Abstract
Background
Gastric cancer is one of the leading causes of malignant disease-related mortality, worldwide. With the use of recently developed anti-tumor agents, the prognoses of patients with unresectable gastric cancer are improving. However, the development of an aggressive treatment strategy for older patients (OPs) remains under debate due to concerns regarding treatment feasibility or patient frailty. We aimed to elucidate whether aggressive chemotherapy has survival benefits for OPs with advanced gastric cancer.
Methods
We analyzed consecutive patients diagnosed with inoperable advanced gastric cancer across seven hospitals from August 2007 to July 2015. We defined OPs as patients aged 75 years or older and compared their survival rates with those of non-older patients (NPs).
Results
A total of 256 OPs and 425 NPs were enrolled. Of the OPs, 152 patients received chemotherapy and 104 patients received best supportive care (BSC). In contrast, among the NPs, 375 patients received chemotherapy and 50 patients received BSC. There was no significant difference of the median survival time between OPs and NPs in the response to BSC (61 vs 43 days) or chemotherapy (312 vs 348 days). Combination chemotherapy significantly improved survival compared to monotherapy in both OPs and NPs groups (382 vs 253 days in OPs, 381 vs 209 days in NPs). Good performance status, combination therapy, and male, but not age, were significant independent prognostic factors.
Conclusion
When the performance status of a gastric cancer patient is good, active chemotherapy may improve survival, regardless of age.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Worldwide, the number of older patients (OPs) requiring medical intervention for malignancies is increasing, particularly in developed countries [1]. Although unresectable malignancies are commonly treated with systemic chemotherapy, many agents are notably toxic or intolerable in OPs, posing a problem when there is an indication for treatment. In addition, OPs often face many issues pertaining to comorbidity, polypharmacy, social support, and life expectancy. Frailty may lead to the avoidance of aggressive treatments by both OPs and doctors. Strikingly, there is not enough clinical evidence concerning the efficacy of chemotherapy for OPs. However, the importance of providing personalized medicine to OPs is being increasingly recognized [2].
Gastric cancer is among the most commonly reported malignancies in many countries [3]. Following the results of the SPIRITS trial in Japan, in 2007, a combination regimen became the standard first-line treatment for patients with unresectable advanced gastric cancer [4]. Combination therapies, such as those including oral 5-fluorouracil (FU) drugs, S-1 or capecitabine, plus platinum, cisplatin, or oxaliplatin, are the standard regimens for first-line treatment. Trastuzumab is added in patients with HER2 positivity. However, in many clinical trials for advanced gastric cancer, OPs were excluded from the protocol or were not referred for chemotherapy, to restrict the target population for the evaluation of efficacy and toxicity. In practice, the drugs used for OPs are chosen based on the presence of frailty, in relation to a person’s physical condition, cognitive function, and laboratory data such as renal function. In addition, quality-of-life (QOL) maintenance and adverse event rate reductions are critical for OPs; however, it is difficult to achieve these owing to the comorbidity.
Here, we performed a multicenter retrospective study (OGF1604) to examine the practical differences between the outcomes of unresectable advanced gastric cancer OPs and non-older patients (NPs).
Patients and methods
Patients
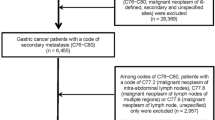
In the present study, we defined patients aged 75 years or older as OPs. From August 2007 to July 2015, a total of 681 consecutive patients was diagnosed with unresectable stage IV (Union for International Cancer Control TNM classification) gastric cancer. These patients were recruited from seven hospitals participating in the Osaka Gut Forum (OGF): Osaka University Hospital, Toyonaka Municipal Hospital, Itami City Hospital, Hyogo Prefectural Hospital, Saiseikai Senri Hospital, Kansai Rosai Hospital, and Osaka Minami National Hospital. The participants comprised 256 OPs and 425 NPs. The patients’ background data are shown in Table 1.
