Abstract
Objectives
To define an expert Delphi consensus on when to intervene in the caries process and on existing carious lesions using non- or micro-invasive, invasive/restorative or mixed interventions.
Methods
Non-systematic literature synthesis, expert Delphi consensus process and expert panel conference.
Results
Carious lesion activity, cavitation and cleansability determine intervention thresholds. Inactive lesions do not require treatment (in some cases, restorations will be placed for reasons of form, function and aesthetics); active lesions do. Non-cavitated carious lesions should be managed non- or micro-invasively, as should most cavitated carious lesions which are cleansable. Cavitated lesions which are not cleansable usually require invasive/restorative management, to restore form, function and aesthetics. In specific circumstances, mixed interventions may be applicable. On occlusal surfaces, cavitated lesions confined to enamel and non-cavitated lesions radiographically extending deep into dentine (middle or inner dentine third, D2/3) may be exceptions to that rule. On proximal surfaces, cavitation is hard to assess visually or by using tactile methods. Hence, radiographic lesion depth is used to determine the likelihood of cavitation. Most lesions radiographically extending into the middle or inner third of the dentine (D2/3) can be assumed to be cavitated, while those restricted to the enamel (E1/2) are not cavitated. For lesions radiographically extending into the outer third of the dentine (D1), cavitation is unlikely, and these lesions should be managed as if they were non-cavitated unless otherwise indicated. Individual decisions should consider factors modifying these thresholds.
Conclusions
Comprehensive diagnostics are the basis for systematic decision-making on when to intervene in the caries process and on existing carious lesions.
Clinical relevance
Carious lesion activity, cavitation and cleansability determine intervention thresholds. Invasive treatments should be applied restrictively and with these factors in mind.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Building an expert Delphi consensus on intervention thresholds
There is an ever-increasing number of strategies available to manage the caries process and its outcome, the carious lesion (ranging from very early signs and symptoms to extended cavitated lesions). These strategies are applied to avoid pain, prevent loss of tooth tissues or entire teeth and retain functionality and aesthetics. Daily, oral health practitioners are faced with the decision of ‘if and when’ to intervene using one of the many approaches available. This decision should be based on the available evidence; the characteristics, needs and wishes of the specific patient; the features of the tooth or carious lesion to be managed (as will be discussed below) and the operator’s experience [1].
Expert consensus can also support decision-making, especially if the existing evidence is limited or of too narrow scope. There have been recent attempts in dentistry, and specifically cariology, to assist clinical decision-making by expert consensus, for example on minimal intervention dentistry (MID) and carious tissue removal [2,3,4,5]. The present paper describes the consensus reached by an expert consensus panel who met in July 2018 in London, UK, and used a structured online Delphi process before and after the meeting to systematically collate experts’ opinion and come to an agreement. The consensus focused specifically on when to intervene in the caries process and on existing carious lesions, rather than caries prevention. A detailed description of the methods can be found in the Appendix; also including the guidance on Conducting and REporting DElphi Studies (CREDES) [6].
Firstly, the contemporary understanding of the caries process and the characteristics of carious lesions will be described. Secondly, the different levels of interventions will be presented and brief examples given. Thirdly, factors determining the decision of when to intervene will be outlined. Finally, consensus recommendations, which emerged from a 2-round Delphi process and a consensus conference involving a panel of more than 20 international experts, will be presented. The panel comprised members of the European Organization for Caries Research (ORCA) and delegates of the European Federation of Conservative Dentistry (EFCD) and international experts from around the world. Note that this document and the resulting consensus recommendations do not aim to update or replace existing (often broader) guidance. The consensus on MID, for example, discussed caries detection and risk assessment, remineralisation and other preventive measures, minimally invasive operative interventions and re-treatments [4]. Instead, this document specifically aims to assist decision-making on when to intervene in the caries process and on existing carious lesions.
Dental caries and carious lesions: contemporary understanding
Dental caries is the most prevalent and ubiquitous non-communicable disease affecting humankind today [7]. It was first understood to be an infectious disease, requiring removal of all plaque (biofilm) from the teeth or from affected carious hard tissues (specific plaque hypothesis). This concept, while debated (i.e. biofilm being cariogenic under certain conditions; non-specific plaque hypothesis), was later modified, suggesting that the mere presence of biofilm is not sufficient for the pathogenesis of caries, but that an overlapping interaction between the host/teeth, substrate and microbiota is needed. Despite being ‘infected’ (or rather, contaminated) with cariogenic bacteria, a cavitated carious lesion will not develop without a cariogenic diet [8, 9]. Marsh (1994) introduced the ecological plaque theory. The microbial composition of the biofilm is stable unless ‘environmental perturbations’ occur which can affect microbial homeostasis, leading to dysbiosis [10, 11]. With respect to caries, diet (mainly free sugars), oral hygiene and salivary factors are the contributing drivers of dysbiosis, leading to a shift in the microbiota towards acidogenic and aciduric microorganisms. There are also wider influences on caries experience [12], but consideration of those is beyond our remit. The initial (naïve) dental biofilm is influenced by both hereditary and environmental factors, but, as time goes on, the types and proportions of micro-organisms acquired early in life are modified by environmental influences [13]. Currently, the extended ecological plaque hypothesis is accepted as an explanation of the pathogenesis of caries [14].
This pathogenesis of dental caries involves organic acids, the by-product of microbial metabolism of dietary free sugars. As the pH of the biofilm decreases, it reaches a point where the biofilm fluid at the surface of the tooth is under-saturated with respect to tooth mineral and dissolution occurs to maintain equilibrium [15]. Initially, the dissolution occurs at the surface of the tooth, but, if conditions persist, and the lesion becomes more extensive, mineral from deeper enamel (and subsequently dentine) will be lost. Caries has a genetic component, with influencing factors including enamel quality and quantity, immune response, dietary preferences and salivary characteristics [16, 17]. In summary, dental caries is a disease characterised by a process of demineralisation of the dental hard tissues, caused by frequent free sugar exposure to the dental biofilm, which shifts the ecological balance towards a cariogenic dysbiosis. For dentine and root caries, cleavage of collagen by bacterial or dentinal enzymes follows early mineral loss and contributes to the loss of the hard tissue [18, 19].
The former and contemporary management of the caries process and carious lesions
The former management of the caries process and carious lesions was influenced by an understanding that caries was a purely infectious disease and could be managed invasively/restoratively by removing all demineralised and ‘contaminated’ tissue. This was grounded in (1) a lack of understanding that the caries process and carious lesions are separate, but related; (2) the incorrect understanding that once a lesion had established and the tooth was ‘infected’, eradication of microbiota was needed; (3) the erroneous concept that lesion progression was inevitable and (4) the fact that the majority of carious lesions dentists encountered in the past were truly ‘decayed’, i.e. cavitated dentine lesions. Therefore, the professional education of dental surgeons concentrated on mechanistic surgical procedures instead of that of dental physicians who manage the disease, and remuneration systems incentivised such invasive/restorative therapies [20, 21]. Notably, the shift away from this approach was initiated decades ago, while consistent current data from all over the globe demonstrates that it has not been fully adopted [22].
