Abstract
Purpose
To verify the possibility that preoperative topical antibiotics are not essential as long as iodine disinfection is performed during surgery.
Study design
Crossover equivalence trial.
Patients and methods
In 204 eyes of 102 patients who underwent routine bilateral cataract surgery, 1 eye was treated with intraoperative iodine, and the other, with preoperative topical antibiotics. For the intraoperative iodine eyes, 5 mL of 0.25% povidone-iodine was applied at 2 stages: (1) just after the placement of the speculum and (2) before intraocular lens (IOL) insertion. For the contralateral eyes, preoperative topical antibiotics were administered 3 days before surgery without intraoperative iodine. Conjunctival samples for culture were obtained at 3 time points: (a) presurgery, (b) beginning of surgery, and (c) postsurgery. Real-time polymerase chain reaction (PCR) samples were obtained at the beginning of surgery and before IOL insertion. Intracameral moxifloxacin was applied in all the cases.
Results
The respective positive bacterial culture rates for intraoperative iodine eyes and preoperative topical antibiotics eyes were 95.1% and 98.0% at (a), 7.8% and 5.9% at (b), and 60.8% and 62.7% at (c). A significant difference in the positive bacterial culture rate was not found at any time point. For the intraoperative iodine eyes, the bacterial DNA copy number at (b) was significantly lower than that for the preoperative topical antibiotics eyes.
Conclusions
The cleanliness of the operative field without using topical antibiotics was revealed to be equivalent to that of the conventional method (using preoperative antibiotics without intraoperative iodine) as long as intraoperative iodine was used.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Postoperative endophthalmitis is a devastating complication of cataract surgery; its causative bacteria are derived mainly from the patient’s eyelids and conjunctiva. In Japan, clinical studies demonstrating that preoperative topical antibiotics significantly reduce bacterial positivity in the surgical field are widely recognized, and preoperative topical antibiotics are commonly used [1]. Conversely, in other countries, including Sweden, Canada, and Australia, preoperative topical antibiotics are not common [2]. Multiple instillations of antibiotics are expensive and complicated for patients [3]. There is also a risk that perioperative antibiotic eye drops induce drug-resistant bacteria [4]. It is beneficial for both patients and physicians if surgery is safely performed without topical antibiotics. Consequently, the concept of dropless cataract surgery, which involves performing surgery without use of instillations, was proposed [5]. In May 2015, the World Health Assembly debated the international response to antimicrobial resistance and issued a Global Action Plan to inspire medical professionals to enhance the awareness and understanding of antimicrobial resistance and to optimize the use of antimicrobials in humans and animals [6].
Worldwide, it is common to use preoperative iodine for periocular disinfection in ophthalmic surgery [2, 7, 8]
In conventional cataract surgery, periocular skin disinfection using 5%–10% iodine, followed by 5% iodine instillations is performed before speculum placement. Recently, the concept of intraoperative iodine has been proposed [9, 10]. Iodine compound is inexpensive, has a strong bactericidal effect, and does not produce drug resistance [8]. In addition, iodine compound is effective in approximately 15 seconds and can be used intraoperatively, whereas antibiotics require more than 60 minutes to show their effect. If intraoperative iodine is used, there is a possibility of attaining an uncontaminated surgical field without preoperative topical antibiotics.
In this study, we sought to verify the possibility that preoperative topical antibiotics are not essential when iodine disinfection is performed during cataract surgery.
Patients and methods
Patient enrollment
This study was conducted at Nojima Hospital between December 2017 and February 2018. Patients without chronic conjunctivitis or blepharitis who were scheduled to undergo routine bilateral cataract surgery were enrolled (102 patients; 204 eyes). The first operated-on eye was assigned to the intraoperative iodine eye without preoperative topical antibiotics, and the contralateral eye, to the eye with preoperative topical antibiotics without intraoperative iodine disinfection. The interval between the surgeries was 3 days. Postoperative topical antibiotics were initiated on the morning of the day after surgery.
