Abstract
Differences among species and their ecological requirements are considered fundamental in determining the outcome of species interactions as well as in coexistence. Thus, species that co-occurs tends to differ in the use of resources as a way to mitigate the effects of interspecific competition, facilitating interactions between pairs of species. So, this study used a set of seven small-sized characid species with similar morphology and feeding strategies, in order to investigate the hypothesis that the coexistence these species is facilitated by the differential use of food resources. Samplings were conducted in the rivers Verde and São Domingos, Upper Paraná River basin, Brazil, in hydrological periods rainy and dry. The analysis of 1055 stomach contents, by the volumetric method, indicated that the species consumed mainly allochthonous items, such as seeds, terrestrial plants and insects. In addition, they showed inter- and intraspecific differences in the diet composition between hydrological periods, which allowed the identification of items that particularise each species and contribute to the trophic segregation between them. Despite the wide variety of food items used, it was not possible to observe a consistent pattern of widening or narrowing of the food spectrum between hydrological periods, as expected. The trophic niche overlap showed intermediate and low values in both periods. In this sense, resource partitioning among species of small characids, facilitated by exploitation of different preferential resources as well as the intraspecific variation in response to seasonal availability of resources, became evident. The alternation of items and proportions of items in the diet as well as changes in feeding behaviour in opportune moments was probably the key for the coexistence of these species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Differences between species in their niches, either their requirements, impacts or both, are considered a key pillar for the niche theory (Chase and Leibold 2003). In this aspect, the competition for food resources has been postulated as an important driving factor in the evolution and diversification of species (Hutchinson 1957; MacArthur 1958). In contrast, species coexisting may differ in the use of resources as a means of mitigating the effects of competition (Pianka 1973; Schoener 1974; Svanbäck and Bolnick 2007; Wiens et al. 2010; Wang et al. 2015). Therefore, resource partitioning mechanisms may arise mainly from the similar ecological requirements and/or facilitation interactions between species pairs (Giacomini 2007; McCreadie and Bedwell 2013; Robertson et al. 2014). In this sense, understanding the processes that govern the partition of resources between morphologically similar and coexisting organisms is essential to the foundation of biological studies as well as conservation actions and management of ecosystems (McCreadie and Bedwell 2013; Mise et al. 2013; Swanson et al. 2015; Silva et al. 2016b). In particular, understanding the trophic niche dimension provides important insights into the coexistence of different species in the same community (Stevens 1989; Ward et al. 2006; Wiens et al. 2010; Sá-Oliveira and Isaac 2013; Quirino et al. 2015; Wang et al. 2015; Mateus et al. 2016).
In this sense, to understand the coexistence of species in the same location, the optimal foraging theory (OFT; Stephens and Krebs 1986) suggests that competition leads the species to change their foraging behaviour, broadening or narrowing the diet, depending on biotic conditions, such as competition (Punchard et al. 2000; Gotelli and McCabe 2002; Ward et al. 2006; Araújo et al. 2011) and predation (Englund et al. 2009; Fernandes et al. 2009) or environmental conditions (Peres-Neto 2004; Mouchet et al. 2013). Thus, the level of trophic segregation between sympatric species may vary considerably over time, reflecting food flexibility and seasonal abundance of prey (Roughgarden 1972; Abelha et al. 2001; Silva et al. 2014).
Tropical lotic environments show a pronounced seasonality resulting from the rainfall regime and fluctuations in water level, which cause cyclical changes in river characteristics and affect all the trophic dynamics of ecosystems (Lowe-Mcconnel 1999; Abujanra et al. 2009; Dodds et al. 2015). As a result, these seasonal changes in the hydrological cycle alter the abundance and availability of food resources for fish (Correa and Winemiller 2014; Silva et al. 2014). Thus, the OFT predicts that under abundance of preferred food items, consumers tend to narrow their diets (Stephens and Krebs 1986). On the other hand, during the food shortage period, the generalist feeding activity may be favoured, with expansion of the trophic niche (Roughgarden 1972; Robinson and Wilson 1998). In this way, temporal diet analysis of the species allows strong inferences on the mechanisms underlying the patterns of organisation and coexistence in fish communities (Ward et al. 2006; Mouchet et al. 2013).
Fish species, especially small-sized species belonging to the family Characidae, are excellent models to assess coexistence patterns. This group is very heterogeneous from a phylogenetic point of view and contributes significantly to the composition of the fish fauna of neotropical rivers, where it is widely distributed (Reis et al. 2003; Langeani et al. 2007; Mirande 2009; Albert and Reis 2011). In lotic environments, small fish preferably inhabit marginal habitats, where they find shelter from predators and favourable conditions for feeding (Lowe-McConnell 1987; Thomaz et al. 2004; Hahn and Fugi 2008). In this context, a growing amount of literature has revealed that small fish species have a high diversity of dietary habits and tactics and can explore the most different tropical environments, such as streams (Casatti et al. 2001; Wolff et al. 2009; Uieda and Pinto 2011; Silva et al. 2012), rivers (Viana et al. 2006; Corrêa et al. 2009) and lakes (Petry et al. 2003; Loureiro-Crippa et al. 2009; Carniatto et al. 2012; Quirino et al. 2015).
Thus, considering that feeding is an essential aspect of the life cycle of an organism (Sih and Christensen 2001), identification and quantification of diet composition are crucial steps to understand the role and interactions of each species in shared environments. For community ecologists, the acquisition of food by forager species plays a key role in determining the dynamics of competition between species (Pianka 1981; Mittelbach and Osenberg 1994), predator–prey interactions (Sih et al. 1985) and indirect interactions with the community (Wilbur and Fauth 1990; Wootton 1992).
In addition, understanding the mechanisms that allow the coexistence of many species with similar ecological requirements in neotropical rivers is still a major challenge in ecological studies (Sá-Oliveira et al. 2014). Therefore, assessing the small fish fauna from the trophic niche perspective is promising. Hence, in this study, we used a set of seven sympatric species of small characins with similar morphology and feeding strategies (Casatti et al. 2001; Mirande 2009; Romero and Casatti 2012), inhabiting two seasonal aquatic lotic environments, the Verde and São Domingos rivers, upper Paraná river Basin, Brazil, to test two general predictions. First, we expected to find differentiation in diet composition between these species, supporting the resource partitioning hypothesis. Secondly, we expected to find a narrow diet breadth during rainy periods. In this case, we assumed one of the premises of optimal foraging theory, which states that individuals would tend to focus their diets on more favourable resources in periods of greater abundance of resources. These predictions are closely related, directly reflecting the degree of overlap of food resources.