Data collection
We collected the following information on patients with advanced unresectable gastric cancer and tumors from medical records created at the time of diagnosis: age, sex, history of previous gastrectomy, performance status (PS), sites of metastasis, strategy, treatment, adverse events (grade 3 or over according to Common Terminology Criteria for Adverse Events ver. 4.0), and survival. Survival duration was defined as the time from clinical diagnosis to death or the last follow-up. PS was evaluated in accordance with the European Cancer Organization Group. We evaluated the efficacy of chemotherapy for OPs compared to that for NPs.
Chemotherapy
In the present study, chemotherapy regimens with doublet cytotoxic agents were defined as combination therapy, and single agent regimens were defined as monotherapy. Trastuzumab was not counted in the number of drugs.
Study approval
The analysis was approved by the Osaka University Hospital Institutional Review Board (No. 13093) and all the other institutions. All participants were provided opportunities to decline participation in this study prior to the initiation of the investigation using the ‘opt-out’ option on our hospital website. Since this study involved previously collected human data, the need for informed consent was waived. This research was performed in accordance with Ethical Guidelines for Medical and Health Research Involving Human Subjects of Ministry of Health, Labor and Welfare, and Ministry of Education, Cultural, Sports, Science and Technology in Japan. All procedures followed were in accordance with the Helsinki Declaration of 1964 and its later versions.
Statistical analysis
We analyzed survival curves using the Kaplan–Meier method, and performed statistical analysis using the log-rank test. The contributions of independent variables to the estimation of survival were analyzed using a Cox proportional hazards model using univariate and multivariate procedures. Differences were considered statistically significant at p < 0.05. Statistical analyses were performed using the JMP statistical software (ver. 13.1. 0, SAS Institute Inc., Cary, NC).
Results
Survival by treatment strategy was equivalent regardless of age
First, we analyzed all the patients regardless of age and treatment strategy. The median survival time (MST) was 273 days (Fig. 1a). The MST was significantly prolonged in NPs (OPs vs NPs; 193 vs 311 days, p = 0.002). Among the 681 patients, 527 (77%) received chemotherapy and 154 (23%) received best supportive care (BSC). For patients receiving chemotherapy, the MST was 341 days (Fig. 1b). There was no difference in the overall survival (OS) between OPs and NPs among those who received chemotherapy (312 vs 348 days) (Fig. 1c). The MST of those with BSC was 55 days (Fig. 1d). There was also no difference between OPs and NPs among those with BSC (61 vs 43 days) (Fig. 1e).
Intensive chemotherapy was efficacious regardless of age
Next, we analyzed the OS in the chemotherapy group. To elucidate whether intensive treatment improves survival in OPs, we compared patients divided into the monotherapy and combination therapy groups. In the NPs group, 77% of the patients received combination therapy, whereas 59% of the OPs received monotherapy. In this cohort, combination therapy was associated with significantly improved survival relative to monotherapy among all the patients (382 vs 239 days, p < 0.001) (Fig. 2a). Consistent with the previous prospective trials conducted among NPs, combination therapy significantly improved survival values compared to monotherapy (381 vs 209 days, p < 0.001) (Fig. 2b). Among OPs, the MST associated with combination therapy was significantly prolonged compared to monotherapy (382 vs 253 days, p = 0.014) (Fig. 2c). The most frequently used first-line protocol in combination chemotherapy was 5-FU (including S-1 and capecitabine) plus platinum (including cisplatin and oxaliplatin), with or without trastuzumab (294/353, 83.3%). To examine whether this toxic protocol was efficacious even among OPs, we compared the survival outcomes between the OPs and NPs who received this treatment. There was no difference in the survival time between the OPs and NPs, and the survival time was generally acceptable (382 vs 401 days, p = 0.901) (Fig. 2d).
