Abstract
Background
The clinical utility of tolvaptan in chronic kidney disease (CKD) patients with heart failure remains uncertain. The level of urine cyclic adenosine monophosphate (AMP) relative to plasma arginine vasopressin (AVP) indicates the residual function of the collecting ducts in response to AVP stimulation and might be a key to predicting response of tolvaptan.
Methods
CKD patients who were hospitalized to treat their congestive heart failure refractory to conventional loop diuretics were considered to receive tolvaptan and included in this prospective study. The impact of urine cyclic AMP/plasma AVP ratio for prediction of response to tolvaptan, which was defined as any increase in urine volume at day 7 from day 0, was investigated.
Results
A total of 30 patients (median 75 years old, 24 men, and median estimated glomerular filtration rate 14.4 mL/min/1.73 m2) were included. As compared to baseline, urine volume increased at day 7 in 17 responders, whereas urine volume decreased at day 7 in 13 non-responders. Baseline urine cyclic AMP/plasma AVP ratio distributed between 0.25 and 4.01 with median 1.90. The urine cyclic AMP/plasma AVP ratio was a significant predictor of response to tolvaptan, which was adjusted for 6 potential confounders with a cutoff of 1.24.
Conclusions
Baseline urine cyclic AMP/plasma AVP ratio is an independent predictor of response to tolvaptan in advanced CKD patients with heart failure.
Clinical trial registration
UMIN000022422
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Management of body fluid overload is challenging in patients with advanced chronic kidney disease (CKD), which often accompanies refractoriness to conventional loop diuretics [1]. Tolvaptan, a vasopressin type-2 receptor antagonist, has been clinically available for the past decade. Tolvaptan can increase urine volume by increasing the excretion of free water in urine, relatively maintaining kidney function [2,3,4]. However, the clinical utility of tolvaptan in patients with advanced CKD remains uncertain.
Clinically, some patients with advanced CKD seem to be responders to tolvaptan with incremental urine volume and amelioration of systemic congestion [5, 6]. Tolvaptan therapy might prolong the free duration from dialysis in some carefully selected patients [7]. On the contrary, others are refractory to tolvaptan with unchanged urine volume and worsening decompensated congestion [8, 9]. In these patients, tolvaptan might increase urine volume just for several days, followed by a considerable decline in urine volume despite continuous tolvaptan therapy. Prediction of response to tolvaptan prior to the initiation of tolvaptan therapy would be of great help to clinicians in constructing therapeutic strategies for those with advanced CKD and systemic congestion.
The stimulation of vasopressin type-2 receptor by incrementally secreted arginine vasopressin (AVP) triggers the expression of cyclic adenosine monophosphate (AMP) in the collecting duct, which activates the synthesis and migration of aquaporin-2 and increases the reabsorption of free water [10]. In patients with CKD, the pathway among cyclic AMP, aquaporin-2, and urine osmolality seems to be preserved, whereas the upstream pathway between AVP and cyclic AMP seems to be impaired in some patients [11]. Thus, the level of urine cyclic AMP per plasma AVP would indicate the residual function of the collecting duct to respond to the AVP stimulation. We hypothesized that the urine cyclic AMP/plasma AVP ratio might be a useful marker to predict response to tolvaptan in patients with advanced CKD. We investigated the prognostic impact of urine cyclic AMP/plasma AVP ratio on response to tolvaptan in this cohort.
Methods
Patient selection
Patients with CKD stage G3–G5 who were hospitalized to treat their congestive heart failure were considered to receive tolvaptan and included in this prospective study. The diagnosis of heart failure was according to Framingham’s criteria. All patients had symptoms or signs of volume overload, including jugular vein distention, lower extremities’ edema, ascites, cardiomegaly, pulmonary congestion, and pleural effusion, which were refractory to conventional loop diuretics.
Of them, patients who received tolvaptan for the first time at the discretion of the attending physicians between March 2016 and December 2019 were included. Tolvaptan was administered at 7.5 mg/day or 15 mg/day and continued for over 7 days at a fixed dose.
Patients dependent on dialysis, those receiving immunosuppressive therapy, those with polycystic kidney, severe valvular diseases, or acute coronary syndrome within the past one month were excluded. Patients under mechanical circulatory supports, those with impaired consciousness with a lack of thirst, and those with hypernatremia with serum sodium concentration > 145 mEq/L were also excluded. Written informed consent was obtained from all participants before the inclusion. The local ethical review board approved this study (R2015144).
