Abstract
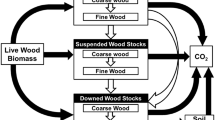
Downed dead wood (DDW) in forest ecosystems is a C pool whose net flux is governed by a complex of natural and anthropogenic processes and is critical to the management of the entire forest C pool. As empirical examination of DDW C net flux has rarely been conducted across large scales, the goal of this study was to use a remeasured inventory of DDW C and ancillary forest attributes to assess C net flux across forests of the Eastern US. Stocks associated with large fine woody debris (diameter 2.6–7.6 cm) decreased over time (−0.11 Mg ha−1 year−1), while stocks of larger-sized coarse DDW increased (0.02 Mg ha−1 year−1). Stocks of total DDW C decreased (−0.14 Mg ha−1 year−1), while standing dead and live tree stocks both increased, 0.01 and 0.44 Mg ha−1 year−1, respectively. The spatial distribution of DDW C stock change was highly heterogeneous with random forests model results indicating that management history, live tree stocking, natural disturbance, and growing degree days only partially explain stock change. Natural disturbances drove substantial C transfers from the live tree pool (≈−4 Mg ha−1 year−1) to the standing dead tree pool (≈3 Mg ha−1 year−1) with only a minimal increase in DDW C stocks (≈1 Mg ha−1 year−1) in lower decay classes, suggesting a delayed transfer of C to the DDW pool. The assessment and management of DDW C flux is complicated by the diversity of natural and anthropogenic forces that drive their dynamics with the scale and timing of flux among forest C pools remaining a large knowledge gap.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Downed dead wood (DDW) C pools in forests may be viewed as a “stage of transition” for C in living woody biomass as it is converted from a living state to the atmosphere, forest floor and soil, or other C pools through the processes of decomposition, combustion, or lateral transfer (Harmon et al. 1986). These processes vary considerably across temporal and spatial scales (Bradford et al. 2014) and ultimately result in individual pieces of DDW serving as sources of C to the atmosphere with emission rates dependent on individual pathways of decomposition, combustion, or physical degradation (Cornwell et al. 2009). With the emergence of bioenergy economies (Malmsheimer et al. 2008, 2011; Hurteau et al. 2013), the need for an understanding of the dynamics of DDW C net flux has increased in importance given that DDW in the early stages of decomposition can be considered a feedstock (Lippke et al. 2011). As a result, the fate of DDW C, whether decomposing in forests versus immediately released to the atmosphere, is one focal point of contemporary forest management (Benjamin et al. 2010; Berger et al. 2013) and bioenergy debates (Schlamadinger et al. 1995; Becker et al. 2009; Sathre and Gustavsson 2011; Zanchi et al. 2012; Nepal et al. 2014). Such policy and management issues require broad-scale assessments of DDW C net flux; however, the limited field observations on DDW C net flux in forest ecosystems over large scales have restricted such efforts.
Despite DDW C net flux being identified as critical to the management of forest ecosystems (McKinley et al. 2011; Harmon et al. 2011a) and associated policies, numerous knowledge gaps remain. While several studies have examined DDW C flux, the majority of this work has been conducted at small scales (e.g., Wang et al. 2002; Jönsson et al. 2011) using empirical information across short time steps or across larger scales with simulation approaches (Bradford et al. 2014). For example, using a laboratory experiment, Wang et al. (2002) found that decay class and temperature, among other factors, were useful surrogates for determining DDW C flux. Forrester et al. (2012) found that DDW C flux in small canopy gaps exceeded that from DDW in closed canopy (over a 2-year period), yet only 23 % of the variation could be explained by temperature and moisture regimes. Jönsson et al. (2011) found that DDW accumulation in Swedish forests was highly episodic due to stochastic stand disturbances. The central focus of most studies has been to identify rates of DDW C pool accretion or depletion over short time steps as an indicator of DDW C net sequestration/emission. A limited number of studies have explored the topic of DDW C net flux over longer time steps and/or larger spatial scales using empirical information. Russell et al. (2014a) estimated ecosystem-level C flux using species-specific estimates of coarse DDW (CDDW) decay, highlighting the usefulness of implementing field observations with modeling approaches to inform DDW dynamics. While these studies have examined the C dynamics within a single ecosystem pool (e.g., DDW), little is known about how C net flux is influenced as woody C transitions into various pools in combination with disturbance events (Harmon et al. 2011a) (e.g., from standing dead to CDDW or branch shedding to fine DDW).
An added complexity in refining the understanding of DDW C net flux is the experimental designs themselves. In many studies, C flux refers to a direct measurement of CO2 efflux from DDW directly to the atmosphere (for examples see Harmon et al. 2011a). In contrast, alternative experimental designs directly measure C stocks at two points in time with the difference serving as an indicator of potential net flux to the atmosphere (Bond-Lamberty and Gower 2008; Woodall 2010). For example, Woodall (2010) conducted an examination of DDW C stock change with a limited number of observations and covariates (e.g., site and stand attributes) across the forests of the upper Midwest in the US. An added complexity with such approaches are the omitted transfers of DDW C to ecosystem components out of the study population (e.g., DDW below a minimum size for measurement) and/or other pools (e.g., forest floor or organic soil) (Russell et al. 2014a). Although a number of studies may restrict assessments of net flux to “terrestrial pool to atmosphere” transfers of C, a coarser yet more inclusive examination of DDW C stock change may provide additional insight into C flux from one pool to another (e.g., lateral transfer from standing live trees to DDW). More recently, spatially extensive forest inventories have begun to measure the C attributes of ecosystem pools that are additional to standing live trees (Woodall et al. 2010; 2013). For example, Gray and Whittier (2014) found that DDW C stocks increased on national forests in the Pacific Northwest of the US in areas that were partially harvested. Barrett (2014) found that DDW C stocks in Chugach and Tongass National Forests in southeastern Alaska increased over time. Examinations of the relationships between patterns found in emerging DDW C inventories with other inventory attributes (e.g., standing live tree C stocks and disturbance history) may prove valuable in refining our understanding of DDW C net flux dynamics, particularly at broad spatial extents.