Nowadays, and building on evidence accrued over several decades, it is clear that (1) the caries process can be controlled by modifying the patient’s caries risk/susceptibility, depending on his/her adherence to behavioural modifications and not only by intervening operatively on carious lesions, yet success/understanding of behavioural interventions on caries control has been limited [23]; (2) the caries process and carious lesions can be managed without removing microorganisms, but by rebalancing the dysbiosis within the tooth surface biofilm and arresting those within the depths of the tissues; (3) active (progressing) lesions can be inactivated [24]; (4) in many high-income countries, the spectrum of carious lesions has been and is shifting, especially in younger people, as there are now more non-cavitated lesions being detected [25, 26] and (5) while dental education in some countries now involves the contemporary understanding of caries and its subsequent carious lesions, remuneration systems for oral healthcare delivery have largely not been adjusted accordingly (there may be exceptions, and admittedly the uptake of a more contemporary approach towards caries and carious lesions has improved over the last three decades).
Hence, the conventional restorative/invasive approach towards managing the caries process and carious lesions is not grounded in the current understanding of the disease and it is also not appropriate for managing the broad spectrum of lesions found in many individuals (from very early to large cavitated). It should also be considered that subsequent interventions on restorations are often necessary [27,28,29]. This is classically known as the ‘restorative death spiral’ [30, 31]. Given these alternative arguments, there is consensus that invasive/restorative interventions alone are not beneficial for managing the caries process and lesions in all situations. Instead, invasive/restorative interventions represent a late stage in the management puzzle, repairing the gross tissue damage and restoring form, function, aesthetics and cleansability, thereby allowing the control of risk of future loss of function. Invasive strategies may also be used to approach acute caries lesions [26]. Invasive/restorative interventions are an important and relevant tool, but they should be complemented by other (non- or micro-invasive) management strategies. These ‘other’ strategies aim to control the caries process and the activity of carious lesions (as will be discussed below). If they are successfully implemented, the presence of bacteria is no longer a problem, since the cariogenic activity and the resulting mineral loss from dental hard tissues can be controlled concomitantly. In order to implement these alternative approaches successfully, there needs to be clear communication between the oral healthcare professional and the patient, with the latter appreciating their role in valuing and respecting their own oral health [20].
Intervention strategies: different levels of invasiveness
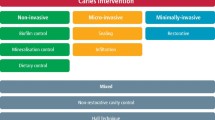
In the present document, we distinguish three levels of invasiveness to classify intervention strategies for ‘treating’ existing carious lesions. They are based on the degree of tissue removal associated with each strategy (Fig. 1).
-
1.
Non-invasive strategies do not remove dental hard tissue and involve, for example, fluorides and other chemical strategies for controlling mineral balance, biofilm control measures and dietary control.
-
2.
Micro-invasive strategies remove the dental hard tissue surface at the micrometre level, usually during an etching step, such as sealing or infiltration techniques.
-
3.
Invasive strategies remove gross dental hard tissue, such as through use of hand excavators, rotary instruments or other devices. In most cases, this process is associated with the placement of restorations.
All of these strategies can be performed in a minimally invasive manner and can be part of MID [4, 32]. Note that some interventions can be regarded as ‘mixed’, not clearly falling into one of these categories.
In the next sections, we will provide an overview of what falls into the three different levels of invasiveness. The individual details pertaining to each strategy will not be discussed, since the aim of this consensus paper was not to give recommendations on how to specifically intervene, but when.
Non-invasive interventions
To arrest existing lesions, several strategies are available; many of which have been developed originally for preventing the development of carious lesions and are now also applied for arresting lesions. These include dietary control, biofilm control and mineralisation control.
Dietary control
With sugars being the driver of biofilm dysbiosis and cariogenic activity, both prevention and lesion arrest should, theoretically, be possible if sugar intake (mainly free sugars) is restricted/regulated [33]. Sugar replacement may also be a valid option. Despite having biological plausibility, dietary control measures, however, have been tested mainly for their preventive efficacy, not for lesion arrest.
Biofilm control
Because dental caries results from dysbiosis in the dental biofilm on a susceptible tooth surface, restoring balance within that biofilm (through the use of mechanical biofilm control, antimicrobials, probiotics, etc.) has been advocated [11]. Regular toothbrushing, interdental hygiene and antimicrobial strategies (including chlorhexidine and polyols) are the most common examples. Toothbrushing in combination with regular provision of fluoride has been investigated specifically for arresting existing active lesions. Most other therapies have been tested mainly for their preventive effect, not necessarily to arrest existing carious lesions [34].
Mineralisation control
Fluoride has been shown to reduce dental caries occurrence consistently in both the primary and permanent dentitions, with the most current evidence strongly suggesting that most of its effect is topical (that is post-eruptive). Examples include dentifrices with fluoride concentrations above 1000 ppm [35,36,37], 5000 ppm fluoride dentifrices [38,39,40,41] and fluoride rinses [42]. Professionally applied fluoride products, such as gels and varnishes, as well as silver fluoride products (such as silver diammine fluoride), have also been tested for arresting non-cavitated lesions [35, 38, 43, 44]. Moreover, a variety of products containing calcium in different forms (e.g. calcium stabilised by casein derivatives, calcium sodium phosphosilicate) or self-assembling peptides [45] have been introduced to aid with remineralisation. The evidence supporting the clinical efficacy of these products is currently limited [34, 46, 47].
Micro-invasive strategies
There are two main strategies falling into this level of invasiveness: sealing and infiltration.
Sealing
A sealant places a diffusion barrier on the susceptible tooth surface and, hence, impedes acid diffusion into and mineral loss from the tooth tissues. Sealing non-cavitated carious lesions on proximal, occlusal or smooth surfaces has been assessed in a range of studies [48,49,50]. There has been a limited assessment of the sealing of cavitated surfaces [49,50,51] and the current data implies a greater risk of sealant failure due to fracture or loss of retention, but further research needs to be undertaken to enable definitive conclusions to be drawn [51,52,53]. The relevance of sealant loss for lesion progression may further differ between different sealant materials (i.e. resin- versus glass ionomer cement–based sealants) [54].
Infiltration
Lesion infiltration involves the penetration of incipient enamel carious lesions by low-viscosity resins after removal of the surface layer (by etching with hydrochloric acid) and drying (using ethanol and air) [55]. A diffusion barrier is created within the dental hard tissue sub-surface, impeding acid diffusion into and mineral loss from the tooth tissue, inactivating the lesion. There is some evidence supporting infiltration of non-cavitated proximal lesions [34, 48]. However, there is currently only one product available on the market for caries infiltration, and nearly all related studies have been sponsored by the manufacturer of that product, with the associated potential for bias in the reported findings.
Mixed strategies
A range of strategies fall in here; we briefly discuss non-restorative cavity control and the Hall Technique.