The procedures conformed to the tenets of the Declaration of Helsinki and were approved by the ethics review committee of Nojima Hospital. Informed consent was obtained from all participants before the surgical procedures, and possible complications were explained to them, including postoperative endophthalmitis. The study was registered on the International Clinical Trial Registry Platform (UMIN Clinical Trials Registry, UMIN000013348; http://www.umin.ac.jp/ctr/index-j.htm).
Disinfection protocol
Preoperative disinfection with povidone-iodine (PVP-I) was performed on both eyes in all patients. The periocular skin was disinfected with 10% PVP-I, followed by two 5% PVP-I instillations at 20-second intervals. The first eye of each patient was treated with intraoperative iodine, and the second eye, with preoperative topical antibiotics.
For the eye with intraoperative iodine, preoperative topical antibiotics were not prescribed. During the surgery, 5 mL of 0.25% PVP-I was applied to the conjunctiva and corneal surface at 2 stages: (1) just after the speculum placement and (2) just before the intraocular lens (IOL) insertion. In the first stage, the surgical field was irrigated twice with 0.25% PVP-I, with a 20-second interval immediately after the placement of the eye speculum and immediately before the initial sclerocorneal incision. The same irrigation procedure was repeated just before the insertion of the IOL.
For the eye with preoperative topical antibiotics, 1.5% levofloxacin (Santen) was administered 3 times a day for 3 days before the surgery and once on the morning of the surgery. In the operation room, iodine disinfection was performed before the placement of the speculum, but not after.
All the patients underwent conventional cataract surgery with phacoemulsification and implantation of an acrylic IOL through a sclerocorneal incision. In all cases, intracameral moxifloxacin with the flushing technique [11] was performed at the end of the surgery. Preoperative, intraoperative, and postoperative surgical field contaminations were compared between the eyes.
Sample collection and bacteriologic examinations
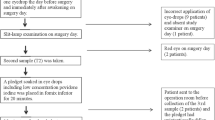
Conjunctival sac scrapings were collected after the conjunctiva was anesthetized with preservative-free oxybuprocaine hydrochloride 0.4% ophthalmic solution via insertion of sterile absorbent sponges (Medical Quick Absorber) moistened with sterile physiological saline into the fornix of the lower eyelid. Conjunctival samples for culture were obtained at the following time points: (a) presurgery, 1–2 weeks before the surgery; (b) beginning of surgery, after placement of the speculum before the first incision; and (c) postsurgery, 2 hours after the surgery. Polymerase chain reaction (PCR) samples were obtained at the beginning of the surgery, just after conjunctival scrapings were obtained for culture, and just before the intraoperative iodine irrigation before IOL insertion. Samples were obtained after iodine irrigation at the beginning of the surgery and before iodine irrigation at pre-IOL insertion (Fig. 1).
Study protocol. The individual steps in the perioperative infection prophylaxis regimen are illustrated. Presurgery 1–2 weeks before surgery, Beginning of surgery after placement of the speculum before the first incision, Pre-IOL insertion before IOL insertion, Postsurgery 2 hours after the surgery, PVI povidone-iodine, IOL intraocular lens
For bacterial culture, sodium thiosulfate (0.01%) was added to the samples to neutralize the germicidal activity of iodine, and the mixture was frozen. The samples were sent to the Research Institute for Microbial Diseases, Osaka University, for aerobic and anaerobic culture and drug susceptibility testing.
At the Osaka University laboratory, the samples were transferred to a thioglycollate medium and incubated at 35°C for 2 weeks to stimulate aerobic and anaerobic bacterial growth. For the growth of anaerobic bacteria, the samples were transferred to Columbia agar enriched with 5% sheep blood and placed in an anaerobic bag at 37°C for 5 days and then incubated at 35°C for 2 days with 5% carbon dioxide. For real-time PCR, the samples were stored at -40°C immediately after the collection.
