Abstract
Uneven skin coloration of early ripening table grapes is characterized by a lack of coloring berries within a cluster and the failure of some berries to develop color even with an uncertain delay of harvest. Biostimulants and plant growth regulators are known to have considerable roles on skin color development in the course of grape maturation. This study was carried out to compare the effects of different biostimulant doses, including 0, 1000, 2000 and 4000 ppm (BSt 0, BSt 1, BSt 2 and BSt 4, respectively) with or without gibberellic acid (GA3; 20 ppm) on grape characteristics and chemical composition of early ripening cv. Cardinal grape. Study results of cv. Cardinal grape revealed that alone effects of biostimulant doses had more effective on grape characteristics, total anthocyanin content and total phenolic compounds content from phytochemical characteristics. Although different foliar spray applications had varying effects on physical and chemical parameters; BSt 2 application had especially more superior effects on grape growth and phytochemical characteristics of cv. Cardinal grape.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Biostimulants and plant growth regulators have become considerable tool in agriculture and viticulture (Jardin 2015; Zabadal and Bukovac 2006) and both groups are used to improve the sizes of grape, cluster and also grape quality in table grape growing.
Biostimulants have favorable effects on both quality and yield of fruits by encouraging and stimulating plant metabolism and stress decrease (Parrado et al. 2008). These are also known to enhance fruit size, appearance and fruit quality by having direct effects on fruit growth and development or indirectly by regulating crop load, plant vigor and canopy architecture in plant (Looney 1993).
Plant growth regulators (PGRs) play crucial roles in regulating physiological processes, plant resistant to biotic and abiotic stress (Mauchi-Mani and Metraux 1998; He et al. 2005). There are five primary plant growth regulators used in global table grape production, including gibberellic acid (GA3), forchlorfenuron (CPPU), ethephon (Ethrel), abscisic acid (S-ABA) and hydrogen cyanamide (H2CN2).
Among these plant growth regulators mentioned above, it is broadly utilized from gibberellic acid to increase grape size and to achieve commercially acceptable grape quality in table grape production for a long time. However, gibberellic acid decreases skin color of grape and delays grape maturity (Peacock and Beede 2004; Zoffoli et al. 2009).
Table grape quality is composed of appearance, physical and chemical characteristics at harvest (Maoz et al. 2016; Kamiloglu 2011). Grape quality is influenced by different viticultural practices, including grapevine pruning, crop load, cluster thinning, girdling, topping and pinching, employing of different foliar spray applications (Prabhu and Singaram 2001; Kok 2011, 2016a, 2016b; Kok et al. 2013; Kok and Bal 2016).
Phytochemicals from chemical characteristics of grape are important quality parameters (Yang et al. 2009) and grapes contain huge amounts of phytochemicals, containing phenolic acids, stilbenes, anthocyanins, proanthocyanidins, all of which are strong antioxidants (Yang and Xiao 2013).
Cv. Cardinal is one of the remarkable early season colored table grape cultivars. Sometimes, insufficient color development and small grape size may detract from characteristics of table grape quality in cv. Cardinal (Nikolaou et al. 2003).
There is limited knowledge about combined effects of biostimulant and gibberellic acid applications on grape growth and phytochemical characteristics. Hence, the objective of this study was to evaluate influences of various doses of biostimulant applications with or without gibberellic acid on grape characteristics and phytochemical profiles of cv. Cardinal table grape.
Materials and Methods
Plant and Site Characteristics
The current research was carried out on a Vitis vinifera cv. Cardinal (Flame Tokay × Alphonse Lavalle) commercial vineyard located in Tekirdağ (NW Turkey; 40°58′23.82″N; 27°27′19.67″E; 80 m. a. s. l.) during the growing season of 2015.
In the study, it was utilized from 20-year-old cv. Cardinal grapevines trained to a gable trellis system grafted on Kober 5 BB (Berlandieri × Riparia Teleki 8B, selection Kober 5BB) rootstock. Grapevines were planted at a spacing of 2.5 m between rows and 1.5 m between grapevines within a row. The vineyard was managed according to standard viticultural practices for cultivar and region.
