Abstract
Purpose
Two-dimensional (2D) analyses of intervertebral disc (IVD) height and foramen measurements following lateral lumbar interbody fusion (LLIF) have been reported. However, three-dimensional (3D) morphometric analysis of intervertebral structure using 3D computed tomography (3D CT) provides increased precision for measuring morphological changes. The purpose of this study was to evaluate 3D changes of lumbar IVD height and foramen diameter in degenerative lumbar disease patients following LLIF.
Methods
Subject-based 3D CT lumbar models were created for 26 patients before and following LLIF. IVD height (whole and five anatomical zones) and foramen diameter (minimum and maximum) were measured based on the model using custom software. The sagittal placement of cages (SPC) and cross-sectional area of the thecal sac (CSA) were measured. Changes in these parameters by LLIF were quantified and statistically analysed.
Results
Following LLIF, disc height increased by an average of 2.9 mm (P < 0.01). Post-operative measurements of both minimum and maximum diameters of the foramen were significantly increased by 1.0 mm and 1.9 mm, respectively (P < 0.01). Change in maximum foramen diameter was significantly correlated with change in disc height (P < 0.05). The SPC was significantly correlated with the changes in disc height and foraminal diameters (P < 0.05, respectively). No significant change between the change in disc height and CSA was found.
Conclusion
This preliminary study quantifies disc height and foramen diameter changes in 3D following LLIF. The presented data provide baseline intervertebral changes for future comparisons with follow-up studies and clinical outcomes.
Level of evidence I
Diagnostic: individual cross-sectional studies with the consistently applied reference standard and blinding.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Lumbar diseases associated with intervertebral disc (IVD) degeneration, such as lumbar canal stenosis, lumbar spondylolisthesis and degenerative scoliosis, can be successfully treated with lumbar interbody fusion surgeries [1].
Lateral lumbar interbody fusion (LLIF) is a minimally invasive spinal fusion surgical approach for lumbar interbody fusion for degenerative lumbar diseases. Traditionally, posterior lumbar interbody fusion (PLIF) or transforaminal lumbar interbody fusion (TLIF) is performed. The goal of LLIF is to provide minimally invasive lumbar fusion while increasing IVD height and foramen size to improve canal and foraminal stenosis by indirect decompression [2, 3]. Two-dimensional (2D) analyses of increases in disc height [2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17] and foraminal height [2,3,4, 7, 9, 10, 12,13,14,15] from radiographs and/or computed tomography (CT) scans following LLIF have also been reported. However, it is known that the quantification of the vertebral dimension differs depending on the three-dimensional (3D) nature of the area measured [18, 19]. Therefore, 3D quantification of LLIF surgical outcomes would be ideal and superior to 2D analyses by CT or radiographs. To date, 3D analyses of resulting disc height and foramen size following LLIF have not been reported.
The purposes of this preliminary study were to evaluate (1) morphometric changes of intervertebral structures, including IVD height and lumbar foramen diameter, following LLIF surgery using 3D CT analysis, and (2) the correlation between changes of these 3D intervertebral structures and LLIF cage placement and indirect thecal sac decompression.
Methods
Patients
Institutional review board approval was obtained for this study. CT scans were obtained from 26 consecutive patients (13 males, 13 females; age range 52–82 years, average age: 67.2 ± 8.9 years old) who had undergone extreme lateral interbody fusion (XLIF®, San Diego, CA, USA) surgery from August 2014 to November 2015 and were the subjects of this study (summarized in Table 1). The surgeries were performed for patients with degenerative lumbar spondylolisthesis (n = 17), degenerative disc diseases (n = 4), degenerative lumbar scoliosis (n = 3) and lumbar canal stenosis with segmental instability (n = 2). Among 26 patients, 12 patients received single-level fusions, 12 patients received two-level fusions, and two patients received three-level fusions. All patients received the LLIF surgery by a left-side approach. A total of 42 IVDs and 84 foramina were analysed.
The sizes of XLIF cages implanted were determined by the surgeon during the procedure, with cage heights ranging from 8 to 14 mm and cage length at 50 mm, 55 mm and 60 mm (summarized in Table 1). The width of all cages used was 18 mm, and the lordosis of all cages used was 10°. The number of cages of each height was as follow: 8 mm (n = 6), 9 mm (n = 13), 10 mm (n = 14), 11 mm (n = 6), 12 mm (n = 2) and 14 mm (n = 1).
