Abstract
In this work, food wastes (FWs) and domestic sewage sludge (DSS) were used as raw materials for energy recovery by anaerobic digestion (AD). AD was carried out with the objective to reduce the emission of greenhouse gases and the production of biogas with high methane yield. FWs were fed in an AD horizontal reactor for co-digesting the organic fraction of waste from a university restaurant with acclimated DSS as inoculum. For this purpose, the pilot-scale reactor was operated with a residual biomass of 12.0 kg (in the proportion of 1:1 of acclimated inoculum and FW), in a semi-batch regime and produced 218 NL, with a maximum methane concentration of 88.0%. Thus, a biogas production potential of 18.16 Nm3/ton is obtained or a specific biogas and methane production of 0.24 and 0.21 Nm3/kg (VS), which can be used to generate electricity or as cooking gas. Finally, between the two kinetic models, the modified Gompertz model was the most suitable model (R2 0.99) to adjust the measured biogas yield and (R2 0.98) for the methane yield that can be used to describe the kinetics of the AD in a more reasonable way.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The growing demand for environmental protection, in face of the damaging effects of using nonrenewable sources of energy, has led to the research of several methods to produce energy from biomass waste [1, 2]. Bearing in mind current concerns related to environmental issues, such global warming, depletion of fossil fuels and greenhouse gases (GHGs), there is an interest in energy production through renewable sources, one of them is the production of biogas by anaerobic digestion (AD). According to Hoornweg and Bhada-Tata [3], the amount of solid waste (SW) generated per person in the cities increased from 0.64 kg per day to more than 1.2 kg daily, between 2003 and 2013. It should be noted that the largest rates of economic growth and urbanization in a country is associated with a greater amount of SW generation [4]. Canada, for example, produces a total of 759 kg per capita/year of solid urban waste, while Peru produces 251 kg/year per capita [5]. In Brazil, the production of municipal solid wastes (MSWs) in 2016 was 78.4 million tons (381 kg/year per capita, based on the population of that year) [6]. This increase in the generation of MSWs represents a global problem that has worsened in the recent years, due to the increase in population, with changes in consumption habits, concentration in urban centers and lack of planning [7]. According to the study by Kaza and coworkers [8], it is cited that 2.01 billion tons of waste is produced in the world annually.
In addition, it is estimated that Brazil has approximately 51.4% biodegradable materials and its per capita generation is 1.07 kg∙day−1 [9]. Today, the most used form for the final destination of MSW in Brazil is the disposal on the ground, that is, in dumps and controlled sanitary landfills. However, dumps and controlled landfills are inadequate forms of final disposal. In view of this, more than 29 million tons of MSWs were improperly discarded in the referred locations in the year 2017 [6]. In the context of environmental sustainability, effective measures for the treatment of solid wastes (SWs) have been carried out by the countries in terms of legislation. In this sense, act No. 12,305/2010, the Brazilian National Solid Waste Police was proclaimed with the objective to establish non/low generation of SWs by public agencies and companies. Moreover, it brought the proposal of practicing sustainable consumption habits and a set of instruments to provide an increase in recycling and reuse of SWs (which has economic value and can be recycled or reused) and the environmentally appropriate disposal of waste (what cannot be recycled or reused) in landfills. Another legal framework for the management of SWs, as a matter of international effort to mitigate global warming, the Paris Agreement was published in 2015, as result of the 21st Conference of the Parties (COP21) of the United Nations Framework Convention on Climate Change [10]. In this sense, political initiatives that deal with the generation of clean energy (with reduction of GHGs) should have been developed worldwide. These policies aim to promote the use of biomass as a substrate for bioenergy, as evidenced by studies carried out in member countries of Brazil-Russia-India-China (BRIC) group [11].
Renewable energy sources have been promoted around the world as a response to the growing concern about climate change and one of the cleanest energy matrices is the biogas [12]. With this, AD plays a paramount role in the achievement of this goal [13, 14]. AD processes include biochemical reactions, such as hydrolysis, acidogenesis, acetogenesis and methanogenesis [1, 15, 16]. In this sense, among the factors that influence biochemical reactions and that can function as a limiting factor in the AD are temperature, pH, C/N ratio and retention time and as accelerators are plant biomass, pure biological culture and inorganic additives [17]. Likewise, Xu and coworkers [18] observed that co-digestion of food wastes (FWs) is a promising process for converting them into clean energy, however, this process is not completely elucidated and further studies are necessary for its optimization. European Union (EU) is the world leader in the production of electricity from biogas, with more than 10.0 GW of installed capacity and a number of 17,400 biogas plants. EU had 66.7% of the global generation capacity (15 GW) of this energy matrix in 2015 [19]. According to the aforementioned source, the organic fraction of municipal solid waste (OFMSW) occupies the penultimate place in the ranking, with 337 MW. Steinmetz and coworkers [20] reported that there is a lack of technical data applicable to agro-industrial plants in Brazil, despite the diverse opportunities for generating biogas from organic waste. Therefore, it can lead to numerous uncertainties and weaknesses for the development of the biomethane chain. The authors also state that the greatest difficulty is obtaining an anaerobic inoculum under stable conditions to perform digestion tests [20]. Among the setbacks in the employment of this type of inoculum (in the case of Brazil) are: great distances between source and the AD plants, lower technological development of the available anaerobic reactors and requirements of inoculum acclimatization prior the substrate treatment. The use of manometric reactors in the evaluation of AD of organic waste in different conditions (with acclimatized inoculum) has been subject of study in several works reported in the literature [20, 21]. Finally, different inocula and their by-products for methane production have been investigated.