Broad-range real-time PCR of bacteria
DNA was extracted from the samples using a QIAamp DNA mini kit (Qiagen) [12]. The number of bacterial DNA copies was determined by real-time PCR targeting 16S ribosomal RNA, as previously described [13].
Statistical analysis
The study protocol was designed as an equivalence trial with positivity of bacterial culture [14]. The primary objective of this study was to examine whether bacterial culture positivity of cataract surgery with intraoperative iodine disinfection was equivalent to that of surgery with preoperative topical antibiotics. The lower and upper equivalence margins to determine the equivalence of surgery with or without preoperative topical antibiotics were set as 80% and 125%, respectively. To determine the required sample size, we used the intersubject coefficient of variation of culture positivity. On the basis of data from our previous study, the coefficient of variation was determined as 0.23 [10]. On the basis of a type I error of 5% and a type II error of 20%, the sample size was calculated as 92 using the FARTSSIE program (Free Analysis Research Tool for Sample Size Iterative Estimation, David Dubins, http://individual.utoronto.ca/ddubins/).
The primary outcome was compared using the Wilcoxon signed rank test, the paired t test, and mixed linear regression analysis, by means of Stata 15 software (StataCorp). Intention-to-treat analyses were conducted. Probability values below .05 were considered significant.
Results
The mean (SD) age of the patients was 74.4 (8.0) years, and 36 of the 102 patients were male. All surgeries were performed by a single surgeon (K.M.), and the operative time was ≤ 15 minutes for each eye. No intraoperative or postoperative complications were observed.
Positive bacterial culture rates at (a) presurgery in the intraoperative iodine eyes and the preoperative topical antibiotics eyes were 95.1% and 98.0%, respectively. Positive bacterial culture rates in the intraoperative iodine eyes and preoperative topical antibiotics eyes were 7.8% and 5.9% at time point (b) (beginning of surgery) and 60.8% and 62.7% at time point (c) (postsurgery), respectively. Significant differences in the positive bacterial culture rate were not found at any time point (Fig. 2).
Positive bacterial culture rate: intraoperative iodine eyes vs preoperative topical antibiotics eyes. The percentages of positive cultures are shown at the different time points (presurgery: 1–2 weeks before the surgery, beginning of surgery: immediately after speculum placement, postsurgery: 2 hours after the surgery). Data are presented as means ± standard error of the means
We examined whether any difference in culture positivity was found between the 2 conditions at the preoperative, intraoperative, and postoperative time points (Fig. 2). At all the time points, 2-sided 95% confidence intervals of the differences in culture positivity crossed the zero outcome difference. All confidence intervals were located within the equivalence range (Fig. 3). The average number of cultured bacterial species per eye was similar and not significantly different (Fig. 4). This indicates that the cleanliness of the surgical field without preoperative topical antibiotics did not differ significantly from that with topical antibiotics.
Equivalence of positive bacterial culture rate between eyes with and without preoperative topical antibiotics. The shaded area indicates equivalence range (-0.20 to 0.25). The circle indicates observed estimate of culture positive rate difference between 2 arms. The error bar indicates 2-sided 95% confidence interval. At all the time points, the 95% confidence interval crossed the 0 outcome difference, and no significant difference in culture positive rates was observed. All confidence intervals were located within the predetermined equivalence range, and the culture positive rate was equivalent between the eyes. Presurgery 1–2 weeks before surgery, Beginning of surgery immediately after speculum placement, Postsurgery 2 hours after surgery
Average number of cultured bacterial species per eye: intraoperative iodine eyes vs preoperative topical antibiotics eyes. The average numbers of bacterial species are shown at the different time points (Presurgery 1–2 weeks before surgery, Beginning of surgery immediately after speculum placement, Postsurgery 2 hours after surgery). Data are presented as means ± standard error of the means
Before surgery (1–2 weeks before surgery), 17.4% of the bacteria were Staphylococcus epidermidis; 22.3%, Propionibacterium acnes; 25.1%, Corynebacterium species; and 8.7%, Streptococcus species (Fig. 5).