Creation of a 3D–CT reconstruction model
Patients were CT-scanned pre-operatively and post-operatively within seven days of receiving LLIF surgery in a SOMATOM Definition Flash CT scanner (Siemens, Munich, Germany) with a 1.0 mm slice thickness and scan resolutions ranging from 260–310 μm. DICOM files from the CT scans were then imported into a commercially available 3D reconstruction software package (Mimics, Materialise Inc., Leuven, Belgium) for 3D reconstruction of the vertebrae. A threshold value was determined for each scan to visualize the cortical bone and was applied to segment each operated vertebra [20, 21] (Fig. 1). Segmentation of the CT data was then converted to point cloud data sets of each vertebra from which disc height distribution (DHD) and foramen dimension measurements could be made (Fig. 1). The average date from the surgery to the post-operative CT was 5.7 ± 0.9 (day 4–day 7).
Measurement of 3D disc height
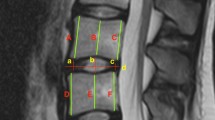
Disc height distribution was calculated using 3D geometric point cloud data of superior and inferior endplates adjacent to the IVDs selected from the 3D CT model, as previously reported [18, 22]. Briefly, each endplate (superior and inferior) was individually selected and isolated from the 3D model of each vertebra; then, a point cloud model of targeted IVD spaces was created using Mimics (Fig. 1).
For measuring the 3D disc height, the least distances from one point in the point cloud model of the inferior endplate to the points in the superior endplate were calculated using a custom-written Visual C + + program [18, 22]. The average of the least distance was calculated by repeating this procedure for all points in the inferior endplate, and then, the averaged distance was defined as the 3D disc height of the whole disc.
Additionally, the IVD was separated into five anatomical areas (posterior, left-lateral [L-lateral], right-lateral [R-lateral], anterior, nucleus pulposus [NP]) that represent the annulus fibrosus and nucleus pulposus footprints ideally (Fig. 2). Then, the 3D disc height of each area was measured as described above.
3D foramen diameter analysis
Foramen diameters (maximum and minimum diameters) were measured for each IVD level before and following surgery. Segmented 3D CT models of the vertebrae adjacent to a targeted IVD were exported as point cloud data files using Mimics, allowing for foramen dimension measurement using a custom-written C + + program as previously reported [19]. In short, foramen dimensions were measured by a virtual cone rotating about an origin within the foramen area to select the boundary points of the foramen in the cross section. The location of the origin was defined as the centre of the pedicle bridge in the axial view, the centre of the pedicle in the coronal view and the centre of the foramen in the sagittal view. The minimum and maximum lengths of the foramen are measured from the boundary points (Fig. 3).
Sagittal placement of LLIF cage
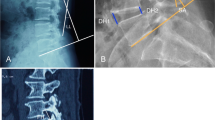
A sagittal CT–multiplanar reconstruction (MPR) was performed using post-operative CT images. According to the method as previously reported [23], the sagittal placement of the cages at superior and inferior endplates was differentially determined. The superior endplate measurement is distance A and the inferior endplate measurement is distance A'. The distance from the anterior edge of the vertebra to the centre of the cage on the superior endplate is B and the distance on the inferior endplate is B' as shown in Fig. 4. All measurements are taken using a clinical image processing software (EV Insite, PSP Corporation, Tokyo, Japan). Sagittal placement of the cages at the superior endplate was calculated as follows: B/A × 100 (%) and those at the inferior endplate as follows: B’/A’ × 100 (%).
Sagittal placement of the lateral lumbar interbody fusion (LLIF) cage. The sagittal placement of the cage (SPC) at the superior and inferior endplates was differentially determined by measuring the anteroposterior distance of the endplate (A or A’) and the distance from the anterior edge of the vertebra to the centre of the cage (B or B’) [23]. The SPC was calculated as follows: B/A (or B’/A’) × 100 (%)
Measurement of the cross-sectional area of the thecal sac (CSA)
T2-weighted images from the axial MRI were used for the analysis of CSA as previously reported [24]. Among 26 subjects, both pre- and post-operative MRI data were obtained from 20 subjects and processed for the CSA analysis. Briefly, the CSA (mm2) was measured by tracing the outline of the thecal sac at the mid-axial slice of corresponding discs using the clinical image processing software (EV Insite) (Fig. 5). The change in CSA was calculated as follows: post-operative CSA—pre-operative CSA (mm2). The average date from the surgery to the post-operative MRI was 8.0 ± 4.7 (day 4–day 21).
