Abstract
Deadwood is a key component of nutrient cycling in natural tropical forests, serving as a globally important carbon storage and habitat for a high number of species. The conversion of tropical forests to agriculture modifies deadwood pools, but we know little about deadwood dynamics in forests recovering from human disturbance. Here we quantified the volume and diversity of coarse woody debris (CWD, ≥ 7 cm diameter) and the mass of fine woody debris (FWD, < 7 cm) along a chronosequence of natural forest recovery in the lowlands of the Ecuadorian Chocó region. We sampled forest plots ranging from 1–37 years of recovery post-cessation of agricultural use as either cacao plantation or cattle pasture, as well as actively managed cacao plantations and cattle pastures, and old-growth forests. In contrast to our expectation, we found no significant increase in deadwood volume with recovery time. The diversity in size, decay stage and type of CWD increased along the recovery gradient, with no effect of previous land use type. The mass of FWD increased overall across the recovery gradient, but these results were driven by a steep increase in former pastures, with no change observed in former cacao plantations. We suggest that the range of sizes and decomposition stages of deadwood found in these two major tropical agricultural systems could provide suitable resources for saproxylic organisms and an overlooked carbon storage outside old-growth forests. Our estimates of deadwood in agricultural systems and recovering forests can help improve global assessments of carbon storage and release in the tropics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Deadwood plays a central role in maintaining forest structure and function (Seibold et al. 2015; Magnusson et al. 2016). It is a key component of the global nutrient cycle, serving as carbon and nitrogen sinks (Hérault et al. 2010). Deadwood decomposes much faster in tropical forests than in temperate or boreal forests; thus > 90% of carbon released from deadwood globally originates within tropical forests (Seibold et al. 2021). Deadwood also provides habitat and food for myriad of facultative and obligate wood depending, so called saproxylic species (Graf et al. 2022), which directly or indirectly depend on dead or dying wood during part of their life cycle. Obligate saproxylics comprise between 6 and 25% of terrestrial biodiversity (Grove 2002; Brin et al. 2011; Stokland et al. 2012) with a high proportion of endangered species in areas with long-term history of intense forest management (Seibold et al. 2015; Stokland et al. 2012). Furthermore, deadwood provides human populations with necessary resources such as firewood and construction materials (Pfeifer et al. 2015). Despite its importance, however, our understanding of the factors that shape deadwood amount and diversity has been identified as a knowledge gap particularly for tropical regions (Seibold et al. 2015). Many deadwood studies have either focused exclusively on coarse pieces or were carried out in primary forests unaffected by logging. As a result, we still know little about the amount of carbon stored in deadwood across recovery gradients (but see Kissing and Powers 2010, for tropical dry forests), which is worrying given that the proportion of tropical forests undergoing selective logging or conversion to agriculture continues to increase worldwide (Fernandez Barrancos et al. 2022; Pfeifer et al. 2015). This lack of information on wood debris stocks and their importance for biodiversity particularly in the tropics (Kissing and Powers 2010; Brown 1997; Seibold et al. 2015) impedes our ability to inform policymakers on ecological problems such as habitat degradation, global warming, and biodiversity loss associated with forest use.
Many tropical forests have suffered and recovered from intermittent periods of disturbance over the last thousand years. For example, while large tracts of the Amazon basin have been continuously covered by forest for at least 5000 years with only small-scale perturbation by fires (Piperno et al. 2021), agricultural land in what is now northern Guatemala likely took 80–260 years to recover into forests after abandonment by the Maya civilization around the year AD 1000 (Mueller et al. 2010). Likewise, land used intensively until the sixteenth century by indigenous populations in Ecuadorian Andean landscapes recovered to old-growth levels in around 130 years (Loughlin et al. 2018). Literature on current anthropogenic impacts attests to the ability of tropical forests to recover from different sources of human intervention within a few decades (Rozendaal et al. 2019). Recovery time is faster when there is a higher cover of primary forest in the surrounding landscape, with a higher abundance and richness of retention trees remaining in the agricultural matrix, when the intensity or duration of disturbance is lower, and with active restoration programs (Crouzeilles and Curran 2016; Palomeque et al. 2017; Pérez-Cárdenas et al. 2021). Recovery time is also impacted by the size of soil carbon and nutrient pools and seed banks (Palomeque et al. 2017; Jakovac et al. 2021). Furthermore, soils in recovering forests may benefit from nutrients originating from deadwood (Holzwarth et al. 2013). While we know that tropical forests post-disturbance recovers relatively fast, our understanding of the trajectories of different forest components and functions, such as the amount of deadwood and its decomposition rate, during recovery remains scant (Jakovac et al. 2021; Fernandez Barranco et al. 2022). Several factors may affect the amount and diversity of deadwood in agricultural lands. For example, some agricultural practices maintain retention trees for shade in pastures or as trees within agro-forestry systems (Harvey et al. 2011; Furtado et al. 1990). Local management practices such as tillage before agricultural use, harvesting of firewood or timber for house building, or logging of specific tree species, may also have legacy effects on deadwood availability (Szymañski et al. 2017). Therefore, specific trajectories of recovering forests should reflect human legacies and site-specific conditions, though these have seldom been investigated concurrently (Jakovac et al. 2021).