Materials and methods
Study area
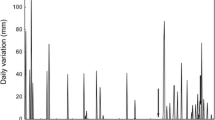
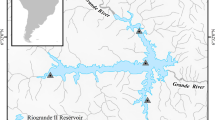
Sampling was carried out in the Verde River, located in the state of Mato Grosso do Sul, which stands out as an important tributary on the right margin of the upper Paraná River and has its mouth in the reservoir of the hydroelectric power plant Sérgio Motta (Porto Primavera), the state of São Paulo and in its main tributary, the São Domingos River, located on its left margin. The study area (20°40′30.61″S, 53°34′4.91″W) is located in the Brazilian Cerrado, which is characterised by plant species with flowers and fruits influenced by seasonal variations in precipitation and humidity, where there is usually loss of leaves in the dry season and seed dispersal at the beginning of the rainy season (Lenza and Klink 2006; Salazar et al. 2012). The climate and hydrological regime (Fig. 1) are characterised by two distinct seasons: dry winters (April to September) and rainy summers (October to March) (Ribeiro et al. 1998; Pagotto and Souza 2006).
Sampling
Six sampling sites were established, distributed in the Verde River (three sites) and São Domingos River (three sites) (Fig. 2). Fish sampling was performed in the rainy period (December 2010 and January 2011 and December 2011 and January 2012) and in the dry period (from May and August 2011 and 2012), totalling 8 months of collecting. We used the following fishing devices: trawls, cast nets and gillnets with simple mesh sizes of 2.4, 3, 4, 5, 6, 7, 8, 9, 10, 12, 14 and 16 cm and trammel nets (locally known as feiticeiras) with mesh sizes of 6, 7 and 8 cm between non-adjacent knots, with 1.5 m in height and 20 m in length in the Verde River and with 10 m in length in the tributary. After capturing the fish, we anesthetised them with a benzocaine solution (250 mg/l) following the recommendations of the American Veterinary Medical Association (Avma 2001), fixed them in plastic bags containing 10% formaldehyde and placed them in polyethylene containers. In the laboratory, we identified the fish following Graça and Pavanelli (2007), measured (total and standard length in cm) and weighed them (g). Specimens of each species were preserved in 70% alcohol and deposited them in the ichthyological collection of the Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (Nupélia), Universidade Estadual de Maringá.
Diet analysis
Only small-sized species (those in which the adults have a standard length <15 cm), as proposed by Castro et al. (2003) and with the number of stomachs >10 were used in this study. The stomachs analysed were those with a degree of repletion >50% of fullness (for scale, see Zavala-Camin 1996). Stomach contents were examined using an optical microscope and a stereoscope, and the items were quantified according to the volumetric method (Hyslop 1980). The food items were identified using Bicudo and Bicudo (1970) for algae and Mugnai et al. (2010) for invertebrates; When necessary, other specific literatures were used. We also used graduated tubes and a glass counting plate to measure the volume of the items (Hellawell and Abel 1971). The food items were classified according to their origin in autochthonous (originating from the aquatic environment) and allochthonous (originating from the terrestrial environment).
Data analysis
Diet composition
To test possible significant differences in the dietary composition of the species among the pre-defined groups (species and hydrological periods), PERMANOVA multivariate permutational variance analysis was used through the Bray-Curtis index obtained with 999 random permutations (Anderson 2001). To evaluate the food items that contributed most to the intraspecific dissimilarity between the hydrological periods, we performed a similarity percentage analysis (SIMPER), using Bray-Curtis dissimilarity (Clarke 1993).
Niche breadth
The test of homogeneity the multivariate dispersion (PERMDISP, Anderson et al. 2008) was used to assess differences in intra- and interspecific niche breadth, between hydrological periods. In our study, the niche breadth was measured through the dispersion of the diet in space. The assumption was that differences in distance between species indicate that some species have more restricted or broader diets than others. By PERMDISP, the distance of the median (i.e. similar to the centroid) of a group defined a priori is calculated, in this case the species/hydrological periods, through a principal coordinate analysis (PCoA). Calculation of the median of the group was performed using the dissimilarity measure of Bray-Curtis, allowing the comparison of the average dissimilarity in n individual observations within the group. To test the null hypothesis that the niche breadth did not differ among the groups, a statistical F was calculated to compare the average distance of each sample to the median of the group. Subsequently, the p value was obtained through 9999 permutations of the residues of least squares (Anderson 2006). Post hoc pairwise comparisons were made by Tukey’s ‘Honest Significant Difference’ method.
Food overlap
The overlap of the diet was calculated per sample for each species pair that co-occurs in time and space (month and site), based on the volume matrix of the food items. We used the Pianka (1973) food overlap index, described by the equation:
where O jk = measure of the Pianka niche overlap between the j and k species; P ij = proportion of a food item i in the diet of the species j; P ik = proportion of the food item i in the diet of the species k; n = total number of food items. The overlap values ranged from 0 (no overlap) to 1 (full overlay) and were defined at the following levels: low (0–0.39), intermediate (0.4–0.6) and high (0.6–1) (modified from Grossman 1986; Corrêa et al. 2011). A one-way ANOVA was applied to data of trophic niche overlap, considering hydrological period (rainy and dry) as a factor.
The PERMANOVA, SIMPER, PERMDISP, ANOVA and statistical analyses were performed in the program R, using vegan and spaa packages (Zhang 2013; R Development 2015). The Pianka index of food overlap was calculated using EcoSim® 7.0 (Gotelli and Entsminger 2006). The figures were created in software STATISTICA 7.0. The level of statistical significance adopted for all analyses was p < 0.05.
Results
Diet composition
We analysed the stomach contents of 1055 individuals belonging the seven most abundant small-sized fish species (Characidae) (Table 1). The analysed species accounted for 83% of the species recorded (Silva et al. 2016a).
