Abstract
A novel mesophilic member of the Thermotogales, strain MesG1.Ag.4.2, was isolated from sediments from Baltimore Harbor, MD, USA. The strain grew optimally at 37 °C with a doubling time of 16.5 h on xylose. Carbohydrates and proteinaceous compounds supported growth and pentoses were preferred over hexoses. The strain was strictly anaerobic and growth was slightly stimulated by thiosulfate, sulfite, and elemental sulfur. The G + C content of its genomic DNA was 45.3 mol%. Strain MesG1.Ag.4.2 and Kosmotoga olearia lipids were analyzed. Strain MesG1.Ag.4.2 contained no long-chain dicarboxylic acids and its major phospholipid was lyso-phosphatidylserine. Long-chain dicarboxylic acids were found in K. olearia and its major phospholipid was cardiolipin, a lipid not yet reported in Thermotogales species. Phylogenetic analyses of its two 16S rRNA genes placed strain MesG1.Ag.4.2 within the bacterial order Thermotogales. Based on the phylogenetic analyses and its low optimal growth temperature, it is proposed that the strain represents a novel species of a new genus within the family Thermotogaceae, order Thermotogales. The name Mesotoga prima gen. nov., sp. nov. is proposed. The type strain of M. prima is MesG1.Ag.4.2 (= DSM 24739 = ATCC BAA-2239).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
All described species of the bacterial order Thermotogales are extreme thermophiles or hyperthermophiles, but 16S rRNA gene sequences amplified from samples from low temperature environments have been found to contain Thermotogales sequences (Nesbø et al. 2010). These sequences are similar enough to one another to the exclusion of other Thermotogales sequences to suggest that they belong to a distinct evolutionary lineage. Because of their unusual environmental affiliation, organisms in this lineage were given the trivial name mesotogas (Nesbø et al. 2006). We have isolated one of these mesotoga species and have demonstrated that, as predicted, it grows at a much lower optimal growth temperature than previously described Thermotogales species. There is a report of the isolation of another member of the mesotogas (Ben Hania et al. 2011), though that isolate was incompletely described. It also apparently grew at a mesophilic temperature. These isolates confirm the predicted mesophilic nature of the mesotoga lineage.
Isolation of strain MesG1.Ag.4.2
The isolate described here was taken from an enrichment culture for polychlorinated biphenyl-reducers inoculated with sediments of the Northwest Branch of Baltimore Harbor, Baltimore, MD, USA (Holoman et al. 1998). The enrichment medium contained short-chain fatty acids as carbon source and was amended with 2,3,5,6-tetrachlorobiphenyl, but not inorganic electron acceptors. After extended incubations at 30 °C through three sequential transfers, cultures were reportedly enriched for a relative of Thermotoga maritima (Holoman et al. 1998). This enrichment was obtained from those authors and used as an inoculum to isolate this mesotoga strain.
The strain was isolated by transferring 1 ml of the PCB-dechlorinating enrichment culture to 20 ml of a standard Thermotogales medium (Takahata et al. 2001) and incubated at room temperature (~22 °C) under nitrogen for about 5 months. One ml of this culture was transferred to fresh anoxic medium and incubated at 45 °C. After 2 months, 1 ml of this culture was transferred into 20 ml fresh medium with 1 ml of 10 % (w/v) glucose added and again incubated at room temperature. A PCR analysis using ‘universal’ 16S rRNA gene primers (16S.27F sequence: 5′-AGA GTT TGA TCC TGG CTC AG-3′ and 16S.1406R: 5′-ACG GGC GGT GTG TRC-3′) indicated that this culture contained mainly ‘mesotoga’ bacteria. We, therefore, made a tenfold dilution series up to a 10−6 dilution and 2 ml of 3 % low melt agarose was added to each Hungate tube of 9 ml inoculated media. The media were then allowed to solidify within the tubes, and the tubes were incubated upside-down. Three white round colonies were picked from the 10−4 dilution after 4 months incubation at room temperature. The 16S rRNA gene sequences of these isolates were identical (not shown). One of these isolates, MesG1.Ag.4.2, was chosen for further characterization.
