Abstract
Objectives
We evaluated the effects of low-level laser therapy (LLLT) using an infrared laser (IRL) and a red laser (RL) on the pulp of molar teeth in rats after dental bleaching to assess inflammation, collagen fiber maturation, and tertiary dentin formation.
Materials and methods
Eighty Wistar rats (Rattus norvegicus, albinus) were randomly divided into eight groups with 10 hemimaxillae in each of the following: control; bleached (Ble, 35% hydrogen peroxide [H2O2]); Ble-1IRL and Ble-1RL (one IRL [808 nm, 30 s, 3 J] or RL [660 nm, 15 s, 1.5 J] application immediately after H2O2); Ble-3IRL and Ble-3RL (three [immediately, 24 h, and 48 h] IRL or RL applications after H2O2); and 3IRL and 3RL (three IRL or RL applications without bleaching). The rats were euthanized after 2 and 30 days for histological evaluation of inflammation (hematoxylin-eosin) and maturation of collagen fibers (picrosirius red). Additionally, the dentin deposition in the specimens obtained at 30 days was quantified via microtomography of the pulp chamber volume. Statistical analyses were performed (P < 0.05).
Results
Initially, severe damages to the pulp were observed in the Ble and Ble-1RL groups. Ble-1IRL and Ble-3RL groups showed lower inflammation. The bleached groups had a greater amount of mature collagen fibers than the control group. The Ble-3IRL group had a greater number of immature fibers than the Ble group. At 30 days, there was an absence of inflammation and equal proportion of mature and immature collagen fibers. All bleached groups showed a reduction in the volume of the pulp chamber.
Conclusion
Three consecutive applications of RL and one IRL application can minimize damage to the pulp of bleached teeth, whereas three IRL applications can minimize pulp fibrosis. However, LLLT did not prevent deposition of tertiary dentin.
Clinical relevance
This study describes LLLT protocols capable of minimizing inflammation and maturation of collagen fibers in pulp tissue after dental bleaching. However, the protocols proved insufficient for reducing the formation of tertiary dentin in bleached teeth.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hydrogen peroxide (H2O2), a constituent of dental bleaching gel, can diffuse through enamel and dentin due to its dissociation into reactive oxygen species and other powerful oxidizing agents [1,2,3,4]. Dental bleaching is capable of causing intense damage to pulp tissue, especially in human mandibular incisor teeth [5, 6]. Pulp tissue damage is related to the number of bleaching sessions [7] and concentration of H2O2 present in the gel [8, 9].
Studies on teeth of dogs and rats have demonstrated that damages occurring in the pulp tissue are reversible [8, 10,11,12,13]. However, studies have also reported accelerated pulp tissue aging because of intense deposition of tertiary dentin [9], expression of mineralization markers [14], and differentiation of new odontoblast-like cells [15]. Maturation of collagen fibers of the pulp, which is another sign of tissue aging, has also been reported after dental bleaching [12].
Furthermore, clinically, intense postoperative dentin hypersensitivity has been reported after bleaching, which decreases over time [16, 17]. Thus, there is a constant search for bleaching agents that do not compromise pulpal tissue and are safe for clinical use. Attempts have been made to reduce tooth sensitivity by concomitant use of anti-inflammatory drugs; however, the results have been inconclusive [16, 18]. Similarly, antioxidants have also been tested, but complete neutralization of the deleterious effects of bleaching has not been achieved [19,20,21].
Low-level laser therapy (LLLT) is being used increasingly in medicine and dentistry owing to its analgesic, anti-inflammatory, and biostimulation effects [22,23,24]. LLLT has been shown to promote regeneration of damaged tissues [25]. Previous studies have reported the use of LLLT to reduce the damage to pulp tissue after bleaching; however, the results are controversial [25,26,27,28,29]. Few clinical studies demonstrated a reduction in post-bleaching sensitivity following LLLT [27, 29], and some LLLT parameters were not effective [28]. A systematic review of the literature revealed that three studies investigated the effects of LLLT on pulp tissue damaged by the bleaching gel with only one being an in vivo study [30, 31]. The authors of the study therefore opined that further studies should be conducted, especially in vivo to clearly analyze the influence of LLLT on the pulp of bleached teeth [31]. Reduction in pulpal sensitivity is important to ensure patient comfort; nonetheless, in vivo studies should evaluate the effect of laser on different parameters such as maturation of collagen fibers or tertiary dentin deposition, which may be altered in response to different aggressors/bleaching agents.
Rat molar teeth were considered for pulp tissue analysis after dental bleaching owing to ease of obtaining and standardization of the specimens and similarity in tissue responses with humans [5, 7]. Thus, the validation of results in humans occurs later according to the ethical principles. Moreover, pulp tissue is surrounded by rigid dentine walls and vital teeth have components such as dentinal fluid, odontoblastic extensions, and antioxidant enzymes that can hinder the penetration of H2O2 [11]. These features highlight the importance of in vivo analysis [11].
This study analyzed the effects of different LLLT application protocols on pulp tissue of rat teeth after dental bleaching using a red laser (RL) and an infrared laser (IRL). The influence of LLLT on pulp tissue inflammation, collagen fiber maturation, and tertiary dentin deposition in bleached teeth was evaluated. The null hypothesis was that LLLT does not influence (a) inflammation generated in the pulp tissue after bleaching, (b) maturation of collagen fibers in the dental pulp of bleached teeth, and (c) tertiary dentin deposition in these teeth.