Non-restorative cavity control
Non-restorative cavity control (NRCC) aims to reinstate the cleansability of cavitated lesions by chiselling, cutting or grinding away overhanging, biofilm-trapping enamel or dentine [56]. Subsequently, patients are encouraged to clean the opened cavity and apply fluoride toothpaste (in addition to professional fluoride varnish application). Currently, the technique has been applied only in primary teeth or root surface lesions. Under optimal conditions, NRCC seems to be a feasible option to manage cavitated lesions, especially in uncooperative children who may not tolerate other treatments [57,58,59,60] or in dependent and systemically compromised older adult patients.
Hall Technique
The Hall Technique involves ‘sealing’ of cavitated carious lesions in primary teeth using preformed metal crowns without any tooth preparation. It combines the biological management of carious lesions (via sealing bacteria and depriving them of nutrition) and the restorative advantages of preformed metal crowns [58, 61, 62].
Invasive strategies
Invasive caries management involves placement of a restoration after selective removal of carious tissue, done to create conditions for long-lasting restorations. Existing adhesive materials have allowed for minimally invasive (conservative) restorations that provide a good seal, and focus on preserving healthy and remineralisable tissue and maintaining pulpal health. An International Caries Consensus Collaboration presented recommendations on terminology and on carious tissue removal and restorative management of cavitated carious lesions. These recommendations support less invasive carious lesion management, delaying entry to, and slowing down, the restorative cycle by preserving tooth tissue (and being selective on need for tissue removal) and retaining teeth long-term [5].
Factors affecting intervention thresholds
A number of factors help to determine intervention thresholds. These are activity, cavitation and cleansability of the lesion.
Activity
The term ‘lesion activity’ reflects the ongoing mineral loss or gain of a lesion. It indicates how likely a lesion is to progress. An inactive (arrested) lesion may be considered as a ‘scar’ and does not require any treatment. In some cases, restorations might be placed for reasons of form, function and/or aesthetics. As discussed, restorations may also be provided in cases of pain to reduce pulpal stress and improve symptoms. Lesion activity is often determined visually; tactile assessment should only be performed gently so as to not damage the surface, using a rounded/ball-ended explorer (forceful probing with a sharp dental explorer is not recommended). For root surfaces, some gentle probing may be undertaken to determine the texture [63].
Since there is currently no technology available that allows an objective longitudinal measurement of lesion activity, the following clinical signs can be used to estimate lesion activity: (1) the presence of biofilm covering the lesion may indicate activity, especially in the presence of high and frequent consumption of sugars; (2) the condition of the gingiva (local gingivitis in proximity to the lesion) can also serve as a proxy measure to determine whether biofilm has been present over time and (3) lesion characteristics, such as texture, hardness and appearance. Smooth enamel lesion surface indicates inactivity, while rough surfaces may indicate activity. Colour-wise, a white chalky lesion colour may indicate activity, but shiny or dark lesions may indicate inactivity [63,64,65,66].
Notably, not all clinical criteria will be always available, e.g. in proximal surfaces or for micro-cavitated lesions (see below). However, in many circumstances, one or more criteria will be assessable, and may be complemented with (4) longitudinal recall data, if available (e.g. via repeated radiographs, or those from fluorescence-assisted systems, visual scales or clinical photographs). These can also be used for activity assessment. Lesion activity is the first main factor to decide intervention thresholds, although evidence is limited and more research is strongly advocated.
Cavitation
Cavitated lesions are those with a surface breach that is clearly detectable to the naked eye or a rounded dental explorer. Most often, this may also involve dentine exposure. Cavitation increases the likelihood of lesion progression [50, 67], because the dental biofilm is protected from self-cleaning and oral hygiene procedures. Moreover, cavitation encourages a more rapid diffusion of acids and carbohydrates, as well as greater bacterial contamination of tissues. In cavitated lesions, the involved dentine is demineralised and the outer portion of the lesion is bacterially contaminated.
A sub-group of cavitated lesions are the micro-cavitated lesions. Sometimes, magnification is needed to detect cavitation in these lesions. Micro-cavitated lesions can show enamel breakdown without visible dentine exposure.
Note that cavitation can be assessed tactile-visually on accessible smooth surfaces (buccal, lingual). Occlusally, their assessment is more complicated. Occlusal carious lesions which radiographically extend deep into the dentine (middle or inner dentine third, D2/3) are usually heavily bacterially contaminated and demineralised. These lesions may require a different management than lesions without such extensive dentine involvement.
On proximal surfaces, detecting cavitation using visual-tactile means is nearly impossible when adjacent teeth are present (except for clear cavitation in more extensive lesions). Orthodontic separators may be used to gain access over some days, but this is not practical in most settings. Hence, most clinicians will rely on further diagnostic aids, mainly bitewing radiography, to gauge the likelihood of cavitation. While radiographs seldom permit accurate detection of cavitation itself, the lesion depth serves as a proxy of how likely cavitation is; deeper radiographic lesions come with a higher chance of being cavitated. A number of studies have assessed the relationship between the radiographic lesion depth and the presence of cavitation, and these have been summarised in a recent review [68]. Lesions confined radiographically to the enamel (outer or inner enamel half, E1/E2) are seldom cavitated, while those into the middle or inner dentine third (D2/D3) are usually cavitated. Some of the lesions radiographically extending into the outer dentine third (D1) may be cavitated, others not. Further detection aids such as near-infrared light transillumination or fluorescence-based systems [69] can be used to corroborate radiographic lesion depth assessment.
Cavitation is the second main factor to determine intervention thresholds.
Cleansability
Cavitation is a major factor for deciding whether and how to intervene, because it serves as an indicator of cleansability and, as described, activity. Nevertheless, sometimes even cavitated lesions can be cleansable, for example in primary anterior teeth with open smooth surface lesions or in root surface lesions. Cleansability is the third main factor to decide intervention thresholds (and closely related to activity, the first factor).
There are further factors which can modify intervention thresholds, which are the patient’s caries risk and age and the dentition.
Caries risk/susceptibility and behavioural adherence
The term caries risk/susceptibility refers to the chances of an individual developing new carious lesions and existing lesions progressing in the future. There are a number of possible aspects to be integrated into caries risk/susceptibility assessment, such as past caries experience (assuming past caries experience to be a robust indicator of risk factors on the behavioural and the genetic level) and factors directly or indirectly related to caries pathogenesis (diet, oral hygiene, saliva) or lesion development (fluoride intake). Past caries experience has been found to have a good predictive value [70], while most other factors have only moderate or low predictive value. Many further factors suggested for caries risk assessment (salivary buffering capacity, bacterial number/concentration in the saliva, to name but two) show limited predictive value [71]. For root caries, the number of surfaces at risk (exposed roots) seems to be a useful robust parameter for risk assessment [72].
There are some established caries risk/susceptibility assessment systems which integrate these factors and weight them. These systems have been found useful to predict coronal and root caries to some degree, but with limited generalisability [73,74,75].
Caries risk/susceptibility assessment helps to identify patient-specific risk factors, which are of value. These should be managed and re-evaluation should be performed regularly throughout any episode of care. If risk factor modification is not successful or re-evaluation is not possible, this should be taken into consideration in determining interventional thresholds.