Physiological characterization of strain MesG1.Ag.4.2
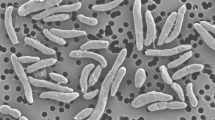
Cells of strain MesG1.Ag.4.2 were grown to exponential phase and samples prepared for microscopy as described by Spurr (1969). Images were acquired using a Philips Morgagni 268 transmission electron microscope (Philips-FEI, Hillsboro, OR, USA) operating at 80 kV with Gatan Orius CCD camera. Strain MesG1Ag.4.2 grew as 1 × 1.5 μm ovoid cells with distinct togas during exponential phase (Fig. 1). Cocci and pleomorphic cells were also observed at all growth stages. Motility was not apparent and no flagella were seen in electron micrographs.
To test the effects on growth of different medium components, a basal medium was prepared that contained (l−1) 40 g NaCl, 0.66 g KCl, 1.8 g MgCl2·2H2O, 2.8 g MgSO4·7H2O, 0.5 g NH4Cl, 0.28 g CaCl2·2H2O, and 0.9 g KH2PO4, 6.7 g PIPES, 5.0 g yeast extract, 0.5 g cysteine, 2.5 g Na2S2O3·5H2O, 0.001 g resazurin, 10 ml trace mineral solution (Balch et al. 1979), and 10 ml vitamin solution (Balch et al. 1979). The pH was adjusted to 6.8 with NaOH, placed in a steam box for 20 min to remove dissolved air, and flushed with N2 for 15–20 min. It was dispensed into Balch tubes in 10 ml aliquots, stoppered and crimp sealed inside a Coy anaerobic chamber (2–4 % H2 in N2), and then autoclaved at 121 °C for 40 min. After autoclaving, the medium was supplemented with 0.5 % (w/v) maltose except for when carbon sources were tested. All tubes were inoculated with a 0.1 ml inoculum. The optical density of triplicate cultures at 660 nm was used to measure growth and results are reported based on maximal optical densities achieved.
Growth was tested at 15, 20, 37, 42, 50, 65, and 80 °C at pH 6.8 and cells grew at 20–50 °C with an optimum of 37 °C. No growth was observed at 15, 65, and 80 °C. The effect of pH on growth was tested using cultures at pH 5.0–9.0 prepared in media buffered with the following at 20 mM concentrations: MES, pH 5.0 and 6.0; PIPES, pH 6.5 and 7.0; HEPES, pH 7.5 and 8.0; and AMPSO, pH 9.0. Inoculated cultures were incubated at 42 °C. Growth was observed over a range of pH 6.5–8.0 with best growth at pH 7.5.
To identify the optimum NaCl concentration, basal medium was adjusted to contain NaCl concentrations from 0 to 60 g l−1. The inoculum was grown in basal medium with 60 g l−1 NaCl and test cultures were incubated at 45 °C. Growth rates were determined in triplicate and growth was measurable between 20 and 60 g l−1 with best growth at 40 g l−1. A modified basal medium containing 40 mg l−1 NaCl was used in the media for all subsequent tests of growth conditions and potential growth inhibitors.
Carbon sources were tested at a final concentration of 0.5 % (w/v) and incubated at 42 °C (Table 1). Growth was measured after inoculation with cultures that had been passed a minimum of three times in the media containing the test carbon source. Yeast extract was required for growth. We routinely used 0.5 % yeast extract and growth occurred on this amount of yeast extract without any added carbon source. Final optical densities were about one-half of the optical densities observed with the best carbon sources. Cells would grow on lower concentrations of yeast extract, but much more slowly. At 0.5 % yeast extract under optimal conditions, cultures typically grew to maximal densities in 2–3 weeks, so for practical purposes, we chose to use the relatively high concentration. Xylose, fructose, ribose, and sucrose stimulated growth of the best above this background growth. Moderate growth stimulation was seen with mannose, galactose, maltose, lactose, Bacto-peptone, tryptone, and casamino acids. Weaker growth stimulation was found with glucose, arabinose, cellobiose, casein, pyruvate, and myo-inositol. Carboxymethyl cellulose, chitin, glycerol, propionate, butanol, methanol, and putrescine did not support growth above the background level. Based on these tests, xylose (0.5 % w/v) was used in the media for all subsequent tests of media conditions and potential growth inhibitors.
