Abstract
This study evaluated the effect of seasonality on diet composition and overlap in fish species inhabiting the littoral zone of a small lake. We analyzed whether the way the species exploit food resources may help explain their coexistence. Fish samples were collected in a lake in the Upper Paraná River floodplain in September (dry period) and December (rainy period) of 2010. The diet of six species was evaluated through stomach content analysis, which showed that the most consumed resources were microcrustaceans, aquatic insects, aquatic higher plant and algae. Diet composition was significantly different between periods for all species studied. Diet overlap values were low in both periods, but trophic segregation was even higher in the rainy period, indicating that with greater abundance of food resources, species could exploit preferential resources. Trophic segregation, evidenced by the low overlap values in both periods, indicates the absence of interspecific competition, which may explain the coexistence of these species in the littoral zone of this small lake.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
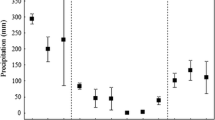
Floodplains have high habitat heterogeneity, including rivers, channels and lakes, which are subjected to a large temporal variability in the hydrological regime (Junk et al. 1989; Thomaz et al. 2007; Agostinho et al. 2009). In the Upper Paraná River floodplain, there is an intense regulation of water flow by dams located upstream, but the flood pulse is still the main driving force determining the structure and functioning of communities in this ecosystem (Agostinho et al. 2004). Therein, the rainy period covers the months from October to February with monthly averages higher than 125 mm, and the dry period, from June to September, with averages lower than 80 mm (Agostinho et al. 2004).
Littoral zones of floodplain lakes are colonized by extensive macrophyte stands that provide structure and high habitat complexity (Dibble et al. 1996; Agostinho et al. 2007; Thomaz et al. 2008; Dibble and Thomaz 2009). Because of this complexity, these areas contain high diversity and abundance of small-sized fish (Delariva et al. 1994; Meschiatti et al. 2000; Neiff et al. 2009; Dibble and Pelicice 2010; Cunha et al. 2011; Gomes et al. 2012) which have particular behavior patterns, such as low swimming activity and high exploitation of interstices among the roots (Priyadarshana et al. 2001). The habitat provided by macrophytes is favorable for these fish owing to the high availability of food resources like algae and invertebrates (Melo et al. 2002; Colon-Gaud et al. 2004; Bickel and Closs 2008; Thomaz et al. 2008; Murakami et al. 2009; Mormul et al. 2010; Colares et al. 2013).
Invertebrates represent an important food resource for fish, and their abundance is affected by seasonality (Takeda and Fujita 2004; Lansac-Tôha et al. 2004; Colares et al. 2013), which may change the diet composition of fish. A trend observed for small-sized aquatic invertivorous species inhabiting the littoral zone of lakes is a high consume of insect larvae in the dry period and microcrustaceans in the rainy period (Casatti et al. 2003; Russo and Hahn 2006). Besides, greater abundance of food resources are often found during rainy periods (Prejs and Prejs 1987; Junk et al. 1989; Esteves and Galetti 1995), when increased availability of terrestrial resources is expected (Abujanra et al. 2009; Novakowski et al. 2008; Walker et al. 2013).
Temporal variation in food availability also influences the partitioning of resources among Neotropical fish (Jepsen et al. 1997), and measures of trophic niche overlap can determine the degree of segregation between species (Corrêa et al. 2011). However, studies have shown different results, such as greater overlap in conditions of higher (Zaret and Rand 1971; Delbeek and Williams 1987; Tófoli et al. 2010) and lower (Goulding 1980; Prejs and Prejs 1987) food availability, and trophic segregation in periods of greater availability of resources (Pelicice and Agostinho 2006; Walker et al. 2013). Despite this controversy, food has been considered the main mechanism of ecological segregation for fish, and food partitioning is an efficient way to explore all food resources in a habitat, allowing the coexistence of species (Gerking 1994).
In Neotropical floodplains, there are several small shallow lakes isolated from the main river channel, which are flooded only during major floods, as in the case of Traíra Lake, where this study was conducted. At this stage of hydrological cycle, the dispersal of species shifts the assemblages of fish that commonly live in the littoral zone of lakes (Gomes et al. 2012). However, these environments are subjected to local influences, such as rainfall, and are characterized by a reduction in area, volume of water and depth during dry period, when it is expected stronger biotic interactions like predation and competition, whereas during rainy period, these interactions can be minimized (Junk et al. 1989; Gomes et al. 2012). In this context, the present study analyzed seasonal variations in the use of food resource by small fish inhabiting littoral zone of a floodplain lake, by comparing the diet between a severe drought (September 2010) and a rainy period (December 2010). We hypothesized that diet composition and overlap are affected by seasonality. We predicted an increase in consume of terrestrial resources and low overlap values during rainy period. This prediction was based on the assumption that the availability of terrestrial resources increases in the rainy period (Abujanra et al. 2009; Novakowski et al. 2008; Walker et al. 2013) and that greater availability of resources in this period provides trophic segregation (Pelicice and Agostinho 2006; Walker et al. 2013).