To perform AD studies with acclimated inoculum, it might be used parameters from private, public or international normative basis, as recommended by the Brazilian Association of Technical Standards (ABNT) [22]. With this, an adaptation was carried out in this work following the recommendation of the German engineering associations [23] which AD of organic materials must be performed under systematic steps: the characterization of substrates, sampling and collection of material data for the production of methane. Furthermore, in an early study carried out by Kainthola and coworkers [2], it was informed that there are several techniques to overcome these challenges of the degradation of lignocellulosic substrate through synergistic interaction. Between them, it is reported the pre-treatment of this raw material, from co-digestion with different substrates, one of which is the efficiency of inoculation it is considered an important factor to increase AD performance.
Despite the diverse opportunities for generating biogas from organic waste, there is a lack of technical data applicable to the Brazilian reality, since there is a lack of standardized and/or reliable data on the kinetics of methane production and this it can lead to numerous uncertainties and weaknesses for the development of the biomethane chain [20]. To suppress this knowledge gap, kinetic assessment of AD of FWs was carried out and the experimental data were modeled. All pilot scale experiments were conducted in a horizontal reactor depicted in Fig. S1, Supplementary Material. Reactor project has a patent application for Registration number: BR1020180073486, INPI (Brazil). Moreover, AD reactor design (horizontal vase with radial agitation and a gas chamber on its top) consists in an innovative concept, since there is no one similar currently reported in the literature, neither in patent registrations. Moreover, the biomass employed is unique, since it is not yet reported the employment of agricultural and livestock wastes plus municipal sewer sludge together in a bioreactor. Furthermore, the process yield and biomethane concentration were calculated and compared with the published data found in the literature.
Experimental section
Collection of substrates and inoculum
The FW used in this study was obtained at the University Restaurant of the Federal University of Pernambuco (UFPE), the same being composed of residues from the kitchen (peels and spoiled pieces of fruits and vegetables) and the cafeteria (rest of processed food). The collections were carried out by the use of 5 bags of 100 L with the aforementioned residues were made available.
The domestic sewage sludge (DSS) from the Upflow Anaerobic Sludge Blanket (UASB) reactor was obtained at the domestic sewage treatment station and was used as inoculants in co-digestion. It is worth mentioning that DSS collection was performed through the reactor bottom discharge valve. DSS was obtained at the sewage treatment station of Dancing Days community, located in Recife/PE, Brazil. In addition to the DSS, bovine manure (BM), collected from the slaughter and corral line, respectively, at the Abatedouro Regional da Paudalho -PE, located in the municipality of same name.
FW and the inoculum were packed in different bags and plastic containers and during transport they were kept at a temperature of approximately 4.0 ℃, following the technique of collecting samples from CETESB report [24]. During the performance of the characterization tests and evaluation of the methanization potential, the inoculants were kept refrigerated at a temperature of approximately 11.0 ℃, and the FW was cooled (nearly − 16.0 ℃), aiming, with this, to avoid the appearance of colonies of fungi, degradation of organic matter and even biogas generation. The BRW was kept at room temperature.
Substrate and inoculum characterization
For the characterization of the FW, a sampling by quartering was carried out in agreement with NBR 10.007 [22], as shown in Fig. 1. Then, the solubilized portion of the residue was extracted using a ratio of 1:20 w/w for the sample mass (FW) and the extractor volume (distilled water), following the methodology reported by Lange and coworkers [25]. The objective was to determine the following parameters: pH (potentiometric method). Such analyzes were also carried out with the fresh samples (liquid) of all inoculum.
Angelidaki and coworkers [26] recommend the use of volatile solids (VS) or volatile suspended solids (VSS) in tests to assess the degradability of sludge of sanitary, industrial origin or the OFMSW, since the determination of chemical oxygen demand (COD, in mg O2/L) and biochemical oxygen demand (BOD, in mg O2/L) in these cases, becomes unviable or subject to uncertainty. In addition, the VDI 4630 guide [23] expresses the results of biogas production, based on the content of VS.
For this reason, such analysis was only performed for FW, since the dosage of alkalizing agent was made based on that analysis. For the other samples (the inoculants), the parameter VS was used to measure the organic load. A fraction of all samples (substrate and inoculum) were sent to oven drying, at a temperature of 105.0 ℃, until the mass consistency, according to the World Health Organization (WHO) [27], aiming at the quantification of the moisture and preparation of samples for tests on dry basis. The dry FW was ground in a knife mill (SP Labor brand, model SP-31 N, series 0017/10), and the inoculants (also dry) were removed with the help of a porcelain mortar and pistil and then the following analyzes were carried out: VS, according to NBR13999 [22]; elementary analysis (carbon, hydrogen, nitrogen and sulfur) using the Pregl–Dumas method (Elemental Analyzer—CHNS-O, Carlo Erba EA1110), in which the samples are subjected to combustion in an atmosphere of pure oxygen and the gases resulting from this combustion are quantified in a thermal conductivity detector (TCD); biochemical analysis, following the Van Soest method (1967) [28], for cellulose, hemicellulose, lignin and Soxhlet's method (1879) [29] for lipids.
