Abstract
Purposes
We use the laparoscopic percutaneous extraperitoneal closure (LPEC) method as the standard procedure for pediatric inguinal hernia. Despite judging there to be no contralateral patent processus vaginalis (PPV) at the time of the first LPEC, we experienced five cases in which metachronous contralateral inguinal hernia (MCH) developed, so we report the characteristics, including the predictors.
Methods
For pediatric inguinal hernia, the LPEC method was used in 1277 cases from 2005 to 2019 in our department. Of these, 374 patients underwent unilateral LPEC, and we compared the 5 patients with MCH onset and the 369 without MCH onset. The items to be examined were the gender, age, presence of a low birth weight, initial-onset side, and contralateral internal inguinal ring classification.
Results
There was no significant difference in the gender, age, initial-onset side, or contralateral internal inguinal ring classification between the two groups. Low-birth-weight infants were significantly more common among those with MCH than among those without MCH.
Conclusions
The only predictor of a contralateral onset after LPEC for pediatric inguinal hernia was a low birth weight. Therefore, for the above-mentioned unilateral LPEC cases, the possibility of a contralateral onset after LPEC due to acquired factors rather than congenital factors should be considered.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The metachronous contralateral onset of pediatric inguinal hernia is an issue with the open (or anterior) approach to unilateral inguinal hernia in children [1, 2]. Laparoscopic percutaneous extraperitoneal closure (LPEC) allows for the laparoscopic observation of the contralateral internal inguinal ring, so the contralateral onset of pediatric inguinal hernia caused by congenital patent processus vaginalis (PPV) can be prevented if contralateral PPV (cPPV) is not identified during surgery. However, it has recently become clear that a contralateral onset occurs even when LPEC is performed without cPPV [3, 4].
We also experienced five cases of a contralateral onset after LPEC at our hospital and present these cases along with their features. The purpose of this study was to examine the characteristics, including the incidence rate, and to identify risk factors for pediatric metachronous contralateral inguinal hernia (MCH) after LPEC.
Methods
Patients
The study population comprised 1277 patients (807 males and 470 females) who underwent LPEC for pediatric inguinal hernia in our hospital from 2005 to 2019. The procedure and technique for LPEC are based on those described previously by Takehara et al. [5]. In cases of unilateral inguinal hernia, the same skilled surgeon observed all cases for the presence or absence of contralateral PPV.
This study was approved by the ethics committee of Tokushima University Hospital (No. 3597) and the corresponding regulatory agencies, and all experiments were carried out in accordance with approved guidelines. All patients provided their written informed consent for surgery and study participation. All patients were followed until December 31, 2020.
Our recent strategy of contralateral PPV
Regarding our contralateral PPV treatment strategy, contralateral PPV was classified into Types 1 to 5 as shown in Fig. 1; Type 1 and Type 2 do not have PPV, Type 3 has moderate excavation with PPV, Type 4 has deep excavation with PPV, and Type 5 has a large hernia sac beyond the outer inguinal ring. Types 1 and 2 were followed up, and Types 3 to 5 received LPEC to prevent a contralateral onset.
Statistical analyses
Data on the age are presented as the mean ± standard deviation. Comparisons of several parameters between the two groups were performed by Student’s t-test and χ2 test. All statistical analyses were performed using a statistical software program (JMP 8.0.1.; SAS, Cary, NC, USA). A P value of less than 0.05 was considered statistically significant.
Results
Incidence rate for pediatric metachronous contralateral inguinal hernia after LPEC
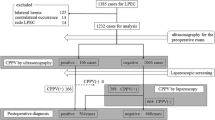
Among 1277 patients who underwent LPEC according to our treatment strategy, 374 of 1134 macroscopic unilateral cases were confirmed to have no contralateral PPV, while 5 experienced metachronous contralateral inguinal hernia (Fig. 2). In addition, the metachronous contralateral inguinal hernia incidence rate was 0.44% in macroscopic unilateral cases and 1.34% in cases without contralateral PPV (Fig. 2).
Case series of pediatric metachronous contralateral inguinal hernia after LPEC
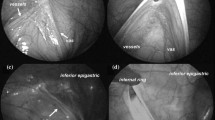
A case of metachronous contralateral inguinal hernia after LPEC is presented in Fig. 3. The contralateral inguinal ring classification at the time of the first unilateral LPEC procedure was type 1, and PPV was not observed, but metachronous contralateral inguinal hernia was identified, as shown in the right panel of Fig. 3.