Materials and methods
The Upper Paraná River floodplain (22°50′S and 53°15′–53°40′W) lies in the upper section of the area corresponding to National Environmental Protection Area of Paraná River, which is the last dam-free stretch of Paraná River in Brazilian territory. It is a system that includes rivers, channels and lakes belonging to the sub-basins of the rivers Paraná, Baía and Ivinhema. Traíra Lake is located in Baía River sub-basin, between the coordinates 22°44′45.6″S; 53°20′21.6″W. It is a small lake (0.47 ha), rounded, with 2.1 m mean depth, 108.9 m length and 292.4 m perimeter. It is an isolated lake, 350 m distant from the main river, having a marginal levee 3 m high. Banks are dominated by grasses (95 %); aquatic vegetation in the littoral zone is abundant, composed of macrophytes rooted with floating leaves, free floating and emergent.
Samplings were conducted in September 2010, period of severe drought, and December 2010, rainy period (Fig. 1). Littoral zones were sampled with seining nets (10 m length, 0.5-cm mesh size). Fish were anesthetized with benzocaine before fixation for further analysis. Fish were identified and measured, and the stomachs were removed and fixed in 4 % formaldehyde. Voucher specimens were deposited in the Fish Collection of the Center for Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (Nupélia), State University of Maringá (Aphyocharax anisitsi NUP 461; Hyphessobrycon eques NUP 14252; Moenkhausia forestii NUP 14257; Psellogrammus kennedyi NUP 14395; Serrapinnus notomelas NUP 1402; Serrapinnus sp. NUP 16470).
Twenty species were caught during the dry period and 13 during the rainy period, from which we selected the six that occurred in both periods and represented 94.5 and 75.0 % of the total caught in the dry and rainy periods, respectively. We analyzed 177 individuals belonging to these species: A. anisitsi Eigenmann and Kennedy 1903 (standard length (SL) range = 2.5–3.0 cm; n = 18); H. eques (Steindachner 1882) (SL = 2.1–2.8 cm; n = 28); M. forestii Benine, Mariguella and Oliveira 2009 (SL = 2.6–3.3 cm; n = 29); P. kennedyi (Eigenmann 1903) (SL = 2.3–4.0 cm; n = 38); S. notomelas (Eigenmann 1915) (SL = 2.0–2.6 cm; n = 31); Serrapinnus sp. (SL = 1.3–2.1 cm; n = 33). The percentage of empty stomachs was low, ranging between 1.4 % in M. forestii and 7.6 % in P. kennedyi, while S. notomelas and Serrapinnus sp. had all stomachs with food. For diet analysis, food items were identified and quantified by the volumetric method (Hyslop 1980), using a glass counting plate (Hellawell and Abel 1971).
To test whether the species diet composition differed between periods (September and December), we applied a permutational multivariate analysis of variance (PERMANOVA), using Bray–Curtis dissimilarity of log-transformed data matrix containing the volumes of food items. Type III sums of squares were used to account for unbalanced statistical design. We used 9999 permutations to estimate the significance of pseudo-F statistics derived from PERMANOVA. Analyses testing differences between groups of samples (periods) in the multivariate space, as PERMANOVA, are sensitive to differences in the dispersion of samples (individuals) between groups (Anderson 2005). Thus, when the results of PERMANOVA showed significant differences in the diet of species between the periods, we used a permutational analysis of multivariate dispersions (PERMDISP; Anderson 2004) on the basis of the distance of samples (individuals) from the mean of the group (period). This analysis indicates the variability of diet between individuals of the same species in each period and evidences significant differences between periods. A permutational ANOVA was run to determine which species differ in relation to individual diet variability. The F statistics of this analysis was tested by Monte Carlo method using 999 randomizations. All multivariate analyses were run in R Programming Environment using the Vegan package (The R Project for Statistical Computing, http://www.r-project.org/).