Study protocol
All included patients were followed following the initiation of tolvaptan for seven days. The day before the initiation of tolvaptan was defined as day 0. Trends in urine volume and body weight during 7-day tolvaptan therapy were monitored. The primary endpoint was any increases in urine volume at day 7 from day 0. A responder has an increased urine volume on day 7 from day 0. A non-responder has a decrease in urine volume on day 7 as compared to day 0.
An independent variable was defined as the baseline urine cyclic AMP/plasma AVP ratio, which was measured at day 0 prior to the initiation of tolvaptan as detailed below. The prognostic impact of this independent variable upon the primary endpoint was investigated. The secondary endpoint was end-stage renal disease that required dialysis and all-cause death.
Data collection
Baseline characteristics including demographics, comorbidity, laboratory (blood and urine samples), echocardiography, and medication data were obtained on day 0. The primary diseases of CKD were determined by the attending physicians. Diabetes mellitus was defined as those receiving medical interventions according to the previous diagnosis of diabetes mellitus. Blood pressure and pulse rate were measured in the sitting position at rest and while fasting.
Blood and urine parameters
Blood and urine samples were obtained from all patients in fasting condition before taking any medications. Blood samples were centrifuged immediately for 20 min and stored at − 80 °C before the assay. Plasma AVP was measured using a radioimmunoassay (Yamasa Shoyu Co., Ltd., Japan). Estimated glomerular filtration rate (GFR) was calculated using the following formula: 194 × (serum creatinine [mg/dL]) −1.094 × (age [years]) −0.287 (× 0.739 only for women) [12].
All urine samples were stored immediately at − 80 °C until the assay. Of note, urine cyclic AMP, aquaporin-2, and osmolality were measured. Urine cyclic AMP was measured by radioimmunoassay in the LSI Medience Co. (Tokyo, Japan). Urine aquaporin-2 was measured using a sandwich enzyme-linked immunosorbent assay (Otsuka Pharmaceutical Co., Ltd., Japan). Urine and serum osmolality was measured by freezing-point depression.
Urine cyclic AMP and aquaporin-2 were adjusted by GFR using the following formulas: urine cyclic AMP (nmol/dL GF) = urine cyclic AMP (nmol/mL) / urine creatinine (mg/dL) × serum creatinine (mg/dL) × 100; urine aquaporin-2 (ng/dL GF) = urine aquaporin-2 (ng/mL) / urine creatinine (mg/dL) × serum creatinine (mg/dL) × 100 [13]. The unit of urine cyclic AMP in this expression was nmol/min per 100 mL GFR, or more correctly, nmol/100 mL GF = nmol/dL GF (where GF was glomerular filtrate). The same was true for the unit of urine aquaporin-2.
Statistical analyses
Continuous variables were stated as median and interquartile and compared between the groups using Mann–Whitney U test. Categorical variables were stated as number and percentage and compared between the groups using Fischer’s exact test.
Logistic regression analysis was performed to investigate the impact of urine cyclic AMP/plasma AVP ratio on the primary endpoint. Its impact was adjusted for 6 clinically potential confounders: age, sex, diabetes mellitus, nephrotic syndrome, estimated GFR, and urine protein by constructing several models. By using receiver operating characteristics analysis, a cutoff of urine cyclic AMP/plasma AVP ratio to predict the primary endpoint was investigated.
The cohort was stratified into two groups using the calculated cutoff of urine cyclic AMP/plasma AVP ratio. Kaplan–Meier analyses were performed to assess the cumulative incidence of the secondary endpoint: requirement of dialysis and all-cause death. A lost follow-up was censored. Two curves were compared by log-rank test.
All statistics were performed using JMP Pro (Ver 16.2.0; SAS Institute Inc., Cary, USA) and EZR (Ver 1.55; Jichi Medical University Saitama Medical Center, Saitama, Japan). Statistical significance was defined as two-tailed p < 0.05.
Results
Baseline characteristics
A total of 30 patients were included (Table 1). Median age was 75 [66–82] years old and 24 were men. Median estimated GFR was 14.4 [10.9–22.7] mL/min/1.73 m2. Of them, 16 were assigned to G5 (estimated GFR < 15 mL/min/1.73 m2). Median urine protein excretion was 3.0 [0.8–6.8] g/g of creatinine. Of them, 11 patients met the diagnostic criteria for nephrotic syndrome. The primary diseases of CKD consisted of 20 diabetic kidney diseases, 9 nephrosclerosis, and 1 IgA nephropathy without active nephritis.