The goal of this study was to explore the dynamics of DDW C net flux in relation to other woody C pools (e.g., standing dead and live trees) and site/stand attributes (e.g., management treatments and stand age) in forests of the Eastern US. Our specific objectives were to:
-
1.
Evaluate how DDW C net flux is influenced by live-tree forest structural conditions [e.g., stand relative density (RD) and live tree abundance].
-
2.
Examine DDW C net flux by DDW piece size and decay class for undisturbed, silviculturally treated, and naturally disturbed sites.
-
3.
Examine C net flux in standing live trees, standing dead trees, and DDW components [fine woody debris (FWD), slightly and highly decayed CDDW] by classes of change in RD for undisturbed, silviculturally treated, and disturbed sites.
Materials and methods
Field sample protocols
Our study relies on data compiled from the USDA Forest Service’s Forest Inventory and Analysis (FIA) program, which is the primary source for information about the extent, condition, status, and trends of forest resources across the US (Smith et al. 2009). FIA applies a nationally consistent sampling protocol using a systematic design covering all ownerships across the US (national sample intensity is one plot per 2,428 ha) using a three-phase inventory (Bechtold and Patterson 2005). To accomplish the objectives of systematic distribution of plots across the nation, the FIA sampling design is based on a tessellation of the US into hexagons approximately 2,428 ha in size with at least one permanent plot established in each hexagon (i.e., national base sample intensity). In phase 1, the population of interest is stratified (e.g., forest canopy cover classes) and plots are assigned to strata to increase the precision of estimates. Remotely sensed data may also be used to determine if plot locations have forest cover; only forested land is measured in the field component of the inventory and is defined as being at least 10 % stocked with tree species, at least 0.4 ha in size, and at least 36.6 m wide (Bechtold and Patterson 2005). In phase 2, tree and site attributes are measured for plots established in each hexagon. FIA inventory plots consist of four, 7.32 m fixed-radius subplots spaced 36.6 m apart in a triangular arrangement with one subplot in the center (US Forest Service 2007a; Woudenberg et al. 2010). All trees (live and standing dead) with a diameter at breast height of at least 12.7 cm are inventoried on forested subplots. A standing dead tree is considered DDW when the lean angle of its central bole is greater than 45° from vertical [US Department of Agriculture (USDA) Forest Service 2007a]. Within each sub-plot, a 2.07 m microplot offset 3.66 m from sub-plot center is established where only live trees with a diameter at breast height between 2.5 and 12.7 cm and seedlings are inventoried.
DDW is sampled on a subset of phase 2 inventory plots during the third phase of FIA’s inventory (USDA Forest Service 2007b; Woodall and Monleon 2008; Woodall et al. 2011b). The national base sample intensity for the phase 3 inventory is 1/16th of phase 2 plots representing 38,848 ha per plot (Woodall et al. 2011b). Given the varied semantics associated with examinations of DDW, we define DDW as detrital components of forest ecosystems comprising fine and coarse woody debris, including coarse woody debris that may be aggregated in piles due to logging activities. CDDW are pieces, or portions of pieces, of DDW with a minimum diameter of at least 7.6 cm at the point of intersection with a sampling transect (assuming line-intercept sampling protocols) and a length of at least 0.91 m. CDDW pieces must be detached from a bole and/or not be self-supported by a root system with a lean angle more than 45° from vertical (Woodall and Monleon 2008). FWD are pieces, or portions of pieces, of DDW with a diameter less than 7.6 cm at the point of intersection with a sampling transect excluding dead branches attached to standing trees, dead foliage, or bark fragments. CDDW is sampled using the line-intersect method on transects radiating from each FIA’s four subplot centers (at angles 30°, 150° and 270°). Each subplot has three 7.32-m transects, totaling 87.8 m for a fully forested inventory plot.
Information collected for every CDDW piece intersected by transects are transect diameter, length, small- and large-end diameters, decay class, and species (Westfall and Woodall 2007). Transect diameter is the diameter of a downed woody piece at the point of intersection with a sampling transect. Decay class is a subjective determination of the stage of decay for an individual log based on presence of branches, bark, collapse, and softness (e.g., Westfall and Woodall 2007). Decay class 1 is the least decayed (freshly fallen log), while decay class 5 is an extremely decayed log. The species of each fallen log is identified through determination of species-specific bark, branching, bud, and wood composition attributes (excluding decay class 5). FWD with transect diameters less than 0.6 cm (small FWD) and 0.6–2.5 cm (medium FWD) are tallied separately on a 1.83-m slope-distance transect (4.27–6.09 m on the 150° transect). FWD with transect diameters of 2.5–7.6 cm (large FWD) are tallied on a 3.05-m slope-distance transect (4.27–7.32 m on the 150° transect).
Data and analysis
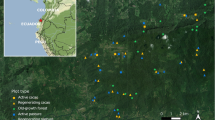
Field data (USDA Forest Service 2007a, b) for this study were taken entirely from the FIA database (Woudenberg et al. 2010; Woodall et al. 2010) using the forest inventory in 37 states of the Eastern US (Fig. 1) for a total of 1,010 plots first established between 2002 and 2005 and remeasured 5 years later from 2005 to 2010. Only fully forested single-condition plots at both measurement times were considered. The associated field data are available for download at the following site: http://fiatools.fs.fed.us (FIA Datamart, USDA 2014).