Age
There are several unique considerations that are important in the management of children’s and older seniors’ (or those with special needs) oral health [76]. The oral health and behaviour of the carer have a marked influence on the oral health, especially in younger children or other heavily dependent individuals, such as the elderly and institutionalised individuals. Also, the over-riding aspect is behavioural capabilities. Especially very young children show only limited cooperation and adherence [77]. In some of these individuals, sedation or general anaesthesia is needed to provide care. Interventional thresholds may be lowered and more invasive treatments chosen in some of these instances.
The dentition
The structure of primary teeth differs from that of permanent teeth. The enamel is thinner and slightly less mineralised. The pulpal space is proportionally larger, and the shape of the teeth is different. The contact areas of the molar teeth, especially between the mandibular molars, are flatter than permanent molars and premolars, and this predisposes to carious lesions developing below the wide contact area, close to the gingival margin. Given this anatomy, conventional restorative approaches (including carious tissue removal and direct restorations) have higher risks of complications (more pulp exposure and restorative complications) in primary than permanent teeth [61, 78]. Moreover, visual-tactile lesion detection on proximal surfaces of primary teeth is difficult because of the wider contact areas [79]. Also, and most importantly, primary teeth exfoliate. The time to exfoliation and the speed of progression of the carious lesion can influence the decision on treatment for a lesion. Pulp therapies for primary molars (especially those with necrotic pulp tissue) may be successful, but technically demanding [80]. In few cases, removing primary molars and (if needed) maintaining the space may be a valid option in some cases to avoid pain or sepsis. In contrast, for permanent teeth, the overarching therapeutic aim is long-term retention in a functional, pain-free and depending on the location in the mouth, aesthetically pleasing condition. Any intervention threshold should consider these aspects.
When to intervene in the caries process
Based on the contemporary understanding of the disease dental caries, the available interventions and the factors determining or modifying intervention thresholds, the consensus group agreed on a number of recommendations for when to intervene non-invasively, micro-invasively or invasively/restoratively in the caries process and for existing carious lesions. These recommendations were the subject of the Delphi process, with experts voting on the verbatim recommendations below.
The recommendations should be adapted to each individual patient and setting and need to be applied with each oral healthcare professional’s individual expertise and practising context in mind. Also note that nearly all recommendations made are not based on strong evidence, but mainly on expert opinion and experience. Hence, clinical judgement remains a key element in deciding the threshold for intervention. Future studies should aim to support such recommendations at a higher evidence level. The recommendations are set out below, together with the level of agreement (mean values on a scale from 1 (do not agree at all) to 10 (fully agree) and standard deviations, SD).
-
1.
Lesion activity should be assessed. An inactive (arrested) lesion is like a ‘scar’ and does not require any treatment (but should be monitored). In some cases, restorations might be placed for reasons of form, function or aesthetics. An active lesion needs management (agreement 9.4, SD 1.1) (Fig. 2).
-
2.
Cavitation increases the likelihood of activity and lesion progression, as the dental biofilm is protected from self-cleaning and oral hygiene procedures. Moreover, cavitation encourages a more rapid diffusion of sugars and acids. Cavitation can be assessed validly on visible/accessible surfaces (9.2, SD 0.8).
-
3.
As a general principle,
-
3.1.
Inactive non-cavitated and cavitated lesions do not require any treatment (except for reasons of form, function or aesthetics) (8.8, SD 1.4).
-
3.2.
Active non-cavitated carious lesions should be managed non- or micro-invasively (9.1, SD 0.9) (Fig. 2).
-
3.3.
Cavitated carious lesions which are cleansable, and active, can also be managed non- or micro-invasively (except for reasons of form, function or aesthetics) (8.8, SD 1.4) (Fig. 2).
-
3.4.
Cavitated carious lesions which are not cleansable, and active, should be managed using invasive/restorative strategies. In specific circumstances, mixed interventions may be applicable (9.1, SD 1.0) (Fig. 2).
-
3.1.
-
4.
On occlusal surfaces, two specific scenarios arise (Fig. 3).
-
a.
Micro-cavitated lesions extending only into enamel. These can be successfully managed using micro-invasive or mixed interventions (8.8, SD 1.5).
-
b.
Micro-cavitated lesions radiographically extending deep into dentine (middle or inner dentine third, D2/3). These are often deeply invaded by bacterial biofilms, demineralised and also cavitated, but the cavitation cannot be detected/accessed given the specific anatomy of the occlusal surface. Lesion arrest using non-invasive means may be unlikely here. Also, the stability of any kind of sealant material placed over these lesions appears to be limited. Hence, such lesions should be managed invasively/restoratively in the majority of cases (9.1, SD 0.8).
-
a.
-
5.
On proximal surfaces, cavitation of early lesions is usually hard to assess tactile-visually. Orthodontic separators may be used to gain visible access, or lesion depth ascertained from bitewing radiography can be used as proxy to determine the likelihood of cavitation (Fig. 4). Lesions extending radiographically into the middle or inner third of the dentine (D2/3) can be assumed to be cavitated, while those restricted to the enamel (E1/2) are usually not cavitated. These lesions should be managed accordingly. For lesions radiographically extending into the outer third of the dentine (D1), a decision problem exists. If possible, these lesions should be managed as if they were non-cavitated, as they are likely to be non-cavitated (i.e. non- or micro-invasively) (8.9, SD 1.1).
-
6.
The caries risk/susceptibility of a patient should be assessed. Identified patient-specific risk factors should be, if possible, managed, and re-evaluation should be performed regularly. If risk factor modification is not successful or re-evaluation is not possible, this should be taken into consideration and may influence subsequent interventional thresholds (9.0, SD 0.8).
-
7.
Interventional thresholds may be lowered and more invasive treatments chosen for treatment requiring sedation or general anaesthesia (e.g. children, disabled patients) (9.2, SD 1.1).
-
8.
In permanent teeth, the overarching therapeutic aim is retaining teeth in a functional, pain-free and—depending on the location in the mouth—aesthetically pleasing condition. In primary teeth, maintaining the space of primary molars and avoiding pain or sepsis are the overarching therapeutic aims. Intervention threshold should consider these aims accordingly (9.0, SD 1.3).