Baseline urine cyclic AMP distributed widely between 0.78 and 10.4 nmol/dL GF with a median value of 4.52 nmol/dL GF (Supplementary Fig. 1). Baseline urine cyclic AMP/plasma AVP ratio distributed between 0.25 and 4.01 with a median value of 1.90 (Supplementary Fig. 2).
Urine cyclic AMP levels and other clinical variables
Among all 30 patients, 9 patients had a lower urine cyclic AMP/plasma AVP ratio ≤ 1.24, which was statistically calculated as detailed below. There were no statistically significant differences in baseline characteristics between the two groups divided by the cutoff, except for several clinical variables (Table 1). Urine cyclic AMP/plasma AVP ratio distributed widely irrespective of the levels of estimated GFR (Fig. 1).
Distribution of urine cyclic AMP/plasma AVP ratio according to the estimated GFR, Urine cyclic AMP/plasma AVP ratio distributed widely irrespective of the levels of estimated GFR. The red dot line indicated urine cyclic AMP/plasma AVP ratio = 1.24, which was a cutoff value to predict the primary endpoint using receiver operating characteristics analysis
Impact of urine cyclic AMP/plasma AVP on the primary endpoint
As compared with day 0, urine volume increased on day 7 in 17 responders (57%), whereas urine volume decreased on day 7 in 13 non-responders (Supplementary Fig. 3). In the non-responders, daily urine volume tended to increase on day 1, followed by gradual decrease in the daily urine volume.
Urine cyclic AMP/plasma AVP ratio tended to have a mild positive correlation with a change in urine volume between day 0 and day 7 (r = 0.33, p = 0.071; Fig. 2). Urine cyclic AMP/plasma AVP levels were significantly higher in the responders than in the non-responders (p = 0.006; Supplementary Fig. 4).
The urine cyclic AMP/plasma AVP ratio was a significant predictor of the primary endpoint using age, male sex, diabetes mellitus, nephrotic syndrome, estimated GFR, and urine protein (p < 0.05 for each model; Table 2).
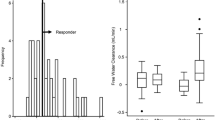
A cutoff of urine cyclic AMP/plasma AVP ratio to predict the primary endpoint was calculated as 1.24 with area under the curve of 0.79 (95% confidence interval 0.61–0.98), sensitivity 1.00, and specificity 0.62 (Fig. 3). The cutoff well-differentiated responders and non-responders in Fig. 2 and supplementary Fig. 4. The predictability of their ratio was superior to each one (Supplementary Fig. 5). Consistently, the change in urine volume from day 0 remained higher in the patients with urine cyclic AMP/plasma AVP ratio > 1.24 compared with those without (Fig. 4).
Changes in urine volume from day 0 stratified by the cutoff of urine cyclic AMP/plasma AVP ratio. Changes in urine volume from day 0 remained positive in patients with higher urine cyclic AMP/plasma AVP ratio. Changes in urine volume from day 0 gradually decreased and got negative from day 4 in patients with lower urine cyclic AMP/plasma AVP ratio. *p < 0.05 by Mann–Whitney U test. Variables were compared between the two groups in each day
Other collecting duct-related variables, including urine aquaporin-2 and urine osmolality, were not significantly associated with the primary endpoint (p > 0.05 for both).
Impact of urine cyclic AMP/plasma AVP on the secondary endpoint
During an observational period for median 5.5 [1.5–10.8] months, 23 patients encountered the secondary endpoint (21 patients had end-stage renal diseases requiring dialysis and 2 patients died due to sepsis). The cutoff could not stratify the cumulative incidence of the secondary endpoints (p = 0.55; Fig. 5).
Discussion
In this study, the response to tolvaptan was defined as any increase in urine volume on day 7 compared with day 0. Baseline urine cyclic AMP/plasma AVP ratio was an independent predictor of response to tolvaptan with a cutoff value of 1.24 among the patients with CKD and volume overload.
How to define response to tolvaptan
How to define response to tolvaptan is challenging. Several definitions have been proposed, including any increase in urine volume on day 1 and any decrease in body weight [8, 14,15,16,17]. However, we sometimes encounter a decline in urine volume several days following the initiation of tolvaptan or body weight reduction due to cardiac cachexia instead of decongestion.