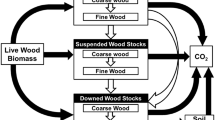
Here we used the distinction of deadwood into fine woody debris (FWD) and coarse wood debris (CWD). Thereby, CWD is defined as standing and fallen dead trees larger than 7 cm in diameter that cannot be captured by littertraps, including sound, and rotting snags, stumps, logs and large branches (Harmon et al. 1986; Idol et al. 2001). Small deadwood pieces, such as small branches and twigs (i.e., fine woody debris, FWD), are an often overlooked component of soil ecology. FWD is a vital ecological resource in agricultural systems, where humans remove pieces of large deadwood CWD as firewood, construction timber, or to make working on the soil possible with animals or machines. Besides functioning as an additional carbon storage, FWD is important for myriad litter-dwelling species, including invertebrates, fungi, and bacteria that benefit from a woody substrate with large surface area to volume ratios (Heilmann-Clausen and Christensen 2004; Brabcova et al. 2022; Ibarra-Isassi et al. 2021). Fungi (and likely the invertebrate fauna feeding upon them) living in FWD within the forest matrix are particularly responsive to microclimate variables, such as those found in forest gaps (Bässler et al. 2010). Then, FWD increases the heterogeneity of forest soil ecosystems (Armbrecht et al. 2004). Despite their high ecological and socio-economic importance, FWD has seldom been quantified in tropical settings and even less so in agricultural landscapes or recovering forests. This study aims to assess the impacts of agricultural activity on the volume and diversity of CWD and the mass of FWD in Canandé, a tropical lowland Chocó rainforest of northern Ecuador. Specifically, we asked how land management legacy in a chronosequence from cacao plantations and pastures to secondary forests recovering ≤ 37 years, to old-growth forests affects the diversity (Siitonen index based on type, diameter category, and decay stage) and volume of CWD (deadwood with ≥ 7 cm diameter) and FWD (deadwood with < 7 cm diameter). Such insights have critical conservation implications for one of the most endangered ecosystems of Ecuador (Myers et al. 2000; Gonzalez-Jaramillo et al. 2016). Based on the general observation that deadwood amount increases with the growing stock of a forest (Christensen 2014; Müller and Bütler 2010), we expect a continuous increase of CWD and FWD from intensive agricultural habitats along the gradient of forest recovery. Furthermore, as old-growth forests comprise large-diameter trees, diverse tree species, and facilitate long-lasting decomposition processes offering a wide range of habitat niches (Grove 2001; Müller et al. 2010) we expect the highest diversity of CWD in old-growth forests. Finally, we expect a steeper gradient of deadwood recovery from pastures to old-growth forests than from cacao plantations due to the higher initial tree density already present in cacao plantations.
Materials and methods
Study area
This study was carried out in Canandé and Tesoro Escondido reserves, Esmeraldas Province, in the Chocó region in northern Ecuador. We conducted our study in the framework of 64 study plots selected by the research unit REASSEMBLY (www.reassembly.de) – a collaborative Ecuadorian-German research approach to study the shift of species networks along a tropical forest recovery gradient. Our plots followed a chronosequence of forest recovery and included (i) active pastures (PA) and cacao plantations (CA) (n = 6 plots of each); (ii) secondary forests originating from formerly used pastures and cacao plantations in an early stage of recovery (pasture recovery PR1, n = 10 plots, mean = 5.8 years, sd = 4, range 0–11 years; and cacao recovery: CR1, n = 11 plots, mean = 6.5 years, sd = 4.1, range 0–14 years), (iii) secondary forests in a late stage of recovery (pasture recovery PR2, n = 8 plots, mean = 25.75 years, sd = 6.8, range 19–37 years; and CR2, n = 7 plots, mean = 27.4 years, sd = 6.8, range 19–37 years), and (iv) old-growth forests OG (n = 16), with no indication of recent use by humans at the time of the study (Figs. 1 and 2 and Table 1). The plots were between 159 to 615 m above sea level, with mean annual temperature of ca. 22–23 °C and annual precipitation of 3000–6000 mm with a dry season between June-July and October–November based on the nearest weather stations 20–50 km away (Santo Domingo: 2800 mm, Luis Vargas Torres: 6200 mm). All recovery processes were natural after abandonment, without active planting.
In each of the 64 plots, we calculated CWD volume by the following formula:
where V was volume (m3) d was diameter (cm), and L was the length (cm) for all objects of CWD ≥ 7 cm diameter. Volume was sampled per area within a circle of 0.1 ha around the center of each plot (Dörfler et al. 2018). Only tree parts within the circle were considered. We estimated CWD diversity per plot using the Siitonen Index (Siitonen et al. 2000), which counts the number of different deadwood types providing habitat for species with different niches. In our case, the Siitonen Index was calculated using five deadwood qualities (1 = fallen whole tree, 2 = standing whole tree, 3 = standing broken tree, 4 = rootstock, 5 = downed branches and trunks), five diameter classes (class 1 = 7–15 cm, 2 = 16–30 cm, 3 = 31–60 cm, 4 = 61–99 cm, 5 = 100–220 cm); and four decay classes (1 = newly dead, 2 = beginning of decomposition, 3 = advanced decomposition, and 4 = strongly decayed) (according to Albrecht 1991). The Siitonen Index (hereafter CWD diversity) of a plot then ranges from 0 (no deadwood) to 14 (presence of deadwood pieces of all qualities, decay and diameter classes). FWD mass was assessed by collecting and weighing all wood pieces in the field (< 7 cm diameter) in a 5 × 5 m square. Unfortunately, we were not able to identify the tree species of the objects because of the decomposition stage and the high tree diversity in general (> 100 tree species/ha). To estimate the mass of deadwood per m3 per hectare in total for further comparisons with literature, we assessed the mass for FWD per ha by as following.
The value 1200 kg/m3 is commonly used in calculations of fresh wood of European beech in calculations of firewood as a rule of thumb. As European beech wood shows very similar density of 0.69 as estimated as species weighted estimate of wood density in Amazonian forests (Fearnside 1997) this assumption seems justified. However, we are fully aware that high density wood is underrepresented in the input of woody debris but overrepresented relative to its input as it decomposes slowly. To compare deadwood amounts reported in the literature as Mg/ha with our estimates in m3/ha and because individual tree species specific density estimates were not available, we divided the m3/ha by 0.271 according to Gora et al. (2019) as measured mean value across tropical tree species in the lowland Chocó forest.
Data analysis
All analyses were conducted using R Statistical Software (v. 4.2.2, R Core Team 2021). First, we used linear models to test our prediction of increasing deadwood (CWD volume and diversity, and FWD mass) along the forest recovery gradient, according to land use. Prior to analyses, we log-transformed the values of CWD volume and FWD mass to reach a normal distribution. We did not include old-growth forests because they do not have a known recovery age. Second, we used linear models to test the relationship between deadwood and recovery categories. For this second set of models, we divided sampling plots into seven recovery categories, from active pasture and cacao (PA and CA) to early recovery plots (PR1 and CR1), to advanced recovery plots (PR2 and CR2), to old-growth plots (OG). We consistently ranked pasture first because of a vast lack of trees compared to cacao. Here the models test if along the seven ranks one of our dependent variables significantly increases following a linear, quadratic or other models. The advantage of this approach was the option to include old-growth with a clear rank but no age into a statistic model. To check the residuals of these models for spatial independence, we used cross-correlograms provided in the package ncf (Bjørnstad 2020).