Inoculum acclimatization
In the pilot reactor
With the objective of the acclimatization process carried out in the horizontal reactor, with the ambient temperature (ranging from 26.8 to 36.1 ℃), 4.0 kg of fresh bovine manure (BM) was added to the reactor (Fig. 1) and 8.0 kg of an anaerobic reactor digestate (DG).
The acclimated inoculum (AIc2), also received a feed of 0.3 g of VS/day L, which happened twice a week with the addition of 10.08 g of dried FW and crushed diluted in approximately 40 mL of distilled water, during a 56-day period (when biogas production was well reduced—about 1L/day).
During the acclimatization process, it was followed restricted monitoring parameters of the pilot reactor, as shown in Table 1, with its respective objectives and periodicities. The aforementioned pilot reactor is under patent process (process number: BR 10 2018 007,348 6). It was manufactured in stainless steel 304 and consists of two parts: the bottom has about 26 L and has a stirring system of the type of pallets (with distance between them of 5.85 cm), where the biomass is placed residual; and the upper one has approximately 17 L and has two duct segments, for fitting the 0 to 1 bar pressure gauge (0.2 scale), the forged brass monobloc ball valve with 1/8″ connections and the nipple for reduction from 1/4″ to 1/8″, where the biogas produced by the digestion process of residual biomass is accommodated and collected. The union of these parts and the tightness of the reactor is provided by a rubber structure (similar to an o-ring) of 5 mm and by screws and nuts of 1/4″.
The reactor agitation system is activated by a Mberle three-phase induction electric motor, 1/3 CV, with 60 Hz frequency, with a voltage of 220 V and a current of 1.45 amps, with a frequency inverter (which transforms the alternating current into continuous) of the brand MEQ, model CFW08, with rotation speed variation from 0 to 1670 rpm. In that experiment, specifically, the agitation was continuous and 100 rpm.
As shown in Table 1, the determination of the volume of biogas produced daily was carried out by measuring the variables: pressure of the reactor manometer (ABC hydraulic mark, with a scale of 0–1.0 kgf/cm2), reactor temperature and pressure atmospheric, according to the methodology of Harries and coworkers [30]. The first two parameters come from the instrumentation of the pilot reactor, while the atmospheric pressure was obtained from the INMET (Brazilian National Institute of Meteorology) website, more specifically, from the Recife/PE automatic station (Recife-A301), considering the atmospheric pressure measured for the approximate time of reading the pressure of the reactor manometers. Regarding the gas analyzer used, Table 2 shows the measurement range, quantification limit and gas error range evaluated by Dräger (Model X-am 7000).
The concentrations of methane and carbon dioxide produced were also analyzed with the aid of a gas chromatograph (APPA Gold), which uses hydrogen as the carrier gas, has a thermal conductivity detector (TCD) and an N2000 Chromatostation data acquisition system. The oven, injector and detector temperatures are 60, 140 and 150 ℃, respectively.
After acclimatization of the inoculum AIc2, a reduction in solids was carried out using a soil sieve No. 10 (2 mm opening), similarly to performed by Steinmetz [20]. Subsequent to this process, laboratory tests were performed to determine the parameters: pH, moisture, total solids (TS), volatile solids (VS) and elementary analysis, as well as counting of anaerobic heterotrophic bacteria, which was performed according to the Pour Plate Technique.
Soon after, 6 kg of the inoculum was added to the horizontal pilot reactor together with 1 kg of waste in natura (only crushed in an industrial blender). One week later, 1 kg of FW was added and this procedure was repeated for the next four weeks, totaling 6 kg of FW (1:1 ratio) and characterizing the system as semi-batch. It is worth noting that the introduction of the FW was done through one of the reactor's duct segments, which has a ball valve attached, with the aid of a funnel and a plastic rod (Fig. 1). The monitoring carried out during co-digestion in the pilot reactor was identical to that of the acclimatization process, which took place in the same reactor. Finally, after 44 days, the analyses of pH, moisture, TS, VS and elementary analysis were repeated.
Kinetics study
The kinetic models are used to adjust the accumulated biogas production curve based on the experimental data. In this sense, it was investigated first order (Eq. 1) and the modified Gompertz (Eq. 2) kinetic models, following steps in the methodology reported by Pramanik and coworkers [31]. The fitting of each model was assessed through statistical parameters (non-linear correlation coefficient, R2; and the chi-square factor, χ2 acquired from the mathematical modeling using a math software. Therefore, the appropriate kinetic models was selected not only to predict the efficiency of AD in the horizontal reactor, but also to correctly analyze the pathways and mechanisms involved during the AD process [32].
First order kinetic model:
Modified Gompertz model:
In which, M is the biogas yield [NmL/g(VS)] with respect to time t (days); Pb is the maximum biogas potential of the substrate (NmL); kh is the hydrolysis rate constant (1/day); t is the time (day); Rm is the maximum biogas production rate [NmL/g(VS)]; λ is the lag phase time (days); e is Euler’s parameter equal to 2.7183.
Results and discussion
Characterization of substrate and inoculum
Table 3 presents the results of the initial characterization of the substrate (FW) and of the inoculum (AIc2), which aims to identify factors that may stimulate or inhibit the action of methanogenic microorganisms.