Characteristics of pediatric metachronous contralateral inguinal hernia after LPEC
Table 1 shows a summary of the five cases of metachronous contralateral inguinal hernia after LPEC. In the MCH cases, male sex, age ≤ 1 year old, left side at the first operation, contralateral inguinal ring classification type 1 at the first LPEC, and the presence of a low birth weight were often observed.
Risk factors for pediatric metachronous contralateral inguinal hernia after LPEC
To identify the risk factors for metachronous contralateral inguinal hernia after LPEC, the 374 patients with type 1 or 2 initial contralateral PPV were divided into 2 groups: 5 patients with MCH and 369 patients without MCH. Values were then compared between these two groups (Fig. 2). There were no marked differences in the gender, age, initial side, or type of initial contralateral PPV; however, a significant difference was noted between the groups in the rate of low-birth-weight infants (Table 2).
The proportion of low-birth-weight infants was 6.1% (23/374 cases), and the incidence of a contralateral onset was 8.7% (2 cases) (Fig. 4a). Furthermore, the contralateral inguinal ring classification at the first time was Type 1 in 240 cases (64.2%) and Type 2 in 134 cases (35.8%), with the incidence of contralateral onset being 1.7% (4 cases) in Type 1 and 0.7% (1 case) in Type 2 (Fig. 4b).
Discussion
More than 20 years have passed since LPEC was first implemented for pediatric inguinal hernia [5], and in recent years LPEC has become the standard surgery for pediatric inguinal hernia [6,7,8]. Because unilateral LPEC allows for the observation of the contralateral side, both sides can be closed if PPV is found on the contralateral side as well. Therefore, it was initially considered that unilateral LPEC could prevent a contralateral onset, which had been a problem with the unilateral open method. However, although the number of contralateral cases is lower with unilateral LPEC than with the open method, contralateral cases do still occur with unilateral LPEC [3], and we have experienced a few ourselves.
The contralateral incidence rate was reported to be 0.3–0.8% at other institutions [3, 4] and was similar at our own institution (0.44%). The contralateral incidence in the unilateral open method was reported to be around 7.0% [1, 2], and although we observed a significant decrease in the contralateral incidence after unilateral LPEC, it did not reach 0% in reality [3, 4, 9].
Hayashi et al. reported that inadequate observation of the area around the contralateral internal inguinal ring during unilateral LPEC, i.e., failure to identify the contralateral PPV, is the cause of a contralateral onset after unilateral LPEC [10]. In addition, Tam et al. also reported that a careful search with grasping forceps around the contralateral inguinal ring is important to ensure that cPPV is not overlooked [11]. Watanabe et al. also reported that, in the contralateral observation during unilateral LPEC, despite the presence of cPPV, membranous structures, such as the peritoneal veil often covered the cPPV and apparently closed the hernia orifice. The action of this veil should prevent the onset of inguinal hernia [12]. This veil flattens the internal inguinal ring and blocks the abdominal contents from entering the hernia sac, and its existence might be unnoticed at the initial LPEC procedure. Changes in the shape of the peritoneal veil on the internal inguinal ring after the initial surgery can occur the patency of the hernia orifice and develop the indirect inguinal hernia and contralateral development may take several years after the initial surgery to complete, as it takes time for the shape of the veil to change. We have also experienced peritoneal veil on the contralateral internal inguinal ring during LPEC. Based on our experience, we believe that there is no bias in our LPEC procedure due to differences in observing surgeons, nor is there any PPV oversight, as the same skilled surgeon carefully and meticulously observes the area around the contralateral internal inguinal ring, including checking the peritoneal veil using forceps, and performs contralateral LPEC when there is a PPV of Type ≥ 3 according to our internal inguinal ring morphology classification.
However, even in the present cases, wherein contralateral PPV was completely ruled out, we experienced contralateral onset after unilateral LPEC. The contralateral internal inguinal ring, which was confirmed laparoscopically to be free of contralateral PPV at the time of the first unilateral LPEC, was found to have a new indirect inguinal hernia without PPV at the time of the second LPEC for contralateral onset. As mentioned above, this contralateral onset might have been de novo inguinal hernia, as overlooking of cPPV by the peritoneal veil was denied, and the congenitally closed processus vaginalis is unlikely to be reopened in an acquired fashion. Although pediatric inguinal hernia has been considered to be caused by congenital PPV, our surgical findings suggest the possibility of an acquired de novo inguinal hernia onset, which is very interesting from the viewpoint of the pathogenesis of pediatric inguinal hernia.
