNA sensing. Nat Immunol 17:1142–1149. https://doi.org/10.1038/ni.3558
Ren J-L, Pan J-S, Lu Y-P et al (2009) Inflammatory signaling and cellular senescence. Cell Signal 21:378–383. https://doi.org/10.1016/j.cellsig.2008.10.011
Kandhaya-Pillai R, Miro-Mur F, Alijotas-Reig J et al (2017) TNFα-senescence initiates a STAT-dependent positive feedback loop, leading to a sustained interferon signature, DNA damage, and cytokine secretion. Aging 9:2411–2435. https://doi.org/10.18632/aging.101328
Patel C, Rojas M, Narayanan SP et al (2013) Arginase as a mediator of diabetic retinopathy. Front Immunol 4:173. https://doi.org/10.3389/fimmu.2013.00173
Ding Q, Hayashi T, Packiasamy AJ et al (2004) The effect of high glucose on NO and O2 – through endothelial GTPCH1 and NADPH oxidase. Life Sci 75:3185–3194. https://doi.org/10.1016/j.lfs.2004.06.005
Du XL, Edelstein D, Dimmeler S et al (2001) Hyperglycemia inhibits endothelial nitric oxide synthase activity by posttranslational modification at the akt site. J Clin Invest 108:1341–1348. https://doi.org/10.1172/JCI11235
Lin F, Yang Y, Wei S et al (2020) Hydrogen Sulfide protects against high glucose-Induced Human Umbilical Vein Endothelial Cell Injury through activating PI3K/Akt/eNOS pathway. Drug Des Devel Ther 14:621–633. https://doi.org/10.2147/DDDT.S242521
Förstermann U, Münzel T (2006) Endothelial nitric oxide synthase in Vascular Disease: from Marvel to Menace. Circulation 113:1708–1714. https://doi.org/10.1161/CIRCULATIONAHA.105.602532
Chandra S, Romero MJ, Shatanawi A et al (2012) Oxidative species increase arginase activity in endothelial cells through the RhoA/Rho kinase pathway. Br J Pharmacol 165:506–519. https://doi.org/10.1111/j.1476-5381.2011.01584.x
Rojas M, Lemtalsi T, Toque HA et al (2017) NOX2-Induced activation of Arginase and Diabetes-Induced Retinal endothelial cell senescence. Antioxid (Basel) 6:43. https://doi.org/10.3390/antiox6020043
Simon HU, Haj-Yehia A, Levi-Schaffer F (2000) Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis 5:415–418. https://doi.org/10.1023/a:1009616228304
Csiszar A, Ungvari Z, Koller A et al (2004) Proinflammatory phenotype of coronary arteries promotes endothelial apoptosis in aging. Physiol Genomics 17:21–30. https://doi.org/10.1152/physiolgenomics.00136.2003
Csiszar A, Labinskyy N, Smith K et al (2007) Vasculoprotective effects of anti-tumor necrosis factor-alpha treatment in aging. Am J Pathol 170:388–398. https://doi.org/10.2353/ajpath.2007.060708
Michaud M, Balardy L, Moulis G et al (2013) Proinflammatory cytokines, aging, and Age-Related diseases. J Am Med Dir Assoc 14:877–882. https://doi.org/10.1016/j.jamda.2013.05.009
Ungvari Z, Orosz Z, Labinskyy N et al (2007) Increased mitochondrial H2O2 production promotes endothelial NF-kappaB activation in aged rat arteries. Am J Physiol Heart Circ Physiol 293:H37–47. https://doi.org/10.1152/ajpheart.01346.2006
Ganz P, Vita JA (2003) Testing endothelial vasomotor function: nitric oxide, a multipotent molecule. Circulation 108:2049–2053. https://doi.org/10.1161/01.CIR.0000089507.19675.F9
Giannotti G, Doerries C, Mocharla PS et al (2010) Impaired endothelial repair capacity of early endothelial progenitor cells in prehypertension: relation to endothelial dysfunction. Hypertension 55:1389–1397. https://doi.org/10.1161/HYPERTENSIONAHA.109.141614
Ungvari Z, Tucsek Z, Sosnowska D et al (2013) Aging-induced dysregulation of dicer1-dependent microRNA expression impairs angiogenic capacity of rat cerebromicrovascular endothelial cells. J Gerontol Biol Sci Med Sci 68:877–891. https://doi.org/10.1093/gerona/gls242
Chang H, Rha SY, Jeung H-C et al (2013) Telomerase- and angiogenesis-related gene responses to irradiation in human umbilical vein endothelial cells. Int J Mol Med 31:1202–1208. https://doi.org/10.3892/ijmm.2013.1300
Ziche M, Morbidelli L, Choudhuri R et al (1997) Nitric oxide synthase lies downstream from vascular endothelial growth factor-induced but not basic fibroblast growth factor-induced angiogenesis. J Clin Invest 99:2625–2634. https://doi.org/10.1172/JCI119451
Kubis N, Richer C, Domergue V et al (2002) Role of microvascular rarefaction in the increased arterial pressure in mice lacking for the endothelial nitric oxide synthase gene (eNOS3pt-/-). J Hypertens 20:1581–1587. https://doi.org/10.1097/00004872-200208000-00021