For the set of species, 21 food items were identified. The resources from autochthonous and allochthonous sources were represented by 10 items each, with the most representative allochthonous items in fish diet (Table 2). Significant differences were observed in diet composition between species (PERMANOVA; pseudo-F = 56.76, p < 0.001) and hydrological periods (pseudo-F = 9.88; p < 0.001), as well, for interaction between the factors (pseudo-F = 17.3; p < 0.001).
Astyanax aff. fasciatus mainly consumed seeds, Hymenoptera and terrestrial plants in the rainy season; in the dry season, terrestrial plants and Hymenoptera were predominant prey items. In the rainy season, the diet of Astyanax lacustris was based on seeds, Hymenoptera and terrestrial plants, while in the dry season, in addition to these items, their diet was supplemented by aquatic plant (Table 2). For Piabarchus sp.1, other aquatic invertebrates, Hymenoptera, Isoptera and terrestrial plants, were substantial items in both periods, except Isoptera in the dry season. Piabarchus stramineus ingested substantial amounts of Isoptera and other aquatic invertebrates in the rainy season and Hymenoptera, other aquatic invertebrates, Plecoptera and Odonata nymphs in the dry season. The diet of Moenkhausia aff. intermedia was based on immature Diptera, other aquatic invertebrates, Ephemeroptera and Hymenoptera in the rainy season and Ephemeroptera and immature Diptera in the dry season. In the diet of Piabina argentea, seeds were predominant in both periods. Serrapinus notomelas ingested larger proportions of algae, detritus and terrestrial plants in the rainy season and mostly aquatic plants in the dry season (Table 2).
The food items that most contributed to the intraspecific differentiation between hydrological periods were seeds for A. aff. fasciatus, A. lacustris and P. argentea, other aquatic invertebrates for Piabarchus sp.1 and P. stramineus; Ephemeroptera for M. aff. intermedia; aquatic plants for S. notomelas (Table 3).
Niche breadth
The species consumed a wide variety of items, which was reflected in intraspecific values in breadth diet (Fig. 3). We did not observe a consistent pattern of expansion or contraction of the food spectrum between hydrological periods (Fig. 3). However, PERMIDISP showed significant differences in trophic niche breadth between species pairs (PERMDISP, F = 9.69, p > 0.001).
Variation in diet breadth among seven small-sized fish species based on food items consumed during the hydrological periods rainy (R) and dry (D) in the Verde and São Domingos River, Upper Paraná River Basin, Brazil, from November 2010 to August 2012. Diet breadth was assessed as species dispersion in diet space using PERMDISP (i.e. greater distance to spatial median indicates larger dispersion and therefore a broader trophic niche). Box lower and upper endpoints represent the 25th and 75th quartiles, respectively. The horizontal bar inside each box represents median diet breadth. Species codes are: Af = A. aff. fasciatus, Al = A. lacustris, Mi = M. aff. intermedia, Pa = P. argentea, Pi = Piabarchus sp.1, Ps = P. stramineus, Sn = S. notomelas
The niche overlap showed overall intermediate and low values (<0.6; Fig. 4). However, there were significant seasonal differences (ANOVA = 3.97, p < 0.05), with an increasing trend in overlap in the rainy season, where 55% of the species pairs with niche overlap values above 0.6 were observed. Higher interspecific overlap values were observed for P. argentea × A. lacustris pairs in the rainy season (seeds partition), S. notomelas × A. aff. fasciatus in the dry season (aquatic plants) and S. notomelas × Piabarchus sp.1 (aquatic and terrestrial plants) (Fig. 4).
Diet overlap values (mean ± SE) for species pairs during the hydrological periods rainy (R) and dry (D) in the Verde and São Domingos River, Upper Paraná River Basin, Brazil, from November 2010 to August 2012. Species codes are: Af = A. aff. fasciatus, Al = A. lacustris, Mi = M. aff. intermedia, Pa = P. argentea, Pi = Piabarchus sp.1, Ps = P. stramineus, Sn = S. Notomelas
Discussion
Our results show that small characids have a diverse diet, especially composed of allochthonous items whose abundance can be influenced by hydrological seasonality. Allochthonous resources were mainly represented by these items (seeds, plants and terrestrial insects). According to Lenza and Klink (2006) and Salazar et al. (2012), the studied region presents great seasonality in the supply of leaves and seeds. In freshwater lotic environments, allochthonous resources are considered the main food sources for fish fauna (Nakano and Murakami 2001; Correa et al. 2007; Trevisan and Hepp 2007; Richardson et al. 2010; Marcarelli et al. 2011). This is mainly observed in sites with riparian vegetation as well as deciduous vegetation, where resources are carried to the aquatic environment, resulting in a strong association between the abundance of the allochthonous items and the rainy season (Prejs and Prejs 1987; Nakano and Murakami 2001; Alvim and Peret 2004; Correa et al. 2007; Pinto and Uieda 2007). Due to declining water levels and, consequently, a reduction in foraging areas in the dry period, some allochthonous resources become less abundant. In this scenario, the fish species tend to consume other items, changing their diets to other items available at that particular time (Correa and Winnemiller 2014).
The intraspecific seasonal variation in the diet was demonstrated in our results. The probable variations in the contribution of resources certainly promoted food spectrum changes in species between the hydrological periods. Thus, this allows us to infer that small species examined here have high trophic plasticity, presenting exploratory and diverse feeding tactics, which represents a recurrent pattern found in the literature (Casatti et al. 2001; Cassemiro et al. 2002; Russo et al. 2004; Casatti and Castro 2006; Viana et al. 2006; Ceneviva-Bastos and Casatti 2007; Wolff et al. 2009; Tófoli et al. 2010; Gandini et al. 2012; Manna et al. 2012, Quirino et al. 2017).
Species with similar morphological characteristics have high competitive potential (Gatz 1979), especially when phylogenetically close, due to what is conceptualised as niche conservatism (Wiens et al. 2010; Wang et al. 2015). Thus, as stated earlier, we expected a high similarity in the diet composition of the species analysed in this study. However, what we have shown is that they segregate from each other in the trophic dimension, supporting our hypothesis of differential resource utilisation. According to the intraspecific information of the diet, even if the species consume some items in common, they are ingested in different proportions, as if there were a rotation of consumption of certain items, between the species and between the hydrological periods.
