Abstract
About 130 seed-propagated walnut trees (Juglans regia L.) grown in the same location were evaluated for 2 years based on 37 quantitative and qualitative traits. The late-leafing trait ranged between 1 April and 14 April (13–27 days after Payne). The first female and male flowering dates ranged from 8 April to 5 May and from 7 to 29 April, respectively. The protandrous, protogynous, and homogamous percentages were 46.72%, 35.24%, and 18.03%, respectively. The harvest time changed from 7 September to 27 September. The bearing type rates were 3.5% for lateral, 13.4% for terminal, and 83.1% for both. The severity of blight and anthracnose disease varied from very low to very severe. The annual growth varied from 3.9 cm to 45.7 cm, while the leaf area ranged from 24.3 cm2 to 48.01 cm2. All types of tree growth habit were observed among the studied genotypes, with the spreading type predominating. The ranges of the fruit attributes were 1.4 mm –5.7 mm for husk thickness, 0.7 mm –2.3 mm for shell thickness, 25.4 mm –41.5 mm for nut length, 25.7 mm –38.5 mm for nut width, 7.27 g –17.73 g for nut weight, 3.68 g –9.49 g for kernel weight, 45.48%–63.86% for kernel percentage, and extra light to amber for kernel color. The highest recorded apomixis rate of the 30 genotypes was 25.55%, whereas this rate was low or even zero in some genotypes. In conclusion, the high diversity found in the studied germplasm resulted in the selection of some superior genotypes that can be considered promising plant materials for future walnut breeding programs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Juglandaceae family consists of 60 species within seven genera, including the genus Juglans, which contains 20 species and of which Persian walnut (Juglans regia L.) is one of the most important economic species. Juglans regia is cultivated in temperate regions for its edible nuts (McGranahan and Leslie 2009) and its multiple uses as an ornamental plant and in the wood industry and medicinal sector (Hayes et al. 2016; Salejda et al. 2016). Walnut species are native to the mountainous regions of Central Asia, and now they are the most widely distributed nut fruit tree in the world (Bayazit et al. 2007; Chen et al. 2014).
Iran is the third highest walnut producer globally after China and the United States, with a total annual production of 356,666 tons (FAO 2020). Walnut is a monoecious tree with male and female flowers. Since ancient times, the dichogamous pollination nature of walnut and its propagation by seeds has helped preserve the phenological, morphological, and pomological variance, even in orchards planted for productive purposes. This practice has enriched the genetic pool of Iranian walnut, with millions of seed-propagated trees (Rezaei et al. 2008).
This variability has led to extensive investigation of germplasm native to the main origin centers. Such investigations have resulted in the selection and introduction of some superior genotypes in Turkey (Akça et al. 2015; Aysen et al. 2019; Bükücü et al. 2020; Sutyemez 2016; Sutyemez et al. 2019, 2021, 2022), Romania (Cosmulescu and Stefanescu 2018), Iran (Fatahi et al. 2010; Hassani et al. 2012, 2020), California (McGranahan and Leslie 2004), China (Liu et al. 2020), and India (Sharma and Sharma 2001; Sharma et al. 2014).
The selection of superior walnut genotypes is mostly based on suitable traits such as late leafing, lateral bearing, lack of pistillate flower abortion, high yield (> 6 t/h), large and relatively smooth nut, sealed suture, high kernel percentage (> 50%) light kernel color, and moderate to high resistance against pests and diseases (Botu et al. 2010; Cosmulescu et al. 2010). Other desirable characteristics in walnut selection include bacterial and anthracnose tolerance, homogamy and protogynous, apomixis, short dormancy in areas with mild winters, winter cold resistance in areas with hard winters, and high-quality fruits and kernels (plumpness, light color, high kernel ratio, easy kernel extraction [Botu et al. 2010; Cosmulescu et al. 2010; Jacimovic et al. 2020; McGranahan and Leslie 2012]).
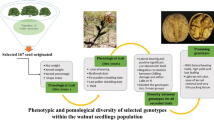
The main objective of this study was to inventory the morphological, phenological, and pomological diversity among seedling genotypes and to select high-quality walnut genotypes for direct cultivation in preparation for studying the possibility of registering them as new cultivars or forming a primary core collection that will allow breeders to have more options in breeding programs.
Material and Methods
Experimental Location and Plant Materials
This research was conducted at the Research Station of the Department of Horticultural Sciences, University of Tehran, Iran, with an altitude of 1320 meters above sea level. In this study, 130 seed-propagated walnut trees (> 15 years) were coded from G1 to G130 and evaluated for 2 years (2016–2017) in terms of some pomological, quantitative, and qualitative traits (Table 1). Nut and kernel traits were measured based on walnut descriptors (IPGRI 1994) with slight changes. Measurements were taken of at least 45 nuts of each genotype (15 nuts in three replications). Thirty leaves were randomly selected from each genotype (10 leaves in three replications) and were used to measure leaf traits in the laboratory. The leaf area was measured using a leaf area meter. Finally, after the information was collected, the data were analyzed using Microsoft Excel and IBM SPSS statistics version 22.0.0 software (SPSS Inc., Chicago, IL, USA).