In our study, we examined the risk factors for the contralateral onset after unilateral LPEC, and a low birth weight was identified. Miyake et al. also examined such risk factors, but they were unable to identify any [13]. However, that report did not consider low-birth-weight infants as a risk factor for contralateral onset. Furthermore, in our study, over 8% of low-birth-weight infants developed MCH after unilateral LPEC, but the incidence of MCH after unilateral open method for low-birth-weight infants is likely to be higher than that after LPEC. In the literature, the contralateral incidence after performing the open method for low-birth-weight infants was reported to be 11–14.3% [14, 15]. Pini Prato et al. reported that the incidence of MCH after the open method was 28% if limited to low-birth-weight infants with a birth weight < 1500 g [16]. In addition, Marulaiah et al. reported that the contralateral incidence after performing the open method was 10.3% in low-birth-weight infants and 5.9% in normal-birth-weight infants, indicating that low-birth-weight infants tended to have a higher incidence of contralateral onset after open method than their normal-weight counterparts [17]. In summary, the incidence of contralateral incidence after open method for low-birth-weight infants reportedly ranges from 10 to 28%, which is still higher than our results concerning the incidence of contralateral onset after LPEC among low-birth-weight infants. These findings seem to support our results.
However, if MCH is de novo, the postoperative recurrence after LPEC is likely to be higher than with the open method. Furthermore, especially in low-birth-weight infants, postoperative recurrence is expected to be extremely high. In fact, there was not only a report that the postoperative recurrence rate after LPEC in normal-birth-weight infants was significantly higher (0.21%) than after the open method (0.04%) [18] but also a report that the postoperative recurrence rate after LPEC in low-birth-weight infants was 5.9% [19]. The postoperative recurrence rate after LPEC in low-birth-weight infants was considerably higher than that after both LPEC and the open method in normal-birth-weight infants. These findings seem to support our proposal.
Although the causal relationship between low-birth-weight infants and a de novo onset is not clear, it may involve the immaturity and fragility of the abdominal wall muscle tissue that is peculiar to low-birth-weight infants, in addition to the natural anatomical weakness of the internal inguinal ring [20]. However, if this is the case, the question arises as to whether the surgical technique for managing contralateral onset should be simply high ligation with LPEC or if reinforcement of the abdominal wall should be added. Since a low birth weight was found to be a risk factor in this retrospective study, we performed high ligation with LPEC, which is the standard surgical technique for pediatric indirect inguinal hernia, in the five cases of contralateral onset after unilateral LPEC that we experienced. No recurrence has been observed in any of the patients so far. However, we will continue to follow the patients in the future.
Although a low birth weight was the only predictor of MCH in this study, focusing on the age at the initial surgery, three out of five patients with MCH were under one year old and did not walk daily. Because “walking is the cause of inguinal hernia” is an anatomical theory based on chronic pressure on the inguinal region, the 374 patients without cPPV were divided into those < 1 year old who had not yet started walking (89 cases) and those ≥ 1 year old who were already walking (285 cases). The comparison between these two groups showed that the incidence of MCH was 3.4% (3 cases) in the group < 1 year old and 0.7% (2 cases) in the group ≥ 1 year old, and the incidence of MCH in the group < 1 year old tended to be higher than that in the group ≥ 1 year old (P = 0.09). In these cases, contralateral congenital PPV was denied at the time of the first unilateral LPEC when subjects were not yet walking, but contralateral onset occurred from the structurally vulnerable internal inguinal ring in patients who had started walking, except for in one case. As a result, morphological changes in the internal inguinal ring that did not depend on congenital PPV after starting walking, i.e., de novo inguinal hernia, were suspected.