Research area is under the influence of a Mediterranean climate and annual mean temperature, sunshine duration per day, relative humidity and total precipitation were respectively recorded as 15.5 °C, 5.0 h, 80.9% and 850.8 mm for 2015 year. On the other hand, general characteristics of vineyard soil in study area was clay-loam with pH of 7.51 and organic matter of 1.2%.
Preharvest Biostimulant and Gibberellic Acid Applications and Application Times
In order to increase grape size and improve table quality characteristics of cv. Cardinal, biostimulant applications in this study were respectively performed at doses of 0, 1000, 2000 and 4000 ppm (BSt 0, BSt 1, BSt 2 and BSt 4) and were applied at 10–14 days after bloom period by repeating three times with 10-day interval. Biostimulant ingredient was presented in Table 1.
However, gibberellic acid (C19H22O6: GA3:) was also applied by combining with biostimulant doses for obtaining stronger effects on grape size and chemical composition of cv. Cardinal. For this purpose, gibberellic acid was prepared at a dose of 20 ppm and was applied at 10–14 days after bloom period by repeating with biostimulant applications at three times with 10-day interval.
Measurement of Physical and Chemical Characteristics
In current research, grape length (cm), grape width (cm), grape weight (g), grape firmness (g mm−1) and cluster length (cm), cluster width (cm), cluster weight (g) were measured as physical characteristics. Moreover, total soluble solids content (%), titratable acidity (g L−1), juice pH, p-value of juice (µW), total phenolic compounds content (mg GAE kg−1 fw), total anthocyanin content (mg GAE kg−1 fw) were also determined as chemical characteristics.
Determination of Grape Harvest Time and Preparation of Grape Sampling for Chemical Analysis
During the grape development period from the verasion to maturation period in cv. Cardinal, grapes on the grapevines were continuously observed and were harvested when the control grapes reached total soluble solids content of 16%.
After the grapes were harvested, samples of 250-fruits were gathered from each treatment and were eventually used to determine total soluble solids content, total acidity, juice pH, p-value of juice. Moreover, it was benefited from 300-fruits samples to appraise total phenolic compounds content and total anthocyanin content. All fruit samples were stored at −25 °C up to analysis of total phenolic compounds content and total anthocyanin content. Before the analysis, fruit samples were removed from −25 °C, allowed to thaw overnight at 4 °C and then homogenized in a commercial laboratory blender for 20 s.
Measurement of Grape Firmness
Grape firmness was measured with an analog penetrometer (FT 02, Wagner Instruments, Riverside, USA) and measurement results were expressed as g mm−1.
Calculation of P-Values
P-value, including redox potential (mV), pH, p-value (μW) and resistivity (Ώ) were formulated with an equation reported by Hoffmann (1991). In present research, p-values in grape juice samples from different foliar spraying applications were calculated according to equation notified above.
Analyses for Determination of Total Phenolic Compounds Content and Total Anthocyanin Content
Total phenolic compounds content and total anthocyanin content from phytochemical characteristics were respectively determined by using spectrophotometric methods clarified by Singleton et al. (1978) and Di Stefano and Cravero (1991). Results of both analyses were expressed as milligrams of gallic acid equivalent per kilogram of fresh weight (mg GAE kg−1 fw).
Statistical Analysis
Significant differences among the foliar sprays applications were assessed by analysis of variance (ANOVA) by using TARIST statistical software. It was utilized from Fisher’s Least Significant Difference (LSD) multiple comparison test for separating the means (p < 0.05) when the ANOVA test was significant.
Results and Discussion
Physical Parameters
Effects of different doses of biostimulant applications alone or in combination with gibberellic acid on physical parameters of cv. Cardinal grape is presented in Table 2.
Berry size is an important factor determining table grape quality (Strydom 2014). In this study, different doses of biostimulant applications alone or in combination with gibberellic acid had no statistically significant effects on grape and cluster characteristics except for grape firmness (Table 2, p < 0.05).
Means of grape length are shown in Table 2 (p < 0.05) and values were not statistically changed depending on doses of foliar spray applications and ranged from 22.93 to 25.27 mm for BSt 0 and BSt 2 application.
As to statistical assessment performed for grape width characteristic (p < 0.05), while BSt 0 was application that caused the lowest value (22.37 mm), the highest value was non-statistically obtained in BSt 2 application (24.92 mm) (Table 2).
