Abstract
Walnut kernels are susceptible to oxidative rancidity and rapid deterioration due to their high oil content. Despite its global importance, little is known about this nut’s optimal storage conditions and shelf life. This research was conducted to evaluate the effect of different temperature (4 °C and 25 °C) and packaging (5% vacuum and air-containing package) on walnut kernels quality during 6 months’ storage. The cultivars ‘Common’ and ‘Claw tip’, both of which are important to the market, were examined. According to the results, the lowest level of peroxide value was observed in stored samples at the temperature of 4 °C under 5% vacuum packaging, while control samples stored at 25 °C with air packaging showed a four-fold increase over the same period in both genotypes. The antioxidant capacity and total phenol of the samples stored in a 5% vacuum at 4 ℃ were significantly higher in both cultivars than in other treatments. In both cultivars, polyphenol oxidase activity was effectively reduced by 5% vacuum packaging and low temperature. The highest L* index was observed in ‘Claw tip’ cultivar in 5% and air-containing package at 4 ℃, while in the ‘common’ cultivar, the highest level of L* was observed in 5% vacuum at 4 ℃. These results showed that low temperature and 5% vacuum significantly improved kernel quality of walnut cultivars ‘Common’ and ‘Claw tip’ during storage.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Walnut (Juglans regia L) is one of the most important nuts in the world. Many countries have been developing walnut cultivation due to the importance of this tree and the high demand on the market (Arzani et al. 2008). Given the importance of natural antioxidants in the food chain for healthy living, the walnut kernel with significant phenolic compounds can have a significant protective effect on various human diseases (Adiletta et al. 2020). On the other hand, antioxidant compounds can increase the shelf life of the products and improve the stability of the fat, thereby preventing the loss of nutritional quality and the taste of the products. However, walnuts contain metals such as copper and iron that can increase the oxidation process. Also, oxidation of vitamins A and E and carotenoids, which are produced by peroxides derived from unsaturated fatty acids, can sometimes occur (Adiletta et al. 2020). Therefore, walnut is susceptible to oxidation because it contains high fatty and unsaturated fatty acids, which can endanger the commercial and nutritional value of this over-strategic product. Studies have shown that walnut storage conditions can have an effective role in maintaining the quality and nutritional value (Adiletta et al. 2020).
Temperature and oxygen availability are among the most important factors affecting the post-harvest quality of nuts during storage. Jensen et al. (2003) reported that high temperatures and the increased oxygen can increase the sensitization of products in terms of fat oxidation (rancidity and volatile matter production) and sensory properties. Christopoulos and Tsantili (2010) investigated the effect of temperature and packaging atmosphere on total antioxidants and the color of walnut during storage. They reported that low temperatures and packaging under N2 or CO2 prevented an increase in the amount of antioxidant and browning. Furthermore, Tsantili et al. (2011) have shown that low temperatures (1 °C instead of 20 °C) and packaging with N2 atmosphere can prevent the reduction of phenolic and antioxidant compounds in eight pistachio genotypes. Currently, there is little information regarding the Iranian walnut kernel traits, such as antioxidant properties (phenol and flavonoids), walnut color and the effect of these traits on the quality of walnut kernel during the storage period; also, not much is known about the interaction of these traits with the peroxide index at the time of storage. In addition, there is a lack of adequate information concerning the effects of postharvest conditions on enzymatic activity (peroxidase, polyphenol oxidase and catalase) and their correlations with other traits. Accordingly, the aim of the present study was to investigate the changes in peroxide index, antioxidants, phenol, flavonoid, color, and enzyme activity; these included polyphenol oxidase, peroxidase, and catalase, as well as the relationship between peroxide index and antioxidant properties, color, and activity of enzymes; the possible correlation between these factors could be useful for optimizing storage conditions.
Materials and Methods
Pre-storage Preparation and Packaging of Samples
Walnut fruits, from the genotypes known as ‘Common’ and ‘Claw tip’ (high-marketability), were collected from a commercial garden in a mountainous region (Rabar), Kerman Province (longitudes 56 ° 45′–57 ° 16′ and latitudes 29 ° 27′–38 ° 54′), Iran. Fruits at full maturity (hull separated easily from the shell) were picked by hand from the trees. After cleaning, the fruits were dried for 4 days in shade through natural air circulation. Then, the walnuts were transferred to the laboratory and their wooden shells were removed using a walnut breaker; the walnut kernels were packaged immediately. About 25 g of walnut kernels in polyethylene films with the thickness of 87 μm (0.087 mm), under a vacuum machine (GSM-DZ410-610) and with a vacuum level of 5% and air-containing packaging as control were packaged and stored at 4 °C and 25 °C for 6 months; their various traits were then evaluated monthly. The schematic illustration of the sample preparation procedures is shown in Fig. 1.