In contrast, in our contralateral exploration, in addition to the above-mentioned prevention of cPPV overlooking, pressure was applied on and around the contralateral internal inguinal ring from both inside and outside the abdominal cavity during unilateral LPEC to evaluate the strength of the contralateral inguinal region. Specifically, from the inside of the abdominal cavity, the strength of the inguinal region in and around the area of the internal inguinal ring was evaluated using forceps. From outside of the abdominal cavity, the strength of the abdominal wall on the contralateral side was evaluated by not only applying pressure to the intra-abdominal cavity but also pulling the scrotum to confirm the morphological change around the internal inguinal ring. However, it was sometimes difficult to assess the strength of the abdominal wall in younger patients [13]. In fact, because three of the five patients who developed MCH in this study were < 1 year old, the evaluation of the abdominal wall may have been insufficient. Morphological changes in the internal inguinal ring independent of congenital PPV after or due to the start of walking and an insufficient assessment for the strength of abdominal wall in the inguinal region due to the relatively young age might have at least partially caused the frequent contralateral onset after unilateral LPEC when the age at the time of initial LPEC was less than one year old, but it was suggested that the contralateral onset after unilateral LPEC might be de novo inguinal hernia. However, our study involves a definite limitation aside from that mentioned above. Since the number of surgeries performed on low-birth-weight infants was small in our case series, we may not be able to conclude that there was a significant difference. In the future, it will be necessary to accumulate more cases of LPEC among low-birth-weight infants.
In the contralateral internal inguinal ring classification that we use, Type ≥ 3 (type 3 to 5) cases with PPV are subject to contralateral LPEC, while Type 2 cases with only mild excavation in the internal inguinal ring without PPV and Type 1 cases with a flat internal inguinal ring are not subjected to LPEC, instead receiving follow-up. We considered the possibility that intra-abdominal pressure might be applied to cases of mild excavation, resulting in contralateral onset (development of acquired de novo inguinal hernia). Indeed, at the beginning of the analysis, we expected that Type 2 cases with mild excavation would be more common among cases of contralateral onset. However, there was no significant difference in the contralateral internal inguinal ring morphological classification, and Type 1 cases with a flat internal inguinal ring were actually more common. For Type 1 cases with a flat contralateral internal inguinal ring, there is no way to prevent contralateral onset, but for Type 2 cases with mild excavation, we are currently conducting a trial to flatten the mild excavation by cauterizing the peritoneum at the bottom of the excavation of the internal inguinal ring with electrocautery, taking care not to damage the vas deferens or testicular vessels, and thereby prevent the contralateral onset after unilateral LPEC. Trials have been started since 2020, and we are currently accumulating cases as well as conducting follow-up studies.
In conclusion, the risk factor of contralateral onset after LPEC for pediatric inguinal hernia was low-birth-weight infants. Therefore, for the above-mentioned unilateral LPEC cases, the possibility of a contralateral onset after LPEC due to acquired factors rather than congenital factors should be considered.
Abbreviations
- cPPV:
-
Contralateral patent processus vaginalis
- LPEC:
-
Laparoscopic percutaneous extraperitoneal closure
- MCH:
-
Metachronous contralateral inguinal hernia
- PPV:
-
Patent processus vaginalis
- SD:
-
Standard deviation
References
Ron O, Eaton S, Pierro A. Systematic review of the risk of developing a metachronous contralateral inguinal hernia in children. Br J Surg. 2007;94(7):804–11.
Staerkle RF, Guglielmetti LC, Bielicki IN, Gaukel S, Frongia G, Hilton S, et al. Is it worth to explore the contralateral side in unilateral childhood inguinal hernia?: A PRISMA-compliant meta-analysis. Medicine (Baltimore). 2020;99(31): e21501.
Fujiogi M, Michihata N, Matsui H, Fushimi K, Yasunaga H, Fujishiro J. Outcomes following laparoscopic versus open surgery for pediatric inguinal hernia repair: analysis using a national inpatient database in Japan. J Pediatr Surg. 2019;54(3):577–81.
Miyake H, Fukumoto K, Yamoto M, Nouso H, Kaneshiro M, Nakajima H, et al. Comparison of percutaneous extraperitoneal closure (LPEC) and open repair for pediatric inguinal hernia: experience of a single institution with over 1000 cases. Surg Endosc. 2016;30(4):1466–72.
Takehara H, Yakabe S, Kameoka K. Laparoscopic percutaneous extraperitoneal closure for inguinal hernia in children: clinical outcome of 972 repairs done in 3 pediatric surgical institutions. J Pediatr Surg. 2006;41(12):1999–2003.