Results
Along the chronosequence of the 64 plots, we recorded a total of 778 coarse woody debris (CWD) pieces with a total volume of 338.1 m3 (mean = 5.28m3/0.1 ha, range 0.06–31.91) and 287 kg of fine woody debris (mean mass = 4.48 kg/25m2, range 0 to 17.6). The relative distribution of CWD quality types varied across the recovery gradient (Fig. 3). CWD pieces found in active agricultural plots consisted mostly of snags and downed branches, with the highest percentage of snags at cacao plantations (43%) and of downed branches at pastures (72%). A small but constant volume of CWD was also present in active agricultural plots in the form of standing trees and rootstock (Fig. 3a), the latter also present in early recovery plots. On the contrary, CWD in the form of fallen trees occupied an increasingly higher percentage of total CWD with increasing age of recovery plots, with the highest percentage (56%) found in old-growth forests. The distribution of the decomposition class of CWD pieces was similar across the recovery gradient, with most CWD pieces in early (stage 2, mean = 55%) and advanced (stage 3, mean = 41%) stages of decomposition (Fig. 3b). Newly dead CWD (stage 1) was only found in early recovery plots, while strongly decayed CWD (stage 4) was mostly present in old-growth forests (Fig. 3b), though both types were only found in minor amounts. Putting these values together, mean CWD diversity was 7.44/0.1 ha (range 2–14) per plot.
CWD volume and diversity did not change across the recovery chronosequence in either former pasture or cacao plots (Fig. 4). FWD showed legacy effects of previous land use, increasing continuously in forests recovering from pasture land uses but remaining constant in forests recovering from cacao plantations (Fig. 4c). Allocation of plots to discrete categories (i.e., active cacao and pasture plots, recovering secondary forests of two age classes with different land use legacies, and old-growth forests) allowed us to include old-growth plots in the model (Fig. 4d–f). Even here, CWD volume did not significantly increase along the ordered succession categories, but CWD diversity and FWD mass did. The spline correlograms of the residuals of our CWD volume, CWD diversity, and FWD mass models support spatial independence (Fig. 5).
Correlation of the coarse woody debris volume and diversity as well as fine woody debris mass with recovery time (a–c), separated by the history of management (cacao or pasture). The old-growth values are added for illustrative purposes and are jittered to increase visibility. They have had no value for years since recovery. For deadwood measures along the ordered categories (d–f) p values are extracted from a linear model using the ordered categories as displayed. All p values correspond to linear models only. Other models were not significant. Note the log transformation of the dependent variable. To avoid zeros in our log-transformed value we added 1 to all FWD values
Discussion
In contrast to our expectation of continuous recovery (i.e., increase) of deadwood along the chronosequence, we found a high variation in both CWD volume and diversity in recovering cacao plantations and pastures over the first decades of recovery. CWD volume did not increase across the recovery gradient even when old-growth forests were included in the analyses, though the diversity of CWD increased. As opposed to CWD, FWD showed a linear increase along the chronosequence in secondary forests that were formerly pastures but not cacao plantations.
Volume of deadwood along the succession gradient
The lack of an apparent increase of CWD volume along our succession gradient might be surprising, considering that in cacao plantations and especially pastures, the volume of wood locked in standing trees is highly reduced compared to old-growth forests. Besides, our results revealed high variability in local volumes of CWD in early succession stages, contrasting with more homogenous volumes of CWD in old-growth forests. In principle, tree mortality shows a U-shaped relationship with tree diameter (Holzwarth et al. 2013), with the lowest mortality found in mid-sized or mid-aged trees, which are more common in mid-aged successional stages. The amount of CWD observed in different succession stages in temperate rainforests in Chile first decreased and then increased over time, with high volumes of deadwood in early successional stages caused by fire and logging disturbance by locals (Carmona et al. 2002). A similar U-shaped pattern was observed along a succession gradient in an evergreen broad-leaved forest in Eastern China, with the lowest values in mid-successional stages (Yan et al. 2007). Deadwood in secondary tropical forests recovering from pastures in Costa Rica increased with active restoration by planting relative to natural regeneration, though there was high variation between recovering forests (Fernandez Barrancos et al. 2022). In contrast to these, our study found a high mean volume of CWD along the whole succession gradient and little support for a U-shaped relationship. This seems to be caused by the origin of deadwood. In contrast to legacy effects in the studies mentioned before, in our plots deadwood objects are not residuals from clearing the old-growth forest, but simply caused by mortality of retention trees in the agriculture area or former retention trees in recovering forests. This more continuous supply together with a tradition not to remove single huge dead trees from farmland due the effort (pers. observation and information by local people) might cause the lack of a trend in CWD along the gradient.
The lack of a clear trend in deadwood volume along the recovery gradient in Canandé could reflect both, the history of anthropogenic disturbance in the study area as well as natural deadwood processes in moist tropical forests. Firstly, the time since recovery in our study landscape might be too short to fully explore patterns of CWD volume in late recovery stages. A decrease in CWD volume may be observed in the phase between 40 and 100 years, when the total amount of growing stock begins to reach that found in old-growth forests. For example, it took more than 50 years for deadwood volumes in naturally regenerating former coffee plantations in a premontane forest landscape in Costa Rica to reach that of old-growth forests (Fernandez Barrancos et al. 2022). However, even tree species composition might affect the pattern significantly with pioneer species such as Ochroma spp. or Cecropia spp. adding deadwood already after 20–30 years. The recent influx of human settlers to Canandé occurred in the 1960s and 70 s; thus, with max 37 years of age, patches of recovering forest measured in this study, and existing naturally within the landscape, are likely to be too young to have reached these later stages. A decrease of CWD later in succession might be supported by the lower median values in the late recovery stage of both agricultural types (Fig. 4d), even though these differences to early succession stages were slight. Secondly, U-shape patterns may be challenging to see in warm and moist tropical forests, which exhibit a much faster regrowth of trees and higher decomposition rates of deadwood coupled with increased activities of insects and fungi. The global study by Seibold et al. (2021) suggests decomposition times below a decade under the climate conditions of our study. It is therefore unlikely, although possible for individual tree species, that the deadwood on our agricultural and early succession plots originates from the clearing phase. Here further research on deadwood in old-growths and clearings is required to clarify the role of residuals from natural forests. In summary, the regular occurrence of dead retention trees in active agriculture and early regrowth forests, together with fast tree growth and rapid decomposition rates may blur the distinct dynamics of deadwood in recovery gradients in the tropics. Similarly considerable amounts of deadwood even in early succession forests have been shown for disturbance gradients in temperate rainforests (Carmona et al. 2002; Yan et al. 2007), or in temperate and boreal forests in general (Hilmers et al. 2018; Priewasser et al. 2013).