At the beginning of surgery, 9 species of bacteria were found in 8 eyes of the intraoperative iodine patients, whereas 7 bacteria were found in 6 eyes of the preoperative topical antibiotic patients. In the intraoperative iodine eyes, 1 S epidermidis, 5 P acnes, 2 Staphylococcus species, and 1 Streptococcus species were identified. Two S epidermidis and 5 P acnes were identified in the preoperative topical antibiotic eyes (Table 1). Postsurgery, 20.3% of the bacteria were S epidermidis; 37.4%, P. acnes; 11.2%, Corynebacterium species; and 4.8%, Streptococcus species (Fig. 5).
Apparent differences were not observed between the groups. At any time point, P acnes was the most frequently detected bacterial species, followed by S epidermidis and Corynebacterium species (Table 1).
For eyes with intraoperative iodine, the number of 16S ribosomal RNAs at time point (a) (beginning of surgery) was 4.7 ± 0.2 × 103 copies, which was significantly lower than that for eyes with preoperative topical antibiotics (5.4 ± 0.2 × 103 copies; P = .000). This indicated that the intraoperative iodine irrigation resulted in a significant reduction in the amount of bacteria at the early stage of surgery. Apparent recontamination was not observed at pre-IOL insertion in either eye (Fig. 6).
Average number of 16S ribosomal RNAs of bacteria: intraoperative iodine eyes vs preoperative topical antibiotics eyes. DNA copy numbers are shown at the different time points intraoperatively (Beginning of surgery immediately after speculum placement, Pre-IOL insertion just before the second intraoperative iodine irrigation before IOL insertion). The asterisk indicates a significant difference between the eyes. Data are presented as means ± standard error of the means
Discussion
In this study, the cleanliness of the operative field without using preoperative topical antibiotics was revealed to be equivalent to that of the conventional method (using preoperative topical antibiotics without intraoperative iodine) as long as intraoperative iodine disinfection was performed. In other words, it was shown that preoperative topical antibiotics are not essential if intraoperative iodine is used. A consensus for using iodine compound before surgery has been universally obtained [2, 8]; however, even if a fair amount of cleanliness is obtained at the beginning of surgery before speculum placement, the possibility of recontamination during surgery cannot be denied. Shimada and colleagues introduced the effectiveness of continuously applying diluted iodine compounds during surgery. Frequent use of iodine compounds, however, is associated with corneal epithelial damage [15].
Therefore, we previously examined the intraoperative use of iodine compounds by limiting it to 2 uses (the twice-iodine method). [10] After speculum placement, the first intraoperative disinfection was performed by iodine irrigation before the first incision was made. Contamination of the operative field was more reduced in the intraoperative iodine eyes than in the noniodine eyes. The second intraoperative iodine irrigation was conducted immediately before IOL insertion because the timing of IOL insertion is most likely to lead to intraocular contamination. Following the intraoperative application of iodine compound, the bacterial culture was negative at preincision and at pre-IOL insertion. The DNA copy number after speculum placement (preincision) was lower in the intraoperative iodine eyes [10]. Intraoperative iodine disinfection in the present study was performed using this twice-iodine method. To evaluate the recontamination during surgery, the sampling for PCR at pre-IOL insertion was conducted before iodine irrigation. The cleanliness was equivalent in both eyes. For the intraoperative iodine eyes, additional iodine irrigation was conducted.
In intravitreal injection, the risk of infection is considered to be higher when perioperative antimicrobial instillation is conducted because antibiotic resistance occurs [16, 17].
Before intravitreal injection, Moss and colleagues also performed iodine disinfection following the placement of the speculum or draping. In their report, comparisons were made with and without topical antibiotics before surgery. The positive culture rate dramatically decreased in both groups after intraoperative iodine was performed, and no significant difference was found between the groups [18].