Apomixis Investigation
Apomixis was studied in 30 walnut genotypes. For this purpose, about 100 female flowers on each genotype were randomly selected. All the catkins on each selected shoot were removed at the differentiation stage. Then, flowers were isolated using double-walled glassine paper bags (15 cm × 25 cm). After 15 days of isolation, and when the stigma had completely dried, the bags were removed, and the percentage of apomixis after 8 weeks of fruit set was determined (Fig. 1). The apomixis rate was calculated as follows:
Results and Discussion
The results showed significant differences among the studied genotypes for all the traits. Therefore, it was possible to select the genotypes for different values of each trait.
Growth and Phenology Characterization
In this research, the coefficient of variation (CV%) of the late leafing trait was 13.94%, and the leafing time ranged from 1 April to 14 April, (i.e., at 27–40 days after 5 March and 14–27 days after Payne). Walnut production is limited not only by cold winters but also by autumn and spring frosts, which cause quantitative and qualitative differences in walnut production (Akça and Ozongun 2004). The most important way to increase cold avoidance in walnuts is to select for late leafing (Haghjooyan et al. 2005). In addition, late-leafing cultivars/genotypes show less infection by anthracnose (Hassani et al. 2011) and blight disease (Forde 1975) because of opening buds in the late dry part of the spring season. Hakan and Akça (2011) reported that the leafing date of te selected genotypes in Turkey ranged from 25 April to 28 April, but in Simsek’s research (2010) it varied from 1 April to 12 April. The heritability of this trait in walnuts is high (greater than 96%) (Hansche et al. 1972), so it can be easily transferred from parents to the next generations by selecting the late-leafing seedlings.
The first female and male flowering dates among the studied genotypes ranged from 8 April to 5 May and from 7 pril to 29 April, respectively (Table 2). The most prolonged female and male flowering periods were 17 days and 16 days, respectively. Akça and Ozongun (2004) showed that the flowering time of promising genotypes was limited from 15 April to 10 May in the Karaman region of Turkey. Simsek (2010) showed that the female and male flowering dates of selected genotypes in the Diyarbakir province of Turkey ranged from 7 April to 18 April and from 11 April to 18 April, respectively, while the results of Oguz and Aşkın (2007) in Ermenek, Turkey, were similar to those of Simsek (2010).
In the present study, the percentages of protandrous, protogynous, and homogamous genotypes were 46.72%, 35.24%, and 18.03%, respectively, i.e., the protandrous type was dominant, which is consistent with the results of Akça and Sen (1995). One of the main goals of walnut breeding programs is selecting homogamous types or obtaining cultivars with high overlap. In this study, about 43.07% of the studied genotypes had suitable homogamy (> 50%), 6.15% had acceptable homogamy (25%–50%), 12.31% had low homogamy (< 25%), and 38.47% had no homogamy.
The harvest time is an essential trait due to its impact on the nutrient content and chemical composition of the nut (Özcan and Lemiasheuski 2020; Matthäus et al. 2018). The harvest time in the current study ranged from 7 September to 27 September, with CV of 3.58%, while it ranged from 15 September to 10 October in the work by Simsik (2010).
Yield production is determined by the fruiting bearing type in walnut trees, which was evaluated based on the ratio of lateral and terminal bearing buds. The flowers usually appear terminally at the ends of the branches shortly after leaves appear. In some cultivars, female flowers also appear on the lateral buds. This type of flowering in walnut is called “lateral bearing” and is associated with high yield in young trees, but the bearing that includes both bearing types (terminal and lateral) is the choice of interest. Among the 130 studied genotypes, about 3.5% had a lateral-bearing type, 13.4% had a terminal-bearing type, and 83.1% had both bearing types, with a CV of 18.57%. However, the ratio of lateral and terminal bearing differed among genotypes, and this was accompanied by a decrease in the percentage of lateral buds over time in old trees (Pinney et al. 1998; Polito 1998).
About four genotypes at the first year and 20 genotypes at the second year had meager fruit set because of the severe abscission of female flowers in the spring, which was probably caused by the pistillate flower abortion phenomenon that is usual in almost all walnut cultivars (McGranahan et al. 1994). Walnut also has an alternate bearing habit that reduces the “off” year’s yield after a productive “on” year.
In the present research, the blight and anthracnose disease severity varied from very low to very severe, with CV of 65.44% and 98.71%, respectively (Fig. 2). Genotypes G95, G96, G97, and G98 were the most resistant to bacterial disease.