Materials and methods
Animals
Eighty healthy male Wistar rats (Rattus norvegicus, albinus variation; weighing between 250 and 300 g) aged 2 months were used. The sample size was defined based on the data of maturation of collagen fibers at 30 days from a previous study [12]. Considering a power sample of 80% and an alpha error of 0.05 to identify a significant difference, a minimum of 10 rats/group was deemed necessary, which was in accordance with previous studies [4, 8, 9]. The animals were housed in a temperature-controlled environment (22 ± 1 °C, 70% relative humidity) with a 12-h light–dark cycle and received water and food (Mogiana Alimentos SA, Campinas, Brazil) ad libitum throughout the experiment period. The rats were maintained in collective polypropylene cages (four per cage). Cage bedding was changed at least three times a week. The animals were kept under observation throughout the experiment. The Ethics Committee of the local university approved the experimental protocol (CEUA 00938) and the entire study was conducted according to ARRIVE guidelines.
Dental bleaching and LLLT treatment
Four operators performed all procedures in an appropriate room in the animal research area. Two operators were responsible for anesthesia and random allocation of rats to groups (J.M.A.L. and J.G.B.). A single calibrated operator (F.B.) was responsible for bleaching and LLLT treatments and was assisted by a fourth operator (A.M.T.). The operator F.B. was calibrated to perform the bleaching procedure based on protocols from previous studies [4, 7,8,9, 11,12,13,14,15]. For LLLT, F.B. underwent training with G.S.A., who has extensive experience in treatment with laser. The rats were anesthetized with intramuscular injection of ketamine (87 mg/kg; Francotar, Virbac do Brasil Ind. e Com. Ltda, Roseira, SP, Brazil) and xylazine (13 mg/kg; Rompum, Bayer SA, São Paulo, SP, Brazil). After application and photoactivation of a resinous gingival barrier (Top Dam; FGM Dental Products, Joinville, SC, Brazil), the right and left maxillary molars were treated randomly, according to the groups described in Table 1. The rats were randomly allocated to groups using a lottery method and the side of upper molars to be treated in each rat was also decided in the same manner.
For the bleaching procedure, a single application of 0.01 mL 35% H2O2 bleaching gel (Whiteness HP Maxx; FGM Dental Products, Joinville, SC, Brazil) was performed on the designated side in each rat. The gel was left on the teeth for 30 min [11]. Subsequently, the tooth surface was wiped with cotton and absorbent paper and rinsed thoroughly with water. To standardize the applied volume of the bleaching gel, 1.0-mL syringes were used [9].
For RL and IRL treatments, a laser duo semiconductor (GaA1As and InGaAlP) was used (MM Optics Ltda, São Carlos, SP, Brazil) after bleaching, at a wavelength of 808 nm for IRL and 660 nm for RL. The device had a fixed power of 100 mW. The beam exit area at the tip of the laser pen was 3 mm2. The tip of the laser pen was kept in contact [24, 25] with the first molars during the procedure. The Ble (bleached)-1RL and Ble-1IRL groups received the LLLT session immediately, while the groups Ble-3RL and Ble-3IRL received an immediate LLLT session, a session at 24 h and another at 48 h after bleaching. The rats were re-anesthetized and resinous gingival barrier was re-applied before the LLLT session at 24 and 48 h. After each session, the rats were housed in the same conditions described earlier and were observed until the effect of the anesthesia wore off.
Histological analyses
Forty animals each were euthanized 2 days and 30 days after the treatment with an overdose of sodium thiopental (240 mg/kg; Thipentax, Cristália, Produtos Químicos Farmacêuticos Ltda, Itapira, Brazil). The hemimaxillae were separated, dissected, and fixed in a solution of 4% buffered formaldehyde (24 h). The specimens were decalcified in 10% ethylenediaminetetraacetic acid (EDTA) (3 months) and subsequently dehydrated, clarified, and embedded in paraffin. Serial histological sections of each specimen were obtained from the site of full longitudinal extension of the mesial root of the first molar. Subsequently, 5-μm sections were created in the vestibular-lingual plane. The blades with histological sections were then stained with hematoxylin-eosin (HE) and picrosirius red (PSR).
A calibrated operator of previous studies and pilot studies analyzed the sections in a blind manner using light microscopy (× 400, DM 4000 B, Leica, Wetzlar, Germany). For the HE analysis, the pulp chamber was divided into occlusal, middle, and cervical thirds [7] and the degree of inflammation was scored in each third as follows: 0, inflammatory cells absent or negligible; 1, mild inflammatory infiltrate (< 25 cells per field); 2, moderate inflammatory infiltrate (25–125 cells per field); 3, severe inflammatory infiltrate (> 125 cells per field); and 4, tissue necrosis [7, 8]. The operator evaluated two histological sections of each blade to identify any differences.
The maturation of collagen fibers was analyzed by PSR staining under a polarized light microscope. Images of the cervical region of the coronal pulp were captured and the LAS program (× 400, Leica LAS 4.12, Leica Microsystems) was used, which allowed selection of corresponding colors for each type of collagen fiber. Greenish-yellow fibers were considered immature and thin, while reddish-yellow fibers were considered mature and thick [12, 32]. After color selection, the program automatically calculated the marked area of each type of collagen fiber. This analysis was also performed by a calibrated operator who evaluated two consecutive histological sections of the same blade and obtained the average value for each type of fiber.