Diversity of deadwood along the succession gradient
Experiments and field surveys have shown that the diversity of deadwood and the associated higher habitat heterogeneity has a greater impact on the biodiversity of saproxylic organisms than the amount of deadwood alone (Heidrich et al. 2020; Seibold et al. 2016). The variation we observed among CWD categories are in accordance with our prior expectations. Higher percentages of CWD in the form of rootstock, snags, and whole dead trees standing in active agricultural and early recovery plots are expected either when trees are felled to make room for cacao and pasture, or when shade-retention and cacao trees die.
The high percentage of CWD surveyed in decay stages 2 and 3 (94%), with freshly fallen wood (decay stage 1, 3% of all CWD) found only in early recovery plots, could support the idea that recovery time in the system might be too short to fully understand deadwood patterns along the successional gradient. While reduced volume of CWD in category 4 is expected in active plantations and early recovery plots (Lachat et al. 2006), the relative lack of highly decomposed wood in late recovery plots and old-growth forests may signal that wood stocks in live trees are still growing (Baker et al. 2007). While some pioneers should already die off before biomass accumulation stops, these decompose so quickly that there is hardly any accumulation of volume (Seibold et al. 2021). Moreover, a relative lack of highly decomposed wood in old-growth relative to early recovery may also indicate greater decomposition rates which may be even higher if wood density at the forest or stand level is low, soils are nutrient rich, and insect activity is high.
The high mean diversity of deadwood between plots as well as across the whole gradient, and especially at very early stages, provides high within-plot heterogeneity and reshapes our understanding of the importance of agricultural landscapes for saproxylic organisms. A variation in deadwood availability between recovering forest plots provides diverse habitats for saproxylic organisms (Müller et al. 2020; Seibold et al. 2022). However, despite the high variation in deadwood in agriculture and early recovering plots, we found on average lower values of deadwood diversity than in old-growth forests, as found in other tropical forests (Fernandez Barrancos et al. 2022; Navarrete et al. 2016). The question arises of how this result was affected by our choice of categories in quantifying the diversity index. We included diameter class, decay stage, and deadwood type; which are all known to significantly affect the composition of saproxylic organisms (Grove 2002; Müller et al. 2020; Seibold et al. 2015). Host identity is another important driver of habitat heterogeneity in deadwood and has been proven relevant for saproxylic beetles and particularly for fungi (Ødegaard 2006; Müller et al. 2020). However, we ignored tree identity because of the difficulty of assessing the identity of decomposed trees in the field. The inventory of tree diversity in our plots is currently underway and has shown an increase in alpha diversity along our succession gradient (Sebastian Escobar, pers. comm.). This suggests that true deadwood diversity in our old-growth forests is even more diverse compared to early succession forests than shown in this study.
The unexpected and generally high diversity of deadwood in small-scale agriculture found in the present study underlines the value even of early succession forest as habitat for saproxylic organisms. In temperate regions, agricultural habitats with a fixed proportion of overmature trees and deadwood have been identified as important for saproxylic organisms that have been able to survive there (Horak 2017; Miklín et al. 2018). In some cases, more endangered deadwood organisms are found in such pastures in Europe today than in production forests (Stokland et al. 2012). In the tropics, these aspects have received little attention so far. Many of the agricultural areas are also still relatively young, and it remains to be seen whether this type of use will also provide valuable deadwood habitats in the long term. This is only possible with a continuous supply of overmature trees in pastures or cacao plantations.
Amount of fine woody debris along the gradient
FWD is regularly understudied in forest ecosystems but is an essential habitat for many species and plays an important role in nutrient cycling. Particularly for fungi it has been shown that the diversity is higher in FWD, when compared to the same amounts of CWD, due to high competition among fungi within deadwood objects (Bässler et al 2010; Heilmann-Clausen and Christensen 2004). Unfortunately, logistics forced us to quantify the FWD in the field measuring the wet FWD, which is imperfect since its mass may change due to changes in daily weather conditions. However, as FWD was in majority in contact with soil and our survey was conducted during the same season and because decay stage, another important variable affecting density, could not be measured, we do not believe significant impacts on the results by our rapid assessment. Of course, drying some subsamples from all plots would be preferred for validation; such data were not available here. Nevertheless, based on our FWD values we found a consistent increase in the amount of FWD along the regeneration gradient, measured as wet mass, in contrast to CWD. Furthermore, we, found a strong effect of land use legacy on the amount of FWD, with a strong increase in FWD found in pastures after abandonment, while FWD in cacao was present throughout the recovery gradient. We suggest several arguments for why FWD increased along the gradient in pastures but not in cacao. First, increasing FWD in pastures might result from increasing tree stem density with recovery time, resulting in higher competition and subsequent higher self-thinning mortality of small-diameter trees (Holzwarth et al. 2013). In contrast, cacao trees already present in active plantations may provide important amounts of FWD to the forest floor as twigs and branches, additionally with mortality of small-diameter trees due to competition, as has been observed in coffee plantations (Ibarra-Isassi et al. 2021). Another reason for the low amount of FWD in active and early recovering pasture plots might be the pressure of domestic animals such as cattle and horses. Both feed not only on grass but also on leaves and shoots of small-diameter trees, and they are much more abundant on pastures than cacao plantations (Grella et al. in prep). Therefore, the active pastures might be rather poor in this resource and would need time during the recovery process for trees to recolonize and start to lose branches or die via competition among trees. In our study, FWD amounts in recovering pastures reached levels similar to old-growth forests after about 30 years. Beyond regeneration dynamics, this study supports the idea that FWD could serve as an essential resource for taxa relevant to ecosystem processes (Lachat et al. 2006; Miklín et al. 2018; Priewasser et al. 2013; Sebek et al. 2013), such as termites (Davies et al. 2003), or ants (Armbrecht et al. 2004; Ibarra-Isassi et al. 2021) that regularly occur or recover fast in both agricultural systems (Hoenle et al. 2022).