It can be observed that the moisture content of the studied substrate and inoculum is elevated; since all FW samples showed moisture content above 80.0%; however, it might change between 60.0 and 95.0%. Similar to the study carried out by Pavi and coworkers [33], who found moisture values of 80.0% for the substrates of the OFMSW, plus the fraction of fruit and vegetables. On the other hand, moisture content must be around 90% in order not to be harmful to digestion. In addition, it is a fundamental mean of nutrients transporting [34].
For the TS parameter, it was necessary to overcome the obstacles of the dilution of high solids contents in the AD process. This is necessary to propose a better productivity and profitability of such a system operating with high TS content (> 15.0%), that is, low moisture content (< 85.0%) according to André and coworkers [35]. In this sense, the high solids content can stagnate the digestion, due to the scarcity of water necessary for the growth of microorganisms.
In addition to having a high TS value, FW1, FW2 (food waste samples, collected at university restaurant), also has a high SV value, demonstrating a high amount of organic matter to be degraded. The VS/TS ratio, also higher for FW1 and FW2 (above 0.9) and lower for the inoculum of 0.58; given that they have already undergone biological degradation due to the acclimatization process. VS/TS values between 0.85 and 0.95 has been reported in characterization of food wastes [36, 37]. Regarding pH, acclimated inoculum 2 (AIc2) (7.71) was close to the range between 6.7 and 7.5; which according to Adekunle and Okolie [38], in which, relates the optimal growth interval of methane-producing microorganisms, especially methanogenic ones. However, the pH of FW1 and FW2, were below this range (6.29 and 5.13), which can inhibit the activity of methanogens.
Elementary contents of carbon, hydrogen, nitrogen and sulfur of the studied substrates and inoculum (FW1, FW2 and AIc1), as well as the relation between some chemical elements which are ideal for methanogenic microorganisms are reported in Table 3. It is noted first that the elementary analysis of the studied is very similar to the standard OFMSW (C: 47.80, H: 6.63, N: 2.54, S: 0.14) [39]. Ideal C/N ratio values should be between 25 and 30, as it is the ideal range for anaerobic mesophilic microorganisms in methanogenesis, according to early reports [20, 37]. In this sense, Zhang and coworkers [36] demonstrated that AD proceeds satisfactorily with the C/N ratio between 15 and 20. Therefore, it can be observed that both the substrate and the inoculum have a lower C/N ratio than the recommended ranges, 11.28 (AIc2), 13.20 (FW1) and 10.69 (FW2), indicating excess nitrogen and carbon deficiency.
The production of methane from AD is affected by the complex composition, the lignocellulosic material is difficult to biodegrade; this difficulty is due to the recalcitrant nature of lignin. According to early reports, lignin physically prevents microorganisms from accessing the most degradable substances, such as cellulose, hemicellulose, proteins, among others [40, 41]. Similarly, lipids in high concentration influence co-digestion, tests show that a concentration greater than 65.0% of the total SV is inhibition concentration [42]. Moreover, the percentage of carbohydrates (sum of cellulose, hemicellulose and lignin) and lignin obtained in this study was very similar to that of Peres and coworkers [43], which was 49.5% and 16.0% for the OFMSW studied.
The lipid content of the substrate was greater than that found by Parra-Orobio and coworkers [7], which was 0.96% for MSW and 0.5% for in the UASB reactor sludge (Inoculum). This fact can be justified by the great variety and difference in the composition existing in the used substrate. In this sense, in relation to the waste, more specifically, the sample analyzed by the authors contained not only FW, but also garden pruning, paper/cardboard, plastic, metals, rubber, among others. From the knowledge of the biochemical compositions of the residues, it is possible to analyze their biodegradability through the relationships of Cellulose + Hemicellulose/Lignin. According to Wang and coworkers [44], a value below 0.24 indicates that the residue is stabilized. It is noted that it has greater biodegradability (already mentioned through the VS/TS ratio).
Performance of the reactor in the acclimatization process
Figure 2a exhibits the biogas generation in the horizontal reactor (Fig. S1) during the acclimatization period, which had an accumulated volume of approximately 99.5 NL (average speed of 1.78 NL/day) and a methane concentration that varied from 13.8 to 62.0%, as shown in Fig. 2b, having stabilized around 64.0% after the 21st day and a Specific Biogas and Methane production of 0.09 and 0.06 Nm3/kg(VS), respectively.
The acclimatization inoculum to be used in the digestion of organic waste was carried out by Santos Filho and coworkers [21]. However, the inoculum was composed of anaerobic sludge from domestic sewage, bovine ruminal residue and goat manure (in the proportion of 5:4:1, in mass), totaling 15.0 kg. The authors reported an accumulated biogas production of 254.35 NL (average speed of 2.12 NL/day), in 120 days, reaching 78.0% of methane (with 41 days). Both the volume of biogas and the maximum concentration of methane in their experiment was higher than in the present study, due to the greater volume of added waste and the higher organic load of the same (only the anaerobic sludge had already been submitted to the reduction in organic matter), in addition to greater hydraulic retention time (HRT).
Steinmetz and coworkers [20] produced an inoculum consisting of UASB sludge from swine manure and from gelatin industry, mixed with fresh bovine manure (in the proportion 1:1:1, in volume). AD was performed during a period of 875 days; however, no biogas generation was quantified throughout the acclimatization process, as specific methanogenic and cellulolytic activities were evaluated.