Diet overlap between species was calculated using Schoener Index: \(S = 1 - 0.5 \, \left( {\sum\nolimits_{i = 1}^{n} {\left| {P_{xi} - P_{yi} } \right|} } \right),\) where P xi and P yi are the proportions (volume) of food item i used by species x and y, and n is the total number of food items. Overlap values range from 0 to 1; values >0.6 are considered biologically significant (Zaret and Rand 1971). These analyses were run in EcoSim version 6.18 (Gotelli and Entsminger 2004).
Results
There were 14 food items identified in the diets of studied species: nine of aquatic origin and five of terrestrial origin. The diet of H. eques, S. notomelas and Serrapinnus sp. was composed solely of aquatic resources regardless of the period (Fig. 2). Aphyocharax anisitsi consumed only aquatic resources in the dry period, and in the rainy period, 20 % of the diet consisted of terrestrial resources. For M. forestii, aquatic resources were the most consumed, but those of terrestrial origin accounted for 30 % of the diet in the dry period and 20 % in the rainy period. Similar proportions of aquatic and terrestrial resources were consumed by P. kennedyi both in dry and rainy periods (Fig. 2e).
Diet composition (aquatic and terrestrial resources) of fish species inhabiting the littoral zone of Traíra Lake, in the dry and rainy periods. a Aphyocharax anisitsi; b Hyphessobrycon eques; c Moenkhausia forestii; d Psellogrammus kennedyi; e Serrapinnus notomelas; f Serrapinnus sp.; Aq = aquatic resources; Te = terrestrial resources
Aphyocharax anisitsi mainly consumed aquatic invertebrates, but there was significant difference in diet between periods (Fig. 3a; Table 1). In the dry period, various items were important in the diet (Copepoda, represented exclusively by Cyclopoida, Chironomidae, Hemiptera and Ephemeroptera), while in the rainy period, Copepoda, predominantly Calanoida, was the dominant item, followed by Araneae. The results of PERMDISP showed that the variation in diet between individuals was relatively high in both periods, but there was no significant difference between periods (Fig. 4a; Table 2). Diet composition of H. eques was significantly different between periods, with dominance of Chironomidae larvae in the dry period and Copepoda (predominantly Calanoida) in the rainy period (Fig. 3b; Table 1). The results of PERMDISP evidenced significant differences in interindividual variability of H. eques diet between periods, higher in the dry period (Fig. 4b; Table 2).
Diet composition of fish species inhabiting the littoral zone of Traíra Lake, in the dry (D) and rainy (R) periods. a Aphyocharax anisitsi; b Hyphessobrycon eques; c Moenkhausia forestii; d Psellogrammus kennedyi; e Serrapinnus notomelas; f Serrapinnus sp.; Cp = Copepoda; Cl = Cladocera; Ch = Chironomidae; Ep = Ephemeroptera; He = Hemiptera; Ap = aquatic plants; Ar = Araneae; Di = others Diptera; Hy = Hymenoptera; Or = Orthoptera; Co = Coleoptera; Tp = terrestrial plants; Te = Testaceae; Ro = Rotifera; Al = algae
Ordination of the diet of individuals (using the first two axes of PCoA) of Aphyocharax anisitsi (a, PCoA explanation = 53 %); Hyphessobrycon eques (b, PCoA explanation = 65 %); Moenkhausia forestii (c, PCoA explanation = 43 %); Psellogrammus kennedyi (d, PCoA explanation = 58 %); Serrapinnus notomelas (e, PCoA explanation = 54 %); Serrapinnus sp. (f, PCoA explanation = 57 %) in the dry (D) and rainy (R) periods
The diet of M. forestii was composed predominantly of invertebrates (volume > 60 %), followed by aquatic plant in both periods, but with significant seasonal difference in diet (Fig. 3c; Table 1). Ephemeroptera, Hemiptera and Coleoptera were consumed exclusively in the dry period, while Cladocera and Orthoptera in the rainy period. There was an increase in the consumption of Chironomidae and a decrease in the consumption of Araneae in the rainy period. The diet of individuals varied in both periods, but PERMDISP detected no significant seasonal variation (Fig. 4c; Table 2). Psellogrammus kennedyi basically consumed plants in the dry and rainy periods, but the diet varied significantly between periods. In the dry period, aquatic and terrestrial plants had similar percentages, and in the rainy period, terrestrial plants made up 49.0 % of the diet and aquatic plants, 20.1 % (Fig. 3d; Table 1). Differences were also found in the consumption of Chironomidae, which represented 3.0 % in the dry and 21.6 % in the rainy period. Diet variability among individuals was not significantly different between dry and rainy periods (Fig. 4d; Table 2).