Deadwood amount in tropical forests of different land use
Deadwood and its related biodiversity remains understudied in the tropics compared to the temperate and boreal biome, and the few available studies reflect variability in CWD volumes across sites. The quantity of deadwood in old-growth plots at our study site (mean = 76.07 m3/ha, range = 15.03–227.31 m3/ha) was lower compared to values found in a separate study in native old-growth plots in Hoja Blanca, located in northwestern Ecuador (range 135.9 ± 18.3 m3/ha; Gale 2000). The higher values of Hoja Blanca with respect to Canandé may be partly due to regional differences, as Gale (2000) sampled CWD only in one area of 10 ha. The volume of CWD found in Canandé is higher than in premontane tropical forests in Costa Rica (45 m3/ha; Fernandez Barrancos et al. 2022) and Peru (44 m3/ha, Baker et al. 2007), but lower than that of found in wet rainforests in Brazil and tropical lowland rainforests in Colombia (range 130–190 m3/ha; Keller et al. 2004; Navarrete et al. 2016). However, the pattern of CWD observed in our study in the NW Ecuadorian Chocó rainforest corresponds well with the detailed assessment of deadwood amounts in Panama representing the northern part of the Chocó rainforest (Gora et al. 2019).
Our study is one of the first to assess CWD and FWD across agricultural lands and recovering forests in Ecuador. Fernandez Barrancos et al. (2022) showed a logistic recovery of deadwood amount in both active and natural recovering plots. Importantly, they found that active recovery plots of ~ 16 years old accumulated 40% of CWD volume compared to old-growth forests, which was much higher than only 1.7% of CWD volume recovered in natural recovery plots. Here the high potential of natural recovery in our study systems seems to show similar effects due to the high number of regrowing trees. However, in contrast to our study, they found no higher amounts of deadwood in the first years of recovery, indicating a lack of overmature retention trees and more consequent removal of trees. In general, several studies show remarkably high amounts of deadwood also in early succession forests with about 60 m3/ha in lowland tropical forests of Australia (Grove 2001) and up to > 300 m3/ha in evergreen moist forests in Brazil (Keller et al. 2004). Even for mid and late-succession tropical forests, high values are reported within 30–50 m3/ha in Australia (Grove 2001) and with more than 200 m3/ha in tropical wet montane forests in Venezuela (Delaney et al. 1998).
Conclusions
Our survey of deadwood along a recovery gradient from cacao plantations and pastures over about four decades of recovery to old-growth forests revealed a surprisingly high amount and diversity of deadwood in agriculture and recovering forests. Our data provide new baselines for estimating carbon released from deadwood in tropical ecosystems. Refining these values in the tropics is important because these ecosystems contribute > 90% of annual carbon release from deadwood globally (Seibold et al. 2021). Moreover, the deadwood resources found in our study underline that deadwood in the tropics should be considered in habitat management in old-growth forests and human-transformed landscapes. Further studies on the importance of the diversity of deadwood objects to saproxylic communities in recovering and old-growth forests are now required. Moreover, these data suggest that deadwood measures might be an important indicator in tropical forests for selecting new conservation areas and for biodiversity credits in the future. Here existing systems for biodiversity monitoring and financial incentives from temperate and boreal forests may serve as a blueprint.
References
Albrecht L (1991) Die Bedeutung des toten Holzes im Wald. Forstw Cbl 110:106–113. https://doi.org/10.1007/BF02741245
Armbrecht I, Perfecto I, Vandermeer J (2004) Enigmatic biodiversity correlations: ant diversity responds to diverse resources. Science 304(5668):284–286. https://doi.org/10.1126/science.1094981
Baker T, Honorio E, Phillips O, Martin J, van der Heijden G, Garcia M, Silva J (2007) Low stocks of coarse woody debris in a southwest Amazonian forest. Oecologia 152:495–504. https://doi.org/10.1007/s00442-007-0667-5
Bässler C, Müller J, Dziock F, Brandl R (2010) Effects of resource availability and climate on the diversity of wood-decaying fungi. J Ecol 98(4):822–832. https://doi.org/10.1111/j.1365-2745.2010.01669.x
Bjørnstad ON (2020) NCF: A package for analyzing spatial (cross-) covariance. https://CRAN.R-project.org/package=ncf
Brabcová V, Tláskal V, Lepinay C, Zrustová P, Eichlerová I, Štursová M, Müller J, Brandl R, Bässler C, Baldrian P (2022) Fungal community development in decomposing fine deadwood is largely affected by microclimate. Front Microbiol 13:835274. https://doi.org/10.3389/fmicb.2022.835274
Brin A, Bouget C, Brustel H, Jactel H (2011) Diameter of downed woody debris does matter for saproxylic beetle assemblages in temperate oak and pine forests. J Insect Conserv 15:653–669. https://doi.org/10.1007/s10841-010-9364-5
Brown S (1997) Estimating biomass and biomass change of tropical forests: a primer. FAO Forestry Paper 134:1–55. https://doi.org/10.1016/j.foreco.2007.06.016
Carmona MR, Armesto JJ, Aravena JC, Perez CA (2002) Coarse woody debris biomass in successional and primary temperate forests in Chiloe Island, Chile. For Ecol Manage 164:265–275. https://doi.org/10.1016/S0378-1127(01)00602-8