Determination of Peroxide Value
Peroxide values (PV) were determined according to the Association of Official Analytical Chemists (AOAC) (2000) method. A total of 5 g of the sample, 0.5 ml of the saturated KI solution, and 30 ml of the solvent mixture consisting of acetic acid and chloroform (3:2) were combined. Titration was carried against 0.1 mol/l Na2S2O3 using 1% starch indicator. By this method, the color of the solution was changed from light yellow to a dark blue color. PV was expressed as milliequivalents of peroxide per kilogram of the sample.
Total Phenolic Content, Total Flavonoids, and Antioxidant Activity
The extraction was carried out according to the Labuckas et al. (2008) method. The total phenolic content (TPC) values of methanolic extracts of walnut kernels were determined using a modified Folin–Ciocalteu colorimetric method (Siddhuraju et al. 2002). Then, 10 mL methanolic extract was diluted with methanol (50%). The reaction mixture contained 0.5 ml of diluted walnut extracts, 2.5 ml of the Folin–Ciocalteu reagent (10:1) and 2 ml of sodium carbonate (7.5% w/v). After 90 min at room temperature, the absorbance at 750 nm was measured using a spectrophotometer (CECIL CE250). A reference curve was constructed, using gallic acid as standard. The results were expressed as milligrams of gallic acid equivalents (GAE) per gram of the extract.
Total flavonoids (TF) were determined using a colorimetric assay according to the methods of Chen and Chen (2004). The absorbance of the solution was measured vs. a blank at 415 nm using a spectrophotometer. The results were expressed as mg of quercetin equivalents (QE) per gram fresh weight (mg g−1 FW).
For the radical scavenging assay, 0.1 mL properly diluted sample of the extract was added to a screw cap tube containing 3.9 mL DPPH solution (2,2-diphenyl-1-picryhydrazyl, 60 µM in methanol). The decrease in absorbance at 515 nm was recorded vs. blank after 30 min incubation in the dark at room temperature (Christopoulos and Tsantili 2011).
Polyphenol Oxidase Activity and Peroxidase Activity
The activity of polyphenol oxidase (PPO) was determined with some modification (Kar and Mishra 1976). The 3 mL reaction mixture contained 25 mM phosphate buffer (pH 7), 0.1 mM pyrogallol, and 0.1 mL enzyme extract and blank without pyrogallol. The absorbance of the purpurogallin formed was recorded at 420 nm, and activity was calculated using the extinction coefficient for purpurogallin.
For the measurement of peroxidase (POD) activity, the assay solution (3 ml) contained 50 mM potassium phosphate buffer (pH 7.0), 20 mM guaiacol, 40 mM H2O2 and 0.1 ml enzyme extract. The reaction was initiated by adding the enzyme extract. The increase in absorbance of the reaction solution at 470 nm was recorded after every 20 s. One-unit POD activity was defined as an absorbance change of 0.01 units min−1 (Chance and Maehly 1955).
Catalase Activity
For measurement of catalase activity, enzyme extract (0.1 mL) was added to the solution containing the phosphate baffer (50 mM, pH = 7) and H2O2 (5.9 mM). The decrease in absorbance of the reaction solution at 240 nm was recorded, after every 20 s. An absorbance change of 0.01 unit’s min−1 was defined as one-unit CAT activity. Enzyme activities were expressed on a protein basis (Chance and Maehly 1955).
Color
Kernel surface color was measured using a chromameter (CR 400, Minolta) which provided (CIE L*, a*, and b* values). L* is color lightness (0 = black and 100 = white). Furthermore, the hue angle (h°) and whiteness index (WI) was calculated according to McGuire (1992) respectively.
Quality Evaluation
As panellists, 21 people associated with the department of pomology were chosen on the basis of availability and motivation. The panelists were advised to chew several kernel pieces, expectorate, and rinse with water between samples. A five-point hedonic scale ranging from 0 to 5, where 5 = extremely like and 0 = extremely dislike was used to evaluate acceptability of sample (bitterness, rancidity, flavor, and aroma).
Statistical Analysis
The experiments were conducted in a randomized factorial designed with three replications. The results were given as means ± standard deviation. Data were subjected to analysis of variance using SAS version 16.0, and means were separated by the Duncan test at 0.05. R software was used to draw clustering heatmap in multivariate analysis.
Results
Peroxide Value
The initial peroxide value of the samples was very low, from 0.023 to 0.001 meq O2/kg, in the ‘Common’ and ‘Claw tip’ genotypes, respectively. Peroxide index was gradually increased during storage (Table 1). Two genotypes showed a significant difference in the peroxide value, and the ‘Claw tip’ genotype had higher oxidation under the same storage conditions. According to the results, the lowest level of peroxide value was observed in both genotypes in stored samples at the temperature of 4 °C and under vacuum. In these conditions, at the last time of storage, the range of peroxide value was equal to 0.18–0.24 meq O2/kg in the ‘Common’ genotypes and ‘Claw tip’, respectively, while the control samples (25 °C and air-containing packages) showed a four-fold increase over the same period in both genotypes.