To test electron acceptors, the basal medium was modified by replacing MgSO4·7H2O with 4.6 g l−1 MgCl2·6H2O and omitting Na2S2O3·5H2O. After autoclave sterilization, sterile (or tyndallized in the case of elemental sulfur) stock solutions of different electron acceptors were added to the xylose media at a final concentration of 2.5 % (w/v). Cystine, sulfate, nitrate, and nitrite did not stimulate growth. Slight growth stimulation was observed in the presence of thiosulfate, sulfite, and elemental sulfur.
Strain MesG1.Ag.4.2 had a doubling time of 16.5 h (specific growth rate = 0.06 h−1), while growing on its best carbon source, xylose, in its optimized medium at 37 °C.
Soluble fermentation products from growth on xylose were determined by gas chromatography using an Agilent GC-quadrupole MS system (Models 7890A GC/5975C MS). Acetic acid was the major product formed by xylose fermentation with much smaller amounts of butyric, isobutyric, isovaleric, and 2-methyl-butyric acid. These compounds were not detected in extracts of uninoculated media.
Antibiotic sensitivities were tested at concentrations of 10, 25, 50, and 100 μg ml−1. Growth in the presence of ampicillin, carbenicillin, kanamycin, streptomycin, and vancomycin was all tested versus a control medium with no additions. For chloramphenicol and rifampicin, control cultures containing ethanol and DMSO were prepared, since those solvents were used to dissolve these antibiotics, respectively. The strain grew in the presence of 100 μg ml−1 of all these antibiotics, except streptomycin. Streptomycin concentrations of 25 μg ml−1 and above completely inhibited its growth.
Oxygen tolerance was tested by removing 2 or 20 ml of the headspace from tubes of sterile media (without resazurin) and replacing the removed headspace with equal volumes of filter sterilized ambient air prior to inoculation. Inocula used to test these conditions were from cultures grown through three successive passages in tubes prepared under identical conditions. Cells were able to grow nearly as well as the control in the tubes containing 2 ml air (2 % oxygen), but grew to a much lesser extent in tubes containing 20 ml air (14 % oxygen).
Lipids of strain MesG1.Ag.4.2 and closest cultivated relative, Kosmotoga olearia were analyzed. Mesotoga prima and K. olearia were grown at 35 and 55 °C, respectively. Total lipids were extracted from 5 mg (wet weight) harvested M. prima and K. olearia cells using a modified Bligh–Dyer method (Bligh and Dyer 1959; White and Ringelberg 1998). M. prima contained branched, mono-unsaturated and saturated fatty acids (predominantly n-C16 and n-C18) (Table 2). Branched-chain fatty acids were not detected in K. olearia, but it contained n-C16 and n-C18 saturated fatty acids.
No long-chain dicarboxylic acids were detected in M. prima, but a long-chain dicarboxylic acid (n-C32) identified as 15, 16-dimethyltriacontanedioic based on mass spectral data reported previously was detected in K. olearia (Damsté et al. 2007; Jung et al. 1993). The lack of long-chain dicarboxylic acids in M. prima, which are thought to be an adaptation to growth at high temperature (Damsté et al. 2007), is consistent with the mesophilic nature of this isolate.
Polar lipids were separated from the total lipids extracted by column chromatography and analyzed by thin layer chromatography (White and Ringelberg 1998). Lipid samples and phospholipid standards were applied to Silica gel 60 plates (Merck) and developed using chloroform–methanol-H2O MiLi Q (65:25:4, v/v/v) solvent system (Novik et al. 2006). The chromatogram was visualized with molybdenum blue spray reagent (Sigma-Aldrich). The major phospholipid of M. prima has a R f value of 0.17 and was identified as lyso-phosphatidylserine (R f 0.18; Avanti polar lipids) (Table 3). It is absent from K. olearia. A phospholipid with R f of 0.17 was also reported for Thermotoga subterranean (Jeanthon et al. 1995). The major phospholipid in K. olearia is cardiolipin (R f of 0.67), which is also present in M. prima, but as a minor phospholipid. Cardiolipin content increases with increased growth temperature of Bacillus megaterium (Rilfors et al. 1978) which might account for the presence of cardiolipin in the facultatively thermophilic K. olearia, but not the mesophilic M. prima. To our knowledge, cardiolipin has not been reported in any Thermotogales species. Furthermore, three other minor phospholipids in K. olearia were tentatively identified, which were not present in M. prima (Table 3). Glycolipids are present in both M. prima and K. olearia.