Significant differences among the foliar spray applications were not statistically pronounced for the grape weight (p < 0.05). The highest grape weight was recorded in the grapevines applied with BSt 2 (9.40 g) and the lowest mean was 6.91 g in BSt application (Table 2).
The firmness of table grapes is a major quality parameter for the commercialization of table grapes (Iwatani et al. 2011). There are significant differences among the foliar spray applications (p < 0.05). The results given in Table 2 clearly revealed that the highest value was 864.00 g mm−1 in BSt 2 + GA3 application and the lowest value was obtained from BSt 2 application (615.25 g mm−1).
Cluster size from visual characteristics is critical for acceptance of table grapes and many factors contribute to cluster characteristics of grapes (Hanoock 2008). Means of cluster length shown in Table 2 were not significantly influenced by the foliar spray applications in present study (p < 0.05). While BSt 2 application was leading to the highest cluster length (20.80 cm), the lowest cluster length was obtained from BSt 0 application (18.41 cm).
Data presented in Table 2 show that the foliar spray applications have no significant effects on cluster width (p < 0.05). BSt 2 application gave rise to the widest cluster (12.73 cm) and the narrowest cluster was recorded in BSt 0 application (9.51 cm).
With respect to cluster weight, it is clear from Table 2 that the foliar spray applications have no statistically effects on cluster weight (p < 0.05). Cluster weight means ranged from 235.81 (BSt 0) to 306.61 g (BSt 2) among the foliar spray applications.
Chemical Parameters
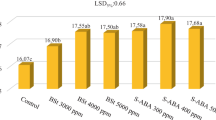
In grape growing, total soluble solids are accepted as an indicator of ripeness and most of commercial table grape cultivars are considered mature when total soluble solids content ranges from 15 to 18% (Munoz-Robredo et al. 2011). In available study, total soluble solids content at harvest was 16.05% in BSt 0 application and means of total soluble solids content varied according to different foliar spray applications (Fig. 1). The value of total soluble solids content from BSt 2 application was significantly higher (18.97%) than that of the other applications (p < 0.05).
The acidity of grapes plays a considerable role in palatability of grapes for table grapes (Laszlo and Saayman 1990). Differences in foliar spray applications were found to be insignificant in terms of titratable acidity (p < 0.05). The titratable acid content was found to be the lowest (3.97 g L−1) in BSt 2 application and the highest (4.80 g L−1) in BSt 4 + GA3 application (Fig. 2).
The juice pH is a measure of the hydrogen ion concentration in grape and is normally related to juice acidity of grape. pH of grape juice affects skin color and palatability of grapes for quality occurrence (Christensen 2000; Celik 2011). In current study, pH means were statistically influenced by foliar spray applications (p < 0.05). The juice pH of BSt 2 application was the highest (3.65), while the juice pH of BSt 4 + GA3 application was the lowest (3.41) (Fig. 3).
P-value is one of the advantageous quality elements used for numerous products and the lower p-value denotes better product quality (Hoffmann 1991; Kara et al. 2008, 2012; Kok 2016b; Kok and Bal 2016, 2017). P value of grape juices from foliar spray applications are shown in Fig. 4 and significant correlations were recorded among the foliar spray applications (p < 0.05). In existing study, foliar spray applications represented significantly variation in terms of p-value, ranging from 90.76 (BSt 2 application) to 112.80 μW (BSt 4 + GA3 application).
Phenolic compounds play key roles on quality and functional attributes of grapes (Majo et al. 2005; Li et al. 2010). The statistical analysis displayed in Fig. 5 shown that total phenolic compounds contents of cv. Cardinal were statistically influenced by foliar spray applications (p < 0.05). Among the foliar spray applications, BSt 1, 2 and BSt 2 + GA3 applications led to the highest total phenolic compounds content in grapes (3368.32, 3680.26 and 3320.33 mg GAE kg−1 fw, respectively) and the lowest mean was obtained from BSt 0 application (1958,60 mg GAE kg−1 fw).