Factors determining intervention thresholds. Activity, cavitation status and cleansability are the main factors determining intervention thresholds (whether and when to intervene). Thresholds need to be adapted to each individual patient and setting, and need to be applied with each dental professional’s individual expertise in mind. There are specific scenarios (Figs. 3 and 4) which require further considerations. Note that as described, not all interventions are applicable in both dentitions or all patients
Factors determining intervention thresholds on occlusal lesions. Activity and cavitation status are the main factors determining intervention thresholds. In addition, radiographic dentine involvement should be considered. Note that as described, not all interventions are applicable in both dentitions or all patients
Factors determining intervention thresholds on proximal lesions. Activity and cavitation status are the main factors determining intervention thresholds. However, cavitation status is clinically often difficult to assess. Hence, radiographic lesion depth is usually used as a proxy to estimate the chances of cavitation. For lesions confined to enamel (E1, 2), cavitation is unlikely. For lesions extending into the middle or inner third of the dentine (D2, 3), cavitation is likely. Lesions extending into the outer third of the dentine (D1) are more likely non-cavitated than cavitated. If a cavitation cannot be detected clinically, dentists should prefer non- or micro-invasive means if possible. Only under specific circumstances, invasive (or mixed) interventions should be applied. Note that as described, not all interventions are applicable in both dentitions or all patients
Conclusions
We have presented a consensus on when to intervene in the caries process and for existing carious lesions using non- or micro-invasive, invasive/restorative or mixed interventions. Lesion activity, cavitation and cleansability are the main factors to be considered to determine intervention thresholds. Inactive lesions do not usually require any treatment (in some cases, restorations might be placed for reasons of form, function and aesthetics); active lesions do. Non-cavitated carious lesions should be managed non- or micro-invasively, as should cavitated carious lesions which are cleansable. Cavitated carious lesions which are not cleansable usually require invasive/restorative management, also to restore form, function and aesthetics of the tooth. In specific circumstances, mixed interventions may be applicable. On occlusal surfaces, cavitated lesions confined to enamel and non-cavitated lesions radiographically extending deep into dentine (middle or inner dentine third, D2/3) may be exceptions to that rule. On proximal surfaces, cavitation is usually hard to assess tactile-visually. Hence, radiographic lesion depth is used to determine the likelihood of cavitation. Lesions extending radiographically into the middle or inner third of the dentine (D2/3) can be assumed to be cavitated, while those restricted to the enamel (E1/2) are usually not cavitated. For lesions radiographically extending into the outer third of the dentine (D1), cavitation status is unclear. These lesions should be managed as if they were non-cavitated unless otherwise indicated. Individual decisions and clinical judgement should consider factors modifying the described intervention thresholds. Comprehensive diagnostics are the basis for systematic decision-making on when to intervene in the caries process and on existing carious lesions. Patients should be informed comprehensively about treatment options and should provide informed consent accordingly.
References
Sackett DL, Rosenberg WMC, Gray JAM, Haynes RB, Richardson WS (1996) Evidence based medicine: what it is and what it isn’t. BMJ 312(7023):71–72. https://doi.org/10.1136/bmj.312.7023.71
Frencken JE, Innes NP, Schwendicke F (2016) Managing carious lesions: why do we need consensus on terminology and clinical recommendations on carious tissue removal? Adv Dent Res 28(2):46–48. https://doi.org/10.1177/0022034516639272
Innes NP, Frencken JE, Schwendicke F (2016) Don’t know, can’t do, won’t change: barriers to moving knowledge to action in managing the carious lesion. J Dent Res 95(5):485–486. https://doi.org/10.1177/0022034516638512
Frencken JE, Peters MC, Manton DJ, Leal SC, Gordan VV, Eden E (2012) Minimal intervention dentistry for managing dental caries - a review: report of a FDI task group. Int Dent J 62(5):223–243. https://doi.org/10.1111/idj.12007
Schwendicke F, Frencken JE, Bjorndal L, Maltz M, Manton DJ, Ricketts D, Van Landuyt K, Banerjee A, Campus G, Domejean S, Fontana M, Leal S, Lo E, Machiulskiene V, Schulte A, Splieth C, Zandona AF, Innes NP (2016) Managing carious lesions: consensus recommendations on carious tissue removal. Adv Dent Res 28(2):58–67. https://doi.org/10.1177/0022034516639271
Junger S, Payne SA, Brine J, Radbruch L, Brearley SG (2017) Guidance on Conducting and REporting DElphi Studies (CREDES) in palliative care: recommendations based on a methodological systematic review. Palliat Med 31(8):684–706. https://doi.org/10.1177/0269216317690685
Kassebaum NJ, Smith AGC, Bernabé E, Fleming TD, Reynolds AE, Vos T, Murray CJL, Marcenes W, Abyu GY, Alsharif U, Asayesh H, Benzian H, Dandona L, Dandona R, Kasaeian A, Khader YS, Khang YH, Kokubo Y, Kotsakis GA, Lalloo R, Misganaw A, Montero P, Nourzadeh M, Pinho C, Qorbani M, Blancas MJR, Sawhney M, Steiner C, Traebert J, Tyrovolas S, Ukwaja KN, Vollset SE, Yonemoto N (2017) Global, regional, and national prevalence, incidence, and disability-adjusted life years for oral conditions for 195 countries, 1990–2015: a systematic analysis for the global burden of diseases, injuries, and risk factors. J Dent Res 96(4):380–387. https://doi.org/10.1177/0022034517693566
Keyes PH (1960) The infectious and transmissible nature of experimental dental caries. Findings and implications. Arch Oral Biol 1:304–320
Keyes PH, Fitzgerald RJ (1962) Dental caries in the Syrian hamster. IX. Arch Oral Biol 7:267–277
Marsh PD (2006) Dental plaque as a biofilm and a microbial community - implications for health and disease. BMC Oral Health 6(S1):S14
Marsh PD (2018) In sickness and in health—what does the oral microbiome mean to us? An ecological perspective. Adv Dent Res 29(1):60–65. https://doi.org/10.1177/0022034517735295
Fisher-Owens SA, Gansky SA, Platt LJ, Weintraub JA, Soobader MJ, Bramlett MD, Newacheck PW (2007) Influences on children’s oral health: a conceptual model. Pediatrics 120(3):e510–e520. https://doi.org/10.1542/peds.2006-3084
Gomez A, Espinoza JL, Harkins DM, Leong P, Saffery R, Bockmann M, Torralba M, Kuelbs C, Kodukula R, Inman J, Hughes T, Craig JM, Highlander SK, Jones MB, Dupont CL, Nelson KE (2017) Host genetic control of the oral microbiome in health and disease. Cell Host Microbe 22(3):269–278.e263. https://doi.org/10.1016/j.chom.2017.08.013