Antioxidant Capacity
The antioxidant capacity of the two varieties of walnuts stored in different conditions during the 6‑month period is shown in Table 2. At first, there was no significant difference in the antioxidant content of the two varieties. The levels of antioxidants in both genotypes (‘Common’ and ‘Claw tip’) were gradually decreased during storage time, but the samples kept under vacuum showed the lowest reduction. At the end of the experiment, the ‘Common’ cultivar stored in vacuum packages showed higher antioxidant levels than the control samples.
Total Phenol and Flavonoid Content
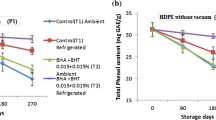
The phenol content was initially 18.2 and 20.2 mg /100 g in the ‘Common’ and ‘Claw tip’, respectively. Generally, total phenol was decreased gradually with increasing storage time. But its intensity varied depending on the genotypes and storage conditions. In the ‘Common’ genotype, temperature and type of packaging had a significant effect on the preservation of phenolic compounds during the storage period. At the end of the experiment, samples under vacuum and at 4 °C had a higher phenol content than the control samples, while in the the ‘Claw tip’ genotype, temperature was more effective than the type of packaging; at the end of the experiment, samples kept at the temperatures of 4 °C and 25 °C showed a significant difference, but there was no significant difference between the vacuum and air samples (Table 3). The flavonoid composition of samples with a similar pattern of phenol showed a decreasing trend during the storage (Table 4). This was more severe in the last 2 months of the test. At the end of the experiment, the ‘Common’ genotype showed a higher flavonoid content than the ‘Claw tip’. However, there was no significant difference between different temperatures and types of packaging.
CAT, PPO, and POD Enzymes
The process of the activity of the enzyme CAT was completely opposite to PPO and POD. By passing storage time, the activity of the CAT enzyme was gradually decreased (Fig. 2a, b). The CAT activity pattern in two varieties was quite similar. According to Fig. 2a, b, the enzyme activity was at the highest level in vacuum and at 4 °C; until the fifth and sixth storage periods, it showed a significant difference during the last maintenance period. But, its activity in control samples (25 °C and air containing) of both varieties was decreased from the third storage time. At the end of the experiment, there was a significant decrease in the samples (4 °C and vacuum). The PPO enzyme was increased gradually over the course of 6 months in two genotypes, and this increase was also observed in the control samples (25 °C and air containing) at a high level; it was significant at the fifth and sixth times (Table 5). At the end of the storage period, the control sample showed a two-fold increase in both genotypes, as compared to the samples kept at 4 °C and under vacuum condition. The POD enzyme with a pattern like the PPO enzyme (Table 6) showed an increasing trend during the 6 months of storage, especially during the fourth, fifth and sixth periods. The two genotypes tested showed a different increase in terms of severity. The ‘Common’ genotype kept under controlled and conventional storage conditions showed only 5% and 26% increase of the enzyme, respectively. But the ‘Claw tip’ genotype showed the 12 and 29% increase under the same conditions, respectively.
Color Parameters
The genotypes did not show any significant differences in Hue and other parameters. According to the results, the temperature showed a higher impact on the Hue than the type of packaging. At the temperature 4 °C, no significant difference was observed between different packaging conditions. But at the end of the experiment, samples packed in air at 25 °C showed a significant difference to the samples stored under vacuum (Tables 7 and 8). Factor L* was significantly reduced in two genotypes during storage, although its severity was more in the ‘Claw tip’ genotype. At the end of the test, the lowest amount of L* was observed in samples at 4 °C under vacuum. Samples kept at the temperature of 4 °C and vacuum packaged during storage did not show any significant difference (Fig. 3).
The quality of the walnut kernel was also influenced by the storage conditions. In the final stages of the experiment, the samples kept at a temperature of 4 °C and under vacuum were superior to the quality control samples. According to Fig. 4 the temperature and type of packaging could play a significant role in changing the quality properties (bitterness, rancidity, flavor, and aroma) of the walnut kernel for 6 months. According to the results, the quality of the walnut kernel began to change gradually from the fourth month of storage; this was more pronounced in the ‘Claw tip’ (data not shown). At the end of the experiment, there was a significant difference between the samples stored at 4 °C and those kept under vacuum, as compared to the control samples.