Microtomographic analyses
Microtomographic analysis was performed by a calibrated operator only on the specimens obtained at 30 days, when formation of tertiary dentin could be observed [12, 15]. After fixation in neutral formalin and before the decalcification procedure in EDTA for histological analysis, these specimens were reduced and placed in distilled water for at least 12 h for scanning [33]. The samples were scanned by the SkyScan 1272 imaging system (Kontich, Belgium). This system allowed scanning at 70 kV and 142 μA with the use of an X-ray detector (11 Mp [2016 × 1344 pixels], 16-bit, with a nominal resolution of 0.45 to 27 mm, scanning space of 75 mm diameter and 70 mm length, and aluminum filter of 0.5 mm). The entire system was connected to a Dell Precision T7610 (Dell Inc. São Paulo, SP, Brazil) computer with Intel (R) XEON (R) CPU E5-2687w v2 3.40 GHz processor.
The volume of interest included space of the pulp chamber. Volumetric data were used to generate three-dimensional reconstructed images [34, 35]. The image volume was oriented such that the long axis of the distal root of the first molar was parallel to the coronal and sagittal planes, in order to quantify the decrease in the pulp chamber volume because of tertiary dentin formation. The volume corresponding to the space filled by pulp tissue was measured in cubic millimeters [36, 37].
Statistical analyses
The data for analysis of inflammatory infiltration (non-parametric data) were subjected to Kruskal–Wallis and Dunn’s tests. The data obtained from PSR staining and microtomographic analyses were subjected to a normality test. The PSR analysis data at 2 days showed normality and were subjected to one-way analysis of variance followed by the Tukey test. The 30-day PSR and microtomographic data distribution were not normal; therefore, the data were subjected to the Kruskal–Wallis test, followed by Dunn’s test. Statistical significance was set at P < 0.05 for all analyses.
Results
Analysis of the inflammatory infiltrate
There was no loss of animals during the course of the experiment. Rats were healthy when the hemimaxillae were removed for further processing and histological analysis. The results of HE analysis are presented in Table 2, and the representative images can be seen in Figs. 1 and 2. At 2 days, the greatest damage was observed in the occlusal third of the coronal pulp in the Ble and Ble-1RL groups, with necrosis observed in a majority of the specimens (P > 0.05). However, despite lesser area of necrosis and the majority of specimens showing moderate to severe inflammation, the results of the Ble-3IRL group were statistically similar to those of the Ble and Ble-1RL groups (P > 0.05) and significantly different from those of the control group (P < 0.05). The Ble-1IRL and Ble-3RL groups showed results similar to the control group (P > 0.05). The Ble and Ble-1RL groups showed the highest amount of tissue alterations with severe inflammation in the middle third (P > 0.05) and moderate inflammation in the cervical third of the pulp (P > 0.05). These findings were significantly different from those of the control group (P < 0.05). Pulp tissue in the other bleached groups showed reduced inflammation, with moderate severity in the middle third (P > 0.05) and mild in the cervical third regions (P > 0.05). The results were statistically similar to those of the control group (P > 0.05).
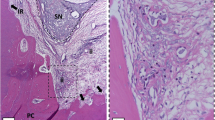
Representative images of the analysis of the inflammatory infiltrate of the groups at 2 days. (A–F) Panoramic microscopic aspect of dental pulp. Cont: (A1, A2) Pulp tissue with no alteration, organized odontoblastic layer, and absence of inflammation. Ble: (B1, B2) Pulp tissue with evident necrosis area and severe inflammatory infiltrate. Ble-1IRL: (C1, C2) Presence of moderate inflammatory infiltrate in pulp tissue and partially disorganized odontoblastic layer. Ble-3IRL: (D1, D2) Presence of moderate inflammatory infiltrate and higher disorganization of pulp tissue. Ble-1RL: (E1, E2) Intense disorganization of pulp tissue and severe inflammatory infiltrate. Ble-3RL: (F1, F2) Disorganized pulp tissue and presence of moderate inflammatory infiltrate (× 50, × 100, × 400; H.E)
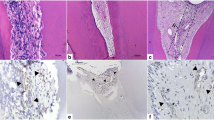
Representative images of the pulp tissue at 30 days. (A, A1) Control group with absence of tertiary dentin and normality of dental pulp, and (B, B1) Ble, (C, C1) Ble-1IRL, (D, D1) Ble-3IRL, (E, E1) Ble-1RL, and (F) Ble-3RL groups with no inflammatory infiltrate, presence of organized pulp tissue and formation of tertiary dentin (× 50, ×100; H.E)
At 30 days, no inflammatory infiltrate was observed. The pulp tissue showed cellular organization with an intact and continuous odontoblastic layer surrounding the entire coronal pulp and the other layers also appeared normal. Pulp tissue in the bleached groups showed intense tertiary dentin formation.
No alterations were observed in the groups that received only LLLT (RL or IRL) at 2 days or 30 days (Fig. 4), and the results were not significant when compared with those of the control group (Table 4).
Analysis of maturation of collagen fibers
The results of PSR analysis are presented in Table 3, and representative images can be seen in Fig. 3. At 2 days, the control group demonstrated higher amount of immature collagen fibers. Pulp tissue in the bleached groups showed higher amount of mature fibers in this period than that in the control group (P < 0.05). However, the group that received three applications of IRL after bleaching (Ble-3IRL) showed more immature fibers than the Ble group and the bleached groups that received RL (P < 0.05). At 30 days, all groups showed similar findings, with equivalent proportions of mature and immature collagen fibers (P > 0.05). Comparison between mature and immature collagen fibers in the same group during each analysis revealed significant differences in the control, Ble-1IRL, and Ble-3IRL groups at 2 days, with the amount of immature fibers being significantly higher (P < 0.05).