Takahashi N, Nyvad B (2011) The role of bacteria in the caries process: ecological perspectives. J Dent Res 90(3):294–303. https://doi.org/10.1177/0022034510379602
Dawes C (2003) What is the critical pH and why does a tooth dissolve in acid? J Can Dent Assoc 69(11):722–724
Vieira AR, Gibson CW, Deeley K, Xue H, Li Y (2015) Weaker dental enamel explains dental decay. PLoS One 10(4):e0124236. https://doi.org/10.1371/journal.pone.0124236
Weber M, Bogstad Sovik J, Mulic A, Deeley K, Tveit AB, Forella J, Shirey N, Vieira AR (2018) Redefining the phenotype of dental caries. Caries Res 52(4):263–271. https://doi.org/10.1159/000481414
Valdebenito B, Tullume-Vergara PO, Gonzalez W, Kreth J, Giacaman RA (2018) In silico analysis of the competition between Streptococcus sanguinis and Streptococcus mutans in the dental biofilm. Mol Oral Microbiol 33(2):168–180. https://doi.org/10.1111/omi.12209
Takahashi N, Nyvad B (2016) Ecological hypothesis of dentin and root caries. Caries Res 50(4):422–431. https://doi.org/10.1159/000447309
Banerjee A (2017) ‘Minimum intervention’ - MI inspiring future oral healthcare? Br Dent J 223(3):133–135. https://doi.org/10.1038/sj.bdj.2017.644
Schwendicke F, Gostemeyer G (2016) Understanding dentists’ management of deep carious lesions in permanent teeth: a systematic review and meta-analysis. Implement Sci 11(1):142. https://doi.org/10.1186/s13012-016-0505-4
Innes N, Schwendicke F (2017) Dentists’ thresholds for restorative intervention in carious lesions: systematic review and meta-analysis journal of dental research submitted
Albino J, Tiwari T (2016) Preventing childhood caries: a review of recent behavioral research. J Dent Res 95(1):35–42. https://doi.org/10.1177/0022034515609034
Domejean S, Banerjee A, Featherstone JDB (2017) Caries risk/susceptibility assessment: its value in minimum intervention oral healthcare. Br Dent J 223(3):191–197. https://doi.org/10.1038/sj.bdj.2017.665
Ismail A (2004) Diagnostic levels in dental public health planning. Caries Res 38(3):199–203. https://doi.org/10.1159/000077755
Kassebaum NJ, Bernabe E, Dahiya M, Bhandari B, Murray CJ, Marcenes W (2015) Global burden of untreated caries: a systematic review and metaregression. J Dent Res 94(5):650–658. https://doi.org/10.1177/0022034515573272
Raedel M, Hartmann A, Bohm S, Priess HW, Samietz S, Konstantinidis I, Walter MH (2017) Four-year outcomes of restored posterior tooth surfaces-a massive data analysis. Clin Oral Investig 21(9):2819–2825. https://doi.org/10.1007/s00784-017-2084-4
Burke FJ, Lucarotti PS, Holder RL (2005) Outcome of direct restorations placed within the general dental services in England and Wales (part 2): variation by patients’ characteristics. J Dent 33(10):817–826. https://doi.org/10.1016/j.jdent.2005.03.007
Schwendicke F, Gostemeyer G, Blunck U, Paris S, Hsu LY, Tu YK (2016) Directly placed restorative materials: review and network meta-analysis. J Dent Res 95:613–622. https://doi.org/10.1177/0022034516631285
Elderton RJ (1990) Clinical studies concerning re-restoration of teeth. Adv Dent Res 4:4–9. https://doi.org/10.1177/08959374900040010701
Brantley C, Bader J, Shugars D, Nesbit S (1995) Does the cycle of rerestoration lead to larger restorations? J Am Dent Assoc 126(10):1407–1413
Tyas MJ, Anusavice KJ, Frencken JE, Mount GJ (2000) Minimal intervention dentistry--a review. FDI Commission Project 1-97. Int Dent J 50(1):1–12
Moynihan PJ, Kelly SAM (2014) Effect on caries of restricting sugars intake: systematic review to inform WHO guidelines. J Dent Res 93(1):8–18
Slayton RL, Urquhart O, Araujo MWB, Fontana M, Guzman-Armstrong S, Nascimento MM, Novy BB, Tinanoff N, Weyant RJ, Wolff MS, Young DA, Zero DT, Tampi MP, Pilcher L, Banfield L, Carrasco-Labra A (2018) Evidence-based clinical practice guideline on nonrestorative treatments for carious lesions: a report from the American Dental Association. J Am Dent Assoc (1939) 149(10):837–849.e819. https://doi.org/10.1016/j.adaj.2018.07.002
Marinho VC, Worthington HV, Walsh T, Clarkson JE (2013) Fluoride varnishes for preventing dental caries in children and adolescents. Cochrane Database Syst Rev 7:Cd002279. https://doi.org/10.1002/14651858.CD002279.pub2
Walsh T, Worthington HV, Glenny AM, Appelbe P, Marinho VC, Shi X (2010) Fluoride toothpastes of different concentrations for preventing dental caries in children and adolescents. Cochrane Database Syst Rev 1:Cd007868. https://doi.org/10.1002/14651858.CD007868.pub2
Wolff MS, Schenkel AB (2018) The anticaries efficacy of a 1.5% arginine and fluoride toothpaste. Adv Dent Res 29(1):93–97. https://doi.org/10.1177/0022034517735298
Wierichs RJ, Meyer-Lueckel H (2014) Systematic review on noninvasive treatment of root caries lesions. J Dent Res 94:261–271. https://doi.org/10.1177/0022034514557330
Baysan A, Lynch E, Ellwood R, Davies R, Petersson L, Borsboom P (2001) Reversal of primary root caries using dentifrices containing 5,000 and 1,100 ppm fluoride. Caries Res 35(1):41–46. https://doi.org/10.1159/000047429
Ekstrand K, Martignon S, Holm-Pedersen P (2008) Development and evaluation of two root caries controlling programmes for home-based frail people older than 75 years. Gerodontology 25(2):67–75. https://doi.org/10.1111/j.1741-2358.2007.00200.x
Ekstrand KR, Poulsen JE, Hede B, Twetman S, Qvist V, Ellwood RP (2013) A randomized clinical trial of the anti-caries efficacy of 5,000 compared to 1,450 ppm fluoridated toothpaste on root caries lesions in elderly disabled nursing home residents. Caries Res 47(5):391–398. https://doi.org/10.1159/000348581
Marinho VC, Chong LY, Worthington HV, Walsh T (2016) Fluoride mouthrinses for preventing dental caries in children and adolescents. Cochrane Database Syst Rev 7:Cd002284. https://doi.org/10.1002/14651858.CD002284.pub2
Gao SS, Zhang S, Mei ML, Lo EC, Chu CH (2016) Caries remineralisation and arresting effect in children by professionally applied fluoride treatment - a systematic review. BMC Oral Health 16:12. https://doi.org/10.1186/s12903-016-0171-6
Fung MHT, Duangthip D, Wong MCM, Lo ECM, Chu CH (2018) Randomized clinical trial of 12% and 38% silver diamine fluoride treatment. J Dent Res 97(2):171–178. https://doi.org/10.1177/0022034517728496
Alkilzy M, Santamaria RM, Schmoeckel J, Splieth CH (2018) Treatment of carious lesions using self-assembling peptides. Adv Dent Res 29(1):42–47. https://doi.org/10.1177/0022034517737025
Fontana M (2016) Enhancing fluoride: clinical human studies of alternatives or boosters for caries management. Caries Res 50(Suppl 1):22–37. https://doi.org/10.1159/000439059
Alkilzy M, Tarabaih A, Santamaria RM, Splieth CH (2018) Self-assembling peptide P11-4 and fluoride for regenerating enamel. J Dent Res 97(2):148–154. https://doi.org/10.1177/0022034517730531
Krois J, Gostemeyer G, Reda S, Schwendicke F (2018) Sealing or infiltrating proximal carious lesions. J Dent 74:15–22. https://doi.org/10.1016/j.jdent.2018.04.026