Multivariate Analysis of Biochemical Traits
Regarding the clustering heatmap, there was no relationship between the two genotypes (Fig. 5). ‘Common’ genotype included cluster I and II. Cluster I contained other treatments in this genotype. ‘Commom’ genotype had the highest color lightness and the lowest PPO enzymes. This genotype was also distinguished by fruit with medium to high phenol. Considering the effects of treatments on ‘Claw tip’ genotype, two clusters were observed. ‘Claw tip’ genotype had also very high values of hue, high whiteness and medium chroma. Comparing two clusters in this genotype, cluster III had the lowest CAT and cluster IV had the lowest PPO enzyme.
Cluster analysis of walnut cultivars ‘Common’ and ‘Claw tip’ based on physical and chemical properties of fruit under two packaging modes, two temperature conditions (gradient from low [red], medium [white] to high [green]). P = packaging method (P1 = Vacuum, P2 = Air), D = temperature (D1 and 2: 4 ℃ and 25 ℃), M = storage time (M1–6: 1, 2, 3, 4, 5 and 6 months, respectively), CAT catalase, PPO polyphenol oxidase, POX peroxidase
Discussion
Throughout the shelf life, walnut fat compositions have a significant impact on flavor, texture, and overall acceptance. In addition to producing an intense flavor and a loss of nutrition (reduced vitamin and essential fatty acid content), the oxidation of fats and oils in foods can also produce toxic and active compounds that pose a serious threat to consumers. A useful indicator of oil rancidity and deterioration, which influences the nutritional and sensory quality of nuts, is the peroxide value, which is determined by evaluating oil oxidation by measuring hydroperoxides, which are a major oil oxidation product (Pakrah et al. 2021). Increased lipoxygenase enzyme activity and increased ROS accumulation, along with an increase in high humidity, oxygen concentration, high temperature, and light, are definite contributors to lipid oxidation and increased rancidity in nuts (Pakrah et al. 2021). The compounds spontaneously, optically, and enzymatically oxidize as a result of the unstable storage conditions, and free radicals are produced. Almonds may have an unpleasant taste and smell due to the production of these radicals as well as lateral compounds like free fatty acids, hydroxides, aldehydes, ketones, and volatile alcohol (Habashi et al. 2019). In this research, peroxide value during storage period was increased in all treatments. The increased amount of peroxide during the maintenance period could be attributed to the high levels of monounsaturated and polyunsaturated fatty acids in the walnut and the presence of peroxides, including oxygen and possibly metal ions. The time required to reach the peroxide of walnuts stored in different conditions to 2 meq O2/kg could be considered as the maximum time of the walnut resistance to oxidative degradation (Lopez and Vidal 2009). In the present study, storage of samples at 4 °C and in vacuum conditions showed an important effect on their oxidation resistance. The peroxide value of these samples (0.28–0.18 meq O2/kg) showed a significant difference, as compared to the control samples (1.05–0.86 meq O2/kg). The two varieties tested did not show the same oxidation trend. In the case of the ‘Claw tip’ genotype, although there was initially a smaller amount of peroxide; at the end of the experiment, under the same conditions, it showed a higher peroxide value than the ‘Common’ genotype. Hosseini et al. (2012) also investigated the effect of contact surface and storage temperature on the oxidative stability of walnut during storage, reporting that storage conditions and storage time played a significant role in the oxidation of walnut.
DPPH is a useful reagent for assessing an antioxidant’s capacity to scavenge radicals. Depending on the extent to which antioxidants can donate hydrogen during their reactions, antioxidants reduce the DPPH radical to a yellow compound (Fu et al. 2016). In general, the ‘Claw tip’ genotype showed a higher antioxidant capacity before and after the experiment than the ‘Common’ genotype. Researchers have reported that the antioxidant activity of plant species can be influenced by different structures of phenolic acids and flavonoids, as well as derivatives of these compounds. Lin and Xing 2008, for example, showed that the antioxidant activity of phenolic acids and their derivatives, like esters, depended on the number of hydroxyl groups in the molecule. The antioxidant activity of the studied ranks was lower than that of the genotypes investigated by Akbari et al. (2014). In evaluating six different walnut genotypes, he reported an antioxidant capacity of between 53.83 and 90.38%. In another study, Chirastoplose and Tsantley (2011) reported the antioxidant potential of some of the important varieties of walnut (‘Chandler,’ ‘Hartley,’ and ‘Lolley’), with the antioxidant potential of about 94.4–181.2 μM TAE on dry weight. However, these differences seem to be related to environmental conditions and varieties.
Chirostoplose and Tessaltli (2011) also examined the effects of temperature and atmospheric packaging on widespread antioxidants over 12 months. They reported that the genotypes, storage conditions, and storage time had a significant effect on antioxidant levels; they observed that the lowest antioxidant potential at high temperatures, and the low temperature and packaging under nitrogen gas or carbon dioxide saved the antioxidant compounds of the samples, as compared to the control samples, showing a significant difference in air temperature. Tsantili et al. (2011) reported similar results in a study on pistachios.