This study used a stock change approach to estimate C net flux where the total stock of C by component (e.g., FWD) was estimated at two points in time with the difference divided by the remeasurement period (in months) serving as an estimate of average annual net flux (C Mg ha−1 year−1). Positive values are taken to indicate sequestration (i.e., assimilation from the atmosphere) or cases where heterotrophic respiration was less than the input of new dead wood C. In contrast, negative values are suggestive of emission and/or high levels of decomposition relative to new dead wood C input. Additionally, the term “net flux” will refer to any movement of C either between pools or to the atmosphere. The C stocks of FWD and CDDW were determined through application of volume estimators detailed in Woodall and Monleon (2008). Briefly, the volume of FWD is estimated per unit area, and then converted to an estimate of necromass using a bulk density and decay reduction factor based on USDA forest type (Woudenberg et al. 2010). An estimate of FWD C is then obtained by multiplying the necromass estimate by 0.5. For CDDW the volume is determined using Smalian’s volume formula for every CDDW piece and then used in an estimator to yield volume per unit area at the plot level. Volume is then converted into necromass and hence C mass through the use of decay reduction factors, bulk density, and C conversion based on a piece’s unique species and decay class (Harmon et al. 2008). To facilitate further evaluation of decay on CDDW C net flux, CDDW C stock changes were summed by slight decay (decay classes 1 and 2) and high decay (decay classes 3, 4, and 5).
Standing dead and live tree C stocks were also calculated in this study using a series of steps based on the national field inventories where they are measured on the same plots. First, the standing dead/live gross volume was calculated based on regional volume models (Woodall et al. 2011a). Second, a tree’s sound volume was calculated based on regional volume models along with merchantable stem deductions due to rough, rotten, and missing cull. Third, the sound volume was converted to bole biomass using species-specific wood density values (Miles and Smith 2009; Woudenberg et al. 2010). For standing dead trees, to account for the reduced wood density due to decay, decay reduction factors by decay class and hardwood/softwood were applied (Harmon et al. 2011b). Fourth, total tree biomass was calculated using the component ratio method (CRM) (Woodall et al. 2011a). Briefly, the CRM facilitates calculation of tree component (e.g., tops and limbs) biomass as a proportion of the bole biomass based on component proportions from Jenkins et al. (2003). For standing dead trees, which may lack some or all of the components calculated using CRM (e.g., loss of limbs), structural reduction factors were applied by decay class and hardwood/softwood (Domke et al. 2011). The fifth and final step was the conversion of standing dead/live total biomass to C mass assuming 50 % C content of woody biomass (or in the case of standing dead trees necromass). Belowground estimates of coarse root C were not included.
To gauge general trends in C net flux, the mean, range, and SD of C net flux by DDW size and CDDW decay class was estimated by USDA forest type group (hereafter referred to as “forest type”) (Woudenberg et al. 2010). To ascertain the most parsimonious approach to quantifying the dynamics associated with DDW C flux, we used nonparametric random forests (Breimen 2001) in R (Liaw and Wiener 2002) to identify site (e.g., elevation), stand attributes (e.g., RD), and climate variables (e.g., Rehfeldt 2006) that explained the most variation in total DDW C flux (for explanations of all variables see Appendix 1). For ecological data, random forests models can offer high classification accuracy and provide a method for assessing the relative importance of predictor variables (Cutler et al. 2007). In this method, classification trees were taken as independently sampled bootstraps of the data (Breimen 2001). We ranked the contribution of each variable in explaining the spatial variability in DDW C flux by examining its importance score (% IncMSE), a measure of the decrease in model accuracy upon removing the variable from an “out-of-bag” sample (Breimen 2001). We employed a full random forest model which included all variables (Table 1). Subsequently, we fit a parsimonious model using the five best-performing variables by dropping the least influential variable (as measured by its % IncMSE) iteratively until five variables remained. We sampled 500 regression trees for each random forests model to ensure an adequate number of bootstrap samples from the DDW data. Given the randomness associated with each regression, we report the mean values for % IncMSE and associated fit statistics after performing 20 random forests regressions.
Based on random forests model results, treatment/disturbance and RD classes were used in subsequent analyses to parse the DDW C net flux dynamics. Plots were assessed as being treated (largely by specific management actions such as pre-commercial or commercial harvest activities), disturbed (largely by natural means such as wind, fire, insects/disease, or floods), or neither treated nor disturbed at time 2 which covers all or part of the remeasurement period [for exact classifications see Woudenberg et al. (2010)]. Due to insufficient sample size (n = 13 observations), plots that were both disturbed and treated were not evaluated in this study (representing 1.3 % of the data). Means and SEs of SDs were tested (α = 0.05) for C net flux (Mg ha−1 year−1) by DDW size class and CDDW decay class for the three classes of disturbance/treatment combinations using linear mixed-effects models. To evaluate DDW C net flux in the context of other forest C pools, means and SEs of annual C net flux (Mg ha−1 year−1) were determined for FWD, low and high decay CDDW, standing dead, and standing live by classes of changes in a stand’s RD and classes of disturbance/treatment combinations, with differences tested using linear mixed-effects models. To assess the influence of forest disturbance and treatment on C net flux values, a generalized linear mixed-model analysis was conducted. Data were analyzed with plot history considered as a fixed effect (i.e., disturbed, treated, or neither). Random effects were specified on the intercepts for each plot to account for variation not explained in fixed effect that might be related to C net flux. The GLIMMIX procedure was used (SAS Institute 2011) to denote SDs. This procedure employed pseudo-likelihood estimation based on linearization where the exponential distribution of the data conditioned on the random effects was specified.