From Table 4, it is evidenced the variation of the parameter moisture, TS, VS, pH and anaerobic heterotrophic bacteria of the AIc2 inoculum (composed of an acclimated inoculum for fruit and vegetable residue—DG and bovine manure, in the proportion of 2:1). These data were acquired before and after the acclimatization process, which lasted only 56 days, since one of the materials had already been acclimated (reduced organic load—TS and VS). It is notorious for an increase in moisture content and a reduction in both TS and VS. However, the VS value was still above that recommended by VDI 4630 [23]; for this reason, this inoculum was also sieved, before co-digestion with FW.
Regarding the pH, it is observed a small decrease, probably due to the degradation of the organic load and production of volatile fatty acids (VFAs). VFAs synthesis took place as the same time as the acclimatization, since its period was much shorter.
Performance of the reactor in the co-digestion process
Figure 3a shows the generation of biogas in the horizontal reactor during the co-digestion period with food waste and AIc2, which had an accumulated volume of approximately 218 NL, that is, the average speed was 3.90 NL/day and a concentration of methane that varied from 14.8 to 88.0%, as shown in Fig. 3b. Dos Santos Filho and coworkers reached an accumulated biogas production of 711.20 NL (average speed of 5.9 NL/day), in the 120-day HRT and a maximum methane concentration was 86.0% (with 73 days), starting from the co-management of 7.5 kg of fruit and vegetable residues and 15.0 kg of acclimated inoculum [21]. In this sense, the volume of biogas from the aforementioned experiment was slightly higher than that of the present study. Mazareli and coworkers also conducted an experiment in horizontal, high-rate, fixed-bed reactors, filled with fruit and vegetable and swine residues in various proportions, obtaining a better methane production with the ratio of 30/70, respectively, which was 1.08 L/day [45]. Moreover, the specific biogas and methane production were 0.24 and 0.21 Nm3/kg(VS), respectively. Such values are near to those found in the literature: 47 and 0.39 Nm3/kg(VS) of biogas and methane, respectively [21] and of 0.26 Nm3/kg(VS) of biogas [46].
In Fig. 3a, it is also possible to observe an increase in the formation of “steps”, specifically on days 6, 13, 20, 27 and 34, when 1.0 kg of FW was added to the semi-batch system. And in Fig. 3b, there is also a reduction in the concentration of methane on the aforementioned days, since with the addition of FW, there is an increase in the production of VFAs, which in turn, reduces the pH, disfavoring methanogenesis. However, as the days go by, VFAs are consumed and converted into other (simpler) compounds, raising the methane concentration again.
Kinetics modeling
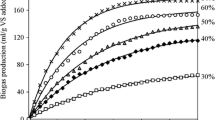
In this study, the first order kinetic models and modified Gompertz were adopted to meet and predict the production of Biogas and Methane for waste co-management. The aforementioned results of the analysis are shown in Table 5 and in Fig. 4 (a and b). These results showed that the Gompertz kinetic model showed a better determination coefficient (R2) of 0.99, with relatively short chi-square factor (χ2 52.0 and 1.5, for biogas and methane production, respectively) in comparison to first order. The first-order kinetic model presented R2 of 0.96 for the studied configuration. Moreover, standard errors were greater for the parameters acquired from first-order model, as can be seen in Table 5. This indicates that the modified Gompertz model presented a more robust estimate and was able to describe the kinetics of biogas and methane production with more than 99.0% of confidence. A similar finding was reported by Zahan and coworkers [47], who observed that the modified Gompertz was the best fit model, followed by the first-order kinetic model. In this sense, as expected Biogas yield (Pb) obtained by the modified Gompertz models was closer to Pb than the first order model.
The hydrolysis rate constant (kh) of this study was determined from the first model order was 0.0012 d1 (1/day). Previous studies found that the k value of AD ranged from 0.13 to 0.56 1/d [48] and pointed out that biogas production and the rate of degradation depends on the k value [17]. In general, faster rates of degradation and biogas production can be achieved with a higher k value [31]. The present study found that a lower k value was related to decreased biodegradability and required longer degradation times to obtain maximum biogas production.
Fruit and vegetable residues showed similar values of kh hydrolysis constant, ranging from 0.02 to 0.10 dL, according to previous reports [48, 49]. The maximum rate of biogas and methane production was (Rm) of 6.94 NmL/g(VS) added and 5.75, was observed for the modified Gompertz model (Table 5). The value of λ is 7.29 1/d; the value of λ low indicates fast initial process speed. Moreover, other works report similar values for fruit residues, ranging from 1.50 to 9.40 dL [49, 50]. However, additional investigation on the AD of these fruit residues are still needed [50]. Furthermore, Pramanik and coworkers reached a value of λ 10.2 1/d, well above this study. Great λ indicates the great synergy of microorganisms with the reaction medium and the capacity to produce biogas within a long period [31].
As described in Table 4, the moisture content increased, while TS and VS parameters of the co-digestion of AIc2 + FW decreased, after 56 days of anaerobic digestion. Similarly to the behavior of acclimatization of the inoculants, an increase in moisture and a reduction in TS and VS can be noted, due to the degradation of organic matter, which culminates both in biogas production and in the formation of water (which occurs via hydrogen trophic) as pH increases [21, 51].