Representative images of the maturation of the collagen fiber. (A, A1) Example images of the Ble group at (A) 2 and (A1) 30 days identifying the captured region for PSR analysis. At 2 days, (B) Cont and (E) Ble-3IRL groups with higher amount of immature fibers, and (C) Ble, (D) Ble-1IRL, (F) Ble-1RL, and (G) Ble-3RL groups with immature and mature collagen fibers. At 30 days, (B1) Cont, (C1) Ble, (D1) Ble-1IRL, (E1) Ble-3IRL, (F1) Ble-1RL, and (G1) Ble-3RL groups, with a balance of mature and immature fibers. The white arrows indicate the areas of tertiary dentin that were excluded from the analysis (× 100, H.E.; × 400, PSR)
No significant alterations in the maturation of collagen fibers were observed in the groups that received only LLLT (RL or IRL) at 2 days or 30 days, compared with that in the control group (Fig. 4, Table 4).
Representative images of groups 3IRL and 3RL. Pulp tissue with normality aspect of groups (A–A3) 3IRL and (B–B3) 3RL at (A, B–A2, B2) 2 and at (A3, B3) 30 days. At 2 days, higher amount of immature fibers in groups (A4) 3IRL and (B4) 3RL, and mature and immature fibers present at 30 days in groups (A5) 3IRL and (B5) 3RL. The white arrows indicate the areas of tertiary dentin that were excluded from the analysis (× 100, × 400 H.E.; × 400 PSR)
Microtomographic analysis
The results of the microtomographic analysis are presented in Table 3. All bleached groups, regardless of the application of LLLT, showed a significant reduction in the volume of pulp chamber at 30 days (P < 0.05), indicating formation of tertiary dentin. The data of the 3IRL and 3RL groups are shown in Table 4.
Discussion
The present in vivo study evaluated the influence of LLLT, using IRL and RL, on the inflammatory process, maturation of collagen fibers, and change in volume of the pulp chamber in bleached teeth. It was observed that a single application of IRL or three applications of RL minimized the inflammatory process in the pulp tissue of bleached teeth, rejecting the null hypothesis (a) that LLLT does not influence the inflammation caused by H2O2 in the dental pulp. In addition, three IRL applications minimized the rate of maturation of collagen fibers in the early stage. Thus, the null hypothesis (b) that LLLT does not influence the maturation of collagen fibers in pulp tissue was also rejected. However, LLLT did not prevent the reduction in the volume of the pulp chamber caused by dental bleaching. Therefore, the null hypothesis (c) that LLLT does not influence tertiary dentin deposition in bleached teeth was accepted.
Studies have reported the deleterious effects of bleaching gels with high concentrations of H2O2 on pulp tissue and cells [4, 8, 9, 38, 39]. Previous studies have reported areas of necrosis in the coronal pulp of rat molar teeth and severe inflammatory infiltrate after one bleaching session with 35% H2O2 for 30 min [7,8,9, 11, 12, 14]. These results are consistent with those observed in the present study. Studies performed on human mandibular incisors have also revealed a similar pulp response, justified by the reduction in thickness of hard tissue in these teeth [5, 6]. Moreover, clinical studies have reported an absolute risk of tooth sensitivity in patients following bleaching, which is barely controlled by anti-inflammatory agents [16,17,18].
Many studies evaluating the effects of LLLT on tissues have shown excellent outcomes such as increased protein synthesis and DNA duplication [40], increased concentration of intracellular calcium [41], and greater control of oxidative stress by enzymatic action [23, 42]. LLLT has also been shown to be effective in inducing cell proliferation by modulating growth factors [40, 42,43,44]. These results indicate that LLLT could minimize the damage to the pulp tissue of bleached teeth and reduce postoperative tooth sensitivity.
A systematic review presented a controversial opinion on the influence of light, including LLLT, on pulp tissue and cellular response [31]. However, two recent studies suggested protocols for clinical use of LLLT. Alencar et al. [27] and De Paula et al. [29] observed that the IRL—with a wavelength of 808 nm—operated at an energy of 1.7 J for 16 s in two consecutive applications (cervical and medial region of teeth) was beneficial in reducing post-bleaching tooth sensitivity. This study used IRL at the same wavelength but at an energy level permitted by the laser device used. Despite the positive clinical results observed in the above studies, there was no comparison between the number of laser applications and the effectiveness of RL.
The laser beam is an electromagnetic wave that differs from ordinary light in its monochromaticity, coherence, and collimation [24]. Laser emits radiation within a small spectral wavelength range in the same direction; therefore, the photons do not diverge significantly [45]. The wavelength of the laser determines its capacity to penetrate tissues, and its consequent performance [45]. It is known that IRL has higher ability to penetrate into tissues than RL [45]. As the laser beam must penetrate through enamel and dentin to act on pulp tissue, better results can be expected with IRL. The presence of enamel and dentin justifies the importance of in vivo studies.
Thus, through novel protocols, we proposed to evaluate the influence of LLLT using one or three applications each of IRL and RL on pulp tissue of bleached teeth. Multiple applications were considered for comparison, as a single session would have limited influence on pulpal response [31]. However, we restricted our experiment to three sessions of LLLT over 2 days for analysis due to intense pulpal inflammation and necrosis in this period [7,8,9].