The diet of S. notomelas consisted of algae, aquatic plants and invertebrates, with significant difference between periods as a result of high and exclusive consumption of Ephemeroptera in the rainy period, and Rotifera in the dry period (Fig. 3e; Table 1). The variability in the diet among individuals was not significantly different between periods (Fig. 4e; Table 2). The diet of Serrapinnus sp. varied significantly between periods (Fig. 3f; Table 1), although algae have been the most consumed item in the dry and rainy periods; Rotifera, Cladocera and Chironomidae were more consumed in the rainy period, and aquatic plants in the dry period. Serrapinnus sp. had the diet with the lowest interindividual variability in both periods, with no significant seasonal difference (Fig. 4f; Table 2).
Diet overlap values were low in both periods, but were even lower in the rainy period (Fig. 5). Of the 15 pairs of species analyzed, 10 exhibited lower overlap values in the rainy period. Values greater than 0.6 were observed between S. notomelas and Serrapinnus sp. in the dry period, and between A. anisitsi and H. eques in the rainy period.
Discussion
The resources most consumed by fish species in the littoral zone of Traíra Lake were aquatic invertebrates, aquatic higher plant and filamentous algae. Seasonality significantly affected the diet composition of all species, indicating the consumption of different food items and/or different proportions of these resources between periods. Nevertheless, the species maintained the same feeding habits in both periods: A. anisitsi and H. eques were invertivorous, M. forestii was omnivorous, P. kennedyi and S. notomelas were herbivorous, and Serrapinnus sp. was algivorous. These results are explained by differences in the abundance of resources between dry and rainy periods, and by feeding habits of each species (Esteves and Galetti 1995; Russo and Hahn 2006; Neiff et al. 2009; Pereira et al. 2011; Carniatto et al. 2012). On the other hand, except for H. eques, there were no significant differences in interindividual variation in diet between periods. These results indicate that although the period affects the availability of food resources for fish (Takeda and Fujita 2004; Colares et al. 2013), the species showed the same pattern of interindividual variation in the use of resources. Thus, seasonal differences are exclusively associated with changes in diet composition of the population between periods. Only H. eques showed significant interindividual variation between periods; this variation was lower in the rainy period when occurred high consumption of Copepoda by most individuals.
Aphyocharax anisitsi and H. eques, which consumed predominantly aquatic invertebrates in both periods, showed pronounced seasonal changes in the diet. The higher consumption of microcrustaceans in the rainy period by these two fish species has been recorded in several lentic environments (Casatti et al. 2003; Russo and Hahn 2006). The remarkable increase in the consumption of Calanoida by A. anisitsi and by H. eques is a strong indication of the increased abundance of these microcrustaceans in the lake during the rainy period. Colares et al. (2013) studied macrophyte stands in this same floodplain and reported greater abundance of microcrustaceans in the rainy period. The temporal availability of this resource may be responsible for the shift in the diet of these species, but the decrease in the consumption of Chironomidae cannot be attributed to a reduced abundance in the environment, because M. forestii and P. kennedyi increased the consumption of this resource in the rainy period. In addition, Chironomidae larvae are among the most abundant insects in floodplain ecosystems (Takeda et al. 2003; Pinha et al. 2013; Ragonha et al. 2013), where they are consumed by fish throughout the year (Mérona and Rankin-de-Mérona 2004; Fugi et al. 2007; Novakowski et al. 2008). These results suggest that A. anisitsi and H. eques have a preference for Calanoida copepods; thus, under conditions of high availability of these microcrustaceans, the consumption of Chironomidae decreases.
In relation to S. notomelas, algae and aquatic higher plants comprised the majority of its diet, which is probably facilitated by the fact that this species preferentially occupy macrophyte stands (Casatti et al. 2003; Pelicice et al. 2005; Dibble and Pelicice 2010; Carniatto et al. 2012), whose submerged portions sustain a high abundance of filamentous algae (Biolo and Rodrigues 2013), which should also occur with Serrapinnus sp. The feeding habits of S. notomelas has been described as essentially algivorous (Casatti et al. 2003; Pelicice and Agostinho 2006; Hahn and Loureiro Crippa 2006). Seasonal differences for both species were mainly associated with the increase in Ephemeroptera in the diet of S. notomelas and a slight increase in the consumption of Cladocera, Rotifera and Diptera by Serrapinnus sp. in the rainy period. In this way, Melo et al. (2004) reported that seasonal changes (dry and wet periods) influence the temporal variation of Ephemeroptera nymphs, as some species migrate to macrophyte roots in the high water period, which thus explains the exclusive and high consumption of this invertebrate by S. notomelas in the rainy period. Seasonal variations in diet of S. notomelas seem to be a consistent pattern related to variation in hydroperiod, because another study on this species also showed high consumption of algae in the dry period and of Cladocera in the rainy period (Carniatto et al. 2012).