Christensen NL (2014) A historical perspective on forest succession and its relevance to ecosystem restoration and conservation practice in North America. For Ecol Manage 330:312–322
Crouzeilles R, Curran M (2016) Which landscape size best predicts the influence of forest cover on restoration success? A global meta-analysis on the scale of effect. J App Ecol 53:440–448. https://doi.org/10.1111/1365-2664.12590
Davies R, Hernández L, Eggleton P, Didham R, Fagan L, Winchester N (2003) Environmental and spatial influences upon species composition of a termite assemblage across Neotropical forest islands. J Trop Ecol 19(5):509–524. https://doi.org/10.1017/S0266467403003560
Dörfler I, Mueller J, Weisser W, Gossner MM, Seibold S (2018) Deadwood enrichment combining integrative and segregative conservation elements enhances biodiversity of multiple taxa in managed forests. Biol Conserv 228:70–78
Delaney M, Brown S, Lug AE, Torres-Lezarna A, Quintero NB (1998) The quantity and turnover of dead wood in permanent forest plots in six life zones of Venezuela. Biotropica 30(1):2–11
Fearnside PM (1997) Wood density for estimating forest biomass in Brazilian Amazonia. For Ecol Manage 90(1997):59–87. https://doi.org/10.1016/S0378-1127(96)03840-6
Fernandez Barrancos EP, Marquis RJ, Reid JL (2022) Restoration plantations accelerate dead wood accumulation in tropical premontane forests. For Ecol Manage 508:120015. https://doi.org/10.1016/j.foreco.2022.120015
Furtado JI, Morgan WB, Pfafflin JR, Ruddle K (1990) Tropical resources: ecology and development, 1st edn. Routledge, Abingdon
Gale N (2000) The aftermath of tree death: coarse woody debris and the topography in four tropical rain forests. Can J for Res 30(9):1489–1493. https://doi.org/10.1139/x00-071
Gonzalez-Jaramillo V, Fries A, Rollenbeck R, Paladines J, Oñate-Valdivieso F, Bendix J (2016) Assessment of deforestation during the last decades in Ecuador using NOAA-AVHRR satellite data. Erdkunde 70:217–235. https://doi.org/10.3112/erdkunde.2016.03.02
Gora EM, Kneale RC, Larjavaara M, Muller-Landau HC (2019) Dead wood necromass in a moist tropical forest: stocks, fluxes, and spatiotemporal variability. Ecosystems 22:1189–1205. https://doi.org/10.1007/s10021-019-00341-5
Graf M, Seibold S, Gossner MM, Hagge J, Ingmar Weiß I, Claus Bässler C, Müller J (2022) Coverage based diversity estimates of facultative saproxylic species highlight the importance of deadwood for biodiversity. For Ecol Manage 517:120275. https://doi.org/10.1016/j.foreco.2022.120275
Grove SJ (2001) Extent and composition of dead wood in Australian woodland tropical rainforest with different management histories. For Ecol Manage 154:35–53. https://doi.org/10.1016/S0378-1127(00)00618-6
Grove SJ (2002) Saproxylic Insect Ecology and the Sustainable Management of Forests. Annu Rev Ecol Sys 33:1–23
Harmon ME, Franklin JF, Swanson FJ, Sollins P, Gregory SV, Lattin JD, Anderson NH, Cline SP, Aumen NG, Sedell JR, Lienkaemper GW (1986) Ecology of coarse woody debris in temperate ecosystems. Adv Ecol Res 15:133–302. https://doi.org/10.1016/S0065-2504(08)60121-X
Harvey CA, Villanueva C, Esquivel H, Gómez R, Ibrahimb M, Lopez M, Martinez J, Munoz D, Restrepo C, Saénz JC, Villacís J, Sinclair FL (2011) Conservation value of dispersed tree cover threatened by pasture management. For Ecol Manage 261(10):1664–1674
Heidrich L, Bae S, Levick S, Seibold S, Weisser W, Krzystek P, Magdon P, Nauss T, Schall P, Serebryanyk A, Wöllauer S, Ammer C, Bässler C, Doerfler I, Fischer M, Gossner MM, Heurich M, Hothorn T, Jung K, Kreft H, Schulze E-D, Simons N, Thorn S, Müller J (2020) Heterogeneity–diversity relationships differ between and within trophic levels in temperate forests. Nat Ecol Evol 4:1204–1212. https://doi.org/10.1038/s41559-020-1245-z
Heilmann-Clausen J, Christensen M (2004) Does size matter? On the importance of various dead wood fractions for fungal diversity in Danish beech forests. For Ecol Manage 201(1):105–117. https://doi.org/10.1016/j.foreco.2004.07.010
Hérault B, Beauchêne J, Muller F, Wagner F, Baraloto C, Blanc L, Martin J (2010) Modeling decay rates of dead wood in a Neotropical forest. Oecologia 164:243–251. https://doi.org/10.1007/s00442-010-1602-8
Hilmers T, Friess N, Bässler C, Heurich M, Brandl R, Pretzsch H, Seidl R, Müller J (2018) Biodiversity along temperate forest succession. J Appl Ecol 55:2756–2766. https://doi.org/10.1111/1365-2664.13238
Hoenle PO, Donoso DA, Argoti A, Staab M, von Beeren C, Blüthgen N (2022) Rapid ant community reassembly in a Neotropical forest: recovery dynamics and land-use legacy. Ecol Appl 32(4):e2559. https://doi.org/10.1002/eap.2559
Holzwarth F, Kahl A, Bauhus J, Wirth C (2013) Many ways to die–partitioning tree mortality dynamics in a near-natural mixed deciduous forest. J Ecol 101:220–230. https://doi.org/10.1111/1365-2745.12015
Horak J (2017) Insect ecology and veteran trees. J Insect Conserv 21(1):1–5
Ibarra-Isassi J, Handa IT, Arenas-Clavijo A, Escobar-Ramírez S, Armbrecht I, Lessard J-P (2021) Shade-growing practices lessen the impact of coffee plantations on multiple dimensions of ant diversity. J Appl Ecol 58:919–930. https://doi.org/10.1111/1365-2664.13842
Idol TW, Figler RA, Pope PE, Ponder FJr, (2001) Characterization of coarse woody debris across a 100-year chronosequence of upland oak-hickory forests. For Ecol Manage 149:153–161. https://doi.org/10.1016/S0378-1127(00)00536-3