Phenols and flavonoids have been found to be significant in nuts, with very high antioxidant effects. The ‘Claw tip’ had higher phenols and lower flavonoids than the ‘Common’ genotype. Although these differences were not statistically significant, according to the results, the temperature of 4 °C under vacuum played an effective role on maintaining the amount of phenol and flavonoid in the walnut. In fact, the prevention of the reduction of phenolic compounds could be attributed to the limitation of oxidation reactions through low temperatures and less oxygen access, because phenolic compounds are susceptible to chemical or enzymatic oxidation (Christopoulos and Tsantili 2015). In our search, although the ‘Claw tip’ had a phenol content higher than that of the ‘Common’ genotype at the time of storage, at the end of the experiment, it almost doubled the ‘Common’ genotype in the same conditions. This could be explained by the varying degrees of the degradation of each phenolic composition due to different phenolic oxidation factors, such as the number and nature of the hydroxyl group attached to the compound (Prabhakar et al. 2020). It has been reported that phenolic and specific phenolic quantities can be affected by genotype, tissue type, and environmental conditions (Gill and Tuteja 2010). There is also a direct relationship between the total phenolic, flavonoid compounds, and antioxidant properties (Yoosefi and Nazoori 2021). A correlation coefficient study showed that there was a positive correlation between antioxidant stenosis and phenol and flavonoid content. These results also suggested the decrease in antioxidant activity, phenol, and flavonoids. Yoosefi and Nazoori (2021) observed that phenolic oxidation resulted in the loss of antioxidant capacity; a similar correlation has been found between phenolic compounds and antioxidant capacity in other studies of nuts. The increase in total phenols during storage could be due to an increase in PAL activity, involved in the biosynthesis of phenolic compounds through the phenylpropanoid pathway, in walnut during cold storage, as demonstrated in other studies (Adiletta et al. 2020). Due to their potent antioxidant properties, flavonoids play a crucial role in the treatment of numerous diseases. According to some authors, flavonoids like quercetin and rutin have antioxidant properties (Fu et al. 2016).
According to the results, the initial activity of the polyphenol oxidase and peroxidase enzyme was very low, but it was slowly increased over the course of 6 months with similar patterns. At the end of the test, samples stored at 25 °C and without vacuum conditions showed the highest levels of PPO and POD enzymes, as compared to others. In general, the ‘Claw tip’ genotype showed a higher enzyme activity than the ‘Common’ genotype. The polyphenol oxidase and peroxidase enzymes are among the well-known enzymes that play a role in the browning process. In fact, surface brownness is usually caused by the polyphenol oxidase enzyme, which turns phenolic compounds into dark pigments; they can affect the sensory and nutritional properties of the product, resulting in the loss of product quality (Christopoulos and Tsantili 2015). Sheikhi et al. (2019) have also reported that polyphenol oxidase substrates are phenolic compounds found in plant tissues and, mainly, flavonoids. Peroxides also catalyze the reduction of hydrogen peroxide by electrons from different molecules such as phenols and lignin precursors (Christopoulos and Tsantili 2015; Sheikhi et al. 2019). However, polyphenol oxidase can stimulate the peroxidase enzyme activity by producing hydrogen peroxide during phenol oxidation (Christopoulos and Tsantili 2015; Sheikhi et al. 2019). The walnut kernel is exposed to enzymatic browns due to its phenolic and flavonoid compounds; consequently, it decreases its quality. In the present study, browning (PPO and POD increase) was observed in samples kept under ambient temperature without vacuum, which had the highest phenol losses. Researchers showed that keeping the product cool and using the appropriate package, which restricted access to oxygen, could reduce the browning of the product. In fact, the increase in temperature caused the destruction of polyphenols as a result of the activity of polyphenol oxidase and peroxidation, leading to the browning process (Chisari et al. 2007). Our data in this experiment showed that the catalase enzyme activity was completely contrary to the peroxidase and polyphenol oxidase activity, in the sense that by decreasing the storage time, the catalase enzyme activity was declined, while the activity of peroxidase and polyphenol oxidase enzymes was increased. The CAT enzyme during storage, such as antioxidant capacity and phenolic compounds, was gradually decreased, so that it was significantly reduced in control samples (25 °C and air package), but at the temperature of 4 °C and vacuum, during the entire storage period, it showed a higher CAT activity. Pre-existing enzyme units were thought to be more active, leading to an increase in PPO activity under cold stress.