In the American Academy of Ophthalmology’s protocol for intravitreous injection, it is noted that preoperative topical antibiotics are not routinely used and that iodine disinfection is recommended after speculum placement [19].
For intravitreal injection, a 30-G needle is commonly used, and the injection site is covered by the conjunctiva. Thus, the cleanliness of the surgical field is necessary only at the moment of penetration of the injection needles. For cataract surgery, however, instruments are inserted into the eye during various procedures throughout the surgery. The timing of pre-IOL insertion is especially critical because the IOL is left in the eye. In other words, even if the surgical field is clean at the beginning of surgery, recontamination is inevitable, which may result in the contamination of the anterior chamber or vitreous. The findings of this study suggest that intraoperative iodine can achieve cleanliness of the surgical field throughout surgery without use of preoperative topical antibiotics.
In cataract surgery, bacteria may invade the eye postoperatively when insufficient self-sealing of the wound occurs. For the purpose of comparing the risk of infection in the early postoperative period, we also compared cultures 2 hours after surgery but found no difference between the eyes. This means that intraoperative iodine can replace the role of preoperative antibiotics even after the early postoperative period. Moreover, long-term use of antimicrobial drugs may induce drug-resistant bacteria. In contrast, when using intraoperative iodine, resistance is less likely to occur. Furthermore, it is also attractive that iodine can be intraoperatively applied by the surgeon him/herself during surgery. In this study, over 60% of the surgical fields were contaminated at 2 hours after surgery. There have been reports of over 20% of surgical fields being contaminated immediately after surgery with removal of the eye speculum despite 3-day preoperative antibiotic instillation [1]. It means that recontamination occurs immediately after surgery even if a fair degree of cleanliness was achieved during the surgery.
To overcome this problem, it is advisable to perform proper wound closure, ensure the wounds is self-sealed, and finish the surgery. Initiating the postoperative topical antibiotics immediately after surgery may be considered. However, it may be risky for patients to instill themselves during the initial period immediately after surgery. The most frequent cause of endophthalmitis is S epidermidis, which is associated with the problem of resistance to antibiotics worldwide [20, 21]. Levofloxacin is the antibacterial agent most frequently used as a perioperative topical antibiotic in Japan; however, the concentration of levofloxacin in the anterior chamber after repeated administration is approximately 1 μg/mL [22, 23]. Thus, the effect of postoperative topical antibiotics on the bacteria introduced into the eye is limited. Therefore, the concept of intraoperative iodine or intracameral antibiotics becomes important. Iodine compound does not have resistant bacteria, and intracameral antibiotics can achieve sufficient intraocular concentration of antibiotics [24]. With intracameral antibiotics, it is not necessary to start antibiotic instillation on the day of surgery because a considerable concentration of antibiotic is supposed to remain in the anterior chamber several hours after surgery.
There are a few limitations to this study. Because the use of iodine compound has not been standardized, there are a myriad of combinations, such as the timing, concentration, and number of uses [8]. Although the twice-iodine method [10] was adopted in the current study, the result may be different if the usage of iodine varies. In addition, a large cohort study is necessary to prospectively confirm whether intraoperative iodine without preoperative topical antibiotics is beneficial in reducing the incidence of endophthalmitis. From the viewpoint of practicality, intraoperative iodine is not affected by patient compliance and is reliably performed by the surgeon during surgery. Although it is necessary to examine the optimal concentration, timing, and number of administrations of intraoperative iodine during intraocular surgery, the findings of this study suggest that preoperative topical antibiotics are not essential as long as intraoperative iodine is used.
References
Inoue Y, Usui M, Ohashi Y, Shiota H, Yamazaki T, Preoperative Disinfection Study G. Preoperative disinfection of the conjunctival sac with antibiotics and iodine compounds: a prospective randomized multicenter study. Jpn J Ophthalmol. 2008;52:151–61. https://doi.org/10.1007/s10384-008-0517-y.