Jakovac CC, Junqueira AB, Crouzeilles R, Peña-Claros M, Mesquita RCG, Bongers F (2021) The role of land-use history in driving successional pathways and its implications for the restoration of tropical forests. Biol Rev 96:1114–1134. https://doi.org/10.1111/brv.12694
Keller M, Palace M, Asner G, Pereira R, Silva J (2004) Coarse woody debris in undisturbed and logged forests in the eastern Brazilian Amazon. Glob Change Biol 10:784–795. https://doi.org/10.1111/j.1529-8817.2003.00770.x
Kissing LB, Powers JS (2010) Coarse woody debris stocks as a function of forest type and stand age in Costa Rican tropical dry forest: long-lasting legacies of previous land use. J Trop Ecol 26:467–471. https://doi.org/10.1017/S0266467410000131
Lachat T, Nagel P, Cakpo Y, Attignon S, Goergen G, Sinsin B (2006) Dead wood and saproxylic beetle assemblages in a semi-deciduous forest in Southern Benin. For Ecol Manage 225:27–38
Loughlin NJD, Gosling W, Mothes P, Montoya E (2018) Ecological consequences of post-Columbian indigenous depopulation in the Andean-Amazonian corridor. Nature Ecol Evol 2:1233–1236. https://doi.org/10.1038/s41559-018-0602-7
Magnusson RI, Tietema A, Cornelissen JHC, Hefting MM, Kalbitz K (2016) Sequestration of carbon from coarse woody debris in forest soils. For Ecol Manage 377:1–15. https://doi.org/10.1016/j.foreco.2016.06.033
Miklín J, Sebek P, Hauck D, Konvicka O, Cizek L (2018) Past levels of canopy closure affect the occurrence of veteran trees and flagship saproxylic beetles. Divers Distrib 24(2):208–218. https://doi.org/10.1111/ddi.12670
Mueller AD, Islebe GA, Anselmetti FS, Ariztegui D, Brenner M, Hodell DA, Hajdas I, Hamann Y, Haug GH, Kennett DJ (2010) Recovery of the forest ecosystem in the tropical lowlands of northern Guatemala after disintegration of classic Maya polities. Geology 38(6):523–526. https://doi.org/10.1130/G30797.1
Müller J, Bütler R (2010) A review of habitat thresholds for dead wood: a baseline for management recommendations in European forests. Eur J for Res 129:981–992. https://doi.org/10.1007/s10342-010-0400-5
Müller J, Noss RF, Bussler H, Brandl R (2010) Learning from a “benign neglect strategy” in a national park: response of saproxylic beetles to dead wood accumulation. Biol Conserv 143(11):2559–2569. https://doi.org/10.1016/j.biocon.2010.06.024
Müller J, Ulyshen M, Seibold S, Cadotte M, Chao A, Bässler C, Vogel S, Hagge J, Weiß I, Baldrian P, Tláskal V, Thorn S (2020) Primary determinants of communities in deadwood vary among taxa but are regionally consistent. Oikos 129:1579–1588. https://doi.org/10.1111/oik.07335
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858. https://doi.org/10.1038/35002501
Navarrete D, Sitch S, Aragão LEOC, Pedroni L, Duque A (2016) Conversion from forests to pastures in the Colombian Amazon leads to differences in dead wood dynamics depending on land management practices. J Environ Manage 171:42–51. https://doi.org/10.1016/j.jenvman.2016.01.037
Ødegaard F (2006) Host specificity, alpha- and beta-diversity of phytophagous beetles in two tropical forests in panama. Biodiv Conserv 15:83–105. https://doi.org/10.1007/s10531-004-3106-5
Palomeque X, Gunter S, Siddons D, Hildebrandt P, Stimm B, Aguirre N, Arias R, Weber M (2017) Natural or assisted succession as approach of forest recovery on abandoned lands with different land use history in the Andes of southern Ecuador. New for 48:643–662
Pérez-Cárdenas N, Mora F, Arreola-Villa F, Arroyo-Rodríguez V, Balvanera P, Flores-Casas R, Navarrete-Pacheco A, Ortega-Huerta MA (2021) Effects of landscape composition and site land-use intensity on secondary succession in a tropical dry forest. For Ecol Manage 482:118818. https://doi.org/10.1016/j.foreco.2020.118818
Pfeifer M, Lefebvre V, Turner E, Cusack J, Khoo MS, Chey VK, Peni M, Ewers RM (2015) Deadwood biomass: an underestimated carbon stock in degraded tropical forests? Environ Res Lett 10(4):044019. https://doi.org/10.1088/1748-9326/10/4/044019
Piperno DR, McMichael CH, Pitman NCA, Guevara Andino JE, Ríos Paredes M, Heijink BM, Torres-Montenegro LA (2021) A 5,000-year vegetation and fire history for tierra firme forests in the Medio Putumayo-Algodón watersheds, northeastern Peru. Proc Nat Acad Sci 118(40):e2022213118. https://doi.org/10.1073/pnas.2022213118
Priewasser K, Brang P, Bachofen H, Bugmann H, Wohlgemuth T (2013) Impacts of salvage-logging on the status of deadwood after windthrow in Swiss forests. E J for Res 132:231–240. https://doi.org/10.1007/s10342-012-0670-1
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rozendaal DMA, Bongers F, Aide TM, Alvarez-Dávila E, Ascarrunz N, Balvanera P, Becknell JM, Bentos TV, Brancalion PHS, Cabral GAL, Calvo-Rodriguez S, Chave J, César RG, Chazdon RL, Condit R, Dalling JS, de Almeida-Cortez JS, de Jong B, de Oliveira A, Denslow JS, Dent DH, DeWalt SJ, Dupuy JM, Durán SM, Dutrieux LP, Espírito-Santo MM, Fandino MC, Fernandes GW, Finegan B, García H, Gonzalez N, Moser VG, Hall JS, Hernández-Stefanoni JL, Hubbell S, Jakovac CC, Hernández AJ, Junqueira AB, Kennard D, Larpin D, Letcher SG, Licona J-C, Lebrija-Trejos E, Marín-Spiotta E, Martínez-Ramos M, Massoca PES, Meave JA, Mesquita RCG, Mora F, Müller SC, Muñoz R, de Oliveira Neto SN, Norden N, Nunes YRF, Ochoa-Gaona S, Ortiz-Malavassi E, Ostertag R, Peña-Claros M, Pérez-García EA, Piotto D, Powers JS, Aguilar-Cano J, Rodriguez-Buritica S, Rodríguez-Velázquez J, Romero-Romero MA, Ruíz J, Sanchez-Azofeifa A, de Almeida AS, Silver WL, Schwartz NB, Thomas WW, Toledo M, Uriarte M, de Sá Sampaio EV, van Breugel M, van der Wal H, Martins SV, Veloso MDM, Vester HFM, Vicentini A, Vieira ICG, Villa P, Williamson GB, Zanini KJ, Zimmerman J, Poorter L (2019) Biodiversity recovery of neotropical secondary forests. Sci Adv 5(3):3114. https://doi.org/10.1126/sciadv.aau3114