Shalaby R, Ismail M, Samaha A, Yehya A, Ibrahem R, Gouda S, et al. Laparoscopic inguinal hernia repair; experience with 874 children. J Pediatr Surg. 2014;49(3):460–4.
McClain L, Streck C, Lesher A, Cina R, Hebra A. Laparoscopic needle-assisted inguinal hernia repair in 495 children. Surg Endosc Other Interv Tech. 2015;29(4):781–6.
Thomas DT, Göcmen KB, Tulgar S, Boga I. Percutaneous internal ring suturing is a safe and effective method for the minimal invasive treatment of pediatric inguinal hernia: experience with 250 cases. J Pediatr Surg. 2016;51(8):1330–5.
Tam YH, Wong YS, Pang KK, Tsui SY, Mou JW, Houben CH, et al. Unexpected metachronous hernia development in children following laparoscopic unilateral hernia repair with negative evaluation for contralateral patent processus vaginalis. J Laparoendosc Adv Surg Tech A. 2013;23(3):287–90.
Hayashi K, Ishimaru T, Kawashima H. Reoperation after laparoscopic inguinal hernia repair in children: a retrospective review. J Laparoendosc Adv Surg Tech A. 2019;29(10):1264–70.
Tam YH, Wong YS, Chan KW, Pang KK, Tsui SY, Mou JW, et al. Simple maneuvers to reduce the incidence of false-negative findings for contralateral patent processus vaginalis during laparoscopic hernia repair in children: a comparative study between 2 cohorts. J Pediatr Surg. 2013;48(4):826–9.
Watanabe T, Nakano M, Endo M. An investigation on the mechanism of contralateral manifestations after unilateral herniorrhaphy in children based on laparoscopic evaluation. J Pediatr Surg. 2008;43(8):1543–7.
Miyake H, Fukumoto K, Yamoto M, Nakajima H, Sekioka A, Yamada Y, et al. Risk factors for recurrence and contralateral inguinal hernia after laparoscopic percutaneous extraperitoneal closure for pediatric inguinal hernia. J Pediatr Surg. 2017;52(2):317–21.
Maillet OP, Garnier S, Dadure C, Bringuier S, Podevin G, Arnaud A, et al. Inguinal hernia in premature boys: should we systematically explore the contralateral side? J Pediatr Surg. 2014;49(9):1419–23.
Steven M, Greene O, Nelson A, Brindley N. Contralateral inguinal exploration in premature neonates: is it necessary? Pediatr Surg Int. 2010;26(7):703–6.
Pini Prato A, Rossi V, Mosconi M, Disma N, Mameli L, Montobbio G, et al. Inguinal hernia in neonates and ex-preterm: complications, timing and need for routine contralateral exploration. Pediatr Surg Int. 2015;31(2):131–6.
Marulaiah M, Atkinson J, Kukkady A, Brown S, Samarakkody U. Is contralateral exploration necessary in preterm infants with unilateral inguinal hernia? J Pediatr Surg. 2006;41(12):2004–7.
Shibuya S, Imaizumi T, Yamada S, Yoshida S, Yamada S, Toba Y, et al. Comparison of surgical outcomes between laparoscopic percutaneous extracorporeal closure (LPEC) and open repair for pediatric inguinal hernia by propensity score methods and log-rank test analysis. Surg Endosc. 2022;36(2):941–50.
Shibuya S, Miyazaki E, Miyano G, Imaizumi T, Mikami T, Ochi T, et al. Comparison of laparoscopic percutaneous extraperitoneal closure versus conventional herniotomy in extremely low birth weight infants. Pediatr Surg Int. 2019;35(1):145–50.
Paran TS. Puri P : Inguinal hernia. In: Puri P, editor. Newborn Surgery. 3rd ed. London: Hodder Arnold; 2011. p. 599–605.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The protocol for the research project was approved by the Ethics Committee of the institution (No. 3597), and it conforms to the provisions of the Declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mori, H., Ishibashi, H., Yokota, N. et al. Risk factors for metachronous contralateral inguinal hernia after laparoscopic percutaneous extraperitoneal closure for unilateral inguinal hernia in children. Surg Today 52, 1491–1496 (2022). https://doi.org/10.1007/s00595-022-02480-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-022-02480-0