Other traits were also studied in this research, including the annual growth, which varied from 3.9 cm to 45.7 cm with a CV of 33.29%. The average leaf area ranged from 24.3 cm2 to 48.01 cm2 with a CV of 14.42%. Keramatlou et al. (2015) reported a close relationship between the leaf area and the nut size and fullness in walnuts. Among the studied genotypes, all types of tree growth habit (vertical, semivertical, and spreading) were observed. A recently introduced genotype is called BD6, which has a semivertical growth habit and semidense branching (Bujdosó et al. 2020).
Nut and Kernel Characterization
The husk thickness in dry and sunny areas is vital to prevent sunburn of the fruit. In this study it ranged between 1.4 mm and 5.7 mm, with a CV of 27.56%.
Nut shape had a high diversity and reached 70.23% (Fig. 3; Table 3). Hence, about 46, 30, and 25 genotypes had round, trapezoidal, and broad elliptical nut shapes, respectively. Trapezoid nuts are easily broken and their kernels easily extracted, which is marketable (Ebrahimi et al. 2010).
The greatest nut length in this study (41.5 mm) is less than the lengths reported by Simsek et al. (2017) (45.78 mm) and Cosmulescu et al. (2017) (53.6 mm), but it is close to the findings of Keles et al. (2014) (42.8 mm) and higher than that reported by Akça et al. (2015) (36.62 mm). The maximum nut diameter obtained in this study (38.5 mm) is higher than that reported by Simsek (2010) (36.71 mm), Keles et al. (2014) (34.77 mm), Akça et al. (2015) (36.15 mm), and Cosmulescu et al. (2017) (37.48 mm), while it is lower than that reported by Sharma et al. (2014) (42.07 g).
In the current study, the nut weight varied from 7.27 g to 17.73 g, and the CV was 19.41%. The optimal nut weight should be 12 g to 18 g (Cosmulescu 2013). The highest nut weight in this study is greater than that found in Turkey by Akça and Sen (2001) (13.93 g), Akça et al. (2015) (15.2 g), Keles et al. (2014) (13.92 g), and Özcan (2009) (10.5 g); lower than that found in the northwestern Himalaya (22.66 g; Shah et al. 2021), India (23.61 g; Sharma and Sharma 2001), and Romania (20.9 g; Cosmulescu et al. 2017); and close to that of Turkish walnut (17.5 g; Asma 2012).
The tightly sealed shell suture is crucial for nut storage (McGranahan and Leslie 1990). During storage, nuts with open seals are exposed to fungal and insect infestation (Forde 1975). The diversity index obtained in this trait was 48.69%, indicating a significant difference among the studied genotypes for this trait (Fig. 3; Table 3).
Shell thickness is a determinant factor in the kernel percentage in walnuts. In the present study, the CV of shell thickness was 23.17% and ranged from 0.7 mm to 2.3 mm. This result is less than the results of Shah et al. (2021) (0.98–2.83 mm) and of Sharma and Sharma (2001) (0.48–2.6 mm) and close to the Turkish genotypes (1.11–2.33 mm [Akça et al. 2015] and 0.23–2.32 mm [Akça and Ozongun 2004]) and Iranian walnut (0.64–2.02 mm [Mahmoodi et al. 2019]), while it is higher than for Turkish walnut (0.95–1.75 mm [Asma 2012], 0.8–1.77 mm [Keles et al. 2014], 0.66–1.4 mm [Simsek et al. 2017]) and Iranian walnut (0.4–1.4 mm [Arzani et al. 2008]).
The kernel weight at an average of 2 years varied from 3.68 g to 9.49 g, with a CV of 21.54%. A kernel weight of 6 g to 10 g is suitable for commercial walnut production, and the kernel percentage should be at least 50% (Cosmulescu 2013; McGranahan and Leslie 1990). The highest kernel weight in the current study (9.49 g) is more than that found in the studies of Akça et al. (2015) (8 g), Asma (2012) (9.1 g), Cosmulescu (2013) (9.07 g), Jacimovic et al. (2020) (6.54 g), Karadağ and Akça (2011) (7.44 g), Keles et al. (2014) (7.36 g), and Simsek et al. (2017) (8.54 g); is close to that obtained in the research of Zeneli et al. (2005) (9.8 g) and Fatahi et al. (2010) (9.83 g); and is less than that reported by Shah et al. (2021) (14.00 g).
The kernel-to-nut ratio is considered an indicator of the yield’s economic performance. According to Hansche et al. (1972), the kernel percentage has a very low heritability of 0.08. In breeding programs, the kernel rate of promising genotypes should be > 48%–50% (Korac et al. 1997; Arzani et al. 2008). In the present study, the kernel percentage as an average of both years varied between 45.48% and 63.86%, with a CV of 9.52%. This result is higher than in the studies by Asma (2012) (60.8%), Jacimovic et al. (2020) (52.25%), and Simsek (2010) (58.04%), and is close to that reported by Karadağ and Akça (2011) (63.16%). The kernel rate in the present study was more than 48% for 73.84% of the evaluated genotypes. Factors that affect this trait include shell thickness, shell hardness, kernel weight, and nut weight. Genotypes with a thin shell and high nut weight have the highest kernel percentage.