Results
Across all Eastern US forest types, mean annual C net flux of DDW was minimal with no clear trends in mean annual net flux by size class or forest type (Table 1). The range in net flux increased as the DDW size class increased. In contrast, C flux of CDDW suggested an increase in this stock for the moderate decay class of 3 across the range of decay examined (Table 2). The largest stock increase was decay class 3 in the oak/gum/cypress forest type (mean and SD = 0.29, 0.79 Mg ha−1 year−1, respectively) while the largest potential emission was decay class 4 in the same type (mean and SD = −0.19, 0.79 Mg ha−1 year−1, respectively). When mean annual C net flux was examined by dead and live pools, standing live trees stocks increased at 0.44 Mg ha−1 year−1 (SD = 2.54 Mg ha−1 year−1) while standing dead was nearly neutral at 0.01 Mg ha−1 year−1 (SD = 0.90 Mg ha−1 year−1) and DDW was a potential emission at −0.14 Mg ha−1 year−1 (SD = 1.12 Mg ha−1 year−1) (Table 3). The oak/gum/cypress forest type had the highest estimated mean net flux for standing live at 1.31 Mg ha−1 year−1 (SD = 2.06 Mg ha−1 year−1) while oak/hickory had the largest estimated range in live flux at −30.05 Mg ha−1 year−1. The only forest type that had a potential increase in its stock was oak/pine at 0.15 Mg ha−1 year−1 (SD = 1.13 Mg ha−1 year−1). When viewed spatially, components of DDW C net flux are highly variable across the eastern forests (Fig. 1). The estimated annual net flux of FWD is approximately normally distributed with no obvious spatial trends (Fig. 1a). The distribution of CDDW annual C net flux is somewhat similar, being normally distributed but with potential reductions in C stocks in Maine and the upper peninsula of Michigan (Fig. 1b). In contrast, the estimated annual C net flux of standing live trees is skewed towards stock increases with only spatially random occurrences of potential emission across the Eastern US (Fig. 1c). Finally, estimated standing dead tree C net flux was approximately normally distributed; however, with greater stock loss relative to gains in regions, such as lower Michigan and areas of Maine.
The results from the random forests models provided a parsimonious approach for further evaluation of DDW C net flux dynamics. The top five variables in the full model (Fig. 2a; root mean square error = 1.11 Mg ha−1 year−1) of all variables in terms of importance scores for explaining DDW C net flux were treatment, aboveground live tree volume, growing degree days, RD, and growing season growing degree days, with importance scores ranging from 8 to 11 (%IncMSE). The top variables in the “five best” variables model (root mean square error = 1.12 Mg ha−1 year−1) were treatment, stand density index, aboveground live tree biomass, aboveground live tree volume, and live trees per hectare, with importance scores ranging from 9 to 22 (% IncMSE) (Fig. 2b). The random forests model estimates did match the majority of DDW C net flux annual observations (±2 Mg ha−1 year−1). When viewed spatially, random forest model residuals across the eastern forests suggest numerous areas where additional factors (not included in our models) may be driving the variability in DDW C net flux dynamics (Fig. 3a, b). The spatial distribution in residuals was random across the Eastern US with a greater range in residuals for the five best variable model reflected spatially with potentially large stochastic C transfer events (e.g., rapid decay, harvest, combustion, or blowdown).
As the random forests model results suggested that treatments and amount of live volume/biomass explained the largest amounts of variation in DDW C net flux, flux was further explored using classes of treatment/disturbance and RD. RD indicates the level of live tree stocking across diverse forest ecosystems (Woodall et al. 2005) and provides a useful metric for understanding DDW dynamics across levels of stocking in contrast to SDI which is an absolute measure of tree density. DDW C net flux was examined by classes of DDW size and the presence/absence of treatment/disturbance (Fig. 4). Large FWD (diameter 2.6–7.6 cm) was estimated as having the largest decreases in C stocks on sites lacking treatment or natural disturbance. In contrast, moderately sized CDDW (diameter 25.4–50.8 cm) was estimated as having the largest increases in C stocks on treated sites. Broadly speaking, on treated sites (where harvests typically occur) DDW C stocks often increased. In contrast, on sites lacking treatment/disturbance DDW C stocks minimally decreased over time. Generalized linear mixed-effects models indicated that DDW C net flux was significantly (p-value <0.05) different from zero in all but the smallest and largest size classes (Table 4).
Coarse DDW C net flux was examined by classes of CDDW decay and treatment/disturbance (Fig. 5). Coarse DDW pieces in advanced stages of decay (decay classes 3–5) were estimated as having slight decreases in their associated C stocks (−0.05 Mg ha−1 year−1) on sites lacking treatment or natural disturbance. In contrast, fresh and lightly decayed (decay classes 1 and 2) CDDW pieces were estimated as having increased in their C stocks (0.15 Mg ha−1 year−1) on treated sites. The largest increase in C stocks was found with moderately decayed CDDW pieces on naturally disturbed sites (0.23 Mg ha−1 year−1). Mixed-effects models indicated that DDW C net flux was significantly (p-value <0.05) different from zero among decay classes (Table 4). When viewed across all classes, the least decayed CDDW pieces often experienced increases in their C stocks over time due to lateral transfer from other pools (e.g., live tree mortality), while CDDW pieces in advanced stages of decay often experienced reductions in their associated C stocks due to decay and lack of transfer of highly decayed material from other pools (e.g., standing dead trees).