Overall survival by monotherapy or combination therapy. a Overall survival curve for patients who underwent chemotherapy, stratified by monotherapy or combination therapy. b Overall survival curve for non-older patients by monotherapy or combination therapy. c Overall survival curve for older patients by monotherapy or combination therapy. d Overall survival curve for patients who underwent the 5-fluorouracil- and platinum-based regimen. MST median survival time, Comb combination therapy, Mono monotherapy, NPs non-older patients, OPs older patients
Performance status affected survival prolongation regardless of age
Next, we analyzed the patients’ PS to elucidate the prognostic factors contributing to prolonged survival. In patients receiving chemotherapy, good PS contributed to survival extension (PS = 0, 1, 2, and 3; MST = 503, 348, 200, and 177 days, respectively) (Fig. 3a). However, in the BSC group patients, the clinical outcomes were poor, regardless of PS (PS = 0, 1, 2, and 3–4; MST = 163, 52, 55, and 47 days, respectively) (Fig. 3b). In NPs receiving chemotherapy, the survival values were better among those with good PS (PS = 0, 1, 2, and 3; MST = 439, 352, 263, and 96 days, respectively) (Figs. 3c). Similarly, in OPs, receiving chemotherapy, the MST was prolonged in accordance with PS (PS = 0, 1, 2, and 3; MST = 599, 323, 193, and 177 days, respectively) (Fig. 3d). Particularly, the MST in OPs with PS 0 who underwent chemotherapy was 599 days. Then, we analyzed the factors contributing to survival in those with chemotherapy. To evaluate the prognostic factors related to prolonged survival in the patients with chemotherapy, we performed univariate and multivariate analyses (Table 2). Male sex, combination chemotherapy, and good PS were significantly and independently related to prolonged survival. Younger age was not a significant factor.
Overall survival by performance status. a Overall survival curve for patients who underwent chemotherapy by performance status (PS). b Overall survival curve for best supportive care patients by PS. c Overall survival curve for non-older patients who received chemotherapy by PS. d Overall survival curve for older patients who received chemotherapy by PS. MST median survival time, BSC best supportive care, NPs non-older patients, OPs older patients
Intensive chemotherapy was efficacious when PS was good, even among OPs
Finally, we evaluated the patients’ survival outcomes by PS 0 and PS 1–3 between patients who underwent monotherapy and combination therapy. For OPs with PS0, combination therapy significantly prolonged survival compared to monotherapy (740 vs 304 days, p = 0.003) (Fig. 4a), but there was no significant difference between monotherapy and combination therapy in NPs with PS0 (439 vs 514 days, p = 0.456) (Fig. 4b). Although there was no significant difference between monotherapy and combination therapy in OPs with PS 1–3 (323 vs 209 days, p = 0.126) (Fig. 4c), combination therapy was significantly efficacious in NPs with PS 1–3 (361 vs 197 days, p < 0.001) (Fig. 4d).
Overall survival by chemotherapy and performance status. a Overall survival curve for older patients, whose performance status (PS) was 0, by monotherapy or combination therapy. b Overall survival curve for non-older patients whose PS was 0, by monotherapy or combination therapy. c Overall survival curve for older patients whose PS was 1–4, by monotherapy or combination therapy. d Overall survival curve for non-older patients, whose PS was 1–4, by monotherapy or combination therapy. OPs older patients, MST median survival time, NPs non-older patients, Comb combination therapy, Mono monotherapy
Discussion
Our results suggest that the survival values were equivalent for OPs and NPs with BSC or chemotherapy, that OPs can benefit from intensive combination chemotherapy, and that the main predictor of prolonged survival is not the absolute age of the patient.
In the decision-making process for OPs with malignancies, the biological features of the cancer, treatment efficacy, patients’ preferences or social circumstances, and drug toxicity are taken into consideration. As doctors often hesitate to use combination chemotherapy in patients with an advanced age, the degree of frailty or vulnerability among OPs should be promptly assessed. This multicenter study demonstrated the clinical outcomes of gastric cancer patients in real-world practice, in which the decision strategy was based on the general condition of patients and not age alone.