Although seasonality has a strong effect on the availability of food for fish (Neiff et al. 2009; Novakowski et al. 2008; Tófoli et al. 2010; Walker et al. 2013), other factors including feeding habits and strategy of each species may have influenced the use of resources in each period. Aphyocharax anisitsi and H. eques, considered invertivorous (Loureiro Crippa et al. 2009; Corrêa et al. 2009; Carniatto et al. 2014), consumed aquatic invertebrates independent of the period, while P. kennedyi consumed higher plants with the increase in terrestrial plants in the rainy period, suggesting that the ability to consume certain prey plays a key role in the use of resources. For example, Calanoida copepods have a well-developed ability to escape from predators (Nassal et al. 1998), which requires that the predator is active and able to capture them, which may be the case of A. anisitsi and H. eques that exploited this resource when it was theoretically more abundant. On the other hand, consumption of plants does not require very active fish, which may be associated with P. kennedyi that consumed more terrestrial plant in the rainy period, when these resources are carried into the aquatic environment and become available and easily consumed by fish (Novakowski et al. 2008; Pereira et al. 2011).
Diet overlap values for the species studied were low in both periods; however, the trophic segregation was even greater in the rainy period, indicating that with greater abundance of food resources, species could exploit the preferential resources. Low overlap values have been found for fish assemblages in lentic environments, both in the rainy and dry periods, indicating tendencies of trophic specialization (Mérona and Rankin-de-Mérona 2004; Novakowski et al. 2008). Nevertheless, some studies have shown trends of higher diet overlap among fish species in conditions of greater food availability (Zaret and Rand 1971; Delbeek and Williams 1987). Trophic segregation is considered a key factor for coexistence of fish species (Schoener 1974; Ross 1986), which may explain the low diet overlap in both periods, preventing interspecific competition. Furthermore, temporal fluctuations in the availability of food resources influence the partitioning of resources among Neotropical fish (Jepsen et al. 1997). In this study, an example was the consumption of Chironomidae by various species but in different periods; in the dry period, these insects were used most by A. anisitsi and H. eques, in the rainy period, by M. forestii and P. kennedyi, which can minimize interspecific competition. In addition, other resources, such as Calanoida copepods, seem to be more available in the rainy period, and the species able to consume them might take advantage of the high seasonal abundance.
Finally, the results showed that seasonality influenced diet composition of all species studied, with the largest changes observed in the proportion of items consumed between periods, while the feeding habits of the species did not change, indicating well-defined diets. The trophic segregation, evidenced by the low overlap values in both periods, indicates the absence of interspecific competition, which may explain the coexistence of these species in the littoral area of this small lake.
References
Abujanra F, Agostinho AA, Hahn NS (2009) Effects of the flood regime on the body condition of fish of different trophic guilds in the Upper Paraná River flooding, Brazil. Braz J Biol 69:469–479. doi:10.1590/S1519-69842009000300004
Agostinho AA, Gomes LC, Thomaz SM, Hahn NS (2004) The upper Paraná River and its floodplain: main characteristics and perspectives for management and conservation. In: Thomaz SM, Agostinho AA, Hahn NS (eds) The upper Paraná River and its floodplain: physical aspects, ecology and conservation. Backhuys Publishers, Leiden, pp 381–393
Agostinho AA, Thomaz SM, Gomes LC, Baltar SLSMA (2007) Influence of the macrophytes Eichhornia azurea on fish assemblage of the upper Paraná River floodplain (Brazil). Aquat Ecol 41:611–619. doi:10.1007/s10452-007-9122-2