Grzybowski A, Schwartz SG, Matsuura K, Ong Tone S, Arshinoff S, Ng JQ, et al. Endophthalmitis prophylaxis in cataract surgery: overview of current practice patterns around the world. Curr Pharm Design. 2017;23:565–73. https://doi.org/10.2174/1381612822666161216122230.
An JA, Kasner O, Samek DA, Levesque V. Evaluation of eyedrop administration by inexperienced patients after cataract surgery. J Cataract Refract Surg. 2014;40:1857–61. https://doi.org/10.1016/j.jcrs.2014.02.037.
Nejima R, Shimizu K, Ono T, Noguchi Y, Yagi A, Iwasaki T, et al. Effect of the administration period of perioperative topical levofloxacin on normal conjunctival bacterial flora. J Cataract Refract Surg. 2017;43:42–8. https://doi.org/10.1016/j.jcrs.2016.10.024.
Lindstrom RL, Galloway MS, Grzybowski A, Liegner JT. Dropless cataract surgery: an overview. Curr Pharm Design. 2017;23:558–64. https://doi.org/10.2174/1381612822666161129150628.
Mendelson M, Matsoso MP. The World Health Organization Global Action Plan for antimicrobial resistance. S Afr Med J. 2015;105:325. https://doi.org/10.7196/samj.9644.
Behndig A, Cochener B, Guell JL, Kodjikian L, Mencucci R, Nuijts RM, et al. Endophthalmitis prophylaxis in cataract surgery: overview of current practice patterns in 9 European countries. J Cataract Refract Surg. 2013;39:1421–31. https://doi.org/10.1016/j.jcrs.2013.06.014.
Grzybowski A, Kanclerz P, Myers WG. The use of povidone-iodine in ophthalmology. Curr Opin Ophthalmol. 2018;29:19–32. https://doi.org/10.1097/icu.0000000000000437.
Shimada H, Arai S, Nakashizuka H, Hattori T, Yuzawa M. Reduction of anterior chamber contamination rate after cataract surgery by intraoperative surface irrigation with 0. 25% povidone-iodine. Am J Ophthalmol. 2011;151:11.e1–17.e1. https://doi.org/10.1016/j.ajo.2010.07.002.
Matsuura K, Miyazaki D, Sasaki SI, Yakura K, Inoue Y, Sakamoto M. Effectiveness of timely intraoperative iodine irrigation during cataract surgery. Jpn J Ophthalmol. 2016;60:433–8. https://doi.org/10.1007/s10384-016-0471-z.
Matsuura K, Suto C, Akura J, Inoue Y. Bag and chamber flushing: a new method of using intracameral moxifloxacin to irrigate the anterior chamber and the area behind the intraocular lens. Graefes Arch Clin Exp Ophthalmol. 2013;251:81–7. https://doi.org/10.1007/s00417-012-2098-1.
Kakimaru-Hasegawa A, Kuo CH, Komatsu N, Komatsu K, Miyazaki D, Inoue Y. Clinical application of real-time polymerase chain reaction for diagnosis of herpetic diseases of the anterior segment of the eye. Jpn J Ophthalmol. 2008;52:24–31. https://doi.org/10.1007/s10384-007-0485-7.
Ikeda Y, Miyazaki D, Yakura K, Kawaguchi A, Ishikura R, Inoue Y, et al. Assessment of real-time polymerase chain reaction detection of Acanthamoeba and prognosis determinants of Acanthamoeba keratitis. Ophthalmology. 2012;119:1111–9. https://doi.org/10.1016/j.ophtha.2011.12.023.
Ahn S, Park SH, Lee KH. How to demonstrate similarity by using noninferiority and equivalence statistical testing in radiology research. Radiology. 2013;267:328–38. https://doi.org/10.1148/radiol.12120725.