Since food is considered the most important niche dimension in the segregation of fish species (Schoener 1974; Ross 1986; Gerking 1994), sympatric species tend to differ in the use of resources to minimise competition (Wootton 1990). Furthermore, species coexistence depends on different ecological responses to similar ecological processes (Hutchinson 1957). In this way, many species tend to select their diet, sharing food resources (Schoener 1974; Gerking 1994). This mechanism seems to be the key factor for trophic segregation between small fish in the Verde River. This idea becomes even more consistent as the analysed species have very similar morphological characteristics (Casatti et al. 2001; Reis et al. 2003; Romero and Casatti 2012) and are exposed to the same biotic and abiotic conditions.
The niche breadth was different between the hydrological periods, however, and does not corroborate with one assumption of the optimal foraging theory, which predicts widening or narrowing of the niche, depending on environmental conditions. Correa and Winemiller (2014), studying Amazonian fish, also described no consistent pattern in niche breadth in response to temporal changes in the quality and availability of food resources, and they suggested that this response to abundant or limited food resources depends on the taxon and ecosystem analysed. This lack of variation in niche breadth relative to seasonality has been reported for various fish species (Ward et al. 2006; Novakowski et al. 2008; Corrêa et al. 2009, 2011; Tófoli et al. 2010; Alves et al. 2011). However, there is no consensus regarding whether the niche is expanded or contracted in the presence of abundant food (Roughgarden 1972; Dyer et al. 2010; Quirino et al. 2017). By means of intraspecific analysis (PERMDISP), we observed that in general, species analysed here, showed high dispersion of food items in diet, regardless of the hydrological period. This supports the hypothesis that species alternate food items and/or their proportions according to preference or availability, but maintain the variety in the diet. According to Sá-Oliveira et al. (2014), wider trophic niches characterise generalist species. In this study, this statement is validated, since the species analysed are identified as typical generalists in the literature (Bennemann et al. 2005; Silva et al. 2012; Moraes et al. 2013; Mise et al. 2013).
The low diet overlap found between most species pairs endorses and confirms the results of niche breadth, where there is no pattern of expansion or contraction of the niche between sampled hydrological periods, although there was an upward trend in the rainy season when seeds and terrestrial insects were probably more available. The importance of fruits and seeds in the fish diet is strongly associated with their availability, as in virtually all ecosystems, fruits and seeds are patchily distributed and seasonally available (Correa et al. 2007), especially in the region studied (Lenza and Klink 2006; Salazar et al. 2012). Although the availability of food resources has not been directly measured in the environment, we rely on the assumption that fish are good samplers of available resources, directly reflecting the resources they can efficiently access (Mérona et al. 2003; Tupinambás et al. 2015). In this sense, the differential exploitation of food resources in the period in which they are more accessible, in this case, in the rainy season, seems to be the key to explain the low diet overlap between most small species analysed. Nevertheless, although some species pairs (e.g. P. argentea × A. lacustris, S. notomelas × A. aff. fasciatus, S. notomelas × Piabarchus sp.1) had higher overlap, mainly because of the great consumption of more abundant resources (seeds, aquatic and terrestrial plants), the general trend confirmed was the trophic differentiation, translated into food resource partitioning.
Since Hutchinson (1957), the mechanisms that allow similar species to partition resources and to coexist are questioned. However, it has been observed that in highly diverse assemblages, the species have reduced the realised niche, revealed through the trophic breadth, to prevent the alleged effects of competition (MacArthur 1972; Robertson et al. 2014). Therefore, the results obtained are consistent with the ecological niche theory (reviewed by Chase and Leibold 2003), which focuses on biotic interactions and dynamics of resources for consumers at more local scales. In this case, it is assumed that correlated species that occupy the same environments tend to show partition of resources (Schoener 1974; Wiens et al. 2010). In this sense, the partition of resources has been widely analysed for neotropical fishes in different environments (Hahn et al. 2004; Mérona and Rankin-de-Mérona 2004; Russo et al. 2004; Novakowski et al. 2008; Brasil-Souza et al. 2009; Corrêa et al. 2009, 2011; Alves et al. 2011; Silva et al. 2012, 2016b); it is also supported by our study.
Moreover, it is important to note that for the coexistence of species in natural systems, differences in resource utilisation must be not only food partitioning, but also the way species use these food resources in response to environmental variations (Giacomini 2007). Thus, the spatial differences in food resource utilisation observed by Silva et al. (2014), for the same species, favour the coexistence of small fish species at larger spatial scales.
In this perspective, our findings demonstrate the existence of partition of resources among species of small characids. Thus, the exploitation of different preferred resources by species, either by feeding habit or tactic, and the intraspecific variation in response to seasonal availability of resources might be essential for trophic segregation in these characid species. Possibly, alternation of items and proportions in the diet and variation in foraging behaviour, at favourable periods, may be plausible explanatory mechanisms for the coexistence and high abundance of these species. However, experimental work analysing the diet of fish species (both at an individual and at the community level) is needed to improve our understanding of the different ways of partition resources.