Agostinho AA, Pelicice FM, Marques EE (2009) Reservatório de Peixe Angical: bases ecológicas para o manejo da ictiofauna. RiMa, São Carlos
Anderson MJ (2004) PERMDISP: a FORTRAN computer program for permutational analysis of multivariate dispersions (for any two-factor ANOVA design) using permutation tests. Department of Statistics, University of Auckland
Anderson MJ (2005) PERMANOVA: a FORTRAN computer program for permutational multivariate analysis of variance. Department of Statistics, University of Auckland
Bickel TO, Closs GP (2008) Fish distribution and diet in relation to the invasive macrophyte Lagarosiphon major in the littoral zone of Lake Dunstan, New Zealand. Ecol Freshw Fish 17:10–19. doi:10.1111/j.1600-0633.2007.00249.x
Biolo S, Rodrigues L (2013) Comparison of the structure of the periphytic community in distinct substrates from a Neotropical floodplain. Int Res J Plant Sci 4:64–75
Carniatto N, Fugi R, Cantanhêde G, Gubiani EA, Hahn NS (2012) Effects of flooding regime and diel cycle on diet of a small sized fish associated to macrophytes. Acta Limnol Bras 24:363–372. doi:10.1590/S2179-975X2013005000007
Carniatto N, Fugi R, Thomaz SM, Cunha ER (2014) The invasive submerged macrophyte Hydrilla verticillata as a foraging habitat for small-sized fish. Nat Conservação 12:30–35. doi:10.4322/natcon.2014.006
Casatti L, Mendes HF, Ferreira KM (2003) Aquatic macrophytes as feeding site for small fishes in the Rosana Reservoir, Paranapanema River, southeastern Brazil. Braz J Biol 63:213–222. doi:10.1590/S1519-69842003000200006
Colares MAM, Bonecker CC, Simões NR, Alves GM, Lansac-Tôha FA (2013) Structure of the zooplankton communities in macrophytes stand of a Neotropical floodplain (the Paraná River, Brazil). Int Rev Hydrobiol 98:89–103. doi:10.1002/iroh.20130147
Colon-Gaud JC, Kelso WE, Rutherford DA (2004) Spatial distribution of macroinvertebrates inhabiting Hydrilla and coontail beds in the Atchafalaya Basin, Louisiana. J Aquat Plant Manag 42:85–91
Corrêa CE, Hahn NS, Delariva RL (2009) Extreme segregation between sympatric fish species: the case of small sized body Aphyocharax in the Brazilian Pantanal. Hydrobiologia 635:57–65. doi:10.1007/s10750-009s9861-2
Corrêa CE, Albrecht MP, Hahn NS (2011) Patterns of niche breadth and feeding overlap of the fish fauna in the seasonal Brazilian Pantanal, Cuiabá River basin. Neotrop Ichthyol 9:237–646. doi:10.1590/S1679-62252011000300017
Cunha ER, Thomaz SM, Evangelista HBA, Carniato J, Souza CF, Fugi R (2011) Small-sized fish assemblages do not differ between a native and a recently established non-indigenous macrophyte in a Neotropical ecosystem. Nat Conservação 9:61–66. doi:10.4322/natcon.2011.007
Delariva RL, Agostinho AA, Nakatani K, Baumgartner G (1994) Ichthyofauna associated to aquatic macrophytes in the Upper Parana River floodplain. Revista UNIMAR 16:41–60
Delbeek JC, Williams DD (1987) Food resource partitioning between sympatric populations of brackishwater stickleback. J Anim Ecol 56:949–967. doi:10.2307/4959
Dibble ED, Pelicice FM (2010) Influence of aquatic plant-specific habitat on an assemblage of small Neotropical floodplain fishes. Ecol Freshw Fish 19:381–389. doi:10.1111/j.1600-0633.2010.00420.x
Dibble ED, Thomaz SM (2009) Use of fractal dimension to assess habitat complexity and its influence on dominant invertebrates inhabiting tropical and temperate macrophytes. J Freshw Ecol 24:93–102. doi:10.1080/02705060.2009.9664269
Dibble ED, Killgore KJ, Harrel SL (1996) Assessment of fish–plant interactions. Am Fish Soc Symp 16:357–372
Esteves KE, Galetti PM Jr (1995) Food partitioning among five characids of a small Brazilian floodplain lake from the Paraná River basin. Environ Biol Fish 42:375–389. doi:10.1007/BF00001468
Fugi R, Hahn NS, Novakowski GC, Balassa GC (2007) Ecologia alimentar da corvina, Pachyurus bonariensis (Perciformes, Sciaenidae) em duas baías do pantanal, Mato Grosso, Brasil. Iheringia 97:343–347. doi:10.1590/S0073-47212007000300020
Gerking SD (1994) Feeding ecology of fish. Academic Press, San Diego