Anthocyanins are a group of the most important phenolic components of red table and wine grape cultivars (Parpinello et al. 2009) and bring about the red and purple coloration of specific grape cultivars. Skin color of table grape cultivars is a crucial quality attributes determining grape’s market value and consumer acceptance. Therefore, anthocyanins are prominent crucial quality factors in both table and wine grapes. Fig. 6 shows effects of foliar spray applications on total anthocyanin content of cv. Cardinal and there were no statistically differences among the foliar spray applications (p < 0.05). The data obtained from foliar spray applications indicated that BSt 2 application was the most effective for stimulating the anthocyanin biosynthesis (306.62 mg GAE kg−1 fw). However, BSt 0 + GA3 application decreased anthocyanin content (40.65 mg GAE kg−1 fw).
Conclusion
Biostimulants and plant growth regulators are remarkable tool used in viticulture and it is broadly utilized from both of them to increase the sizes of grape and cluster and also improve grape quality in table grape growing. In growing of early table grape cultivars, insufficient color development and small grape size may occasionally diminish quality attributes of grape. In present research, it was observed that different doses of biostimulant applications alone or in combination with gibberellic acid affected physical and chemical characteristics at varying levels in early ripening cv. Cardinal grape. The results obtained in this study confirmed that the influences of alone effects of biostimulant doses were found to be more effective than combined effects of biostimulant and gibberellic acid. As a result, BSt 2 application from biostimulant doses with or without gibberellic acid led to the highest means in physical and phytochemical properties of cv. Cardinal grape.
References
Celik S (2011) Bağcılık (Ampeloloji), 3rd edn. vol I. Avcı Ofset, Istanbul, p 428
Christensen LP (2000) Raisin production manual. University of California, Los Angeles, p 295
Hanoock JF (2008) Temperate fruit crop breeding: germplasm to genomics. University of Michigan, Ann Arbor, p 454
He Y, Liu Y, Cao W, Hua M, Xu B, Huang B (2005) Effects of salicylic acid on heat tolerance associated with antioxidant metabolism in Kentucky blue grass. Crop Sci 45:988–955
Hoffmann M (1991) Elektrochemische Merkmale zur Differenzierung von Lebensmitteln. In: Meier-Ploeger A, Vogtmann H (eds) Lebensmittelqualitat – Ganzheitliche Methoden und Konzepte – Alternative Konzepte, vol 66. CF Müller, Karlsruhe, pp 67–86
Iwatani SI, Yakushiji H, Mitani N, Sakurai N (2011) Evaluation of grape flesh texture by an acoustic vibration method. Postharvest Biol Technol 62:305–309
Jardin PD (2015) Plant biostimulants: definition, concept, main categories and regulation. Sci Hortic 196:3–14
Kamiloglu O (2011) Influence of some cultural practices on yield, fruit quality and individual anthocyanins of table grape cv. Horoz Karasi. J Anim Plant Sci 21:240–245
Kara Z, Akay A, Ateş F, Yağmur B (2008) The P‑value as A tool for quality measurements in grape industry. 31. World Congress of Vine and Wine, 6. General Assembly of the O.I. V., Growing Techniques. Sustainability and Specificity of Production, Verona, 15–20 June 2008 ,
Kara Z, Ateş F, Sabir A (2012) Some quality parameters investigated in Sultani Çekirdeksiz (Vitis vinifera L.) clones during ripening period. 47th Croatian and 7th International symposium on Agriculture, Opatija-Croatia, 13–17 February 2012 (Hrvatska)
Kok D (2011) Influences of pre- and post-verasion cluster thinning treatments on grape composition variables and monoterpene levels of Vitis vinifera L. Cv. Sauvignon Blanc. J Food Agric Environ 9(1):22–26
Kok D (2016a) Effects of foliar seaweed and humic acid treatments on monoterpene profile and biochemical properties of cv. Riesling berry (V. vinifera L.) throughout the maturation period. J Tekirdag Agric Fac 13(2):67–74
Kok D (2016b) Variation in total phenolic compounds, anthocyanin and monoterpene content of ‘Muscat Hamburg’ table grape variety (V. vinifera L.) as affected by cluster thinning and early and late period basal leaf removal treatments. Erwerb Obstbau 58(4):241–246