References
Abelha MCF, Agostinho AA, Goulart E (2001) Plasticidade trófica em peixes de água doce. Acta Sci Biol Sci 23:425–434. doi:10.4025/actascibiolsci.v23i0.2696
Abujanra F, Agostinho AA, Hahn NS (2009) Effects of the flood regime on the body condition of fish of different trophic guilds in the Upper Paraná River floodplain, Brazil. Braz J Biol 69:469–479. doi:10.1590/S1519-69842009000300003
Albert JS, Reis RE (2011) Historical biogeography of neotropical freshwater fishes. University of California Press, London
Alves GHZ, Tófoli RM, Novakowski GC, Hahn NS (2011) Food partitioning between sympatric species of Serrapinnus (Osteichthyes, Cheirodontinae) in a tropical stream. Acta Sci Biol Sci 33:153–159. doi:10.4025/actascibiolsci.v33i2.7593
Alvim MCC, Peret AC (2004) Food resources sustaining the fish fauna in a section of the upper São Francisco River in Três Marias, MG, Brazil. Braz J Biol 64:195–202. doi:10.1590/S1519-69842004000200003
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46. doi:10.1111/j.1442-9993.2001.01070
Anderson MJ (2006) Distance-based tests for homogeneity of multivariate dispersions. Biometrics 62:245–253. doi:10.1111/j.1541-0420.2005.00440.x
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA+ for PRIMER: guide to software and statistical methods. PRIMER-E Ltd, Plymouth
Araújo MS, Bolnick DI, Layman CA (2011) The ecological causes of individual specialization. Ecol Lett 14:948–958. doi:10.1111/j.1461-0248.2011.01662.x
Avma Panel on Euthanasia (2001) Report of the Avma panel on euthanasia. J Am Vet Med Assoc 218:669–696. doi:10.1016/0892-0362(92)90004-T
Bennemann ST, Gealh AM, Orsi ML, Souza LM (2005) Ocorrência e ecologia trófica de quatro espécies de Astyanax (Characidae) em diferentes rios da bacia do rio Tibagi, Paraná, Brasil. Iheringia Sér Zool 95:247–254. doi:10.1590/S0073-47212005000300004
Bicudo CEM, Bicudo RMT (1970) Algas de águas continentais brasileiras chave ilustrada para identificação de gêneros. Fundação Brasileira para o Desenvolvimento do Ensino de Ciências, São Paulo
Brazil-Sousa C, Marques RM, Albrecht MP (2009) Segregação alimentar entre duas espécies de Heptapteridae no Rio Macaé, RJ. Biota Neotrop 9:31–37. doi:10.1590/S1676-06032009000300002
Carniatto N, Fugi R, Cantanhêde G, Gubiani EA, Hahn NS (2012) Effects of flooding regime and diel cycle on diet of a small sized fish associated to macrophytes. Acta Limnol Bras 24:363–372. doi:10.1590/S2179-975X2013005000007
Casatti L, Castro RMC (2006) Testing the ecomorphological hypothesis in a headwater riffles fish assemblage of the rio São Francisco, southeastern Brazil. Neotrop ichthyol 4:203–214. doi:10.1590/S1679-62252006000200006
Casatti L, Langeani F, Castro RMC (2001) Peixes de riacho do Parque Estadual Morro do Diabo, Bacia do Alto rio Paraná, SP. Biota Neotrop 1:1–15. doi:10.1590/S1676-06032001000100005
Cassemiro FAS, Hahn NS, Fugi R (2002) Avaliação da dieta de Astyanax altiparanae Garutti and Britski, 2000 (Osteichthyes, Tetragonopterinae) antes e após a formação do reservatório de Salto Caxias, Estado do Paraná, Brasil. Acta Sci Biol Sci 24:419–425. doi:10.4025/actascibiolsci.v24i0.2314
Castro RMC, Casatti L, Santos HF, Ferreira KM, Ribeiro AC, Benine RC, Dardis GZP, Melo ALA, Stopiglia R, Abreu TX, Bockmann FA, Carvalho M, Gibran FZ, Lima FCT (2003) Estrutura e composição da ictiofauna de riachos do rio Paranapanema, sudeste e sul do Brasil. Biota Neotrop 3:01–14. doi:10.1590/S1676-06032003000100007
Ceneviva-Bastos M, Casatti L (2007) Oportunismo alimentar de Knodus moenkhausii (Teleostei, Characidae): uma espécie abundante em riachos do noroeste do Estado de São Paulo, Brasil. Iheringia Sér Zool 97:7–15. doi:10.1590/S0073-47212007000100002
Chase JM, Leibold MA (2003) Ecological niches: linking classical and contemporary approaches. University of Chicago Press, Chicago
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Aust J Ecol 18:117–143. doi:10.1111/j.1442-9993.1993.tb00438.x
Correa SB, Winemiller KO (2014) Niche partitioning among frugivorous fishes in response to fluctuating resources in the Amazonian floodplain forest. Ecology 95:210–224. doi:10.1890/13-0393.1
Correa SB, Winemiller KO, López-Fernández H, Galetti M (2007) Evolutionary perspectives on seed consumption and dispersal by fishes. Bioscience 57:748–756. doi:10.1641/B570907
Corrêa CE, Petry AC, Hahn NS (2009) Influência do ciclo hidrológico na dieta e estrutura trófica da ictiofauna do rio Cuiabá, Pantanal Mato-Grossense. Iheringia Sér Zool 99:456–463. doi:10.1590/S0073-47212009000400018
Corrêa CE, Albrecht MP, Hahn NS (2011) Patterns of niche breadth and feeding overlap of the fish fauna in the seasonal Brazilian Pantanal, Cuiabá River basin. Neotrop ichthyol 9:637–646. doi:10.1590/S1679-62252008000400004
Dodds WK, Gido K, Whiles MR, Daniels MD, Grudzinski BP (2015) The stream biome gradient concept: factors controlling lotic systems across broad biogeographic scales. Freshw Sci 34:1–19. doi:10.1086/679756
Dyer LA, Walla TR, Greeney HF, Stireman JO, Hazen RF (2010) Diversity of interactions: a metric for studies of biodiversity. Biotropica 42:281–289. doi:10.1111/j.1744-7429.2009.00624.x
Englund G, Johansson F, Olofsson P, Salonsaari J, Ohman J (2009) Predation leads to assembly rules in fragmented fish communities. Ecol Lett 12:663–671. doi:10.1111/j.1461-0248.2009.01322.x