We observed a reduction in the inflammatory process after dental bleaching following three sessions of RL (each session at 1.5 J for 15 s) similar to that seen after a single session of IRL at energy of 3 J for 30 s. A study performed by Dantas et al. [25] showed that LLLT was able to increase cellular metabolism when IRL or RL was applied at energy of 10 J for 10 s. In contrast, Lima et al. [26] reported that the application of laser near the infrared wavelength (780 nm) at 4, 10, and 15 J for 300, 800, and 1200 s, respectively, did not demonstrate any beneficial effects. Moreover, 15 J of energy resulted in more significant damage [26]. These results corroborate the present study because low intensity of LLLT was found to be most beneficial for the pulp tissue. The beneficial response was associated with lower penetration of the laser and a greater number of sessions, or higher penetration and fewer sessions. In addition, our results corroborate recent clinical studies that reported reduced sensitivity after dental bleaching following one session of IRL therapy [27, 29].
Yamakawa et al. [46] reported that LLLT modulates cell proliferation, apoptosis, and gene expression in dental pulp cells. These effects may minimize the inflammatory tissue response, which is consistent with the present study. However, to the best of our knowledge, the effects of LLLT on collagen fibers of pulp tissue of bleached teeth have not yet been studied. The connective tissue of dental pulp is composed of approximately 34% collagen fibers [47]. Therefore, the analysis of these fibers after bleaching is justified, considering that reactive oxygen species from the bleaching gel can cause significant damage to pulp tissue [3, 12, 48].
In the present study, the control group showed higher amount of immature fibers at 2 days, while the bleached groups had a significant amount of mature fibers during this period. This observation may indicate accelerated maturation of pulp tissue after dental bleaching. Similar findings were observed in a previous study, even in diabetic rats [12]. Another study revealed increased expression of proteins associated with mineralization, such as osteocalcin and osteopontin, in the pulp tissue of bleached teeth [14]. According to Dong and Ma [49], the expression of these proteins is related to the process of tissue fibrosis. Findings reported in these studies corroborate with the present study.
However, LLLT was unable to prevent the advancement of tissue fibrosis after bleaching. A previous study revealed that LLLT resulted in increased synthesis of fibronectin in pulp cells and maturation of the extracellular matrix [50], which may explain the reduction in inflammation of the pulp tissue, but it does not prevent long-term fibrosis. In contrast, 2 days after treatment, the Ble-3IRL group showed a significantly lower amount of mature collagen fibers than the Ble and RL groups. These findings may indicate that IRL has the potential to reduce tissue fibrosis caused by bleaching, while such an effect is not observed with RL. At 30 days, as organization of the pulp tissue was complete, no significant difference in fibrosis was observed among the groups. It may be speculated that the damage caused by the bleaching gel was very intense and thus irreversible by three applications of IRL in later stages. Therefore, in addition to LLLT, a reduction in H2O2 concentration in the bleaching gel may be required to achieve optimum results in relation to pulp tissue. Further studies must be conducted to assess such changes. Fibrosis is related to tissue aging; therefore, therapies that reduce the same should be chosen [12].
In the present study, an equilibrium between the amount of mature and immature collagen fibers was observed in all the groups, including the control group, at 30 days. Thus, it may be suggested that tissue fibrosis in rat teeth occurs throughout this period, as reported previously [12]. This finding can be explained by the accelerated metabolism in rats [11]. Thus, the present study underlines the importance of IRL application to minimize accelerated aging in bleached teeth. Other analysis periods prior to 30 days could have provided more information regarding the maturation of collagen fibers.
This study analyzed the data at only 2 and 30 days after bleaching. The second day following treatment was chosen for analysis as previous studies have evaluated changes during this period and have reported significant inflammation and necrosis in the bleached groups [7,8,9]. Other studies reported ongoing repair at 7 and 15 days after bleaching and complete organization of pulp tissue by 30 days [14, 15]. Nevertheless, despite apparent repair of the pulp tissue, significant changes in collagen fibers were observed at 30 days [12]. Moreover, tertiary dentin formation was more significant at 30 days; thus, the effects of LLLT on reducing dentin formation could be studied during this period.
Previous studies have reported significant deposition of tertiary dentin after dental bleaching. The amount of deposition was directly proportional to inflammation generated by the bleaching gel [9, 11, 12]. Therefore, the volume of the pulp chamber was measured using computed microtomography in this study, which allowed three-dimensional reconstruction of the area to be analyzed. This analysis can be considered ideal for evaluating reduction in the volume of the pulp chamber, unlike histological analysis, which may be inconclusive because of variations in each section owing to pulp chamber irregularities [8].
Microtomographic analyses were performed 30 days after the bleaching, when it was possible to observe deposition of tertiary dentin [11, 14]. The pulp chamber volume of the first molar was measured. Significant reduction in the volume of the pulp chamber was observed in all bleached groups, regardless of application of LLLT. Studies have indicated that LLLT stimulates protein synthesis of the mineralization process [51, 52]. Yamakawa et al. [46] revealed that LLLT induces mineralization in dental pulp cells and tissues. Another study reported that LLLT helps in osteogenic differentiation of stem cells in pulps of deciduous teeth [53]. These findings might explain the negative effect of LLLT on the reduction of tertiary dentin deposition, while minimizing inflammation.
A limitation of this study is the use of rats. Each species of animals shows variations in repair capacity of pulp tissue and detailed biocompatibility studies are needed to elucidate this aspect [54, 55]. However, the biological response and healing process of rat pulp tissue are comparable with those of other mammals [56]. In addition, histological and biological characteristics of rat molar teeth are similar to those of humans [56, 57]. The present study as well as previous literature on molar teeth in rats demonstrated changes in pulp tissue following application of H2O2 [11, 12]. The results of the present study should not be directly extrapolated to human teeth because rat molars also have small areas of exposed dentin on the occlusal surface [58]. This exposed dentin can intensify the penetration of H2O2, as well as the action of LLLT. Thus, future studies should evaluate these protocols on human teeth.