In this study, the mean kernel color rank was 2.19 out of 4, with a CV of 38.81%, indicating that most genotypes had a light to light amber kernel color. About 21 and 39 genotypes had extra light and light kernel colors, respectively (Fig. 3; Table 3). Rezaei et al. (2018) reported that about 51.72%, 22.41%, and 25.86% of evaluated genotypes had light, very light, and amber color kernels, respectively. A clear and uniform kernel is an important trait in commercial cultivars (McGranahan and Leslie 1990).
Shell roughness varied from very smooth to very rough, with a CV of 69.04%, which indicates high diversity among the studied genotypes. The shell color ranged from very light to very dark, and the CV of this trait was 42.27%. A high diversity was recorded in the shell hardness trait (40.96%), which affects the ease of kernel extraction. Fruit with a hard shell that is smooth and thin, with a closed aperture seal and light kernel color is desirable in breeding programs (McGranahan and Leslie 1990). The high diversity of fruit characteristics indicates the high potential of genotypes in the region to select superior genotypes based on the goals of the breeding program (Cosmulescu and Botu 2012).
As a result, genotypes G23, G46, G56, and G126 seem to be among the most promising genotypes. These genotypes were late-leafing (13 to 15 April, i.e., about 10 days after Chandler [4 April] and 27 days after Payne [18 March]), with a high lateral-bearing rate, high yield, and ease of kernel extraction. But one of the disadvantages of G56 was its poor shell sealing, although its kernel percentage was high (62%). Genotypes G9, G22, G25, G31, G51, G108 had high yield, high lateral-bearing, high kernel weight (> 7 g), and high kernel percentage (> 58.5%). Genotype G51 had a light kernel color, well-sealed shell, high kernel percentage (63.72%), high kernel weight (8.93 g), shell thickness of 1.2 mm, and easy kernel extraction. Genotype G31 had very good traits (kernel weight 9.1 g, nut weight 15.68 g, kernel rate 58.02%); however, it was characterized by an amber kernel color and medium plumpness. Genotypes G108, G22, G126, and G9 were superior in terms of kernel weight, nut weight, kernel color, and kernel percentage (Fig. 4; Table 4).
In the case of late-leafing genotypes, it was observed that genotypes G23, G46, and G76 were homogeneous, but genotype G23 had the best overlap with other late-leafing genotypes as a donor of pollen grains. Some selected genotypes in this study were homogeneous (G9, G46, G23), some genotypes were protogynous (G22, G21, G51, G25, and G51), and some others were protandrous (G108, G126). It was found that homogamy was extremely low in some superior genotypes (G22), or there was no homogamy at all (G56). Therefore, it is possible to plant the genotypes in overlapping pairs to ensure a high fruit set (Fig. 5).
Principal Component Analysis
Using principal component analysis (PCA), different attributes can be discussed in the form of components, each of which includes several attributes. This analysis can show the main differentiating factors between the studied genotypes. In this study, PCA was able to express 26 evaluated traits as eight main components, among which the first, second, and third components had the largest share in justifying total variance. A total of eight main and independent components whose variance values were greater than 1 were able to explain 75.37% of the total variance (Table 5).
The first component, PC1, consisted of four significantly contributed traits, including nut length, width, weight, and kernel weight, which explained 22.92% of the variance contribution. The second component, PC2, included the traits of leafing date, first female and male flowering, and harvest date, which accounted for 13.36% of the variance (Fig. 6). Traits including shell color, roughness, integrity, kernel fill, and middle blade thickness were in PC3, with a variance of 9.28%. The PC4 included the shell thickness, ease of kernel extraction, and kernel rate, which explained 7.92% of the variance. In PC5, kernel fullness, plumpness, shrivel, and color were included and explained 6.50% of the variance. These components played a major role in distinguishing the genotypes studied. The PC6 explained 5.63% of the variance and correlated with the severity of leaf and tree blight disease. The shell seal and nut shape were found in PC7 and explained 5.54% of the variance, and PC8 explained 4.18% of the variance and correlated with severity of anthracnose disease and bearing type (Table 6). Haghjooyan et al. (2005) showed that the first two components, cumulatively, explained 99.87% of the changes in the initial data and included nut length, width, weight, shell percentage, and kernel rate. In the study of Pop et al. (2013) of 20 walnut cultivars in Romania, PC1 and PC2 explained 29.2% and 17.53% of the total variance, respectively. In research by Mousavi et al. (2015), a total of seven main components (with 21 traits) were able to explain 73.78% of the total variance: PC1 (nut weight, length, width, and diameter) was 22.04%, PC2 (kernel weight, rate and size, shell weight, and percentage) was 14.17%, PC3 (adhesion of shell to kernel and maturity heterogeneity) was 10.42%, PC4 (flowering habit, fruit ripening time and yield) was 9.11%, PC5 (shell thickness and tree shape) was 7.94%, PC6 (nut shape) was 6.75%, and PC7 (leafing time) was 3.35%. The first and second components in the study by Ahandani et al. (2014) explained about 69.6% of all the variances. In the studies by Pop et al. (2013) and Mousavi (2015), nut and kernel traits were the effective traits in the first factor.