Conclusion
In this work, food wastes (FW) from restaurant and domestic sewage sludge were employed in AD process to produce biomethane in an anaerobic horizontal reactor. FW samples were characterized and showed pH values and a C/N ratio below the propitious range for methane production, on the other hand, the high moisture, organic load and biodegradability, can be beneficial for digestion. Regarding the acclimated inoculum, it showed high moisture content, lower organic load and the ideal pH for methanogenesis. In this perspective, an acclimatized inoculum was used in this research and over the period of this acclimatization process it had a production of 99.5 NL of biogas and a production of 61.70 NL of methane (considering a final methane concentration of 62.0%). The result was a decrease in the levels of total and volatile solids (enhanced by the addition of water and sieving). Regarding the pH, the inoculum is acclimated with a slight increase.
The pilot reactor was operated with a residual biomass of 12.0 kg (in the proportion of 1:1, of acclimated inoculum and FW), in a semi-batch regime, and produced 218 NL, with a maximum methane concentration of 88.0%. Thus, obtaining a biogas production potential of 18.16 Nm3/ton, or a Specific Production of Biogas and Methane of 0.24 and 0.21 Nm3/kg(VS), which can be used to generate electricity or as cooking gas. Besides its satisfactory biogas production and methane yield, the innovative concept of the reactor employer in this work brings new perspectives to the literature regarding the development of AD reactors.
Finally, between the two kinetic models, the modified Gompertz model was the most suitable model (R2 0.99) to adjust the measured biogas yield and (R2 0.98) for the methane yield that can be used to describe the kinetics of AD more reasonably. The calculated parameters showed that AD of food residue co-digestion with the acclimated inoculum has a relatively low hydrolysis rate due to λ (7.29 and 9.13) for biogas and methane. With this, the modified Gompertz model can be used for applications to optimize AD process parameters. These data are significant in face of the lack of kinetic data reported in Brazilian database for AD of food wastes.
Data availability
Any complementary material, as well as the research data, might be provided by contacting any of the authors.
References
Singh B, Szamosi Z, Simenfálvi Z (2019) State of the art on mixing in an anaerobic digester: a review. Renew Energy 141:922–936. https://doi.org/10.1016/j.renene.2019.04.072
Kainthola J, Shariq M, Kalamdhad AS, Goud VV (2019) Electrohydrolysis pretreatment methods to enhance the methane production from anaerobic digestion of rice straw using graphite electrode. Renew Energy 142:1–10. https://doi.org/10.1016/j.renene.2019.04.083
Hoornweg D, Bhada-Tata P (2012) What a waste: a global review of solid waste management, Washington. https://openknowledge.worldbank.org/handle/10986/17388. Accessed 07 July 2019
Arıkan E, Şimşit-Kalender ZT, Vayvay Ö (2017) Solid waste disposal methodology selection using multi-criteria decision making methods and an application in Turkey. J Clean Prod 142:403–412. https://doi.org/10.1016/j.jclepro.2015.10.054
D-Waste, D-Waste (2017) Waste management for everyone. https://www.d-waste.com. Accessed 5 May 2020
ABRELPE (2020) Panorama 2010/2019. http://abrelpe.org.br. Accessed 10 Feb 2021
Parra-Orobio BA, Torres-Lozada P, Marmolejo-Rebellón LF (2017) Anaerobic digestion of municipal biowaste for the production of renewable energy: effect of particle size. Brazilian J Chem Eng 34:481–491. https://doi.org/10.1590/0104-6632.20170342s20150331
Kaza S, Yao L, Bhada-Tata P, Van Woerden F (2018) What a waste 2.0: a global snapshot of solid waste management to 2050, Washington. https://openknowledge.worldbank.org/handle/10986/30317. Accessed 7 July 2019
Alfaia RGSM, Costa AM, Campos JC (2017) Municipal solid waste in Brazil: a review. Waste Manage Res 35:1195–1209. https://doi.org/10.1177/0734242X17735375
UNFCCC (2015) Adoption of the Paris Agreement (FCCC/CP/2015/L.9/Rev.1), United Nations Framework Convention on Climate Change, Paris. https://unfccc.int/resource/docs/2015/cop21/eng/l09.pdf. Accessed 20 June 2019
Danish W, Z, (2019) Does biomass energy consumption help to control environmental pollution? Evidence from BRICS countries. Sci Total Environ 670:1075–1083. https://doi.org/10.1016/j.scitotenv.2019.03.268
Winquist E, Rikkonen P, Pyysiäinen J, Varho V (2019) Is biogas an energy or a sustainability product?—Business opportunities in the Finnish biogas branch. J Clean Prod 233:1344–1354. https://doi.org/10.1016/j.jclepro.2019.06.181
Siddique MNI, Wahid ZA (2018) Achievements and perspectives of anaerobic co-digestion: a review. J Clean Prod 194:359–371. https://doi.org/10.1016/j.jclepro.2018.05.155