In this study, we assessed urine volume increase on day 7 to investigate the sustained effects of tolvaptan. Consistently, urine volume remained higher than day 0 during the whole 7-day tolvaptan therapy in the responders. Ineffective continuation of tolvaptan in the non-responders might not be encouraged.
Conventional indicators of response to tolvaptan
Several researchers attempted to find predictors of response to tolvaptan in the CKD cohort, but no definite variables have been found thus far. As confirmed in our study, the existence of hypoalbuminemia and proteinuria did not disturb response to tolvaptan [18, 19].
Collecting duct function and response to tolvaptan
Given the previous studies, we focused on the residual function of the collecting duct [11, 20]. Tolvaptan antagonizes the vasopressin type-2 receptors, which are located on the collecting duct, and suppresses the reabsorption of free water. Thus, a residual function of the collecting duct should be a key to responding to tolvaptan. Of note, the whole kidney function and collecting duct function are not necessarily parallel in the CKD patients [11]. In other words, the residual function of the collecting duct is preserved even in some patients with advanced CKD.
Urine cyclic AMP/plasma AVP ratio and response to tolvaptan
Water restriction test and water intake test are applied to assess urine concentration and urine dilution ability, respectively [21, 22]; whereas, these invasive tests would not be feasible in patients with volume overload.
As non-invasive indicators, several markers have been proposed to assess collecting duct function: AVP, cyclic AMP, aquaporin-2, and urine osmolality [11, 22]. Cyclic AMP is located on the upstream of the signal cascade in the collecting duct. In patients with CKD, in which the downstream of signal cascade is relatively attenuated, the levels of downstream markers including urine aquaporin-2 and urine osmolality are decreased [11, 22]. Thus, urine cyclic AMP was the most powerful prognostic marker to predict renal outcome compared with other collecting duct-associated parameters among those with CKD [20]. The value of urine cyclic AMP would be affected by the degree of AVP stimulation. Given all together, we defined urine cyclic AMP/plasma AVP ratio as an independent variable to predict response to tolvaptan.
Clinical implication
Given a high sensitivity, a urine cyclic AMP/plasma AVP ratio would be particularly useful to discriminate non-responders (i.e., those with low value is highly expected to be non-responders). Other modalities including dialysis might be required soon and tolvaptan therapy might not be recommended in such patients. Long-term impact of tolvaptan therapy upon renal outcomes in patients with advanced CKD remains the next concern.
Limitations
This study included a small sample cohort obtained from a single center. Several comparisons might have reached statistical significance when the sample sizes were larger. We could not show the long-term advantage of tolvaptan in the responders, which requires further studies. We did not measure plasma cyclic AMP levels, which might also affect the levels of urine cyclic AMP. Other parameters including GFR and stimulation of parathyroid hormone might also affect the levels of urine cyclic AMP [13].
Conclusions
Baseline urine cyclic AMP/plasma AVP ratio is an independent predictor of response to tolvaptan in patients with advanced CKD and heart failure.
References
Wilcox CS. New insights into diuretic use in patients with chronic renal disease. J Am Soc Nephrol. 2002;13:798–805.
Gheorghiade M, Konstam MA, Burnett JC Jr, Grinfeld L, Maggioni AP, Swedberg K, et al. Short-term clinical effects of tolvaptan, an oral vasopressin antagonist, in patients hospitalized for heart failure: the EVEREST clinical status trials. JAMA. 2007;297:1332–43.
Kinugawa K, Sato N, Inomata T, Yasuda M, Shimakawa T, Fukuta Y. Real-world effectiveness and tolerability of tolvaptan in patients with heart failure - final results of the Samsca post-marketing surveillance in heart failure (SMILE) study. Circ J. 2019;83:1520–7.
Kinugawa K, Sato N, Inomata T. Effects of tolvaptan on volume overload in patients with heart failure. Int Heart J. 2018;59:1368–77.
Inomata T, Ikeda Y, Kida K, Shibagaki Y, Sato N, Kumagai Y, et al. Effects of additive tolvaptan vs. increased furosemide on heart failure with diuretic resistance and renal impairment - Results from the K-STAR study. Circ J. 2017;82:159–67.