Investigation of Apomictic Genotypes
Apomixis is a natural phenomenon of asexual reproduction in plants in which the embryo is formed without the fusion of male and female gametes. Apomict seeds are genetically similar to the female parent (Ulukan 2009). The apomixis percentage in this study varied between 0 and 25.75%. Genotypes G2, G11, and G9 recorded the highest apomixis rate (25.75%, 20%, and 10.44%, respectively), while it was the lowest in G87, G1, G22, and G53 (5.55%, 5.19%, 5%, and 3.75%, respectively). Many researchers have reported a low apomixis rate in walnuts (Laiko 1990; Şan and Dumanoğlu 2006; Valdiviesso 1990), which in some cases was almost nonexistent. Laiko (1990), Guo-liang et al. (2007), and Solar et al. (1995) reported a high apomixis rate in some genotypes (23.5% to 81.2%). The apomixis rate may vary from year to year depending on the environmental conditions (Guo-liang et al. 2007). Therefore, the rate of apomixis cannot be accurately predicted. Guo-liang et al. (2010) introduced a new walnut cultivar called ‛Qinquan 1’ that was selected from genotypes in northern China with an apomixis rate of 24.7%.
Cluster Analysis
Based on the Euclidean distance, the genotypes were divided into four main groups, and each group was divided into smaller clusters with more shared characteristics (Fig. 7).
The first group included three subgroups that included traits such as high annual growth, early pollination, intermediate fruit ripening, both flowering types, high kernel percentage, very thin to thin middle blade, ease of kernel extraction, thin shell to papery, kernel weight from 5 g to 7.81 g, plump kernel, and medium to good kernel fullness.
The second group included three subgroups with traits such as high annual growth, late leafing, short female flowering period (9–12 days), early pollination, both flowering types, and medium to good kernel fullness.
Three subgroups were found in the third group, which included traits such as high annual growth, early leafing, long female flowering period (12–14 days), both flowering types, zero anthracnose percentage, seed length and diameter greater than 29 mm, high nut weight, rough to very rough shell, and high shell integrity.
The fourth group consisted of three subgroups that had traits such as a very long female flowering period (12–17 days), nut diameter less than 33.5 mm, and nut length less than 37.5 mm.
Conclusions
Walnut propagation by seeds has long been standard in Iran. The long juvenile period of walnuts and their large size rendered the genetic evaluation difficult. Therefore, identifying, collecting, and evaluating superior genotypes from native walnut landraces and seed-propagated orchards or nurseries are essential for walnut breeding (McGranahan et al. 1998).
Although it is difficult, due to environmental influences, to distinguish different genotypes based on morphological characteristics, these studies are necessary for evaluation, selection, and breeding programs. In the current study, about 130 seed genotypes were evaluated as part of an integrated project for walnut breeding in Iran to improve local cultivars or to develop new cultivars.
At the end of this research, several promising genotypes were determined to carry some important morphological, phenological, and morphological traits that are important plant materials that can be used in walnut breeding programs. A core collection of superior genotypes was formed, and a comprehensive study is currently being conducted on them, including their chemical composition (crude protein, free fatty acids, nutritional content, etc.), with an in-depth study of the phenomenon of apomixes.