Schwendicke F, Jager AM, Paris S, Hsu LY, Tu YK (2015) Treating pit-and-fissure caries: a systematic review and network meta-analysis. J Dent Res 94(4):522–533. https://doi.org/10.1177/0022034515571184
Griffin SO, Oong E, Kohn W, Vidakovic B, Gooch BF, Bader J, Clarkson J, Fontana MR, Meyer DM, Rozier RG, Weintraub JA, Zero DT (2008) The effectiveness of sealants in managing caries lesions. J Dent Res 87(2):169–174. https://doi.org/10.1177/154405910808700211
Fontana M, Platt JA, Eckert GJ, Gonzalez-Cabezas C, Yoder K, Zero DT, Ando M, Soto-Rojas AE, Peters MC (2014) Monitoring of sound and carious surfaces under sealants over 44 months. J Dent Res 93(11):1070–1075. https://doi.org/10.1177/0022034514551753
Hesse D, Bonifacio CC, Mendes FM, Braga MM, Imparato JC, Raggio DP (2014) Sealing versus partial caries removal in primary molars: a randomized clinical trial. BMC Oral Health 14:58. https://doi.org/10.1186/1472-6831-14-58
Bakhshandeh A, Qvist V, Ekstrand K (2012) Sealing occlusal caries lesions in adults referred for restorative treatment: 2–3 years of follow-up. Clin Oral Investig 16(2):521–529. https://doi.org/10.1007/s00784-011-0549-4
Mickenautsch S, Yengopal V (2013) Validity of sealant retention as surrogate for caries prevention--a systematic review. PLoS One 8(10):e77103. https://doi.org/10.1371/journal.pone.0077103
Paris S, Hopfenmuller W, Meyer-Lueckel H (2010) Resin infiltration of caries lesions. J Dent Res 89(8):823–826. https://doi.org/10.1177/0022034510369289
Gruythuysen R (2010) Non-restorative cavity treatment. Managing rather than masking caries activity. Nederlands Tijdschrift Voor Tandheelkunde 117(3):173–180
Mijan M, de Amorim RG, Leal SC, Mulder J, Oliveira L, Creugers NH, Frencken JE (2014) The 3.5-year survival rates of primary molars treated according to three treatment protocols: a controlled clinical trial. Clin Oral Investig 18(4):1061–1069. https://doi.org/10.1007/s00784-013-1077-1
Santamaria RM, Innes NPT, Machiulskiene V, Schmoeckel J, Alkilzy M, Splieth CH (2017) Alternative caries management options for primary molars: 2.5-year outcomes of a randomised clinical trial. Caries Res 51(6):605–614. https://doi.org/10.1159/000477855
Hansen NV, Nyvad B (2017) Non-operative control of cavitated approximal caries lesions in primary molars: a prospective evaluation of cases. J Oral Rehabil 44(7):537–544. https://doi.org/10.1111/joor.12508
Lo EC, Schwarz E, Wong MC (1998) Arresting dentine caries in Chinese preschool children. Int J Paediatr Dent/ the British Paedodontic Society [and] the International Association of Dentistry for Children 8(4):253–260
Hickel R, Kaaden C, Paschos E, Buerkle V, García-Godoy F, Manhart J (2005) Longevity of occlusally-stressed restorations in posterior primary teeth. Am J Dent 18(3):198–211
Innes NP, Evans DJ, Stirrups DR (2011) Sealing caries in primary molars: randomized control trial, 5-year results. J Dent Res 90(12):1405–1410. https://doi.org/10.1177/0022034511422064
Nyvad B, Machiulskiene V, Baelum V (1999) Reliability of a new caries diagnostic system differentiating between active and inactive caries lesions. Caries Res 33(4):252–260
Braga M, Mendes F, Martignon S, Ricketts D, Ekstrand K (2009) In vitro comparison of Nyvad’s system and ICDAS-II with lesion activity assessment for evaluation of severity and activity of occlusal caries lesions in primary teeth. Caries Res 43:405–412
Braga MM, Martignon S, Ekstrand KR, Ricketts DN, Imparato JC, Mendes FM (2010) Parameters associated with active caries lesions assessed by two different visual scoring systems on occlusal surfaces of primary molars - a multilevel approach. Community Dent Oral Epidemiol 38(6):549–558. https://doi.org/10.1111/j.1600-0528.2010.00567.x
Nyvad B, Fejerskov O (1997) Assessing the stage of caries lesion activity on the basis of clinical and microbiological examination. Community Dent Oral Epidemiol 25(1):69–75
Ferreira Zandona A, Santiago E, Eckert GJ, Katz BP, Pereira de Oliveira S, Capin OR, Mau M, Zero DT (2012) The natural history of dental caries lesions: a 4-year observational study. J Dent Res 91(9):841–846. https://doi.org/10.1177/0022034512455030
Wenzel A (2014) Radiographic display of carious lesions and cavitation in approximal surfaces: advantages and drawbacks of conventional and advanced modalities. Acta Odontol Scand 72(4):251–264. https://doi.org/10.3109/00016357.2014.888757
Tassery H, Levallois B, Terrer E, Manton DJ, Otsuki M, Koubi S, Gugnani N, Panayotov I, Jacquot B, Cuisinier F, Rechmann P (2013) Use of new minimum intervention dentistry technologies in caries management. Aust Dent J 58(Suppl 1):40–59. https://doi.org/10.1111/adj.12049
Broadbent JM, Foster Page LA, Thomson WM, Poulton R (2013) Permanent dentition caries through the first half of life. Br Dent J 215(7):E12. https://doi.org/10.1038/sj.bdj.2013.991
Mejare I, Axelsson S, Dahlen G, Espelid I, Norlund A, Tranaeus S, Twetman S (2013) Caries risk assessment. A systematic review. Acta Odontol Scand 72(2):81–91. https://doi.org/10.3109/00016357.2013.822548
Lopez R, Smith PC, Gostemeyer G, Schwendicke F (2017) Ageing, dental caries and periodontal diseases. J Clin Periodontol 44(Suppl 18):S145–s152. https://doi.org/10.1111/jcpe.12683
Bratthall D, Hansel Petersson G (2005) Cariogram--a multifactorial risk assessment model for a multifactorial disease. Community Dent Oral Epidemiol 33(4):256–264. https://doi.org/10.1111/j.1600-0528.2005.00233.x
Hayes M, Da Mata C, McKenna G, Burke FM, Allen PF (2017) Evaluation of the Cariogram for root caries prediction. J Dent 62:25–30. https://doi.org/10.1016/j.jdent.2017.04.010
Domejean S, White JM, Featherstone JD (2011) Validation of the CDA CAMBRA caries risk assessment--a six-year retrospective study. J Calif Dent Assoc 39(10):709–715
Innes NP, Manton DJ (2017) Minimum intervention children’s dentistry - the starting point for a lifetime of oral health. Br Dent J 223(3):205–213. https://doi.org/10.1038/sj.bdj.2017.671
Leal SC (2014) Minimal intervention dentistry in the management of the paediatric patient. Br Dent J 216(11):623–627. https://doi.org/10.1038/sj.bdj.2014.449
Manhart J, Chen H, Hamm G, Hickel R (2004) Buonocore Memorial Lecture. Review of the clinical survival of direct and indirect restorations in posterior teeth of the permanent dentition. Oper Dent 29(5):481–508
Ribeiro AA, Purger F, Rodrigues JA, Oliveira PR, Lussi A, Monteiro AH, Alves HD, Assis JT, Vasconcellos AB (2015) Influence of contact points on the performance of caries detection methods in approximal surfaces of primary molars: an in vivo study. Caries Res 49(2):99–108. https://doi.org/10.1159/000368562
Smail-Faugeron V, Glenny AM, Courson F, Durieux P, Muller-Bolla M, Fron Chabouis H (2018) Pulp treatment for extensive decay in primary teeth. Cochrane Database Syst Rev 5:Cd003220. https://doi.org/10.1002/14651858.CD003220.pub3
Funding
The conference was kindly sponsored by DMG (Hamburg, Germany). This included travel, accommodation and conference costs for panel members.