rRNA gene and genome sequence evidence for a new Thermotogales lineage
The genome sequence of strain MesG1.Ag.4.2 has been determined and from those data the G + C content of its genomic DNA was found to be 45.3 mol%. The phylogenetic position of strain MesG1.Ag.4.2 was determined by comparing the sequence of its 16S rRNA genes with those of other members of the Thermotogales. A maximum likelihood tree was constructed from Thermotogales 16S rRNA gene sequences using PhyML (GTR model). 16S rRNA genes were amplified using the universal primers 16S.27F and 16S.1406R, listed above. The isolate encodes two non-identical 16S rRNA genes that are 99.1 % similar (Fig. 2). The presence of two 16S rRNA genes was confirmed by inspecting the unfinished genome sequence of the isolate. Neither of the two genes is identical to the 16S rRNA gene in the fosmid clone sequenced by Nesbø et al. (2006) (Fig. 2), thus at least two strains of MesG1.Ag.4.2 are present in the original enrichment cultures generated by Holoman et al. (1998). The two 16S rRNA gene sequences from MesG1.Ag.4.2 were deposited in GenBank as accession numbers HM003108 and HM003109.
Maximum likelihood tree constructed from Thermotogales 16S rRNA sequences using PhyML (GTR model + γ +Ι). AM184116 is the 16S rRNA gene sequence detected in the fosmid clone that was sequenced in Nesbø et al. (2006) and FN611033 is the ‘Mesotoga’ sequence reported in Ban Hania et al. (2011). The Thermotogales tree was rooted on the branch leading to Aquifex and the Firmicutes. Mesotoga prima strain MesG1.Ag.4.2 encodes two non-identical 16S rRNA genes that are 99.1 % similar
The draft sequence of strain MesG1.Ag.4.2 and the complete sequence of its closest cultivated relative, K. olearia, were used to calculate the average nucleotide identity (ANI) of their shared genes and this was 68.5 %, a value well below the ~94 % suggested to distinguish species (Konstantinidis and Tiedje 2005). The 16 rRNA A gene sequence of strain MesG1.Ag.4.2 shows 89.2 % similarity to that of K. olearia, while the sequence of its16S rRNA B gene shows 88.9 % similarity to the K. olearia sequence.
Strain MesG1.Ag.4.2 belongs to a lineage previously identified only through 16S rRNA gene sequences culled from samples taken from low temperature environments (Briones et al. 2007; Chouari et al. 2005; Dollhopf et al. 2001; Enright et al. 2007; Fernandez et al. 2008; Holoman et al. 1998; Leven et al. 2007; Pearson et al. 2008; Pham et al. 2009; Rowe et al. 2008; Watts et al. 2001; Wu et al. 2000; Yan et al. 2006; Yoshida et al. 2005; Zhao et al. 2008). Consequently, these phylotypes were called mesotogas to reflect their likely mesophilic growth temperatures (Nesbø et al. 2006, 2010). Strain MesG1.Ag.4.2 is the first characterized isolate in this group.
On the basis of the rRNA gene sequence evidence and evaluation of the ANI with its closest cultured relative, strain MesG1.Ag.4.2 represents a novel species within a previously unknown genus of the Thermotogales, which we propose to name M. prima gen. nov., sp. nov. Table 1 summarizes the differences between Mesotoga and composite phenotypes of the closest Thermotogales genera.
Description of Mesotoga gen. nov
Mesotoga (me.so.to’ga Gr. adj. mésos middle; L. fem. n. toga Roman outer garment; Mesotoga, a “garment in the middle,” referring to its moderate optimal growth temperature and the presence of a ‘toga’-like sheath).
Cells are round to ovoid, short rods with a sheath-like outer envelope that extends beyond the cytoplasmic aspect of cells in some phases of growth. Optimal growth temperature is in the mesophile range. Grows as a chemo-organotroph fermenting sugars and proteinaceous substrates. The 16S rRNA gene sequence comparisons place it in the order Thermotogales. The type species is M. prima strain MesG1.Ag.4.2.
Description of Mesotoga prima sp. nov.
Mesotoga prima (pri’ma. L. fem. prima, first; referring to the fact that this is the first characterized and named mesophilic representative of the Thermotogales).