Given the differences in C net flux between the size and state of decay of DDW, mean annual C net flux in FWD, slight decay CDDW, high decay CDDW, standing dead trees, and live trees was examined by classes of treatment/disturbance and changes in a stand’s RD (Fig. 6). For undisturbed sites, the live tree pool experienced the only C stock change of note with nearly −2 Mg ha−1 year−1 of C stock loss on sites that had a reduction in RD of more than 0.1, while a concomitant C stock gain of 2 Mg ha−1 year−1 on sites that had RD increase by more than 0.1 (Fig. 6a). On treated sites, the live tree pool had the largest C stock reduction at more than −6 Mg ha−1 year−1 in sites where RD decreased by more than 0.1 (Fig. 6b). On such sites the lightly decayed CDDW had a slight concomitant increase in C stocks of 0.2 Mg ha−1 year−1. On sites that were naturally disturbed and had a reduction of RD exceeding 0.1, the live tree pool had the largest C stock loss at approximately −4 Mg ha−1 year−1 but with a comparable C stock increase of standing dead trees of nearly 3 Mg ha−1 year−1 (Fig. 6c). For treatment and disturbance classes, mixed-effect model results indicated that only standing dead and live tree C net flux was significantly different from zero (p-value <0.05) (Table 5). For changes in a stand’s RD, results indicated that C net flux was significantly different from zero for the pools of slight decay CDDW and standing dead/live trees (p-value <0.05).
Discussion
The C dynamics of the DDW C pool, not unlike other forest ecosystem C pools, is a complex of processes that exchange C between the atmosphere, living organisms, dead biomass, and soils (Harmon et al. 2011a; Bradford et al. 2014). Akin to the live tree biomass pool which receives C fixed by leaves (i.e., foliage pool), processes that increase DDW C stocks are lateral transfers from other pools (live and dead biomass) (Krankina and Harmon 1995; Gough et al. 2007; Bond-Lamberty and Gower 2008). Processes that decrease DDW C stocks are combustion, heterotrophic respiration [i.e., interaction with the saproxylic community (Stokland et al. 2012)], and lateral transfer to other pools (e.g., litter) through processes such as physical degradation and biological transformation (Harmon et al. 1986; Bond-Lamberty and Gower 2008; Cornwell et al. 2009). Numerous results from this study highlight this complex nature of DDW C net flux especially in disturbed or treated stands where processes such as heterotrophic respiration are vast knowledge gaps (Harmon et al. 2011a). The results also highlight the difficulty in determining the emission or sequestration status (i.e., net flux) of the DDW C pool. Scale is an important aspect of DDW C flux dynamics. At the scale of an individual piece of DDW one can assume a net emission of C over time as the piece decays or combusts. However, at the population scale DDW C stocks may greatly increase due to natural disturbances representing a C transfer from live pools. Such transfers potentially increase the uncertainty associated with the C flux status of a forest landscape. It can be suggested that refining the understanding of transfer, combustion, biological transformation, and heterotrophic respiration of the DDW C pool is key to a more comprehensive assessment of the forest C cycle. The stochastic nature of DDW C stock changes both over time and space, highlighted by this work, and indicates that managing DDW C stocks does not solely include increasing associated C stocks, as this C comes from lateral transfer of C already sequestered from the atmosphere. Instead, we propose exploring management paradigms focused on reducing the rate and/or risk of C emissions from DDW (i.e., increasing the residence time of DDW). Such a paradigm would need to be evaluated in the context of secondary effects on other critical ecosystem functions such as decomposers (Stokland et al. 2012), fire risk, and nutrient cycling that could constrain forest net primary productivity.
Despite the complexities associated with the DDW C cycle, the results of our study indicated that DDW C net flux varies at a landscape scale by very general metrics of stand and site attributes such as treatments, disturbance, and RD (i.e., stage of stand development). Woodall and Liknes (2008) and Russell et al. (2014b) found that DDW attributes across the entire US varied by climatic regions, while both Sturtevant et al. (1997) and Woodall and Westfall (2009) found that stand stocking metrics indicated self-thinning mortality as important inputs to the DDW pool. However, as noted by random forests results in this study (i.e., minimal reduction in model error and the associated map of residuals), DDW C net flux at small scales are highly variable and dependent on unique site and stand history factors. This is consistent with concepts advanced by Bradford et al. (2014), who suggest that local scale attributes such as fungal communities are primary controls over decomposition rates and subsequent forest C cycling dynamics. Moreover, Russell et al. (2014a) demonstrates that attributes of individual DDW pieces are primary factors in the residence time of DDW C as residence time often is one of the largest sources of uncertainty in C cycle models (Friend et al. 2014).
Our findings add to the growing body of knowledge that DDW attributes at the stand level are highly heterogeneous owing to stochastic management and disturbance events (Fraver et al. 2002; Bond-Lamberty and Gower 2008; Aakala 2011; Bradford et al. 2014). However, at very broad spatial scales, trends in DDW C net flux align with previous findings where DDW C net flux was found to be relatively minimal (Gough et al. 2007) when compared to the standing live pool (while at the scale of a DDW individual piece there could be substantial flux due to heterotrophic respiration). In addition, our study found standing dead tree C to have a lower rate of C net flux than DDW. This may be the result of potentially slower rates of heterotrophic respiration associated with standing dead trees due to lower moisture content and fungal activity compared to DDW that often contacts the ground (Bond-Lamberty and Gower 2008).