If hydrogen peroxide is present, POD is responsible for the brown color because it undergoes single-electron oxidation. Plant tissues only contain very low levels of hydrogen peroxide. However, by producing hydrogen peroxide during phenol oxidation, PPO may increase POD activity. Under molecular oxygen, PPO causes monophenols to be hydroxylated into o‑diphenols and then o‑quinones, which in turn make brown pigments. It is vital, however, that PPO plays an immediate part in tyrosine digestion, keeping away from the tyramine collection and cell passing, as well as in the biosynthesis of hydroxycoumarin esculetin in green leaves of pecan tree (Christopoulos and Tsantili 2015; Sheikhi et al. 2019). CAT enzymes are antioxidant enzymes in the plant that can cause the oxygenation of water to decompose. Oxygen water is a toxic molecule that can oxidase and regenerate organic matter in the cell. Catalase destroys this toxicity by decomposing oxygenated water into water and molecular oxygen (Van Doorn and Ketsa 2014). Catalase also plays an important role in oxidative stress and increases the antioxidant activity in the cell (Yuan et al. 2010). It can, therefore, be effective in maintaining the nutritional value and quality of the walnut kernel quality. There is not much information about the enzymatic activity of walnut or even nuts in the storage. Generally, little is known about the enzyme activity of the walnut kernel and even nuts during the storage time.
Color is one of the most important aspects of dry fruits. It influences their acceptability by the consumer. Therefore, abnormal colors, especially those caused by fat oxidation, or by the loss of phenolic compounds and their antioxidant capacity, will reduce the acceptability of the product by consumers. Walnut pellicle (the skin of the kernel) variety, as an actual boundary, is one of the vital quality criteria that determines buyer inclination and furthermore is a sign of the tactile quality (Pakrah et al. 2021; Donis-González et al. 2020). In general, the color of the walnut kernel ranges from bright to amber. In the present study, the samples showed a significant difference in the type of kernel color, and this color difference could be related to genetic factors. According to the results, storage conditions could have a significant effect on the color of the walnut. In this experiment, the highest browning (minimum WI, L *, h) was observed in the samples stored at 25 °C by air packaging, such that they had the highest peroxide value and the lowest amounts of antioxidant and phenolic compounds. The results of this study were consistent with those of Christopoulos et al. (2010), and with Pakrah et al. (2021) on walnut and Guine et al. (2015) on hazelnut. They studied three varieties of walnut in modified atmospheric treatments (air-containing packages, N2-containing packages, and Co2-containing packs) and temperatures (1 and 20 °C), indicating that Hue, L* and WI were gradually decreased during storage. However, this decrease was more intense in the samples air-bagged at a temperature of 20 °C. These results agreed with Guiné et al. (2014). It was stated that the browning of walnut kernel could be the product of enzymatic activity or phenolic chemical oxidation. The walnut pellicle’s higher color index makes it more popular with consumers (Fuentealba et al. 2017). In addition, the walnut pellicle color index is significant because it may indicate the rate of browning of the pellicle (Habibie et al. 2019). The browning of the walnut pellicle during drying may be caused by oxidative stress brought on by a greater accumulation of reactive oxygen species (ROS) and the activation of the lipoxygenase enzyme, both of which increased lipid oxidation and peroxidation as well as nonenzymatic browning (Pakrah et al. 2021; Ortiz et al. 2019).
Sensory properties are the main factors in accepting or rejecting many products by consumers and obtaining their satisfaction. The oxidation compounds affect the flavors of the oils; if oxidation of compounds is carried out at high levels, they will be unusable (Akbari et al. 2022). In addition to color parameters, other sensory properties such as flavor, appearance, and texture can also affect the acceptance of the product. Therefore, sensory evaluation of samples is necessary to achieve the above objective. The results of this study showed that storage time and storage condition could play a significant role in determining the orangeloptic characteristics of the walnut kernel depending on the genotype. The highest quality loss was observed in samples with a temperature of 25 °C and air content, especially at the fifth and sixth time of storage, so that samples kept at 4 °C and vacuum packaged at the end of the experiment showed a significant difference, as compared to the control sample. At the end of the experiment, the ‘Common’ genotype showed a better sense of quality than the ‘Claw tip’ genotype. The results were consistent with those of other researchers. Jensen et al. (2003) also reported that increasing temperature and access to oxygen enhanced the sensitivity of products to fatty oxidation (rinsing and producing the volatile matter) and sensory properties. The stability of the sensory quality as well as the physio-chemistry profiles of the kernel are also important aspects of the storage tolerance of walnut fruit. The sensory quality score represented the cumulative effects of storage time and the commercial value of each genotype (Yuan et al. 2019). Generally, according to these results, walnut kernel vacuum packaging and 4 °C have an important role in preventing phenol oxidization and preserving the quality of walnut kernel for 6 months. The present work showed that the deterioration of antioxidants was reduced during storage when nuts were kept in vacuum and at the lower temperature.