Ashraf M, Sampani K, Rageh A et al (2020) Interaction between the Distribution of Diabetic Retinopathy Lesions and the Association of Optical Coherence Tomography Angiography Scans with Diabetic Retinopathy Severity. JAMA Ophthalmol 138:1291–1297. https://doi.org/10.1001/jamaophthalmol.2020.4516
Tan B, Chua J, Lin E et al (2020) Quantitative microvascular analysis with wide-field optical coherence tomography angiography in eyes with Diabetic Retinopathy. JAMA Netw Open 3:e1919469. https://doi.org/10.1001/jamanetworkopen.2019.19469
López-Luppo M, Catita J, Ramos D et al (2017) Cellular Senescence is Associated with Human retinal microaneurysm formation during aging. Invest Ophth Vis Sci 58:2832–2842. https://doi.org/10.1167/iovs.16-20312
Stitt AW, Gardiner TA, Archer DB (1995) Histological and ultrastructural investigation of retinal microaneurysm development in diabetic patients. Br J Ophthalmol 79:362–367. https://doi.org/10.1136/bjo.79.4.362
An D, Tan B, Yu D-Y, Balaratnasingam C (2022) Differentiating Microaneurysm Pathophysiology in Diabetic Retinopathy through Objective Analysis of Capillary Nonperfusion, inflammation, and Pericytes. Diabetes 71:733–746. https://doi.org/10.2337/db21-0737
López-Luppo M, Nacher V, Ramos D et al (2017) Blood vessel basement membrane alterations in human retinal microaneurysms during Aging. Invest Ophth Vis Sci 58:1116–1131. https://doi.org/10.1167/iovs.16-19998
Binet F, Cagnone G, Crespo-Garcia S et al (2020) Neutrophil extracellular traps target senescent vasculature for tissue remodeling in retinopathy. Science 369:eaay5356. https://doi.org/10.1126/science.aay5356
Acosta JC, O’Loghlen A, Banito A et al (2008) Chemokine signaling via the CXCR2 receptor reinforces senescence. Cell 133:1006–1018. https://doi.org/10.1016/j.cell.2008.03.038
Kuilman T, Michaloglou C, Vredeveld LCW et al (2008) Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 133:1019–1031. https://doi.org/10.1016/j.cell.2008.03.039
Han JR, Ju WK, Park IW (2004) Spontaneous regression of neovascularization at the disc in diabetic retinopathy. Korean J Ophthalmol 18:41–46. https://doi.org/10.3341/kjo.2004.18.1.41
Bandello F, Gass JD, Lattanzio R, Brancato R (1996) Spontaneous regression of neovascularization at the disk and elsewhere in diabetic retinopathy. Am J Ophthalmol 122:494–501. https://doi.org/10.1016/s0002-9394(14)72108-7
Chaib S, Tchkonia T, Kirkland JL (2022) Cellular senescence and senolytics: the path to the clinic. Nat Med 28:1556–1568. https://doi.org/10.1038/s41591-022-01923-y
Singer M, Sansonetti PJ (2004) IL-8 is a key chemokine regulating neutrophil recruitment in a new mouse model of Shigella-induced colitis. J Immunol 173:4197–4206. https://doi.org/10.4049/jimmunol.173.6.4197
Arroba AI, Alcalde-Estevez E, García-Ramírez M et al (2016) Modulation of microglia polarization dynamics during diabetic retinopathy in db/db mice. Biochim Biophys Acta 1862:1663–1674. https://doi.org/10.1016/j.bbadis.2016.05.024
Zeng H, Green WR, Tso MOM (2008) Microglial activation in human diabetic retinopathy. Arch Ophthalmol 126:227–232. https://doi.org/10.1001/archophthalmol.2007.65
Klotzsche-von Ameln A, Sprott D (2022) Harnessing retinal phagocytes to combat pathological neovascularization in ischemic retinopathies? Pflugers Arch 474:575–590. https://doi.org/10.1007/s00424-022-02695-7
Korovina I, Neuwirth A, Sprott D et al (2020) Myeloid SOCS3 Deficiency regulates angiogenesis via enhanced apoptotic endothelial cell engulfment. J Innate Immun 12:248–256. https://doi.org/10.1159/000502645
Korovina I, Neuwirth A, Sprott D et al (2019) Hematopoietic hypoxia-inducible factor 2α deficiency ameliorates pathological retinal neovascularization via modulation of endothelial cell apoptosis. FASEB J 33:1758–1770. https://doi.org/10.1096/fj.201800430R
Joussen AM, Poulaki V, Mitsiades N et al (2003) Suppression of Fas-FasL-induced endothelial cell apoptosis prevents diabetic blood-retinal barrier breakdown in a model of streptozotocin-induced diabetes. FASEB J 17:76–78. https://doi.org/10.1096/fj.02-0157fje
Kessenbrock K, Krumbholz M, Schönermarck U et al (2009) Netting neutrophils in autoimmune small-vessel vasculitis. Nat Med 15:623–625. https://doi.org/10.1038/nm.1959