Kok D, Bal E (2016) Seedless berry growth and bioactive compounds of cv. ‘Recel Uzümü’ (V. vinifera L.) as affected by application doses and times of pre-harvest Thidiazuron. Erwerb Obstbau 58(4):253–258
Kok D, Bal E (2017) Electrochemical properties and biochemical composition of cv. Shiraz wine grape (V. vinifera L.) depending on various dose and application time of foliar microbial fertilizer treatment. Erwerb Obstbau. https://doi.org/10.1007/s10341-017-0319-9
Kok D, Bal E, Celik S (2013) Influences of various canopy management techniques on wine grape quality of V. vinifera L. cv. Kalecik Karası. Blg J Agric Sci 19(6):1247–1252
Laszlo JC, Saayman D (1990) Optimum harvesting stage for Sultanina as table grape. Decid Fruit Grow 40(3):101–105
Li Z, Pan Q, Cui X, Duan C (2010) Optimization on anthocyanins extraction from wine grape skins using orthogonal test design. Food Sci Biotechnol 19:1047–1053
Looney NE (1993) Improving fruit size, appearance and other aspects of fruit crop quality with plant bioregulating chemicals. Acta Hortic 329:120–127
Majo DD, Giammanco M, Guardia ML, Tripoli E, Giammanco S, Finotti E (2005) Flavanones in citrus fruit: Structure-antioxidant activity relationships. Food Res Int 38:1161–1166
Maoz I, Beno Moualem D, Kaplunov T, Lewinsohn E, Lichter A (2016) Uneven distribution of flavour components in table grape berries. Aust J Grape Wine Res 22:343–349
Mauchi-Mani B, Metraux J (1998) Salicylic acid and systemic acquired resistance to pathogen attack. Ann Bot 82:535–540
Munoz-Robredo P, Robledo P, Manriquez D, Molina R (2011) Characterization of sugar and organic acids in commercial varieties of table grapes. Chilean J Agric Res 71(3):452–458
Nikolaou N, Zioziou E, Stavrakas D, Patakas A (2003) Effects of ethephon, methanol and girdling treatments on berry maturity and colour development in Cardinal table grapes. Aust J Grape Wine Res 9:12–14
Parpinello GP, Versari A, Chinnici F, Galassi S (2009) Relationships among sensory descriptors, consumer preference and color parameters of Italian Novello red wines. Food Res Int 42:1389–1395
Parrado J, Bautista J, Romero EJ, Garcia Martinez AM, Friaza V, Tejada M (2008) Production of a carob enzymatic extract: Potential use as a biofertilizer. Bioresour Technol 99:2312–2318
Peacock B, Beede B (2004) Improving maturity of Thompson seedless for raisin production. Grape Notes – University of California Cooperative Extension 1(2):1–4
Prabhu PC, Singaram P (2001) Effect of micronutrients on growth and yield of grapes cv Muscat. Madras Agric J 88:1–3
Singleton VL, Timberlake CF, Kea L (1978) The phenolic cinnamates of white grapes and wine. J Sci Food Agric 29:403–410
Di Stefano R, Cravero MC (1991) Metodi per lo studio deipolifenolidell’uva. Riv Vitic Enol 2:37–45
Strydom J (2014) The effect of foliar potassium and seaweed products in combination with a leonardite fertigation product on Flame Seedless grape quality. S Afr J Enol Vitic 35(2):283–291
Yang J, Xiao YY (2013) Grape phytochemicals and associated health benefits. Crit Rev Food Sci Nutr 53(11):1202–1225
Yang J, Martinson TE, Liu RH (2009) Phytochemical profiles and antioxidant activities of wine grapes. Food Chem 116(1):332–339
Zabadal TJ, Bukovac MJ (2006) Effect of CPPU on fruit development of selected seedless and seeded grape cultivars. HortScience 41(1):154–157
Zoffoli JP, Latorre BA, Naranjo P (2009) Preharvest application of growth regulators and their effect on postharvest quality of table grapes during cold storage. Postharvest Biol Technol 51:183–192
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
D. Kok declares that he has no competing interests.
Rights and permissions
About this article
Cite this article
Kok, D. Grape Growth, Anthocyanin and Phenolic Compounds Content of Early Ripening Cv. Cardinal Table Grape (V. vinifera L.) as Affected by Various Doses of Foliar Biostimulant Applications with Gibberellic Acid. Erwerbs-Obstbau 60, 253–259 (2018). https://doi.org/10.1007/s10341-018-0366-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10341-018-0366-x