Fernandes R, Gomes LC, Pelicice FM, Agostinho AA (2009) Temporal organization of fish assemblages in floodplain lagoons: the role of hydrological connectivity. Environ Biol Fish 85:99–108. doi:10.1007/s10641-009-9466-7
Gandini CV, Boratto IA, Fagundes DC, Pompeu PS (2012) Estudo da alimentação dos peixes no rio Grande à jusante da usina hidrelétrica de Itutinga, Minas Gerais, Brasil. Iheringia Sér Zool 102:56–61. doi:10.1590/S0073-47212012000100008
Gatz AJ (1979) Community organization in fishes as indicated by morphological features. Ecology 60:711–718. doi:10.2307/1936608
Gerking SD (1994) Feeding ecology of fishes. Academic Press, San Diego
Giacomini HC (2007) Os mecanismos de coexistência de espécies como vistos pela teoria ecológica. Oecol Bras 11:521–543. doi:10.4257/oeco.2007.1104.05
Gotelli NJ, Entsminger GL (2006) EcoSim: nullmodels software for ecology. Jericho, Acquired Intelligence Inc., Kesey-Bear, version 7. http://www.garyentsminger.com/ecosim/index.htm. Accessed 06 Sept 2016
Gotelli NJ, McCabe D (2002) Species co-occurrence: a meta-analysis of J. M. Diamond’s assembly rules model. Ecology 83:2091–2096. doi:10.2307/3072040
Graça WJ, Pavanelli CS (2007) Peixes da planície de inundação do alto rio Paraná e áreas adjacentes. Eduem, Maringá
Grossman GD (1986) Food resources partitioning in a rocky intertidal fish assemblage. J Zool 1:317–355. doi:10.1111/j.1096-3642.1986.tb00642.x
Hahn NS, Fugi R (2008) Environmental changes, habitat modifications and feeding ecology of freshwater fish. In: Cyrino JEP, Bureau DP, Kapoor BG (eds) Feeding and digestive functions of fishes. Science Publishers, New Hampshire, pp 35–65
Hahn NS, Fugi R, Andrian IF (2004) Trophic ecology of the fish assemblages. In: Thomaz SM, Agostinho AA, Hahn NS (eds) The Upper Paraná River and its floodplain: physical aspects, ecology and conservation. Backhuys Publishers, Leiden, pp 247–269
Hellawell JM, Abel RA (1971) Rapid volumetric method for the analysis of the food of fishes. J Fish Biol 3:29–37. doi:10.1111/j.1095-8649.1971.tb05903.x
Hutchinson GE (1957) Concluding remarks. Cold Spring Harb Symp Quant Biol 22:415–427. doi:10.1101/SQB.1957.022.01.039
Hyslop EJ (1980) Stomach contents analysis: a review of methods and their application. J Fish Biol 17:411–429. doi:10.1111/j.1095-8649.1980.tb02775.x
Koerber S, Litz TO, Mirande JM (2017) Checklist of the freshwater fishes of Argentina (CLOFFAR). Ichthyol Contrib Peces Criollos 47:1–9
Langeani F, Castro CRM, Oyakawa OT, Shibatta OA, Pavanelli CS, Casatti L (2007) Diversidade da ictiofauna do Alto Rio Paraná: composição atual e perspectivas futuras. Biota Neotrop 7:181–197. doi:10.1590/S1676-06032007000300020
Lenza E, Klink CA (2006) Comportamento fenológico de espécies lenhosas em um cerrado sentido restrito de Brasília, DF. Rev Bras Bot 29:627–638. doi:10.1590/S0100-84042006000400013
Loureiro-Crippa VE, Hahn NS, Fugi R (2009) Food resource used by small-sized fish in macrophyte patches in ponds of the Upper Paraná river floodplain. Acta Sci Biol Sci 31:119–125. doi:10.4025/actascibiolsci.v31i2.3266
Lowe-McConnell RH (1987) Ecological studies in tropical fish communities. Cambridge University Press, Cambridge
Lowe-McConnel RH (1999) Estudos ecológicos de comunidade de peixes tropicais. EDUSP, São Paulo
MacArthur RH (1958) Population ecology of some warblers of northeastern coniferous forests. Ecology 39:65–70. doi:10.1111/izy.12000
MacArthur RH (1972) Geographical ecology: patterns in the distribution of species. Harper and Row, New York
Manna LR, Rezende CF, Mazzoni R (2012) Plasticity in the diet of Astyanax taeniatus in a coastal stream from south-east Brazil. Braz J Biol 72:919–928. doi:10.1590/S1519-69842012000500020
Marcarelli AM, Baxter CV, Mineau MM, Hall RO Jr (2011) Quantity and quality: unifying food web and ecosystem perspectives on the role of resource subsidies in freshwaters. Ecology 92:1215–1225. doi:10.1890/10-2240.1
Mateus L, Ortega J, Mendes A, Penha J (2016) Nonlinear effect of density on trophic niche width and between-individual variation in diet in a neotropical cichlid. Austral Ecol 41:492–500. doi:10.1111/aec.12335
Mccreadie JW, Bedwell CR (2013) Patterns of co-occurrence of stream insects and an examination of a causal mechanism: ecological checkerboard or habitat checkerboard? Insect Conserv Divers 6:105–113. doi:10.1111/j.1752-4598.2012.00191.x
Mérona B, Rankin-de-Mérona J (2004) Food resource partitioning in a fish community of the central Amazon floodplain. Neotrop ichthyol 2:75–84. doi:10.1590/S1679-62252004000200004
Mérona B, Vigouroux R, Horeau V (2003) Changes in food resources and their utilization by fish assemblages in a large tropical reservoir in South America (Petit-Saut Dam, French Guiana). Acta Oecol 24:147–156. doi:10.1016/S1146-609X(03)00065-1
Mirande JM (2009) Weighted parsimony phylogeny of the family Characidae (Teleostei: Characiformes). Cladistics 25:574–613. doi:10.1111/j.1096-0031.2009.00262.x
Mise FT, Fugi R, Pagotto JPA, Goulart E (2013) The coexistence of endemic species of Astyanax (Teleostei: Characidae) is propitiated by ecomorphological and trophic variations. Biota Neotrop 13:21–28. doi:10.1590/S1676-06032013000300001
Mittelbach GG, Osenberg CW (1994) Using foraging theory to study trophic interactions. In: Stouder DJ, Fresh KL, Feller RJ (eds) Theory and application in fish feeding ecology. University of South Carolina Press, South Carolina, pp 45–59