This study showed that a single application of IRL has the potential to reduce inflammation. However, three applications were needed to minimize fibrosis of pulp tissue caused by bleaching. Nevertheless, specific studies are required to evaluate inflammation and other cellular mechanisms in the dental pulp at a molecular level after LLLT in bleached teeth. Moreover, other therapeutic protocols may be needed to prevent the formation of tertiary dentin.
Clinically, the need for three IRL therapy sessions may hinder patient involvement. However, patient participation may be ensured if the treatment reduces post-bleaching hypersensitivity. To the best of our knowledge, this is the first study to histologically evaluate the effect of these LLLT protocols on pulp tissue of bleached teeth. Further studies should be performed to better adapt this protocol to clinical practice. IRL therapy combined with a bleaching gel with lower concentration of H2O2 can also provide cost-effective results and maybe need few sessions of LLLT.
Conclusion
LLLT has the potential to minimize the damage caused by the bleaching agent, H2O2. Consecutive applications of RL after bleaching produce similar effects as that of one application of IRL. Three applications of IRL have the potential to minimize accelerated fibrosis of pulp tissue caused by dental bleaching. However, the LLLT protocols used in the present study were unable to prevent marked deposition of tertiary dentin.
References
Korytowski W, Sarna T (1990) Bleaching of melanin pigments. Role of copper ions and hydrogen peroxide in autooxidation and photoxidation of synthetic dopa-melanin. J Biol Chem 265:12410–12416
Marson FC, Gonçalves RS, Silva CO, Cintra LT, Pascotto RC, Santos PH, Briso ALF (2015) Penetration of hydrogen peroxide and degradation rate of different bleaching products. Oper Dent 40:72–79
Cintra LTA, Benetti F, Ferreira LL, Gomes-Filho JE, Ervolino E, Gallinari MO, Rahal V, Briso ALF (2016) Penetration capacity, color alteration and biological response of two in-office bleaching protocols. Braz Dent J 27:169–175
Barbosa JG, Benetti F, de Oliveira Gallinari M, Carminatti M, da Silva ABD, Lopes INI, Briso ALF, Cintra LTA (2020) Bleaching gel mixed with MI Paste Plus reduces penetration of H2O2 and damage to pulp tissue and maintains bleaching effectiveness. Clin Oral Investig 24:1299–1309
Costa CAS, Riehl H, Kina JF, Sacono NT, Hebling J (2010) Human pulp responses to in-office tooth bleaching. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 109:e59–e64
Roderjan DA, Stanislawczuk R, Hebling J, Costa CA, Reis A, Loguercio AD (2015) Response of human pulps to different in-office bleaching techniques: preliminary findings. Braz Dent J 26:242–248
Cintra LTA, Benetti F, Facundo ACS, Ferreira LL, Gomes-Filho JE, Ervolino E, Rahal V, Briso ALF (2013) The number of bleaching sessions influences pulp tissue damage in rat teeth. J Endod 39:1576–1580
Benetti F, Gomes-Filho JE, Ferreira LL, Ervolino E, Briso ALF, Sivieri-Araújo G, Cintra LTA (2017) Hydrogen peroxide induces cell proliferation and apoptosis in pulp of rats after dental bleaching in vivo. Arch Oral Biol 81:103–109
Benetti F, Gomes-Filho JE, Ferreira LL, Sivieri-Araújo G, Ervolino E, Briso ALF, Cintra LTA (2018) Concentration-dependent effect of bleaching agents on the immunolabelling of interleukin-6, interleukin-17 and CD5-positive cells in the dental pulp. Int Endod J 51:789–799
Seale NS, McIntosh JE, Taylor AN (1981) Pulpal reaction to bleaching of teeth in dogs. J Dent Res 80:948–953
Cintra LTA, Benetti F, Ferreira LL, Rahal V, Ervolino E, Jacinto RC, Gomes Filho JE, Briso ALF (2016) Evaluation of an experimental rat model for comparative studies of bleaching agents. J Appl Oral Sci 24:95–104
Cintra LTA, Ferreira LL, Benetti F, Gastélum AA, Gomes-Filho JE, Ervolino E, Briso ALF (2017) The effect of dental bleaching on pulpal tissue response in a diabetic animal model. Int Endod J 50:790–798
Ferreira LL, Gomes-Filho JE, Benetti F, Carminatti M, Ervolino E, Briso ALF, Cintra LTA (2018) The effect of dental bleaching on pulpal tissue response in a diabetic animal model: a study of immunoregulatory cytokines. Int Endod J 51:347–356
Benetti F, Briso ALF, Carminatti M, de Araújo Lopes JM, Barbosa JG, Ervolino E, Gomes-Filho JE, Cintra LTA (2019) The presence of osteocalcin, osteopontin and reactive oxygen species-positive cells in pulp tissue after dental bleaching. Int Endod J 52:665–675
Benetti F, Briso ALF, de Araújo Lopes JM, Carminatti M, Conti LC, Gallinari MO, Ervolino E, Cintra LTA (2019) In vivo analysis of the presence of heme oxygenase-1, transcription factor Jun-D and CD90+/CD73+/CD105+/CD45- cells in the pulp of bleached teeth. Int Endod J 52:1723–1737