References
Adiletta G, Magri A, Albanese D, Liguori L, Sodo M, Di Matteo M, Petriccione M (2020) Overall quality and oxidative damage in packaged freshly shelled walnut kernels during cold storage. J Food Meas Character. https://doi.org/10.1007/s11694-020-00589-9
Akbari V, Heidari R, Jamei R (2014) Fatty acid compositions and nutritional value of six walnut (Juglans regia L.) genotypess grown in Iran. Adv Herb Med 1(1):36–41
Akbari B, Baghaei‐Yazdi N, Bahmaie M, Mahdavi Abhari F (2022) The role of plant‐derived natural antioxidants in reduction of oxidative stress. BioFactors 48(3):611–633
AOAC (2000) Peroxide value of oils and fats. Official methods of analysis of AOAC international, 17th edn.
Arzani K, Mansouri-Ardakan H, Vezvaei A, Roozban MR (2008) Morphological variation among Persian walnut (Juglans regia) genotypes from central Iran. N Z J Crop Hortic Sci 36:159–168
Chance B, Maehly AC (1955) Assay of catalases and peroxidases. Methods Enzymol 11:764–755
Chen G, Chen H (2011) Extraction and deglycosylation of flavonoids from sumac fruits using steam explosion. Food Chem 126(4):1934–1938
Chisari M, Barbagallo RN, Spagna G (2007) Characterization of polyphenol oxidase and peroxidase and influence on browning of cold stored strawberry fruit. J Agricultural Food Chem 55(9):3469–3476
Christopoulos MV, Tsantili E (2011) Effects of temperature and packaging atmosphere on total antioxidants and colour of walnut (Juglans regia L.) kernels during storage. J Sci Hortic 131:49–57
Christopoulos MV, Tsantili E (2015) Participation of phenylalanine ammonia-lyase (PAL) in increased phenolic compounds in fresh cold stressed walnut (Juglans regia L.) kernels. Postharvest Biol Technol 104:17–25. https://doi.org/10.1016/j.postharvbio.2015.03.003
Christopoulos MV, Tsantili E, Papageorgiou V, Komaitis M, Rouskas D (2010) Effects of package atmosphere and temperature on phenolics, total antioxidant capacity and colour in kernels of ‘Franquette’ walnuts during 8‑month storage. Acta Hortic 858:75–81
Donis-González IR, Bergman SM, Sideli GM, Slaughter DC, Crisosto CH (2020) Color vision system to assess English walnut (Juglans Regia) kernel pellicle color. Postharvest Biol Technol 167:111199. https://doi.org/10.1016/j.postharvbio.2020.111199
Fu M, Qu Q, Yang X, Zhang X (2016) Effect of intermittent oven drying on lipid oxidation, fatty acids composition and antioxidant activities of walnut. LWT Food Sci Technol 65:1126–1132. https://doi.org/10.1016/j.lwt.2015.10.002
Fuentealba C, Hernández I, Saa S, Toledo L, Burdiles P, Chirinos R, Pedreschi R et al (2017) Colour and in vitro quality attributes of walnuts from different growing conditions correlate with key precursors of primary and secondary metabolism. Food Chem 232:664–672
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48(12):909–930
Guiné RPF, Almeida CFF, Correia PMR (2014) Influence of packaging and storage on some properties of hazelnuts. J Food Meas Charact 9(1):11–19. https://doi.org/10.1007/s11694-014-9206-3
Guiné RP, Almeida CF, Correia PM (2015) Influence of packaging and storage on some properties of hazelnuts. J Food Measure Characterization 9:11–19
Habashi RT, Zomorodi S, Talaie A, Jari SK (2019) Evaluation of shelf life of walnut kernel coated by antioxidants in combination with packaging under different storage conditions. J Postharvest Technol 7(3):87–95
Habibie A, Yazdani N, Saba MK, Vahdati K (2019) Ascorbic acid incorporated with walnut green husk extract for preserving the postharvest quality of cold storage fresh walnut kernels. Scientia Horticulturae 245:193–199
Hosseini H, Ghorbani M, Sadeghi Mahoonak A, Maghsoudlou Y (2012) Estimation of walnuts oxidative stability using an accelerated shelf-life testing approach. Iran Food Sci Technol Res J 9(4):348–358
Jensen MD (2003) Fate of fatty acids at rest and during exercise: regulatory mechanisms. Acta Physiologica Scandinavica 178(4):385–390
Kar MD, Mishra D (1976) Catalase, peroxidase, and polyphenoloxidase activities during rice leaf senescence. Plant Physio 57:315–319
Labuckas DO, Maestri DM, Perelló M, Martínez ML, Lamarque AL (2008) Phenolics from walnut (Juglans regia L.) kernels: Antioxidant activity and interactions with proteins. Food Chem 107(2):607–612