References
Ahandani EA, Ramandi HD, Sarmad J, Samani MA, Yavari A (2014) Evaluation of morphological diversity among somepersian walnut accessions (Juglans regia L.) in Guilan, Northern Iran. Int J Plant Biol Res 2(3):10–15
Akça Y, Ozongun S (2004) Selection of late leafing, late flowering, laterally fruitful walnut (Juglans regia) types in Turkey. N Z J Crop Hortic Sci 32(4):337–342
Akça Y, Sen MS (1995) The relationship between dichogamy and yield-nut characteristics in Juglans regia L. In: III International Walnut Congress, vol 442, pp 215–216
Akça Y, Sen SM (2001) Study on the selection of superior walnut trees in Hizan (Bitlis) populations. Acta Hortic 544:115–118
Akça Y, Bilgen Y, Ercisli S (2015) Selection of superior persian walnut (Juglans regia L.) from seedling origin in Turkey. ACTA Sci Polonorum Hortic 14(3):103–114
Arzani K, Mansouri-Ardakan H, Vezvaei A, Roozban MR (2008) Morphological variation among persian walnut (Juglans regia) genotypes from central Iran. N Z J Crop Hortic Sci 36(3):159–168. https://doi.org/10.1080/01140670809510232
Asma B (2012) Pomological and phenological characterization of promising walnut (Juglans regia L.) genotypes from Malatya, Turkey. Acta Sci Pol Hortorum Cultus 11(4):169–178
Aysen KOC, Keles H, Ercisli S (2019) Some pomological properties of promising seed propagated walnut genotypes from inner Turkey. Notulae Bot Horti Agrobot Cluj Napoca 47(4):1094–1099
Bayazit S, Kazan K, Gülbitti S, Cevik V, Ayanoğlu H, Ergül A (2007) AFLP analysis of genetic diversity in low chill requiring walnut (Juglans regia L.) genotypes from Hatay, Turkey. Sci Hortic 111(4):394–398
Botu M, Tudor M, Papachatzis A (2010) Evaluation of some walnut cultivars with different bearing habits in the ecological conditions of Oltenia–Romania. Acta Hortic 861:119–126
Bujdosó G, Fodor A, Karacs-Végh A (2020) BD6 Walnut. HortScience 55(8):1393–1394. https://doi.org/10.21273/HORTSCI15148-20
Bükücü Ş, Özcan A, Sütyemez M, Yildirim E (2020) Determınation in the phenological difference levels of seedlings of some walnut genotypes (Juglans regia L.). Appl Ecol Environ Res 18(3):4807–4815
Chen L, Ma Q, Chen Y, Wang B, Pei D (2014) Identification of major walnut cultivars grown in China based on nut phenotypes and SSR markers. Sci Hortic 168:240–248
Cosmulescu S (2013) Phenotypic diversity of walnut (Juglans regia L.) in Romania-opportunity for genetic improvement. South Western J Hortic Biol Environ 4(2):117–126
Cosmulescu S, Botu M (2012) Walnut biodiversity in south-western Romania resource for perspective cultivars. Pak J Bot 44(1):307–311
Cosmulescu S, Stefanescu D (2018) Morphological variation among persian walnut (Juglans regia) genotypes within the population and depending on climatic year. Sci Hortic 242:20–24
Cosmulescu S, Botu M, Trandafir I (2010) Mineral composition and physical characteristics of walnut (Juglans regia L.) cultivars originating in Romania. Selcuk Tarım Gıda Bilim Derg 24:33–37
Cosmulescu S, Stefanescu D, Ionescu MB (2017) Genetic diversity among juglans regia genotypes based on morphological characters of nut. Erwerbs-Obstbau 60(2):137–143. https://doi.org/10.1007/s10341-017-0347-5
Ebrahimi A, FttahiMoghadam M, Zamani Z, Vahdati K (2010) An investigation on genetic diversity of 608 Persian walnut accessions for screening of some genotypes of superior traits. Iran J Hortic Sci 40(4):83–94
FAO (2020) FAO statistical yearbook. Agricultural production. Food and Agriculture Organization of the United Nations. http://faostat.fao.org/site/291/default.aspx. Accessed 17 Feb 2022
Fatahi R, Ebrahimi A, Zamani Z (2010) Characterization of some Iranians and foreign walnut genotypes using morphological traits and RAPD markers. Hortic Environ Biotechnol 51(1):51–60
Forde HI (1975) Walnuts, pp 439–455
Guo-Liang W, Yan-Hui C, Peng-Fei Z, Jun-Qiang Y, Yu-Qin S (2007) Apomixis and new selections of walnut. Acta Hortic 760(2):541
Guo-liang W, He L, Qunlong L, Yong W, Pengfei Z (2010) ‘Qinquan 1’, a new apomixis walnut cultivar. Fruits 65(1):39–42
Haghjooyan R, Ghareriazi B, Sanei-Sharuat-Panahi M, Khalighy A (2005) Investigation Genetic Diversity of Walnut Genotype in Different Region of Country by Quantaitives Morphological Marker. Pajhoohesh and Sazandegi, No 69
Hakan K, Akça Y (2011) Phenological and pomological properties of promising walnut (Juglans regia L.) genotypes from selected native population in Amasya Province. African J Biotechnol Vic Isl 10(74):16763–16768