PS is a classic and important clinical factor that indicates one’s general condition [5]. In our study, PS was clearly associated with prognoses in both the NPs and OPs receiving chemotherapy. Concerning the MST associated with chemotherapy, OPs with PS = 0 had a longer MST than NPs with PS = 0 (599 vs 439 days). The MST of OPs with PS = 1 was equivalent to that of NPs with PS = 1 (323 vs 352 days), but OPs with PS = 2 had a shorter MST than NPs with PS = 2 (193 vs 263 days). Regarding NPs, the standard chemotherapy regimen used is of the combination type. It is not known why the efficacy of monotherapy for NPs with PS = 0 was equivalent to that in combination therapy. Our results suggest that a sufficient survival benefit was obtained from chemotherapy, even monotherapy, in OPs. Geriatric assessment tools have been developed to evaluate frailty in OPs [6, 7]. However, a cancer-specific geriatric assessment tool has not yet been established. If a useful assessment tool is developed in addition to PS, the decision-making strategy for vulnerable OPs may become easier.
In Japan, S-1 plus cisplatin combination therapy was established as the standard chemotherapy regimen for relapsed or metastatic gastric cancer, according to a phase III trial performed among patients aged 20–74 years. However, limited existing data on 70–74-year-old patients do not show that S-1 plus cisplatin is significantly superior [4]. A phase II trial that was performed in patients older than 76 years with a PS = 0–1 demonstrated that S-1 plus cisplatin yielded good OS values (12.3 months) [8]. Some pooled analyses have focused on OPs with gastric cancer. For instance, the efficacies of S-1 and S-1 plus cisplatin were compared in patients aged 70 years or older [9]. The median OS was 10.4 months in the S-1 group and 17.8 months in the S-1 plus cisplatin group. Oxaliplatin has a lower toxicity than cisplatin and is a potentially useful agent for OPs. A secondary analysis of the G-SOX trial [10] demonstrated that the S-1 and oxaliplatin combination was noninferior to S-1 plus cisplatin, as no difference in OS was shown between the two among OPs aged 70 years or older [11]. An interim report of a phase III trial comparing capecitabine and capecitabine plus oxaliplatin (XELOX) in OPs with gastric cancer was recently published. A survival benefit for patients aged 70 or older was shown in association with XELOX compared to capecitabine alone [12]. Targeted agents seem to be the optional treatment option for OPs due to their reduced toxicity [13]. The efficacy of trastuzumab was shown for HER2-positive gastric cancer in the ToGA trial [14]. That phase II study, in which S-1 plus trastuzumab was administered to patients aged over 76 years with PS = 0–2, reported a median OS of 15.8 months [15]. A phase II study of S-1 monotherapy that included 35 patients with gastric cancer aged older than 75 years revealed a median OS duration of 14.6 months [16]. Available data suggest that combination chemotherapy can be considered for non-frail OPs, while monotherapy may be considered for frail or vulnerable OPs [17]. In the present study, intensive combination chemotherapy was significantly efficacious when the general condition of the patient was good. The analysis conducted among NPs revealed that intensive chemotherapy significantly extended patients’ survival duration, even in the presence of poor PS.
This study has several limitations. First, due to its retrospective nature, we had no input pertaining to the proper treatment strategy, and these decisions were made based on the individual assessment of the treating physician. Toxicities might affect the outcome of chemotherapy. There were differences of the rate of severe adverse events between combination therapy and monotherapy in both OPs and NPs. However, the profile of toxicities was equivalent between the OPs and NPs. Our results indicate that intensive chemotherapy can be administered to OPs with good PS. Second, some indices, such as the prognostic nutrition index and Charlson comorbidity index, have recently been proven to be useful in such settings [5, 18]; as ours was a retrospective multicenter study, we could not evaluate these indicators. In addition, blood examination data such as tumor markers at the time of diagnosis or the reasons of chemotherapy discontinuation were not investigated. Third, the drugs used in practice have been changing recently, with nivolumab being applied with good results. However, the present cohort did not include new agents and investigate regimens after secondary treatment. The importance of precision medicine is increasing. While chronological age is an important factor, it is not absolute. Our results suggest that a combination of cytotoxic agents can be considered for OPs with good PS, and monotherapy can also have a survival benefit in OPs. In the future, the development of targeted agents or immune checkpoint inhibitors may be promising for OPs with gastric cancer owing to the reduced toxicity. This study is of value, as it highlights the need to consider intensive treatments for both OPs and NPs, particularly among those with a good general condition.