Fuhrmann-Stroissnigg H, Ling YY, Zhao J et al (2017) Identification of HSP90 inhibitors as a novel class of senolytics. Nat Commun 8:422. https://doi.org/10.1038/s41467-017-00314-z
Chan CM, Jing X, Pike LA et al (2012) Targeted inhibition of src kinase with Dasatinib blocks thyroid Cancer Growth and Metastasis. Clin Cancer Res 18:3580–3591. https://doi.org/10.1158/1078-0432.CCR-11-3359
Zhu Y, Tchkonia T, Pirtskhalava T et al (2015) The Achilles’ heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell 14:644–658. https://doi.org/10.1111/acel.12344
Zhu Y, Tchkonia T, Fuhrmann-Stroissnigg H et al (2016) Identification of a novel senolytic agent, navitoclax, targeting the Bcl-2 family of anti-apoptotic factors. Aging Cell 15:428–435. https://doi.org/10.1111/acel.12445
Zhu Y, Doornebal EJ, Pirtskhalava T et al (2017) New agents that target senescent cells: the flavone, fisetin, and the BCL-XL inhibitors, A1331852 and A1155463. Aging 9:955–963. https://doi.org/10.18632/aging.101202
Kirkland JL, Tchkonia T (2020) Senolytic drugs: from discovery to translation. J Intern Med 288:518–536. https://doi.org/10.1111/joim.13141
Guarente L, Sinclair DA, Kroemer G (2024) Human trials exploring anti-aging medicines. Cell Metab 36:354–376. https://doi.org/10.1016/j.cmet.2023.12.007
Muñoz-Espín D, Rovira M, Galiana I et al (2018) A versatile drug delivery system targeting senescent cells. EMBO Mol Med 10:e9355. https://doi.org/10.15252/emmm.201809355
Guerrero A, Guiho R, Herranz N et al (2020) Galactose-modified duocarmycin prodrugs as senolytics. Aging Cell 19:e13133. https://doi.org/10.1111/acel.13133
Cai Y, Zhou H, Zhu Y et al (2020) Elimination of senescent cells by β-galactosidase-targeted prodrug attenuates inflammation and restores physical function in aged mice. Cell Res 30:574–589. https://doi.org/10.1038/s41422-020-0314-9
Yin K, Patten D, Gough S et al (2022) Senescence-induced endothelial phenotypes underpin immune-mediated senescence surveillance. Genes Dev 36:533–549. https://doi.org/10.1101/gad.349585.122
Amor C, Feucht J, Leibold J et al (2020) Senolytic CAR T cells reverse senescence-associated pathologies. Nature 583:127–132. https://doi.org/10.1038/s41586-020-2403-9
Wang T-W, Johmura Y, Suzuki N et al (2022) Blocking PD-L1–PD-1 improves senescence surveillance and ageing phenotypes. Nature 611:358–364. https://doi.org/10.1038/s41586-022-05388-4
Song S, Tchkonia T, Jiang J et al (2020) Targeting senescent cells for a healthier aging: challenges and opportunities. Adv Sci (Weinh) 7:2002611. https://doi.org/10.1002/advs.202002611
Wang D, DuBois RN (2015) Immunosuppression associated with chronic inflammation in the tumor microenvironment. Carcinogenesis 36:1085–1093. https://doi.org/10.1093/carcin/bgv123
Huang J, Zhao Q, Li M et al (2019) The effects of endothelium-specific CYP2J2 overexpression on the attenuation of retinal ganglion cell apoptosis in a glaucoma rat model. Faseb J 33:11194–11209. https://doi.org/10.1096/fj.201900756R
Tsuruda P, Chaney S, Dejda A et al (2021) UBX1325, a small molecule inhibitor of Bcl-xL, attenuates vascular dysfunction in two animal models of retinopathy. Invest Ophth Vis Sci 62:1163–1163
Crespo-Garcia S, Fournier F, Diaz-Marin R et al (2024) Therapeutic targeting of cellular senescence in diabetic macular edema: preclinical and phase 1 trial results. Nat Med 30:443–454. https://doi.org/10.1038/s41591-024-02802-4
Hayashi T, Matsui-Hirai H, Miyazaki-Akita A et al (2006) Endothelial cellular senescence is inhibited by nitric oxide: implications in atherosclerosis associated with menopause and diabetes. Proc Natl Acad Sci USA 103:17018–17023. https://doi.org/10.1073/pnas.0607873103
Funding
This research was supported by the National Natural Science Foundation of China (Nos. 82371071, 81970814).
Author information
Authors and Affiliations
Contributions
Conceptualization, YL.L and GR.D; writing—original draft preparation, YL.L and GR.D; writing—review and editing, YL.L, YF.F, JX.S and GR.D; supervision, GR.D; funding acquisition, GR.D. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liao, YL., Fang, YF., Sun, JX. et al. Senescent endothelial cells: a potential target for diabetic retinopathy. Angiogenesis (2024). https://doi.org/10.1007/s10456-024-09943-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10456-024-09943-7