Cells are ovoid with cocci and pleomorphic forms evident at all growth stages. Gram-negative, growing as a slightly aerotolerant fermentative chemo-organotroph. Grows on sugars and some proteinaceous substrates. Acetic acid is the major soluble fermentation product. Thiosulfate, sulfite, and elemental sulfur slightly stimulate growth. Grows optimally at 37 °C, but not over 50 °C. Grows best at pH 7.5 and 4 % NaCl. The G + C content is 45.3 % (from genome sequence).
Isolated from Baltimore Harbor, MD, USA sediments. The type strain is MesG1.Ag.4.2 (=DSM 24739, =ATCC to be inserted when accession number is assigned).
References
Balch WE, Fox GE, Magrum LJ, Woese CR, Wolfe RS (1979) Methanogens: reevaluation of a unique biological group. Microbiol Rev 43:260–296
Ben Hania W, Ghodbane R, Posteca A, Brochier-Armanetc C, Hamdi M, Fardeau M-L, Ollivier B (2011) Cultivation of the first mesophilic representative (“mesotoga”) within the order Thermotogales. Syst Appl Microbiol 34:581–585
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Briones AM, Daugherty BJ, Angenent LT, Rausch KD, Tumbleson ME, Raskin L (2007) Microbial diversity and dynamics in multi- and single-compartment anaerobic bioreactors processing sulfate-rich waste streams. Environ Microbiol 9:93–106
Chouari R, Le Paslier D, Daegelen P, Ginestet P, Weissenbach J, Sghir A (2005) Novel predominant archaeal and bacterial groups revealed by molecular analysis of an anaerobic sludge digester. Environ Microbiol 7:1104–1115
Damsté JS, Rijpstra WI, Hopmans EC, Schouten S, Balk M, Stams AJ (2007) Structural characterization of diabolic acid-based tetraester, tetraether and mixed ether/ester, membrane-spanning lipids of bacteria from the order Thermotogales. Arch Microbiol 188:629–641
Dollhopf SL, Hashsham SA, Tiedje JM (2001) Interpreting 16S rDNA T-RFLP data: application of self-organizing maps and principal component analysis to describe community dynamics and convergence. Microb Ecol 42:495–505
Enright AM, Collins G, O’Flaherty V (2007) Temporal microbial diversity changes in solvent-degrading anaerobic granular sludge from low-temperature (15 degrees C) wastewater treatment bioreactors. Syst Appl Microbiol 30:471–482
Fernandez N, Diaz EE, Amils R, Sanz JL (2008) Analysis of microbial community during biofilm development in an anaerobic wastewater treatment reactor. Microb Ecol 56:121–132
Holoman TR, Elberson MA, Cutter LA, May HD, Sowers KR (1998) Characterization of a defined 2,3,5, 6-tetrachlorobiphenyl-ortho-dechlorinating microbial community by comparative sequence analysis of genes coding for 16S rRNA. Appl Environ Microbiol 64:3359–3367
Jeanthon C, Reysenbach AL, Lharidon S, Gambacorta A, Pace NR, Glenat P, Prieur D (1995) Thermotoga subterranea sp. nov., a new thermophilic bacterium isolated from a continental oil reservoir. Arch Microbiol 164:91–97
Jung S, Lowe SE, Hollingsworth RI, Zeikus JG (1993) Sarcina ventriculi synthesizes very long chain dicarboxylic acids in response to different forms of environmental stress. J Biol Chem 268:2828–2835
Konstantinidis KT, Tiedje JM (2005) Genomic insights that advance the species definition for prokaryotes. Proc Natl Acad Sci USA 102:2567–2572
Leven L, Eriksson AR, Schnurer A (2007) Effect of process temperature on bacterial and archaeal communities in two methanogenic bioreactors treating organic household waste. FEMS Microbiol Ecol 59:683–693
Nesbø CL, Dlutek M, Zhaxybayeva O, Doolittle WF (2006) Evidence for existence of “mesotogas,” members of the order Thermotogales adapted to low-temperature environments. Appl Environ Microbiol 72:5061–5068
Nesbø CL, Kumaraswamy R, Dlutek M, Doolittle WF, Foght J (2010) Searching for mesophilic Thermotogales bacteria: “mesotogas” in the wild. Appl Environ Microbiol 76:4896–4900
Novik GI, Astapovich NI, Grzegorzewicz A, Gamian A (2006) Analysis of phospholipids in Bifidobacteria. Microbiol 75:29–34