Sebek P, Altman J, Platek M, Cizek L (2013) Is active management the key to the conservation of saproxylic biodiversity? Pollarding promotes the formation of tree hollows. PlosOne 8:e60456
Seibold S, Bässler C, Brandl R, Gossner MM, Thorn S, Ulyshen MD, Müller J (2015) Experimental studies of dead-wood biodiversity—a review identifying global gaps in knowledge. Biol Conserv 191:139–149. https://doi.org/10.1016/j.biocon.2015.06.006
Seibold S, Bässler C, Brandl R, Büche B, Szallies A, Thorn S, Ulyshen MD, Müller J (2016) Microclimate and habitat heterogeneity as the major drivers of beetle diversity in dead wood. J Appl Ecol 53:934–943. https://doi.org/10.1111/1365-2664.12607
Seibold S, Rammer W, Hothorn T, Seidl R, Ulyshen MD, Lorz J, Cadotte MW, Lindenmayer DB, Adhikari YP, Aragón R, Bae S, Baldrian P, Varandi HB, Barlow J, Bässler C, Beauchêne J, Berenguer E, Bergamin RS, Birkemoe T, Boros G, Brandl R, Brustel H, Burton PJ, Cakpo-Tossou YT, Castro J, Cateau E, Cobb TP, Farwig N, Fernández RD, Firn J, Gan KS, González G, Gossner MM, Habel JC, Hébert C, Heibl C, Heikkala O, Hemp A, Hemp C, Hjältén J, Hotes S, Kouki J, Lachat T, Liu J, Liu Y, Luo Y, Macandog DM, Martina PE, Mukul SA, Nachin B, Nisbet K, O’Halloran J, Oxbrough A, Pandey JN, Pavlíček T, Pawson SM, Rakotondranary JS, Ramanamanjato J-B, Rossi L, Schmidl J, Schulze M, Seaton S, Stone MJ, Stork NE, Suran B, Sverdrup-Thygeson A, Thorn S, Thyagarajan G, Wardlaw TJ, Weisser WW, Yoon S, Zhang N, Müller J (2021) The contribution of insects to global forest deadwood decomposition. Nature 597:77–81. https://doi.org/10.1038/s41586-021-03740-8
Seibold S, Weisser WW, Ambarli D, Gossner MM, Mori AS, Cadotte MW, Hagge J, Bässler C, Thorn S (2022) Drivers of community assembly change during succession in wood-decomposing beetle communities. J Anim Ecol 92:1–14. https://doi.org/10.1111/1365-2656.13843
Siitonen J, Martikainen P, Punttila P, Rauh J (2000) Coarse woody debris and stand characteristics in mature managed and old-growth boreal mesic forests in southern Finland. For Ecol Manage 128:211–225. https://doi.org/10.1016/S0378-1127(99)00148-6
Stokland JN, Siitonen J, Jonsson BG (2012) Biodiversity in dead wood. Cambridge University Press, Cambridge
Szymañski C, Fontana G, Sanguinetti J (2017) Natural and anthropogenic influences on coarse woody debris stocks in Nothofagus-Araucaria forests of northern Patagonia, Argentina. Austral Ecol 42:48–60. https://doi.org/10.1111/aec.12400
Yan ER, Wang XH, Huang JJ, Zeng FR, Gong L (2007) Long-lasting legacy of forest succession and forest management: Characteristics of coarse woody debris in an evergreen broad-leaved forest of Eastern China. For Ecol Manage 252:98–107. https://doi.org/10.1016/j.foreco.2007.06.016
Acknowledgements
Fieldwork: We thank the Fundación Jocotoco (Martin Schaefer) and Fundación Tesoro Escondido (Citlalli Morelos-Juarez) for logistic support and permission to do research on their reserves. We would like to especially acknowledge local support from the staff in the Canandé and Tesoro Escondido reserves: Katrin Krauth (manager of the Chocó Lab); Bryan Tamayo (plot manager); Lady Condoy, Leonardo de la Cruz, Franklin Quintero, Jefferson Tacuri, Silvia Vélez; Patricio Encarnacion, Ariel Villigu and Patricio Paredes. We especially thank Yadira Giler for her support. Project coordination: We thank Nico Blüthgen, Martin Schaefer, María-José Endara, Juan Guevara, Sebastián Escobar, Julieta Muñoz, and Karin Römer for project coordination and administration. We thank Markku Larjavaara and one anonymous reviewer for valuable comments on an earlier version of our manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was supported by the Deutsche Forschungsgemeinschaft (DFG) funded Research Unit REASSEMBLY (FOR 5207; sub-project SP7, with grants FE631/13-1 and MU3621/10-1).
Author information
Authors and Affiliations
Contributions
Author Contributions Ana Falconí López: Conceptualization, Methodology, Formal analysis, Investigation, Data Curation, Writing - Original Draft, Writing - Review & Editing, Visualization Nina Grella: Investigation, Data Curation, Writing - Review & Editing David A. Donoso: Conceptualization, Writing - Review & Editing Heike Feldhaar: Supervision, Writing - Review & Editing Constance J. Tremlett: Writing - Review & Editing Jörg Müller: Conceptualization, Methodology, Supervision, Formal analysis, Writing - Review & Editing
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Gediminas Brazaitis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Falconí-López, A., Grella, N., Donoso, D.A. et al. Patterns of deadwood amount and deadwood diversity along a natural forest recovery gradient from agriculture to old-growth lowland tropical forests. Eur J Forest Res (2024). https://doi.org/10.1007/s10342-024-01671-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10342-024-01671-3