Within the DDW pool itself, a number of general trends of C net flux were found. First, FWD appears to exhibit rapid accumulation on an annual basis [e.g., branch shedding (Oliver and Larson 1996)]. Although Lee et al. (1997) did not explicitly examine FWD, they found the accumulation of post-fire DDW was in part driven by senescence and self-thinning processes. In contrast, as seen in our study, the rapid accumulation of larger sizes of DDW, such as CDDW, often followed treatments/disturbance. C stocks of more highly decayed CDDW typically exhibited reductions over time, while fresh CDDW pieces on disturbed/treated sites tended to increase their associated C stocks. In undisturbed forests, the accumulation of CDDW C stocks may be from fallen standing dead trees in advanced stages of decay as a result of stand development (i.e., self-thinning mortality) (Sturtevant et al. 1997; Lee et al. 1997; Woodall and Westfall 2009). This was found in our study on undisturbed sites that had reductions in RD but with an input of CDDW in advanced stages of decay. This was in contrast to treated sites in our study where reductions in RD and live tree C stocks only resulted in a slight increase in CDDW C stocks. These patterns highlight the need for refined C stock accounting to fully account for the C implications of harvested wood products (Lippke et al. 2011), whereby live tree C did not transfer to the DDW pool as was clearly seen in naturally disturbed sites. One more dynamic of DDW C net flux can be hypothesized. The relationship between the CDDW C flux of highly decayed versus slightly decayed pieces appears to be negative on treated sites (i.e., considerable input of fresh CDDW during harvest activities). Such a metric could serve as an indicator of stand dynamics (i.e., natural versus anthropocentric disturbances) that drives a component of DDW C net flux.
Given the results from this study, management treatments and natural disturbances had the most pronounced effect on DDW C stock changes (i.e., net flux). Given the expectation that forests will face more extreme disturbance events (Joyce et al. 2014) in the future, coupled with pressure for active forest management, we can hypothesize that DDW C stocks in the near future will increase. This can already be seen in the northern Rocky Mountains where standing dead tree C stocks may have increased due to regional tree mortality (Woodall et al. 2012; Wilson et al. 2013). If this current trend is sustained, then the importance of maintaining DDW C stocks may be elevated in the context of offsetting greenhouse gas emissions. If future forest production (i.e., live tree C stock accumulation) is limited due to droughts (Zhao and Running 2010) or climate extremes, then research efforts should focus on refining our understanding of DDW C stock residence (Friend et al. 2014) and associated forest management activities. Towards these ends, C flux should be examined over longer time periods and/or with intensified plot networks to facilitate greater statistical power (Westfall et al. 2013) while refined assessments of C in the forest floor and soil should augment our knowledge of C transfer in and out of the DDW C pool.
Conclusion
C storage is just one of many ecosystem services (e.g., habitat for saproxylic organisms or substrate for tree establishment) provided by DDW. The results of our study offer a complementary paradigm: the diversity of these services and the large number of organisms involved contribute to the complex dynamics of DDW C flux. Some components of the DDW C pool, such as FWD, may represent a small stock but are highly dynamic over short time steps. Other components, such as large CDDW, can be a considerable stock especially following disturbance, which suggests active monitoring of their residence times and emission rates especially if tree mortality is expected to increase under future scenarios of climate change. Regardless, all components of DDW C stocks are affected by stand development and disturbance processes (e.g., mortality events and wildfires) that interact with a community of decomposers and other C pools that ultimately determine their resulting net flux. Assessments of DDW C stocks and associated net flux may be broadly estimated at large scales. However, their assessment and management at the stand level would be greatly improved by addressing the knowledge gaps of scale and timeframes of DDW C net flux to the atmosphere and laterally to other pools.
References
Aakala T (2011) Temporal variability of deadwood volume and quality in boreal old-growth forests. Silva Fenn 45:969–981
Barrett TM (2014) Storage and flux of carbon in live trees, snags, and logs in the Chugach and Tongass National Forests. USDA Forest Service general technical report PNW-889. USDA, Portland, OR
Bechtold WA, Patterson PL (Eds) (2005) The enhanced forest inventory and analysis program—national sampling design and estimation procedures. USDA Forest Service general technical report SRS-80. USDA, Asheville, NC
Becker DR, Skog K, Hellman A, Halvorsen KE, Mace T (2009) An outlook for sustainable forest bioenergy production in the Lake States. Energy Policy 37:5687–5693
Benjamin JG, Lilieholm RJ, Coup CE (2010) Forest biomass harvesting in the northeast: a special-needs operation? North J Appl For 27:45–49
Berger AL, Palik B, D’Amato AW, Fraver S, Bradford JB, Nislow K, King D, Brooks RT (2013) Ecological impacts of energy-wood harvests: lessons from whole-tree harvesting and natural disturbance. J For 111:139–153