Fernandes M, Pathengay A. Reduction of anterior chamber contamination rate after cataract surgery by intraoperative surface irrigation with 0.25% povidone-iodine. Am J Ophthalmol. 2011;152:320. https://doi.org/10.1016/j.ajo.2011.03.030(author reply-1).
Yin VT, Weisbrod DJ, Eng KT, Schwartz C, Kohly R, Mandelcorn E, et al. Antibiotic resistance of ocular surface flora with repeated use of a topical antibiotic after intravitreal injection. JAMA Ophthalmol. 2013;131:456–61. https://doi.org/10.1001/jamaophthalmol.2013.2379.
Storey P, Dollin M, Pitcher J, Reddy S, Vojtko J, Vander J, et al. The role of topical antibiotic prophylaxis to prevent endophthalmitis after intravitreal injection. Ophthalmology. 2014;121:283–9. https://doi.org/10.1016/j.ophtha.2013.08.037.
Moss JM, Sanislo SR, Ta CN. A prospective randomized evaluation of topical gatifloxacin on conjunctival flora in patients undergoing intravitreal injections. Ophthalmology. 2009;116:1498–501. https://doi.org/10.1016/j.ophtha.2009.02.024.
Michelle EW, Adrienne WS. How to give intravitreal injections. 2013. https://www.aao.org/eyenet/article/how-to-give-intravitreal-injections. Accessed Apr 2013.
Miller DM, Vedula AS, Flynn HW Jr, Miller D, Scott IU, Smiddy WE, et al. Endophthalmitis caused by Staphylococcus epidermidis: in vitro antibiotic susceptibilities and clinical outcomes. Ophthalmic Surg Lasers Imaging. 2007;38:446–51.
Stringham JD, Relhan N, Miller D, Flynn HW Jr. Trends in fluoroquinolone nonsusceptibility among coagulase-negative Staphylococcus isolates causing endophthalmitis, 1995-2016. JAMA Ophthalmol. 2017;135:814–5. https://doi.org/10.1001/jamaophthalmol.2017.1826.
Colin J, Simonpoli S, Geldsetzer K, Ropo A. Corneal penetration of levofloxacin into the human aqueous humour: a comparison with ciprofloxacin. Acta Ophthalmol Scand. 2003;81:611–3.
Koch HR, Kulus SC, Roessler M, Ropo A, Geldsetzer K. Corneal penetration of fluoroquinolones: aqueous humor concentrations after topical application of levofloxacin 0.5% and ofloxacin 0.3% eyedrops. J Cataract Refract Surg. 2005;31:1377–85. https://doi.org/10.1016/j.jcrs.2004.12.063.
Arshinoff SA, Modabber M. Dose and administration of intracameral moxifloxacin for prophylaxis of postoperative endophthalmitis. J Cataract Refract Surg. 2016;42:1730–41. https://doi.org/10.1016/j.jcrs.2016.10.017.
Acknowledgements
Grant-in-Aid for Scientific Research (to S.S., 15K20261) from the Ministry of Education, Culture, Sports, Science and Technology of Japan. Y.I. is a medical advisor to Senju Pharmaceutical Company, Japan. No conflicting relationship exists for the other authors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
K. Matsuura, None; D. Miyazaki, Lecture fee (Senju, Santen, Pfizer); S. Sasaki, None; Y. Inoue, Grant (Santen, Alcon), Consultant fee (Senju); Y. Sasaki, None; Y. Shimizu, None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Corresponding Author: Kazuki Matsuura
About this article
Cite this article
Matsuura, K., Miyazaki, D., Sasaki, Si. et al. Effectiveness of intraoperative iodine in cataract surgery: cleanliness of the surgical field without preoperative topical antibiotics. Jpn J Ophthalmol 64, 37–44 (2020). https://doi.org/10.1007/s10384-019-00703-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-019-00703-5