Gomes LC, Bulla CK, Agostinho AA, Vasconcelos SP, Miranda LE (2012) Fish assemblage dynamics in a Neotropical floodplain relative to aquatic macrophytes and the homogenizing effect of a flood pulse. Hydrobiologia 685:97–107. doi:10.1007/s10750-011-0870-6
Gotelli NJ, Entsminger GL (2004) EcoSim: Null models software for ecology. Version 7. Acquired Intelligence Inc. & Kesey-Bear. Jericho, VT 05465, Available at: http://www.garyentsminger.com/ecosim/index.htm. Accessed 5 Aug 12
Goulding M (1980) The fishes and the forest: exploration in Amazonian natural history. University of California Press, Los Angeles
Hahn NS, Loureiro Crippa VE (2006) Estudo comparativo da dieta, hábitos alimentares e morfologia trófica de duas espécies simpátricas, de peixes de pequeno porte, associados à macrófitas aquáticas. Acta Scientiarum 28:359–364. doi:10.4025/actascibiolsci.167
Hellawell JM, Abel R (1971) A rapid volumetric method for the analysis of the food of fishes. J Fish Biol 3:29–37. doi:10.1111/j.1095-8649.1971.tb05903.x
Hyslop EJ (1980) Stomach content analysis: a review of methods and their applications. J Fish Biol 17:411–429. doi:10.1111/j.1095-8649.1980.tb02775.x
Jepsen TM, Winemiller KO, Taphorn DC (1997) Temporal patterns of resource partitioning among Cichla species in a Venezuelan blackwater. J Fish Biol 51:1085–1108. doi:10.1111/j.1095-8649.1997.tb01129.x
Junk WJ, Bayley PB, Sparks RE (1989) The flood pulse concept in river-floodplain systems. Can J Fish Aquat Sci 106:110–127
Lansac-tôha FA, Bonecker CC, Velho LFM (2004) Composition, species richness and abundance of zooplankton community. In: Thomaz SM, Agostinho AA, Hahn NS (eds) The Upper Paraná River and its floodplain: physical aspects, ecology and conservation. Backhuys Publishers, Leiden, pp 145–190
Loureiro Crippa VE, Hahn NS, Fugi R (2009) Food resource used by small-sized fish in macrophyte patches in ponds of the upper Paraná River floodplain. Acta Scientiarum 31:119–125. doi:10.4025/actascibiolsci.v31i2.3266
Melo SM, Takeda AM, Monkolski A (2002) Seasonal dynamics Callibaetis willineri (Ephemeroptera, Baetidae) associated with Eichhornia azurea (Pontederiaceae) in Guaraná Lake of the Upper Paraná River, Brazil. Hydrobiologia 470:57–62. doi:10.1023/A:1015617102463
Melo SM, Takeda AM, Gryzbkowska M, Monkolski A (2004) Distribution of ephemeropteran nymphs associated with different stolon sections of Eichhornia azurea (Schwartz) in two floodplain lakes of the Upper Paraná River (Brazil). Pol J Ecol 52:369–376
Mérona B, Rankin-de-Mérona J (2004) Food resource partitioning in a fish community of the central Amazon floodplain. Neotrop Ichthyol 2:75–84. doi:10.1590/S1679-62252004000200004
Meschiatti AJ, Arcifa MS, Fenerich-Verani N (2000) Fish communities associated with macrophytes in Brazilian floodplain lakes. Environ Biol Fish 58:133–143. doi:10.1023/A:1007637631663
Mormul RP, Thomaz SM, Higuti J, Martens K (2010) Ostracod (Crustacea) colonization of a native and a non-native macrophyte species of Hydrocharitaceae in the Upper Paraná floodplain (Brazil): an experimental evaluation. Hydrobiologia 644:185–193. doi:10.1007/s10750-010-0112-3
Murakami EA, Bicudo DC, Rodrigues L (2009) Periphytic algae of the Garças Lake, Upper Paraná River floodplain: comparing the years 1994 and 2004. Braz J Biol 69:459–468. doi:10.1590/S1519-69842009000300002
Nassal B, Burghard W, Maier G (1998) Predation by juvenile roach on the calanoid copepod Eudiaptomus gracilis and the cyclopoid copepod Cyclops vicinus: a laboratory investigation with mixed and single prey. Aquat Ecol 32:335–340. doi:10.1023/A:1009995432725
Neiff JJ, Neiff AP, Verón MBC (2009) The role of vegetated areas on fish assemblage of the Paraná River floodplain: effects of different hydrological conditions. Neotrop Ichthyol 7:39–48. doi:10.1590/S1679-62252009000100006
Novakowski GC, Hahn NS, Fugi R (2008) Diet seasonality and food overlap of the fish assemblage in a pantanal pond. Neotrop Ichthyol 6:567–576. doi:10.1590/S1679-62252008000400004
Pelicice FM, Agostinho AA (2006) Feeding ecology of fishes associated with Egeria spp. patches in a tropical reservoir, Brazil. Ecol Freshw Fish 15:10–19. doi:10.1111/j.1600-0633.2005.00121.x