Kumanowska E, Uruñuela Saldaña M, Zielonka S, Oechsner H (2017) Two-stage anaerobic digestion of sugar beet silage: the effect of the pH-value on process parameters and process efficiency. Bioresour Technol 245:876–883. https://doi.org/10.1016/j.biortech.2017.09.011
Li Y, Chen Y, Wu J (2019) Enhancement of methane production in anaerobic digestion process: a review. Appl Energy 240:120–137. https://doi.org/10.1016/j.apenergy.2019.01.243
Mayer F, Enzmann F, Lopez AM, Holtmann D (2019) Performance of different methanogenic species for the microbial electrosynthesis of methane from carbon dioxide. Bioresour Technol 289:121706. https://doi.org/10.1016/j.biortech.2019.121706
Mao C, Feng Y, Wang X, Ren G (2015) Review on research achievements of biogas from anaerobic digestion. Renew Sustain Energy Rev 45:540–555. https://doi.org/10.1016/j.rser.2015.02.032
Xu F, Li Y, Ge X, Yang L, Li Y (2018) Anaerobic digestion of food waste—challenges and opportunities. Bioresour Technol 247:1047–1058. https://doi.org/10.1016/j.biortech.2017.09.020
Scarlat N, Dallemand J-F, Fahl F (2018) Biogas: developments and perspectives in Europe. Renew Energy 129:457–472. https://doi.org/10.1016/j.renene.2018.03.006
Steinmetz RLR, Mezzari MP, da Silva MLB, do Kunz Amaral AAC, Tápparo DC, Soares HM (2016) Enrichment and acclimation of an anaerobic mesophilic microorganism’s inoculum for standardization of BMP assays. Bioresour Technol 219:21–28. https://doi.org/10.1016/j.biortech.2016.07.031
dos Santos Filho DA, Oliveira LRG, Fraga TJM, da Motta Sobrinho MA, Jucá JFT (2020) Development of a horizontal reactor with radial agitation to synthesize bio-methane from biomass waste and domestic sewage sludge. J Clean Prod 257:120616. https://doi.org/10.1016/j.jclepro.2020.120616
ABNT (2004) NBR 10007: amostragem de resíduos sólidos (sampling of solid waste), 2nd edn. ABNT—Associação Brasileira de Normas Técnicas, Rio de Janeiro
Verein Deutscher Ingenieure (2006) VDI 4630 - Fermentation of organic materials, Characterisation of Substrate, Sampling, Collection of Material Data, Fermentation Tests. Verein Deutscher Ingenieure, Düsseldorf
CETESB (2011) Guia nacional de Coletas e Preservação de Amostras Água, sedimentos, comunidades aquáticas e efluentes líquidos, National collection and preservation guide—water, sediments, aquatic communities and liquid effluents, 1st edn. Agência Nacional de Águas (ANA), Brasília
Lange LC, Simões GF, Ferreira CFA, Santana DWEA, Garcia LN (2002) Estudo comparativo de metodologias empregadas para a análise de resíduos sólidos urbanos, comparative study of methodologies used for the analysis of urban solid waste (in Portuguese). AIDIS-Fe FEMISCA, Mexico City
Angelidaki I, Alves M, Bolzonella D, Borzacconi L, Campos JL, Guwy AJ, Kalyuzhnyi S, Jenicek P, van Lier JB (2009) Defining the biomethane potential (BMP) of solid organic wastes and energy crops: a proposed protocol for batch assays. Water Sci Technol 59:927–934. https://doi.org/10.2166/wst.2009.040
WHO (1979) Methods of analysis of sewage sludge solid wastes and compost. World Health Organization (WHO), Zurich
Van Soest PJ (1967) Development of a comprehensive system of feed analysis and its application to forage. J Anim Sci 26:119–128. https://doi.org/10.2527/jas1967.261119x
Soxhlet F (1879) Die gewichtsanalytische bestimmung des milchfettes, The weight analysis of milk fat (in German). Dinglers Polytech J 232:461–465
Harris PW, Schmidt T, McCabe BK (2017) Evaluation of chemical, thermobaric and thermochemical pre-treatment on anaerobic digestion of high-fat cattle slaughterhouse waste. Bioresour Technol 244:605–610. https://doi.org/10.1016/j.biortech.2017.07.179
Pramanik SK, Suja FB, Porhemmat M, Pramanik BK (2019) Performance and kinetic model of a single-stage anaerobic digestion system operated at different successive operating stages for the treatment of food waste. Processes 7:600. https://doi.org/10.3390/pr7090600
Nguyen DD, Jeon B-H, Jeung JH, Rene ER, Banu JR, Ravindran B, Vu CM, Ngo HH, Guo W, Chang SW (2019) Thermophilic anaerobic digestion of model organic wastes: evaluation of biomethane production and multiple kinetic models analysis. Bioresour Technol 280:269–276. https://doi.org/10.1016/j.biortech.2019.02.033
Pavi S, Kramer LE, Gomes LP, Miranda LAS (2017) Biogas production from co-digestion of organic fraction of municipal solid waste and fruit and vegetable waste. Bioresour Technol 228:362–367. https://doi.org/10.1016/j.biortech.2017.01.003
Rizzoni LB, Tobias ACT, Del Bianchi M, Dias Garcia JÁ (2012) Biodigestão anaeróbia no tratamento de dejetos de suínos, anaerobic biodigestion in the treatment of swine manure (In Portuguese). Rev Científica Eletrônica Med Veterinária 17400–0. www.revista.inf.br. Accessed 20 October 2019