Lin D, Xing B (2008) Adsorption of phenolic compounds by carbon nanotubes: role of aromaticity and substitution of hydroxyl groups. Environ Sci Techmol 42(19):7254–7259
Lopez-Duarte AL, Vidal-Quintanar RL (2009) Oxidation of linoleic acid as a marker for shelf life of corn flour. Food Chem 114(2):478–483
McGuire RG (1992) Reporting of objective color measurements. HortScience 27:1254–1255
Ortiz CM, Vicente AR, Fields RP, Grillo F, Labavitch JM, Donis-Gonzalez I, Crisosto CH (2019) Walnut (Juglans regia L.) kernel postharvest deterioration as affected by pellicle integrity, genotype, and oxygen concentration. Postharvest Biotechnol 156:110948. https://doi.org/10.1016/j.postharvbio.2019.110948
Pakrah S, Rahemi M, Nabipour A, Zahedzadeh F, Kakavand F, Vahdati K (2021) Sensory and nutritional attributes of Persian walnut kernel influenced by maturity stage, drying method, and cultivar. J Food Process Preservation 45(6):e15513
Prabhakar H, Sharma S, Kong F (2020) Effects of postharvest handling and storage on pecan quality. Food Rev Int. https://doi.org/10.1080/87559129.2020.1817066
Sheikhi A, Mirdehghan SH, Karimi HR, Ferguson L (2019) Effects of passive- and active-modified atmosphere packaging on physio-chemical and quality attributes of fresh in-hull pistachios (Pistacia vera L.cv. Badami). Foods 8(564):1–15. https://doi.org/10.3390/foods8110564
Siddhuraju P, Mohan PS, Becker K (2002) Studies in the antioxidant activity of Indian laburnum (Cassia fistula L.): A preliminary assessment of crude extracts from stem bark, leaves, flowers and fruit pulp. Food Chem 79:61–67
Tsantili E, Konstantinidis K, Christopoulos MV, Roussos PA (2011) Total phenolics and flavonoids and total antioxidant capacity in pistachio (Pistachia vera L.) nuts in relation to genotypess and storage conditions. Sci Hortic 129:694–701
Van Doorn WG, Ketsa S (2014) Cross reactivity between ascorbate peroxidase and phenol (guaiacol) peroxidase. Postharvest Biol Technol 95:64–69
Yoosefi M, Nazoori F (2021) Investigating the packaging effect on the quality of fresh pistachio fruit during storage. Pistachio Health J 4(3):63–74
Yuan G, Sun B, Yuan J, Wang Q (2010) Effect of 1-methylcyclopropene on shelf life, visual quality, antioxidant enzymes and health-promoting compounds in broccoli florets. Food Chem 118(3):774–781
Yuan X, Huang S, Ma H, Huang N, Ye N (2019) Differential responses of walnut genotypes to cold storage and their correlation with postharvest physiological parameters. HEB. https://doi.org/10.1007/s13580-019-00126-8
Acknowledgements
The authors would like to thank the Agriculture and Natural Resources College, University of Hormozgan, Bandar Abbas, Iran, for help in processing this project.
Author information
Authors and Affiliations
Contributions
Azam Shojaee: Carrying out the research, Formal analysis, Writing—Original Draft; Somayeh Rastegar: Investigation, Supervision, Project administration, Writing, Review and Editing; Behjat Tajeddin: Investigation, Review and Editing; Pegah Sayyad-Amin: Writing—Review, Editing, Visualization and Validation
Corresponding author
Ethics declarations
Conflict of interest
A. Shojaee, S. Rastegar, B. Tajeddin, and P. Sayyad-Amin declare that they have no competing interests.
Additional information
Data availability
Data will be made available on request.
Rights and permissions
Springer Nature oder sein Lizenzgeber (z.B. eine Gesellschaft oder ein*e andere*r Vertragspartner*in) hält die ausschließlichen Nutzungsrechte an diesem Artikel kraft eines Verlagsvertrags mit dem/den Autor*in(nen) oder anderen Rechteinhaber*in(nen); die Selbstarchivierung der akzeptierten Manuskriptversion dieses Artikels durch Autor*in(nen) unterliegt ausschließlich den Bedingungen dieses Verlagsvertrags und dem geltenden Recht.
About this article
Cite this article
Shojaee, A., Rastegar, S., Tajeddin, B. et al. Quality Preservation of Walnut Kernels: Effect of Storage Temperature and Vacuum Packaging. Erwerbs-Obstbau 65, 2407–2418 (2023). https://doi.org/10.1007/s10341-023-00944-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10341-023-00944-2