Author information
Authors and Affiliations
Contributions
FS, DJM: Organised the meeting, administered the consensus process and drafted parts of the first version of this document
AB, MF, SP: Drafted parts of the first version of this document
All authors: Provided input into the draft document, participated in the consensus process, revised the document and are accountable for the final version
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article did not involve undertaking of any study of humans or animals.
Informed consent
For this type of study, formal consent is not required.
Disclaimer
The sponsor had no role in design or conduct of the conference or the content of this manuscript. No honoraria were given to any of the panel members.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
The expert group represented members of the European Association for Caries Research (ORCA) and the European Federation of Conservative Dentistry (EFCD) as well as (mainly overseas) non-members. The group was organised and the process was led by two members, FS and DJM. These members also organised financial support for the meeting. The members of the expert group were chosen based on their clinical and scientific expertise, allowing sufficient breadth of experience, as well as geographic aspects. All experts were familiar to one or both organisers. Note that some experts came from the same institution; no weighting or adjustment during the consensus was performed for this, as any kind of possible bias introduced by this was assumed to be limited and was accepted, but also as no valid rules for such weighting or adjustment are available.
Both ORCA and EFCD approved and supported the initiative, its aims and the meeting, and the then-president-elect of ORCA and the then-acting-president of EFCD were members of the group. As described, all members of the group provided a conflict of interest declaration and no member was found to be subject to relevant conflict of interest related to the consensus statement.
Prior to the meeting, a working paper, which also formed the basis for the present consensus document, was drafted by a smaller group of members, whose task it was to sum up and synthesise the available evidence for the different levels of invasiveness (NI, MI, invasive) as well as the evidence base towards possible intervention thresholds. Note that no systematic review process was performed, but existing reviews were considered. The compiled draft was sent to the overall group, who commented on it extensively, in two rounds. The resulting manuscript was the basis of the following steps and included consensus recommendations. Only these recommendations were voted on during the subsequent Delphi process; the text itself (excluding the recommendations) was not submitted to any further consensus process as we felt the core of the consensus was the recommendations.
A two-staged confidential e-Delphi survey was then undertaken. Between the two Delphi rounds, the consensus panel meeting was held. The reporting for this Delphi follows the guidance on Conducting and REporting DElphi Studies (CREDES) [6], with all points being laid out below once more for reasons for clarity.
Rationale for the choice of the Delphi technique
-
1.
Justification: A stepwise approach of coming to a consensus on a set of evidence-grounded statements, after discussion first via e-mail/text, then in a form of a meeting, was decided to be built on the Delphi technique. This technique is transparent, anonymous in voting and accepted by the community. Further, it was feasible and fitted to the specific design of this consensus process. By combining an open-ended approach with a Delphi, we aimed to allow a systematic but nevertheless comprehensive approach.
Planning and design
-
2.
Planning and process. The consensus rules (see below) were agreed to by the panel via e-mail before starting the Delphi process. Modifications are described below. The Delphi asked for an agreement to each consensus statement (as can be found in the consensus recommendations section of the main paper), with a scale of 1–10 (do not at all agree to agree fully) being used. A multi-stage Delphi was planned, without removal of any items prior to concluding at maximum three rounds. Each round closed after a 4-week period. One reminder via e-mail was sent for each round. Panellists were allowed to comment on each item. The survey was conducted via Delphi Manager 3.0, University of Liverpool, UK, and Surveyjet (Calibrum, https://calibrum.com), and survey data was analysed descriptively.
-
3.
Definition of consensus. The following consensus rules applied. (1) Agreement to an item was defined by marking grades 7–10 on a scale from 1 to 10. (2) Minimum 70% of all participants needed to agree to an item for this to be consensually accepted. Items which did not meet these criteria after the planned 2 rounds were to be dropped (no item was eventually dropped). For reasons of transparency, we additionally report on the mean agreement and the standard deviation
Study conduct
-
4.
Informational input: The material provided to the panel is described in the main text. Its attainment has been described above.
-
5.
Prevention of bias: To identify possible risk of bias, all members filled out a conflict of interest form. To prevent bias, a systematic, evidence-grounded approach was chosen. Note that the topic itself does only limitedly lend itself for financial/commercial bias. The planning and conduct were performed independent from the sponsor.
-
6.
Interpretation and processing of results: There was, as discussed, stable agreement to all items after the second round.
-
7.
External validation: No external validation was sought.
Reporting
-
8.
Purpose and rationale: These have been provided.
-
9.
Expert panel: The criteria for the selection of experts were provided.
-
10.
Description of the methods: Preparatory steps, synthesis of the evidence, piloting of the statements, survey rounds and conference have been described.
-
11.
Procedure: The Delphi steps have been described.
-
12.
Definition and attainment of consensus: The following consensus rules applied. (1) Agreement to an item was defined by marking grades 7–10 on a scale from 1 to 10. (2) Minimum 70% of all participants needed to agree to an item for this to be consensually accepted.
-
13.
Results: The results are reported in the main text. Note that between steps, at the panel meeting, a discussion on all items was held; these discussions had not been planned a priori but found necessary after the first round and the revision of the manuscript. Some items, mainly those showing low agreement in round 1, were revised in language or content, and all items provided to the group in the second round. A consensus was reached on all items in the second Delphi round. All panellists except one took part in both Delphi rounds.
-
14.
Discussion of limitations: A limited group of people have been invited and came to this consensus, which is a limitation. Moreover, and as laid out, most statements are not supported by strong evidence, as this is missing.
-
15.
Adequacy of conclusions: The conclusions reflect the outcomes of the Delphi and aim for applicability of the deduced guidance points.
-
16.
Publication and dissemination: The consensus paper will be translated in various languages and published in national journals for dissemination.
Rights and permissions
About this article
Cite this article
Schwendicke, F., Splieth, C., Breschi, L. et al. When to intervene in the caries process? An expert Delphi consensus statement. Clin Oral Invest 23, 3691–3703 (2019). https://doi.org/10.1007/s00784-019-03058-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-019-03058-w