Pelicice FM, Agostinho AA, Thomaz SM (2005) Fish assemblages associated with Egeria in a tropical reservoir: investigating the effects of plant biomass and diel period. Acta Oecol 27:9–16. doi:10.1016/j.actao.2004.08.004
Pereira JO, Silva MT, Vieira LJS, Fugi R (2011) Effects of flood regime on the diet of Triportheus curtus (Garman, 1890) in an Amazonian floodplain lake. Neotrop Ichthyol 9:623–628. doi:10.1590/S1679-62252011005000029
Pinha GD, Alessio PC, Gurski FA, Sacramento PA, Pezentini TA, Takeda AM (2013) Spatial distribution of the assemblage of Chironomidae larvae (Diptera) in five floodplain lakes from Ilha Grande National Park (Paraná—Mato Grosso do Sul State, Brazil). Acta Scientiarum 35:169–177. doi:10.4025/actascibiolsci.v35i2.10799
Prejs A, Prejs K (1987) Feeding of tropical freshwater fishes: seasonality in resource availability and resource use. Oecologia 71:397–404. doi:10.1007/BF00378713
Priyadarshana T, Asaeda T, Manatunge J (2001) Foraging behaviour of planktivorous fish in artificial vegetation: the effects on swimming and feeding. Hydrobiologia 442:231–239. doi:10.1023/A:1017578524578
Ragonha FH, Pinha GD, Bilia CG, Silva RG, Tramonte RP, Takeda AM (2013) The influence of shoreline availability on the density and richness of Chironomid larvae in Neotropical floodplain lakes. Bioikos 27:67–77
Ross ST (1986) Resource partitioning in fish assemblages: a review of field studies. Copeia 1986:352–388
Russo MR, Hahn NS (2006) Importance of zooplankton in the diet of a small fish in lagoons of the upper Paraná River floodplain, Brazil. Acta Limnol Bras 18:357–366
Schoener TW (1974) Resource partitioning in ecological communities. Science 185:27–39
Takeda AM, Fujita DS (2004) Benthic invertebrates. In: Thomaz SM, Agostinho AA, Hahn NS (eds) The Upper Paraná River and its floodplain: physical aspects, ecology and conservation. Blackhuys Publishers, The Netherlands, pp 191–208
Takeda AM, Souza-Franco GM, Melo SM, Monkolski A (2003) Invertebrados associados às macrófitas aquáticas da planície de inundação do Alto Rio Paraná (Brasil). In: Thomaz SM, Bini LM (eds) Ecologia e manejo de macrófitas aquáticas. Eduem, Maringá, pp 243–260
Thomaz SM, Bini LM, Bozelli RL (2007) Floods increase similarity among aquatic habitats in river-floodplain systems. Hydrobiologia 579:1–13. doi:10.1007/s10750-006-0285-y
Thomaz SM, Dibble ED, Evangelista LR, Higuti J, Bini LM (2008) Influence of aquatic macrophyte habitat complexity on invertebrate abundance and richness in tropical lagoons. Freshw Biol 53:358–367. doi:10.1111/j.1365-2427.2007.01898.x
Tófoli RM, Hahn NS, Alves GHZ, Novakowski GC (2010) Uso do alimento por duas espécies simpátricas de Moenkhausia (Characiformes, characidae) em um riacho da Região Centro-Oeste do Brasil. Iheringia 100:201–206. doi:10.1590/S0073-47212010000300003
Walker RH, Kluender ER, Inebnit TE, Adams SR (2013) Differences in diet and feeding ecology of similar-sized spotted (Lepisosteus oculatus) and shortnose (Lepisosteus platostomus) gars during flooding of a south-eastern US river. Ecol Freshw Fish 22:617–625. doi:10.1111/eff.12066
Zaret TM, Rand AS (1971) Competition in tropical stream fishes: support for the competitive exclusion principle. Ecology 52:336–342. doi:10.2307/1934593
Acknowledgments
We thank S.M. Thomaz (Universidade Estadual de Maringá) who greatly improved this manuscript. B.A. Quirino and N. Carniatto acknowledge the Brazilian Council of Research (CNPq) for providing grants. This study was supported by CNPq, a Brazilian organization focused on the development of human resources.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Thomas Mehner.
Rights and permissions
About this article
Cite this article
Quirino, B.A., Carniatto, N., Gaiotto, J.V. et al. Seasonal variation in the use of food resources by small fishes inhabiting the littoral zone in a Neotropical floodplain lake. Aquat Ecol 49, 431–440 (2015). https://doi.org/10.1007/s10452-015-9535-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-015-9535-2