Moraes M, Rezende CF, Mazzoni R (2013) Feeding ecology of stream-dwelling Characidae (Osteichthyes: Characiformes) from the upper Tocantins River, Brazil. Zoologia 30:645–651. doi:10.1590/S1984-46702013005000003
Mouchet MA, Burns MDM, Garcia AM, Vieira JP, Mouillot D (2013) Invariant scaling relationship between functional dissimilarity and co-occurrence in fish assemblages of the Patos Lagoon estuary (Brazil): environmental filtering consistently overshadows competitive exclusion. Oikos 122:247–257. doi:10.1111/j.1600-0706.2012.20411.x
Mugnai R, Nessimian JL, Baptista DF (2010) Manual de identificação de macroinvertebrados aquáticos do Estado do Rio de Janeiro. Technical Books, Rio de Janeiro
Nakano S, Murakami M (2001) Reciprocal subsidies: Dynamic interdependence between terrestrial and aquatic food webs. PNAS 98:166–170. doi:10.1073/pnas.98.1.166
Novakowski GC, Hahn NS, Fugi R (2008) Diet seasonality and food overlap of the fish assemblage in a pantanal pond. Neotrop ichthyol 6:567–576. doi:10.1590/S1679-62252008000400004
Pagotto CS, Souza PR (2006) Biodiversidade do Complexo Aporé-Sucuriú: subsídios à conservação e ao manejo do Cerrado: área prioritária 316-Jauru. UFMS, Campo Grande
Peres-Neto PR (2004) Patterns in the co-occurrence of fish species in streams: the role of site suitability, morphology and phylogeny versus species interactions. Oecologia 140:352–360. doi:10.1007/s00442-004-1578-3
Petry P, Bayley PB, Markle DF (2003) Relationships between fish assemblages, macrophytes and environmental gradients in the Amazon River floodplain. J Fish Biol 63:547–579. doi:10.1046/j.1095-8649.2003.00169.x
Pianka ER (1973) The structure of lizard communities. Annu Rev Ecol Syst 4:53–74. doi:10.1146/annurev.es.04.110173.000413
Pianka ER (1981) Competition and niche theory. In: May RM (ed) Theoretical ecology, 2nd edn. Blackwell Scientific, Oxford, pp 167–196
Pinto TLF, Uieda VS (2007) Aquatic insects selected as food for fishes of a tropical stream: are there spatial and seasonal differences in their selectivity? Acta Limnol Bras 19:67–78
Prejs A, Prejs K (1987) Feeding of tropical freshwater fishes: seasonality in resource availability and resource use. Oecologia 71:97–404. doi:10.1007/BF00378713
Punchard NT, Perrow MR, Jowitt AJD (2000) Fish habitat associations, community, struture, density and biomass in natural and channelised lowland streams in the catchment of the River Wensum, UK. In: Cowx IG (ed) Management and ecology of river fisheries. Fishing News Books, Malden, pp 143–157
Quirino BA, Carniatto N, Gaiotto JV, Fugi R (2015) Seasonal variation in the use of food resources by small fishes inhabiting the littoral zone in a neotropical floodplain lake. Aquat Ecol 49:431–440. doi:10.1007/s10452-015-9535-2
Quirino BA, Carniatto N, Guglielmetti R, Fugi R (2017) Changes in diet and niche breadth of a small fish species in response to the flood pulse in a neotropical floodplain lake. Limnologica 62:126–131. doi:10.1016/j.limno.2016.10.005
R Development Core Team (2015) R, a language and environment for statistical computing. Version 3.2.3. R Foundation for Statistical Computing, Austria
Reis RE, Kullander SO, Ferraris CJ Jr (2003) Checklist of the freshwater fishes of south and central America. Edipucrs, Porto Alegre
Ribeiro JF, Walter BMT, Sano SM, Almeida SD (1998) Fitofisionomias do bioma Cerrado. In: Sano SM, Almeida SP (eds) Cerrado: ambiente e flora. EMBRAPA-CPAC, Planaltina, pp 89–166
Richardson JS, Zhangb AY, Marczakc LB (2010) Resource subsidies across the land-freshwater interface and responses in recipient communities. River Res Appl 26:55–66. doi:10.1002/rra.1283
Robertson GS, Bolton M, Grecian WJ, Wilson LJ, Davies W, Monaghan P (2014) Resource partitioning in three congeneric sympatrically breeding seabirds: foraging areas and prey utilization. Ornithol Adv 131:434–446. doi:10.1642/AUK-13-243.1
Robinson BW, Wilson DS (1998) Optimal foraging, specialization, and a solution to Liem’s Paradox. Am Nat 151:223–235. doi:10.1086/286113
Romero RM, Casatti L (2012) Identification of key microhabitats for fish assemblages in tropical Brazilian savanna streams. Int Rev Hydrobiol 97:526–541. doi:10.1002/iroh.201111513
Ross ST (1986) Resource partitioning in fish assemblages: a review of field studies. Copeia 1986:352–388. doi:10.2307/1444996
Roughgarden J (1972) Evolution of niche width. Am Nat 106:683–718
Russo MR, Hahn NS, Pavanelli CS (2004) Resource partitioning between two species of Bryconamericus Eigenmann, 1907 from the Iguaçu river basin, Brazil. Acta Sci Biol Sci 26:431–436. doi:10.4025/actascibiolsci.v26i4.1534
Salazar A, Goldstein G, Franco AC, Miralles-Wilhelm F (2012) Differential seedling establishment of woody plants along a tree density gradient in neotropical savannas. J Ecol 100:1411–1421. doi:10.1111/j.1365-2745.2012.02028.x
Sá-Oliveira JC, Isaac VJ (2013) Diet breadth and niche overlap between Hypostomus plecostomus (Linnaeus, 1758) and Hypostomus emarginatus (Valenciennes, 1840) (Siluriformes) in the Coaracy Nunes hydroelectric reservoir, Ferreira Gomes, Amapá-Brazil. Biota Amazôn 3:16–125. doi:10.18561/2179-5746/biotaamazonia.v3n2p116-125
Sá-Oliveira JC, Angelini R, Isaac-Nahum VJ (2014) Diet and niche breadth and overlap in fish communities within the area affected by an Amazonian reservoir (Amapá, Brazil). An Acad Bras Ciênc 86:383–405. doi:10.1590/0001-3765201420130053
Schoener TW (1974) Resource partitioning in ecological communities. Science 185:27–39