References
Pilleron S, Sarfati D, Janssen-Heijnen M et al (2019) Global cancer incidence in older adults, 2012 and 2035: a population-based study. Int J Cancer 144:49–58
Walko CM, McLeod HL (2014) Personalizing medicine in geriatric oncology. J Clin Oncol 32:2581–2586
Bertuccio P, Chatenoud L, Levi F et al (2009) Recent patterns in gastric cancer: a global overview. Int J Cancer 125:666–673
Koizumi W, Narahara H, Hara T et al (2008) S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): a phase III trial. Lancet Oncol 9:215–221
Sachlova M, Majek O, Tucek S (2014) Prognostic value of scores based on malnutrition or systemic inflammatory response in patients with metastatic or recurrent gastric cancer. Nutr Cancer 66:1362–1370
Extermann M, Hurria A (2007) Comprehensive geriatric assessment for older patients with cancer. J Clin Oncol 25:1824–1831
Rodin MB, Mohile SG (2007) A practical approach to geriatric assessment in oncology. J Clin Oncol 25:1936–1944
Sasaki Y, Iwasa S, Okazaki S et al (2017) A phase II study of combination therapy with oral S-1 and cisplatin in elderly patients with advanced gastric cancer. Gastric Cancer 21:439–445
Terazawa T, Iwasa S, Takashima A et al (2013) Impact of adding cisplatin to S-1 in elderly patients with advanced gastric cancer. J Cancer Res Clin Oncol 139:2111–2116
Koizumi W, Takiuchi H, Yamada Y et al (2010) Phase II study of oxaliplatin plus S-1 as first-line treatment for advanced gastric cancer (G-SOX study). Ann Oncol 21:1001–1005
Bando H, Yamada Y, Tanabe S et al (2016) Efficacy and safety of S-1 and oxaliplatin combination therapy in elderly patients with advanced gastric cancer. Gastric Cancer 19:919–926
Hwang IG, Ji JH, Kang JH et al (2017) A multi-center, open-label, randomized phase III trial of first-line chemotherapy with capecitabine monotherapy versus capecitabine plus oxaliplatin in elderly patients with advanced gastric cancer. J Geriatr Oncol 8:170–175
Kelly CM, Power DG, Lichtman SM (2014) Targeted therapy in older patients with solid tumors. J Clin Oncol 32:2635–2646
Bang YJ, Van Cutsem E, Feyereislova A et al (2010) Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 376:687–697
Kimura Y, Fujii M, Masuishi T et al (2018) Multicenter phase II study of trastuzumab plus S-1 alone in elderly patients with HER2-positive advanced gastric cancer (JACCRO GC-06). Gastric Cancer 21:421–427
Imamura H, Kishimoto T, Takiuchi H et al (2014) Phase II study of S-1 monotherapy in patients over 75 years of age with advanced gastric cancer (OGSG0404). J Chemother 26:57–61
Kim HS, Kim JH, Kim JW et al (2016) Chemotherapy in elderly patients with gastric cancer. J Cancer 7:88–94
Kim ST, Park KH, Oh SC et al (2012) Is chemotherapy in elderly patients with metastatic or recurrent gastric cancer as tolerable and effective as in younger patients? Asia Pac J Clin Oncol 8:194–200
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All author declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Hayashi, Y., Nishida, T., Tsutsui, S. et al. Efficacy of chemotherapy for older patients with gastric cancer: a multicenter retrospective cohort study. Int J Clin Oncol 24, 1377–1384 (2019). https://doi.org/10.1007/s10147-019-01501-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-019-01501-2