Faria-E-Silva AL, Nahsan FP, Fernandes MT, Martins-Filho PR (2015) Effect of preventive use of nonsteroidal anti-inflammatory drugs on sensitivity after dental bleaching: a systematic review and meta-analysis. J Am Dent Assoc 146:87–93
Rezende M, Loguercio AD, Kossatz S, Reis A (2016) Predictive factors on the efficacy and risk/intensity of tooth sensitivity of dental bleaching: a multi regression and logistic analysis. J Dent 45:1–6
Charakorn P, Cabanilla LL, Wagner WC, Foong WC, Shaheen J, Pregitzer R, Schneider D (2009) The effect of preoperative ibuprofen on tooth sensitivity caused by in-office bleaching. Oper Dent 34:131–135
Sasaki RT, Flório FM, Basting RT (2009) Effect of 10% sodium ascorbate and 10% alpha-tocopherol in different formulations on the shear bond strength of enamel and dentin submitted to a home-use bleaching treatment. Oper Dent 34:746–752
May LG, Salvia AC, Souza RO, Michida SM, Valera MC, Takahashi FE, Bottino MA (2010) Effect of sodium ascorbate and the time lapse before cementation after internal bleaching on bond strength between dentin and ceramic. J Prosthodont 19:374–380
Vargas FDAS, Soares DG, Basso FG, Hebling J (2014) Dose-response and time-course of α-tocoferol mediating the cytoprotection of dental pulp cells against hydrogen peroxide. Braz Dent J 25:367–371
Reddy GK (2004) Photobiological basis and clinical role of low-intensity lasers in biology and medicine. J Clin Laser Med Surg 22:141–150
Silveira PCL, Streck EL, Pinho RA (2007) Evaluation of mitochondrial respiratory chain activity in wound healing by low-level laser therapy. J Photochem Photobiol B 86:279–282
Moosavi H, Arjmand N, Ahrari F, Zakeri M, Maleknejad F (2016) Effect of low-level laser therapy on tooth sensitivity induced by in-office bleaching. Lasers Med Sci 31:713–719
Dantas CM, Vivan CL, Ferreira LS, Freitas PM, Marques MM (2010) In vitro effect of low intensity laser on the cytotoxicity produced by substances released by bleaching gel. Braz Oral Res 24:460–466
Lima AF, Ribeiro AP, Basso FG, Bagnato VS, Hebling J, Marchi GM, de Souza Costa CA (2014) Effect of low-level laser therapy on odontoblast-like cells exposed to bleaching agent. Lasers Med Sci 29:1533–1538
Alencar CM, De Paula BLF, Araújo JLN, Alves EB, De Albuquerque Jassé FF, Silva CM (2018) Effect of low-level laser therapy combined with 5000 parts per million fluoride dentifrice on post bleaching sensitivity: a clinical, randomized, and double-blind study. J Esthet Restor Dent 30:352–359
Calheiros APC, Moreira MS, Gonçalves F, Aranha ACC, Cunha SR, Steiner-Oliveira C, Eduardo CP, Ramalho KM (2017) Photobiomodulation in the prevention of tooth sensitivity caused by in-office dental bleaching. A randomized placebo preliminary study. Photomed Laser Surg 35:415–420
de Paula B, Alencar C, Ortiz M, Couto R, Araújo J, Silva C (2019) Effect of photobiomodulation with low-level laser therapy combined with potassium nitrate on controlling post-bleaching tooth sensitivity: clinical, randomized, controlled, double-blind, and split-mouth study. Clin Oral Investig 23:2723–2732
Caviedes-Bucheli J, Ariza-García G, Restrepo-Méndez S, Ríos-Osorio N, Lombana N, Muñoz HR (2008) The effect of tooth bleaching on substance P expression in human dental pulp. J Endod 34:1462–1465
Benetti F, Lemos CAA, Oliveira Gallinari M, Terayama AM, Briso ALF, Castilho Jacinto R, Sivieri-Araújo G, Cintra LTA (2018) Influence of different types of light on the response of the pulp tissue in dental bleaching: a systematic review. Clin Oral Investig 22:1825–1837
Junqueira LC, Montes GS, Sanchez EM (1982) The influence of tissue section thickness on the study of collagen by the Picrosirius-polarization method. Histochemistry 74:153–156
Martins CM, Sasaki H, Hirai K, Andrada AC, Gomes-Filho JE (2016) Relationship between hypertension and periapical lesion: an in vitro and in vivo study. Braz Oral Res 30:e78
Aghaloo TL, Kang B, Sung EC, Shoff M, Ronconi M, Gotcher JE, Bezouglaia O, Dry SM, Tetradis S (2011) Periodontal disease and bisphosphonates induce osteonecrosis of the jaws in the rat. J Bone Miner Res 26:1871–1882
Kang SY, Deshpande SS, Donneys A, Rodriguez JJ, Nelson NS, Felice PA, Chepeha DB, Buchman SR (2013) Parathyroid hormone reverses radiation induced hypovascularity in a murine model of distraction osteogenesis. Bone 56:9–15
Sun Z, Wang L, Peng B (2014) Kinetics of glycogen synthase kinase (GSK)3β and phosphorylated GSK3β (Ser 9) expression in experimentally induced periapical lesions. Int Endod J 47:1107–1116
Kalatzis-Sousa NG, Spin-Neto R, Wenzel A, Tanomaru-Filho M, Faria G (2017) Use of micro-computed tomography for the assessment of periapical lesions in small rodents: a systematic review. Int Endod J 50:352–366
Soares DG, Basso FG, Scheffel DS, Hebling J, de Souza Costa CA (2015) Responses of human dental pulp cells after application of a low-concentration bleaching gel to enamel. Arch Oral Biol 60:1428–1436