Bond-Lamberty B, Gower ST (2008) Decomposition and fragmentation of coarse woody debris: revisiting a boreal black spruce chronosequence. Ecosystems 11:831–840
Bradford MA, Warren RJ II, Baldrian P, Crowther TW, Maynard DS, Oldfield EE, Wieder WR, Wood SA, King JR (2014) Climate fails to predict wood decomposition at regional scales. Nat Clim Change 4:625–630
Breimen L (2001) Random forests. Mach Learn 45:5–32
Cornwell WK et al (2009) Plant traits and wood fates across the globe. Glob Change Biol 15:2431–2449
Cutler DR, Edwards TC, Beard KH, Cutler A, Hess KT, Gibson J, Lawler JJ (2007) Random forests for classification in ecology. Ecology 88:2783–2792
Domke GM, Woodall CW, Smith JE (2011) Accounting for density reduction and structural loss in standing dead trees: implications for forest biomass and carbon stock estimates in the US. Carbon Balance Manage 6:14
Forrester JA, Mladenoff DJ, Gower ST, Stoffel JL (2012) Interactions of temperature and moisture with respiration from coarse woody debris in experimental forest canopy gaps. For Ecol Manage 265:124–132
Fraver S, Wagner RG, Day M (2002) Dynamics of coarse woody debris following gap harvesting in the Acadian forest of central Maine, USA. Can J For Res 32(12):2094–2105
Friend AD, Lucht W, Rademacher TT, Keribin R, Betts R, Cadule P, Ciais P, Clark DB, Dankers R, Falloon PD, Ito A, Kahana R, Kleidon A, Lomas MR, Nishina K, Ostberg S, Pavlick R, Peylin P, Schaphoff S, Vuichard N, Warszawski L, Wilshire AM, Woodward FI (2014) Carbon residence time dominates uncertainty in terrestrial vegetation responses to future climate and atmospheric CO2. Proc Natl Acad Sci USA 111:3280–3285
Gough CM, Vogel CS, Kazanski C, Nagel L, Flower CE, Curtis PS (2007) Coarse woody debris and the carbon balance of a north temperate forest. For Ecol Manage 244:60–67
Gray AN, Whittier TR (2014) Carbon stocks and changes on Pacific Northwest national forests and the role of disturbance, management, and growth. For Ecol Manage 328:167–178
Harmon ME, Franklin JF, Swanson FJ, Sollins P, Gregory SV, Lattin JD, Anderson NH, Cline SP, Aumen NG, Sedell JR, Lienkaemper GW, Cromack K Jr, Cummins KW (1986) Ecology of coarse woody debris in temperate ecosystems. Adv Ecol Res 15:133–302
Harmon ME, Woodall CW, Fasth B, Sexton J (2008) Woody detritus density and density reduction factors for tree species in the US: a synthesis. General technical report 29. USDA Forest Service, Northern Research Station
Harmon ME, Bond-Lamberty B, Tang J, Vragas R (2011a) Heterotrophic respiration in disturbed forests: a review with examples from North America. J Geophys Res 116:G00K04
Harmon ME, Woodall CW, Fasth B, Sexton J, Yatkov M (2011b) Differences between standing and downed dead tree wood density reduction factors: a comparison across decay classes and tree species. Research paper 15. Northern Research Station, USDA Forest Service
Hurteau MD, Hungate BA, Koch GW, North MP, Smith GR (2013) Aligning ecology and markets in the forest carbon cycle. Front Ecol Environ 11:37–42
Jenkins JC, Chojnacky DC, Heath LS, Birdsey RA (2003) National scale biomass estimators for US tree species. For Sci 49:12–35
Jönsson MT, Fraver S, Jonsson BG (2011) Spatio-temporal variation of coarse woody debris input in woodland key habitats in central Sweden. Silva Fenn 45:957–967
Joyce LA, Running SW, Breshears DD, Dale VH, Malmsheimer RW, Sampson RN, Sohngen B, Woodall CW (2014) National climate assessment of US 2014. Ch 7: forests. In: Melillo JM, Richmond TC, Yohe GW (eds) Climate change impacts in the US: the third national climate assessment. US Global Change Research Program, pp 175–194
Krankina ON, Harmon ME (1995) Dynamics of the dead wood carbon pool in northwestern Russian boreal forests. Water Air Soil Pollut 82:227–238
Lee PC, Crites S, Nietfeld M, Nguyen HV, Stelfox JB (1997) Characteristics and origins of deadwood material in aspen-dominated boreal forests. Ecol Appl 7:691–701
Liaw A, Wiener M (2002) Classification and regression by randomForest. R News 2:18–22
Lippke B, Oneil E, Harrison R, Skog K, Gustavsson L, Sathre R (2011) Life cycle impacts of forest management and wood utilization on carbon mitigation: knowns and unknowns. Carbon Manage 2:303–333
Malmsheimer RW, Heffernan P, Brink S, Crandall D, Deneke F, Galik C, Gee E, Helms JA, McClure N, Mortimer M, Ruddell S, Smith M, Stewart J (2008) Forest management solutions for mitigating climate change in the US. J For 106:115–171
Malmsheimer RW, Bowyer JL, Fried JS, Gee E, Izlar RL, Miner RA, Munn IA, Oneil E, Stewart WC (2011) Managing forests because carbon matters: integrating energy, products, and land management policy. J For 109:S7–S50
McKinley DC, Ryan MG, Birdsey RA, Giardina CP, Harmon ME, Heath LS, Houghton RA, Jackson RB, Morrison JF, Murray BC, Pataki DE, Skog KE (2011) A synthesis of current knowledge on forests and carbon storage in the US. Ecol Appl 21:1902–1924
Miles PD, Smith WB (2009) Specific gravity and other properties of wood and bark for 156 tree species found in North America. Research note NRS-38. Northern Research Station, USDA Forest Service, Newtown Square, PA
Nepal P, Wear DN, Skog KE (2014) Net change in carbon emissions with increased wood energy use in the US. GCB Bioenergy (in press)
Oliver CD, Larson BC (1996) Forest stand dynamics.Update edition. Wiley, New York
Rehfeldt GE (2006) A spline model of climate for the western US. General technical report RMRS-165. USDA Forest Service