Sih A, Christensen B (2001) Optimal diet theory: when does it work, and when and why does it fail? Anim Behav 61:379–390. doi:10.1006/anbe.2000.1592
Sih A, Crowley P, McPeek M, Petranka J, Strohmeier K (1985) Predation, competition, and prey communities: a review of field experiments. Ann Rev Ecol Syst 16:269–311. doi:10.1146/annurev.es.16.110185.001413
Silva JC, Delariva RL, Bonato KO (2012) Food-resource partitioning among fish species from a first-order stream in northwestern Paraná, Brazil. Neotrop ichthyol 10:389–399. doi:10.1590/S1679-62252012005000008
Silva JC, Gubiani EA, Delariva RL (2014) Use of food resources by small fish species in neotropical rivers: responses to spatial and temporal variations. Zoologia 31:435–444. doi:10.1590/s1984-46702014000500004
Silva JC, Gubiani EA, Piana PA, Delariva RL (2016a) Effects of a small natural barrier on the spatial distribution of the fish assemblage in the Verde River, Upper Paraná River Basin, Brazil. Braz J Biol 76:851–863. doi:10.1590/1519-6984.01215
Silva NCS, Costa AJL, Louvise J, Soares BE, Reis VCS, Albrecht MP, Caramaschi EP (2016b) Resource partitioning and ecomorphological variation in two syntopic species of Lebiasinidae (Characiformes) in an Amazonian stream. Acta Amazon 46:25–36. doi:10.1590/1809-4392201501024
Stephens DW, Krebs JR (1986) Foraging theory. Princeton University Press, New Jersey
Stevens GC (1989) The latitudinal gradient in geographical range: how so many species coexist in the tropics. Am Nat 133:240–256
Svanbäck R, Bolnick DI (2007) Intraspecific competition drives increased resource use diversity within a natural population. Proc R Soc B 274:839–844. doi:10.1098/rspb.2006.0198
Swanson HK, Lysy M, Power M, Stasko AD, Johnson JD, Reist JD (2015) A new probabilistic method for quantifying n-dimensional ecological niches and niche overlap. Ecology 96:318–324. doi:10.1890/14-0235.1
Thomaz SM, Agostinho AA, Hahn NS (2004) The Upper Paraná River and its floodplain: physical aspects, ecology and conservation. Backhuys Publishers, Leiden
Tófoli RM, Hahn NS, Alves GHZ, Novakowski GC (2010) Uso do alimento por duas espécies simpátricas de Moenkhausia (Characiformes, characidae) em um riacho da Região Centro-Oeste do Brasil. Iheringia Sér Zool 100:201–206. doi:10.1590/S0073-47212010000300003
Trevisan A, Hepp LU (2007) Dinâmica de componentes químicos vegetais e fauna associada ao processo de decomposição de espécies arbóreas em um riacho no norte do Rio Grande do Sul, Brasil. Neotrop Biol Conserv 2:55–60
Tupinambás TH, Pompeu PS, Gandini CV, Hughes RM, Callisto M (2015) Fish stomach contents in benthic macroinvertebrate assemblage assessments. Braz J Biol 75:157–164. doi:10.1590/1519-6984.09913
Uieda VS, Pinto TLF (2011) Feeding selectivity of ichthyofauna in a tropical stream: space-time variations in trophic plasticity. Commun Ecol 12:31–39. doi:10.1556/ComEc.12.2011.1.5
Viana LF, Santos SL, Lima-Junior SE (2006) Variação sazonal na alimentação de Pimelodella cf. gracilis (Osteichthyes, Siluriformes, Pimelodidae) no rio Amambai, Estado do Mato Grosso do Sul. Acta Sci Biol Sci 28:123–128. doi:10.4025/actascibiolsci.v28i2.1028
Wang M, Liu F, Lin P, Yang S, Liu H (2015) Evolutionary dynamics of ecological niche in three Rhinogobio fishes from the upper Yangtze River inferred from morphological traits. Ecol Evol 5:567–577. doi:10.1002/ece3.1386
Ward AJW, Webster MM, Hart PJB (2006) Intraspecific food competition in fishes. Fish Fish 7:231–261. doi:10.1111/j.1467-2979.2006.00224.x
Wiens JJ, Ackerly DD, Allen AP, Anacker BL, Buckley LB, Cornell HV, Wiens JJ, Damschen EI, Davies TJ, Grytnes JA, Harrison SP, Hawkins BA, Holt RD, McCain CM, Stephens PR (2010) Niche conservatism as an emerging principle in ecology and conservation biology. Ecol Lett 13:1310–1324. doi:10.1111/j.1461-0248.2010.01515.x
Wilbur HM, Fauth JE (1990) Experimental aquatic food webs: interactions between two predators and two prey. Am Nat 135:176–204. doi:10.1086/285038
Wolff LL, Abilhoa V, Rios FS, Donatti L (2009) Spatial, seasonal and ontogenetic variation in the diet of Astyanax aff. fasciatus (Ostariophysi: Characidae) in an Atlantic Forest river, Southern Brazil. Neotrop ichthyol 7:257–266. doi:10.1590/S1679-62252009000200018
Wootton RJ (1990) Ecology of teleost fishes. Chapman and Hall, London
Wootton JT (1992) Indirect effects, prey susceptibility, and habitat selection: impacts of birds on limpets and algae. Ecology 73:981–991. doi:10.2307/1940174
Zavala-Camin LA (1996) Introdução aos estudos sobre alimentação natural em peixes. Eduem/Nupélia, Maringá
Zhang J (2013) spaa: SPecies Association Analysis. R package version 0.2.1. http://CRAN.R-project.org/package=spaa. Accessed 24 June 2016
Acknowledgements
We thank the Grupo de pesquisas em Recursos Pesqueiros e Limnologia (GERPEL) for their help with fieldwork. Eletrosul Centrais Elétricas S.A. funded our research. The Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) granted us a research fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Piet Spaak.
Rights and permissions
About this article
Cite this article
da Silva, J.C., Gubiani, É.A., Neves, M.P. et al. Coexisting small fish species in lotic neotropical environments: evidence of trophic niche differentiation. Aquat Ecol 51, 275–288 (2017). https://doi.org/10.1007/s10452-017-9616-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-017-9616-5