Hansche PE, Beres V, Forde HI (1972) Estimated of quantitative genetic properties of walnut and their implications for cultivar improvement. Am Soc Hort Sci J 97:279–285
Hassani D, Aqaei MJ, Dastjerdi R, Sorkhi B, Damyar S (2011) Guideline for evaluation and determination of factors causing damages in walnut orchards
Hassani D, Atefi J, Haghjooyan R, Dastjerdi R, Keshavarzi M, Mozaffari MR, Malmir A (2012a) Cultivar release: damavand, a new persian walnut cultivar as a pollinizer for Iranian walnut cultivars and genotypes, cultivar. Seed Plant Improv J 28(3):529–531
Hassani D, Mozaffari MR, Soleimani A, Dastjerdi R, Rezaee R, Keshavarzi M, Atefi J (2020) Four new Persian walnut cultivars of Iran: persia, caspian, chaldoran, and alvand. HortScience 55(7):1162–1163
Hayes D, Angove MJ, Tucci J, Dennis C (2016) Walnuts (Juglans regia) chemical composition and research in human health. Crit Rev Food Sci Nutr 56(8):1231–1241
IPGRI (1994) Descriptors for walnut (Juglans spp.). International Plant Genetic Resources Institute, Rome
Jacimovic V, Adakalić M, Ercisli S, Božović D, Bujdoso G (2020) Fruit quality properties of walnut (Juglans regia L.) genetic resources in Montenegro. Sustainability 12(23):9963
Karadağ H, Akça Y (2011) Phenological and pomological properties of promising walnut (Juglans regia L.) genotypes from selected native population in Amasya Province. Afr J Biotechnol 10(74):16963–16968
Keles H, Akca Y, Ercisli S (2014) Selection of promising walnut genotypes (Juglans regia L.) from inner Anatolia. Acta Sci Pol Hortorum Cultus 13(3):167–173
Keramatlou I, Sharifani M, Sabouri H, Alizadeh M, Kamkar B (2015) A simple linear model for leaf area estimation in Persian walnut (Juglans regia L.). Sci Hortic 184:36–39
Korac M, Cerovic S, Golosin B, Miletic R (1997) Collecting, evaluation and utilization of walnut (Juglans regia L.) in Yugoslavia. Plant Genet Res Newsl 111:72–74
Laiko RE (1990) Apomixis of walnut. Acta Hortic 284:233–236
Liu B, Zhao D, Zhang P, Liu F, Jia M, Liang J (2020) Seedling evaluation of six walnut rootstock species originated in China based on principal component analysis and cluster analysis. Sci Hortic 265:109212
Mahmoodi R, Dadpour MR, Hassani D, Zeinalabedini M, Vendramin E, Micali S, Nahandi FZ (2019) Development of a core collection in Iranian walnut (Juglans regia L.) germplasm using the phenotypic diversity. Sci Hortic 249:439–448
Matthäus B, Özcan MM, Al Juhaimi F, Adiamo OQ, Alsawmahi ON, Ghafoor K, Babiker EE (2018) Effect of the harvest time on oil yield, fatty acid, tocopherol and sterol contents of developing almond and walnut kernels. J Oleo Sci 67(1):39–45
McGranahan G, Leslie C (2004) Robert Livermore’, a Persian walnut cultivar with a red seedcoat. HortScience 39(7):1772
McGranahan G, Leslie C (2009) Breeding walnuts (Juglans regia). In: Breeding plantation tree crops: temperate species. Springer, New York, pp 249–273
McGranahan GH, Leslie C (1990) Walnuts (Juglans). Genet Resour Temp Fruit Nut Crop 290:907–974
McGranahan GH, Leslie C (2012) Walnut. In: Badenes M, Byrne D (eds) Fruit breeding. Springer, Berlin Heidelberg
McGranahan GH, Voyiatzis DG, Catlin PB, Polito VS (1994) High pollen loads can cause pistillate flower abscission in walnut. J Am Soc Hortic Sci 119:505–509
McGranahan GH, Charles A, Leslie CA, Philips HA, Dandaker A (1998) Propagation. In: Walnut production manual
Mousavi SA, Tatari M, Moradi H, Hasani D (2015) Evaluation of genetic diversity among the superior walnut genotypes based on pomological and phenological traits in Chahar Mahal va Bakhtiari province. Seed Plant Improv J 31(2):365–389
Oguz Hİ, Aşkın A (2007) A study on the selection of walnut (Juglans regia L.) in Ermenek. YYU J Agric Sci 17(1):21–28
Özcan M (2009) Some proximate characteristics of fruit and oil of walnut (Juglans regia L.) growing in Turkey. Iran J Chem Chem Eng 28(1):57–62
Özcan MM, Lemiasheuski V (2020) The effect of harvest times on mineral contents of almond and walnut kernels. Erwerbs-Obstbau 62(4):455–458
Pinney K, Labavich J, Polito VA (1998) Fruit growth and development. In: Walnut production manual. University of California Division of Agriculture and Natural Resources, Publication No. 3373, pp 139–143
Polito V (1998) Floral biology: flower structure, development and pollination. In: Walnut Production Manual. University of California. Division of Agriculture and Natural Resources. Publication, 3373, pp 127–132