Russell MB, Woodall CW, Fraver S, D’Amato AW, Domke GM, Skog KE (2014a) Residence times and decay rates of downed woody debris biomass/carbon in Eastern US forests. Ecosystems 17:765–777
Russell MB, Woodall CW, D’Amato AW, Fraver S, Bradford JB (2014b) Technical note: Linking climate change and downed woody debris decomposition across forests of the eastern United States. Biogeosciences (in press)
SAS Institute (2011) SAS/STAT(R) 9.3 user’s guide. SAS Institute, Cary, NC
Sathre R, Gustavsson L (2011) Time-dependent climate benefits of using forest residues to substitute fossil fuels. Biomass Bioenery 35:2506
Schlamadinger B, Spitzer J, Kohlmaier GH, Ludeke M (1995) Carbon balance of bioenergy from logging residues. Biomass Bioenergy 8:221–234
Smith WB, Miles PD, Perry CH, Pugh SA (2009) Forest resources of the US, 2007. General technical report WO-GTR-78. US Forest Service
Stokland JN, Siitonen J, Jonsson BG (2012) Biodiversity in dead wood. Cambridge University Press, Cambridge
Sturtevant BR, Bissonette JA, Long JN, Roberts DW (1997) Coarse woody debris as a function of age, stand structure, and disturbance in boreal Newfoundland. Ecol Appl 7:702–712
USDA Forest Service (2007) Forest inventory and analysis national core field guide, vol I: field, data collection procedures for phase 2 plots, version 4.0. USDA Forest Service, Forest Inventory and Analysis, Washington, DC. URL: http://www.fia.fs.fed.us/library/. Accessed December 2010
USDA Forest Service (2007) Forest inventory and analysis phase 3 guide—downed woody materials, version 4.0. USDA Forest Service, Forest Inventory and Analysis, Washington, DC. URL: http://www.fia.fs.fed.us/library/. Accessed January 2011
USDA Forest Service (2014) Forest inventory and analysis national program—data and tools–FIA data mart, FIADB version 5.1. USDA Forest Service, Washington, DC. http://apps.fs.fed.us/fiadb-downloads/datamart.html . Accessed 24 June 2014
Wang C, Bond-Lamberty B, Gower ST (2002) Environmental controls on carbon dioxide flux from black spruce coarse woody debris. Oecologia 132:374–381
Westfall JA, Woodall CW (2007) Measurement repeatability of a large-scale inventory of forest fuels. For Ecol Manage 253:171–176
Westfall JA, Woodall CW, Hatfield MA (2013) A statistical power analysis of woody carbon flux from forest inventory data. Clim Change 118:919–931
Wilson BT, Woodall CW, Griffith D (2013) Imputing forest carbon stock estimates from inventory plots to a nationally continuous coverage. Carbon Balance Manage 8:1
Woodall CW (2010) Carbon flux of down woody materials in forests of the north central US. Int J For Res 2010:413703
Woodall CW, Liknes GC (2008) Climatic regions as an indicator of forest coarse and fine woody carbon stocks in the US. Carbon Balance Manage 3:5
Woodall CW, Monleon VJ (2008) Sampling, estimation, and analysis procedures for the down woody materials indicator. General technical report NRS-22. USDA Forest Service, Newtown Square, PA
Woodall CW, Westfall JA (2009) Relationships between the stocking levels of live trees and dead tree attributes in forests of the US. For Ecol Manage 258:2602–2608
Woodall CW, Miles PD, Vissage JS (2005) Determining maximum stand density index in mixed species stands for strategic-scale stocking assessments. For Ecol Manage 216:367–377
Woodall CW, Conkling BL, Amacher MC, Coulston JW, Jovan S, Perry CH, Schulz B, Smith GC, Will-Wolf S (2010) The forest inventory and analysis database version 4.0: description and users manual for phase 3. General technical report NRS-61. USDA Forest Service, Northern Research Station, Newtown Square, PA
Woodall CW, LS Heath, G Domke, M Nichols (2011a) Methods and equations for estimating volume, biomass, and carbon for trees in the US forest inventory, 2010. General technical report NRS-GTR-88. US Forest Service
Woodall CW, Amacher MC, Bechtold WA, Coulston JW, Jovan S, Perry CH, Randolph KC, Schulz BK, Smith GC, Tkacz B, Will-Wolf S (2011b) Status and future of the forest health indicators program of the US. Environ Monit Assess 177:419–436
Woodall CW, Domke GM, MacFarlane DW, Oswalt CM (2012) Comparing field- and model-based standing dead tree carbon stock estimates across forests of the US. Forestry 85:125–133
Woodall CW, Walters BF, Oswalt SN, Domke GM, Toney C, Gray AN (2013) Biomass and carbon attributes of downed woody materials in forests of the US. For Ecol Manage 305:48–59
Woudenberg SW, Conkling BL, O’Connell BM, LaPoint EB, Turner JA, Waddell KL (2010) The forest inventory and analysis database: database description and users manual version 4.0 for phase 2. General technical report RMRS-GTR-245. USDA Forest Service, Rocky Mountain Research Station, Fort Collins, CO
Zanchi G, Pena N, Bird N (2012) Is woody bioenergy carbon neutral? A comparative assessment of emissions from consumption of woody bioenergy and fossil fuel. GCB Bioenergy 4:761–772
Zhao M, Running SW (2010) Drought-induced reduction in global terrestrial net production from 2000 to 2009. Science 329:940–943
Acknowledgments
Special thanks to Andrew Gray, Mark Harmon, and John Stanovick for valuable reviews and insights into DDW dynamics.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Ram Oren.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Woodall, C.W., Russell, M.B., Walters, B.F. et al. Net carbon flux of dead wood in forests of the Eastern US. Oecologia 177, 861–874 (2015). https://doi.org/10.1007/s00442-014-3171-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-014-3171-8