Pearson A, Kraunz KS, Sessions AL, Dekas AE, Leavitt WD, Edwards KJ (2008) Quantifying microbial utilization of petroleum hydrocarbons in salt marsh sediments by using the 13C content of bacterial rRNA. Appl Environ Microbiol 74:1157–1166
Pham VD, Hnatow LL, Zhang S, Fallon RD, Jackson SC, Tomb JF, DeLong EF, Keeler SJ (2009) Characterizing microbial diversity in production water from an Alaskan mesothermic petroleum reservoir with two independent molecular methods. Environ Microbiol 11:176–187
Rilfors L, Wieslander A, Ståhl S (1978) Lipid and protein composition of membranes of Bacillus megaterium variants in the temperature range 5 to 70 degrees C. J Bacteriol 135:1043–1052
Rowe AR, Lazar BJ, Morris RM, Richardson RE (2008) Characterization of the community structure of a dechlorinating mixed culture and comparisons of gene expression in planktonic and biofloc-associated “Dehalococcoides” and Methanospirillum species. Appl Environ Microbiol 74:6709–6719
Spurr AR (1969) A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26:31–43
Takahata Y, Nishijima M, Hoaki T, Maruyama T (2001) Thermotoga petrophila sp. nov. and Thermotoga naphthophila sp. nov., two hyperthermophilic bacteria from the Kubiki oil reservoir in Niigata, Japan. Int J Syst Evol Microbiol 51:1901–1909
Watts JE, Wu Q, Schreier SB, May HD, Sowers KR (2001) Comparative analysis of polychlorinated biphenyl-dechlorinating communities in enrichment cultures using three different molecular screening techniques. Environ Microbiol 3:710–719
White DC, Ringelberg DB (1998) Signature lipid biomarker analysis. In: Burlage RS, Atlas R, Stahl D, Geesey G, Sayler G (eds) Techniques in microbial ecology. Oxford University Press, New York, pp 255–272
Wu Q, Sowers KR, May HD (2000) Establishment of a polychlorinated biphenyl-dechlorinating microbial consortium, specific for doubly flanked chlorines, in a defined, sediment-free medium. Appl Environ Microbiol 66:49–53
Yan T, LaPara TM, Novak PJ (2006) The reductive dechlorination of 2,3,4,5-tetrachlorobiphenyl in three different sediment cultures: evidence for the involvement of phylogenetically similar Dehalococcoides-like bacterial populations. FEMS Microbiol Ecol 55:248–261
Yoshida N, Takahashi N, Hiraishi A (2005) Phylogenetic characterization of a polychlorinated-dioxin-dechlorinating microbial community by use of microcosm studies. Appl Environ Microbiol 71:4325–4334
Zhao Y, Ren N, Wang A (2008) Contributions of fermentative acidogenic bacteria and sulfate-reducing bacteria to lactate degradation and sulfate reduction. Chemosphere 72:233–242
Acknowledgments
This work was supported by funds to KMN from the NASA Exobiology program (NNX08AQ10G), the US Department of Energy Office of Biological and Environmental Research (DE-PS02-08ER08-12), and the National Science Foundation Assembling the Tree of Life program (DEB0830024) and by funds to CLN from the Norwegian research council (project no. 180444/V40). The authors thank Joy Watts at the University of Portsmouth, United Kingdom for the enrichment cultures from which M. prima was isolated, Dr. Randy Mandryk, University of Alberta, for assistance with TEM, and Dr. Olga Zhaxybayeva, West Virginia University, for assistance with genome analysis, and Lauren Bradford for technical assistance. We also thank anonymous reviewers of an earlier manuscript for their helpful comments and suggestions on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. da Costa.
The GenBank accession numbers for the 16S rRNA gene sequence of Mesotoga prima strain MesG1.Ag.4.2 are HM003108 and HM003109.
Rights and permissions
About this article
Cite this article
Nesbø, C.L., Bradnan, D.M., Adebusuyi, A. et al. Mesotoga prima gen. nov., sp. nov., the first described mesophilic species of the Thermotogales. Extremophiles 16, 387–393 (2012). https://doi.org/10.1007/s00792-012-0437-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-012-0437-0