Komiya S, Katsumata M, Ozawa M, Haze T, Kawano R, Ohki Y, et al. Efficacy of tolvaptan on advanced chronic kidney disease with heart failure: a randomized controlled trial. Clin Exp Nephrol. 2022;26:851–8.
Tanaka A, Hiramatsu E, Watanabe Y, Ito C, Shinjo H, Otsuka Y, et al. Efficacy of long-term treatment with tolvaptan to prolong the time until dialysis initiation in patients with chronic kidney disease and heart failure. Ther Apher Dial. 2019;23:319–27.
Sato E, Nakamura T, Amaha M, Nomura M, Matsumura D, Yamagishi H, et al. Effect of tolvaptan in patients with chronic kidney disease due to diabetic nephropathy with heart failure. Int Heart J. 2014;55:533–8.
Kakeshita K, Koike T, Imamura T, Fujioka H, Yamazaki H, Kinugawa K. Expression of aquaporin-2 in the collecting duct and responses to tolvaptan. CEN Case Rep. 2021;10:69–73.
Noda Y, Sasaki S. Updates and perspectives on aquaporin-2 and water balance disorders. Int J Mol Sci. 2021;22:12950.
Kakeshita K, Koike T, Imamura T, Fujioka H, Yamazaki H, Kinugawa K. Altered arginine vasopressin-cyclic AMP-aquaporin 2 pathway in patients with chronic kidney disease. Clin Exp Nephrol. 2022;26:788–96.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
Broadus AE, Mahaffey JE, Bartter FC, Neer RM. Nephrogenous cyclic adenosine monophosphate as a parathyroid function test. J Clin Invest. 1977;60:771–83.
Imamura T, Kinugawa K, Shiga T, Kato N, Muraoka H, Minatsuki S, et al. Novel criteria of urine osmolality effectively predict response to tolvaptan in decompensated heart failure patients - association between non-responders and chronic kidney disease. Circ J. 2013;77:397–404.
Imamura T, Kinugawa K, Fujino T, Inaba T, Maki H, Hatano M, et al. Increased urine aquaporin-2 relative to plasma arginine vasopressin is a novel marker of response to tolvaptan in patients with decompensated heart failure. Circ J. 2014;78:2240–9.
Iwatani H, Kawabata H, Sakaguchi Y, Yamamoto R, Hamano T, Rakugi H, et al. Urine osmolarity predicts the body weight-reduction response to tolvaptan in chronic kidney disease patients: a retrospective, observational study. Nephron. 2015;130:8–12.
Katsumata M, Hirawa N, Sumida K, Kagimoto M, Ehara Y, Okuyama Y, et al. Effects of tolvaptan in patients with chronic kidney disease and chronic heart failure. Clin Exp Nephrol. 2017;21:858–65.
Masuda T, Ohara K, Nagayama I, Matsuoka R, Murakami T, Nakagawa S, et al. Impact of serum albumin levels on the body fluid response to tolvaptan in chronic kidney disease patients. Int Urol Nephrol. 2019;51:1623–9.
Takagi K, Sato N, Ishihara S, Sone M, Tokuyama H, Nakama K, et al. Effects of tolvaptan on urine output in hospitalized heart failure patients with hypoalbuminemia or proteinuria. Heart Vessels. 2018;33:413–20.
Kakeshita K, Koike T, Imamura T, Fujioka H, Yamazaki H, Kinugawa K. Prognostic impact of urine cyclic AMP levels in patients with chronic kidney disease. Clin Exp Nephrol. 2022;26:1194–9.
Davis BB, Zenser TV. Evaluation of renal concentrating and diluting ability. Clin Lab Med. 1993;13:131–4.
Pedersen EB, Thomsen IM, Lauridsen TG. Abnormal function of the vasopressin-cyclic-AMP-aquaporin2 axis during urine concentrating and diluting in patients with reduced renal function. A case control study BMC Nephrol. 2010;11:26.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors have declared no competing interest.
Ethical approval
This study was approved by our institutional review board (IRB approval number R2015144) and carried out following the Declaration of Helsinki. Clinical trial registration: UMIN000022422.
Informed consent
Written informed consent was obtained from all patients before the inclusion in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Kakeshita, K., Koike, T., Imamura, T. et al. Impact of urine cyclic AMP relative to plasma arginine vasopressin on response to tolvaptan in patients with chronic kidney disease and heart failure. Clin Exp Nephrol 27, 427–434 (2023). https://doi.org/10.1007/s10157-023-02325-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-023-02325-1