Pop FI, Vicol CA, Botu M, Raica AP, Vahdati K, Pamfila D (2013) Relationships of walnut cultivars in a germplasm collection: Comparative analysis of phenotypic and molecular data. Sci Hortic 153:124–135
Rezaei R, Hasani G, Hasani D, Vahdati D (2008) Morphological characteristics of some newly selected walnut genotypes from seedling collection in Kahriz-Orumia. Iran J Hortic Sci Technol 9(3):205–214 (https://www.sid.ir/en/journal/ViewPaper. aspx?id=151927)
Rezaei Z, Khadivi A, ValizadehKaji B, Abbasifar A (2018) The selection of superior walnut (Juglans regia L.) genotypes as revealed by morphological characterization. Euphytica 214(4):69
Salejda AM, Janiewicz U, Korzeniowska M, Kolniak-Ostek J, Krasnowska G (2016) Effect of walnut green husk addition on some quality properties of cooked sausages. LWT Food Sci Technol 65:751–757
Şan B, Dumanoğlu H (2006) Determination of the apomictic fruit set ratio in some Turkish walnut (Juglans regia L.) genotypes. Turk J Agric For 30(3):189–193
Shah RA, Bakshi P, Sharma N, Jasrotia A, Itoo H, Gupta R, Singh A (2021) Diversity assessment and selection of superior Persian walnut (Juglans regia L.) trees of seedling origin from North-Western Himalayan region. Resour Environ Sustain 3:100015
Sharma OC, Sharma SD (2001) Genetic divergence in seedling trees of Persian walnut (Juglans regia L.) for various metric nut and kernel characters in Himachal Pradesh. Sci Hortic 88:163–171
Sharma RM, Kour K, Singh B, Yadav S, Kotwal N, Rana JC, Anand R (2014) Selection and characterization of elite walnut (Juglans regia L.) clone from seedling origin trees in north western himalayan region of india. Aust J Crop Sci 8(2):257–262
Simsek M (2010) Determination of walnut genotypes with high fruit bearing and quality in Dicle, Hani, Egil and Kocaköy townships. GOU J Agric Fac 27(1):85–93
Simsek M, Gulsoy E, Beyhan O, Smanoglu A, Turgut Y (2017) Determination of some botanical, phenological, physical and chemical characteristics of walnut (Juglans regia L.) genotypes grown in Turkey. Appl Ecol Eenvironment Res 15(3):1279–1291
Solar A, Smole J, Simoncic S (1995) The ability of apomictic fruit setting in five walnut cultivars (Juglands regia L.). Zbornik Biotehniske Fakultete, Universe v Ljubljani, Kmetijstvo 65, pp 103–110
Sutyemez M (2016) New walnut cultivars: Maras 18, Sutyemez 1, and Kaman 1. HortScience 51(10):1301–1303
Sutyemez M, Bukucu ŞB, Özcan A (2019) Maraş 12: a walnut cultivar with cluster-bearing habit. HortScience 54(8):1437–1438
Sütyemez M, Bükücü ŞB, Özcan A (2021) ‘Helete Güneşi’, a new walnut cultivar with late leafing, early harvest date, and superior nut traits. Agriculture 11(10):991
Sütyemez M, Özcan A, Yılmaz A, Yıldırım E, Bükücü ŞB (2022) Determining phenological and genetic variation in genotypes obtained from open-pollinated seeds of ‘Maraş 12’ walnut (Juglans regia L.) cultivar. Genet Resour Crop Evol 69(2):823–838
Ulukan H (2009) A new alternative for plant breedings, biodiversity and environmental sustainability: apomictics. J Biol Sci 9(8):788–795
Valdiviesso T (1990) Apomixis in Portuguese walnut varieties. Acta Hortic 284:279–283
Zeneli G, Kola H, Dida M (2005) Phenotypic variation in native walnut populations of Northern Albania. Sci Hortic 105(1):91–100
Acknowledgements
We greatly appreciate the Iran National Science Foundation and the Center of Excellence for Temperate Fruit Trees for funding this research.
Funding
This study was supported by the Iran National Science Foundation and the Center of Excellence for Temperate Fruit Trees.
Author information
Authors and Affiliations
Contributions
Experiments, methodology, data curation, data analysis, writing of original draft, and writing review and editing were performed by Adnan Sallom. Design and experiments, supervision, investigation, data analysis, and writing review and editing were performed by Dr. Reza Fatahi, and Dr. Zabihollah Zamani. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
A. Sallom, R. Fatahi, and Z. Zamani declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Sallom, A., Fatahi, R. & Zamani, Z. Morphological, Phenological, and Pomological Diversity Among 130 Seed-Propagated Walnut (Juglans regia L.) Trees and Apomixis Study in Some Selected Genotypes. Erwerbs-Obstbau 65, 101–113 (2023). https://doi.org/10.1007/s10341-022-00695-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10341-022-00695-6