Louzada LM, Briso ALF, Benetti F, Vieira LB, Castilho Jacinto R, Dezan-Júnior E, Cintra LTA (2019) Anti-inflammatory potential of a carvedilol gel in the pulpal tissue of rats after dental bleaching: a histopathological evaluation. J Investig Clin Dent 10:e12401
Yu HS, Chang KL, Yu CL, Chen JW, Chen GS (1996) Low-energy helium-neon lasers irradiation stimulates interleukin-1 alpha and interleukin-8 release from cultured human keratinocytes. J Invest Dermatol 107:593–596
Lubart R, Friedmann H, Sinyakov M, Cohen N, Breibart H (1997) Changes in calcium transport in mammalian sperm motichondria and plasma membranes caused by 780 nm irradiation. Lasers Surg Med 21:493–499
Jori G, Schindl L, Schindl A, Polo N (1996) Novel approaches towards a detailed control of the mechanism and efficiency of photosensitized process in vivo. J Photochem Photobiol A 102:101–107
Moore P, Ridgway TD, Higbee RG, Howard EW, Lucroy MD (2005) Effect of wavelength on low-intensity laser irradiation- stimulated cell proliferation in vitro. Lasers Surg Med 36:8–12
Hawkins V, Abrahamse H (2006) Effect of multiple exposures of low-level laser therapy on the cellular responses of wounded human skin fibroblasts. Photomed Laser Surg 24:705–714
Buchalla W, Attin T (2007) External bleaching therapy with activation by heat, light or laser-a systematic review. Dent Mater 23:586–596
Yamakawa S, Niwa T, Karakida T, Kobayashi K, Yamamoto R, Chiba R, Yamakoshi Y, Hosoya N (2018) Effects of Er:YAG and diode laser irradiation on dental pulp cells and tissues. Int J Mol Sci 19:E2429
Abrahão IJ, Martins MD, Katayama E, Antoniazzi JH, Segmentilli A, Marques MM (2006) Collagen analysis in human tooth germ papillae. Braz Dent J 17:208–212
Benetti F, Briso ALF, Ferreira LL, Carminatti M, Álamo L, Ervolino E, Dezan-Júnior E, Cintra LTA (2018) In vivo study of the action of a topical anti-inflammatory drug in rat teeth submitted to dental bleaching. Braz Dent J 29:555–561
Dong J, Ma Q (2017) Osteopontin enhances multi-walled carbon nanotube-triggered lung fibrosis by promoting TGF-β1 activation and myofibroblast differentiation. Part Fibre Toxicol 14:18
Garrido PR, Pedroni ACF, Cury DP, Moreira MS, Rosin F, Sarra G, Marques MM (2019) Effects of photobiomodulation therapy on the extracellular matrix of human dental pulp cell sheets. J Photochem Photobiol B 94:149–157
Pyo SJ, Song WW, Kim IR, Park BS, Kim CH, Shin SH, Chung IK, Kim YD (2013) Low-level laser therapy induces the expressions of BMP-2, osteocalcin, and TGF-β1 in hypoxic-cultured human osteoblasts. Lasers Med Sci 28:543–550
de Oliveira LSS, de Araújo AA, de Araújo Júnior RF, Barboza CAG, Borges BCD, da Silva JSP (2017) Low-level laser therapy (780 nm) combined with collagen sponge scaffold promotes repair of rat cranial critical-size defects and increases TGF-β, FGF-2, OPG/RANK and osteocalcin expression. Int J Exp Pathol 98:75–85
Pinheiro CCG, de Pinho MC, Aranha AC, Fregnani E, Bueno DF (2018) Low power laser therapy: a strategy to promote the osteogenic differentiation of deciduous dental pulp stem cells from cleft lip and palate patients. Tissue Eng Part A 24:569–575
Hebling J, Giro EMA, Costa CAS (1999) Biocompatibility of an adhesive system applied to exposed human dental pulp. J Endod 25:676–682
Costa CA, Oliveira MF, Giro EM, Hebling J (2003) Biocompatibility of resin-based materials used as pulp-capping agents. In Endod J 36:831–839
Dammaschke T (2010) Rat molar teeth as a study model for direct pulp capping research in dentistry. Lab Anim 44:1–6
Sasaki T, Kawamata-Kido H (1995) Providing an environment for reparative dentine induction in amputated rat molar pulp by high molecular-weight hyaluronic acid. Arch Oral Biol 40:209–219
Hunt HR, Rosen S, Hoppert CA (1970) Morphology of molar teeth and occlusion in young rats. J Dent Res 49:508–514
Funding
This study was supported by a grant (2016/20271-7) from the Fundação de Amparo à Pesquisa do Estado de São Paulo, São Paulo, SP, Brazil and by a grant (311650/2018-0) from the Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq, São Paulo, SP, Brazil.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors. The in vivo (CEUA 00938) study using rat molars was approved by the Ethics Committee of the institution in which the study was performed and conducted in accordance with its ethical standards.
Informed consent
For this type of study, informed consent is not required.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Terayama, A.M., Benetti, F., de Araújo Lopes, J.M. et al. Influence of low-level laser therapy on inflammation, collagen fiber maturation, and tertiary dentin deposition in the pulp of bleached teeth. Clin Oral Invest 24, 3911–3921 (2020). https://doi.org/10.1007/s00784-020-03258-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-020-03258-9