André L, Pauss A, Ribeiro T (2018) Solid anaerobic digestion: State-of-art, scientific and technological hurdles. Bioresour Technol 247:1027–1037. https://doi.org/10.1016/j.biortech.2017.09.003
Zhang C, Su H, Baeyens J, Tan T (2014) Reviewing the anaerobic digestion of food waste for biogas production. Renew Sustain Energy Rev 38:383–392. https://doi.org/10.1016/j.rser.2014.05.038
Yadvika S, Sreekrishnan TR, Kohli S, Rana V (2004) Enhancement of biogas production from solid substrates using different techniques––a review. Bioresour Technol 95:1–10. https://doi.org/10.1016/j.biortech.2004.02.010
Adekunle KF, Okolie JA (2015) A review of biochemical process of anaerobic digestion. Adv Biosci Biotechnol 06:205–212. https://doi.org/10.4236/abb.2015.63020
Pecorini I, Rossi E, Iannelli R (2020) Bromatological, proximate and ultimate analysis of OFMSW for different seasons and collection systems. Sustainability 12:2639. https://doi.org/10.3390/su12072639
Pellera F-M, Gidarakos E (2018) Chemical pretreatment of lignocellulosic agroindustrial waste for methane production. Waste Manag 71:689–703. https://doi.org/10.1016/j.wasman.2017.04.038
Fernández-Cegrí V, Ángeles De la Rubia M, Raposo F, Borja R (2012) Effect of hydrothermal pretreatment of sunflower oil cake on biomethane potential focusing on fibre composition. Bioresour Technol 123:424–429. https://doi.org/10.1016/j.biortech.2012.07.111
Sun Y, Wang D, Yan J, Qiao W, Wang W, Zhu T (2014) Effects of lipid concentration on anaerobic co-digestion of municipal biomass wastes. Waste Manag 34:1025–1034. https://doi.org/10.1016/j.wasman.2013.07.018
Peres CS, Sanchez CR, Matumoto C, Schmidell W (1992) Anaerobic biodegradability of the organic components of municipal solid wastes (OFMSW). Water Sci Technol 25:285–293. https://doi.org/10.2166/wst.1992.0160
Wang Y-S, Byrd CS, Barlaz MA (1994) Anaerobic biodegradability of cellulose and hemicellulose in excavated refuse samples using a biochemical methane potential assay. J Ind Microbiol 13:147–153. https://doi.org/10.1007/BF01583999
Mazareli RCS, Duda RM, Leite VD, de Oliveira RA (2016) Anaerobic co-digestion of vegetable waste and swine wastewater in high-rate horizontal reactors with fixed bed. Waste Manag 52:112–121. https://doi.org/10.1016/j.wasman.2016.03.021
Martí-Herrero J, Soria-Castellón G, Diaz-de-Basurto A, Alvarez R, Chemisana D (2019) Biogas from a full scale digester operated in psychrophilic conditions and fed only with fruit and vegetable waste. Renew Energy 133:676–684. https://doi.org/10.1016/j.renene.2018.10.030
Zahan Z, Othman MZ, Muster TH (2018) Anaerobic digestion/co-digestion kinetic potentials of different agro-industrial wastes: a comparative batch study for C/N optimization. Waste Manag 71:663–674. https://doi.org/10.1016/j.wasman.2017.08.014
Li Y, Zhang R, Liu G, Chen C, He Y, Liu X (2013) Comparison of methane production potential, biodegradability, and kinetics of different organic substrates. Bioresour Technol 149:565–569. https://doi.org/10.1016/j.biortech.2013.09.063
Zhao C, Yan H, Liu Y, Huang Y, Zhang R, Chen C, Liu G (2016) Bio-energy conversion performance, biodegradability, and kinetic analysis of different fruit residues during discontinuous anaerobic digestion. Waste Manag 52:295–301. https://doi.org/10.1016/j.wasman.2016.03.028
dos Santos LA, Valença RB, da Silva LCS, Holanda SHB, da Silva AFV, Jucá JFT, Santos AFMS (2020) Methane generation potential through anaerobic digestion of fruit waste. J Clean Prod 256:120389. https://doi.org/10.1016/j.jclepro.2020.120389
Gueri MVD, De Souza SNM, Kuczman O, Schirmer WN, Buratto WG, Ribeiro CB, Besinella GB (2017) Digestão anaeróbia de resíduos alimentares utilizando ensaios BMP, anaerobic digestion of food waste using BMP assays (in Portuguese). BIOFIX Sci J 3:08. https://doi.org/10.5380/biofix.v3i1.55831
Funding
This work was funded by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) [Grant numbers 311133/2015-0 and 312128/2018-5] and the Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco (FACEPE) [Grant Numbers IBPG-0604-3.01/13, IBPG-0664-3.01/13, IBPG-1917-3.06/16 and APQ-1086-3.06/15].
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by [LRGdO] and [DAdSF]. The first draft of the manuscript was written by [DAdSF] and all authors commented on previous versions of the manuscript. The visualization and revisions of graphics were carried out by [DAdSF] and [TJMF]. The supervision of experimental protocols and results was carried out by [JFTJ] and [MAdMS]. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
de Oliveira, L.R.G., dos Santos Filho, D.A., Fraga, T.J.M. et al. Kinetics assessment and modeling of biogas production by anaerobic digestion of food wastes and acclimated sewage sludge. J Mater Cycles Waste Manag 23, 1646–1656 (2021). https://doi.org/10.1007/s10163-021-01248-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10163-021-01248-x