Cross-sectional area of the thecal sac (CSA). Pre-operative and post-operative T2-weighted images from the axial magnetic resonance imaging (MRI) closest to the date of surgery were used for the analysis of the CSA [26]. The CSA (mm2) was measured by tracing the outline of the thecal sac (pink-coloured dotted line) at the mid-axial slice of the corresponding discs. The representative pre-operative (A: Pre-OP) and post-operative (B: Post-OP) measurements of CSA are indicated
Statistical analysis
The paired Student's t-test was used for two-group comparisons between pre- and post-operation data of 3D disc height and 3D foraminal diameter. One-way analysis of variance (ANOVA) was used for the area comparisons of 3D disc height with Bonferroni post hoc tests (p < 0.05). Pearson's product–moment correlation coefficient was used to test for correlations between measurements. The data are expressed as mean ± standard deviation (SD). A P-value of less than 0.05 was regarded as statistically significant.
Results
3D disc height
Pre- and post-operative measurements of 3D disc height are summarized in Table 2. The averaged whole 3D disc height was significantly increased following LLIF surgery by an average of 2.9 ± 1.2 mm (P < 0.01). The percent change ([Post-OP data–Pre-OP data]/Pre-OP data] × 100) was 65.8 ± 9.2%.
Pre-operative data of 3D disc height evaluated for each disc area showed that the anterior area was highest followed by the NP area, L-lateral area, R-lateral area and posterior area. Following LLIF surgery, the 3D disc height of all disc areas was significantly increased compared to that before surgery (P < 0.01 vs. Pre-op). However, there were no significant differences in the changes (Post-op data–Pre-op data) in 3D disc height among the five different disc areas. When the changes in 3D disc height were evaluated by the percentage of increase, no significant differences were also found among the five different areas.
3D disc height analysis with different cage heights
The change of 3D disc height with cage heights (from 8 to 11 mm) documented in Table 1 was separately evaluated and statistically analysed. Pre-operative 3D disc height showed no significant trend to be higher with the height of the cage inserted into the fusion levels (Fig. 6a). After insertion of cages, 3D disc height showed a trend to be higher by cage height (Fig. 6a). A post-hoc test showed that the post-operative 3D disc height of 11 mm cages was significantly higher than that of 8 mm (P < 0.01) and 9 mm (P < 0.05) cages. The post-operative 3D disc height of 10 mm cages was significantly higher than that of 8 mm cages (P < 0.05) (Fig. 6a).
However, no significant differences were identified on changes of 3D disc height among the four different cage heights (8 mm: 2.5 ± 0.9 [mm]; 9 mm: 3.1 ± 0.4; 10 mm: 3.0 ± 0.3; 11 mm: 2.9 ± 0.4, P = 0.47, one-way ANOVA, Fig. 6b). When the data were separately analysed by disc area, there were also no significant changes in 3D disc height among the five areas with the four different cage heights.
Foramen diameter measurement
Pre- and post-operative measurements of foramen diameter are summarized in Table 3. Post-operative measurements of both minimum and maximum diameters of the foramen were significantly higher than pre-operative measurements (P < 0.01, respectively). On average, the minimum diameter and maximum diameter significantly increased by 1.0 mm and 1.9 mm, respectively. The change of maximum diameter was significantly greater than that of minimum diameter (P < 0.01). On the other hand, the percent change of the minimum diameter was greater (19.8%) than that of the maximum diameter (13.3%); however, no significant difference was found between the minimum and maximum diameter (P = 0.1). The Pearson’s correlation test showed that there was a weak correlation between the changes in minimum and maximum foramen diameters (r [correlation coefficient] = 0.27, P < 0.01).
Correlation between changes in 3D disc height and 3D foramen diameter
Pearson’s correlation test showed that there was no significant correlation between changes in minimum foramen diameters and whole 3D disc height (P = 0.48). When the data were analysed in each disc area, a weak but significant correlation was identified between changes in minimum foramen diameters and the change in 3D disc height at the posterior area (r = 0.28, P < 0.01).
Change in maximum foramen diameter following LLIF surgery was significantly, but weakly correlated with change in whole 3D disc height (r = 0.24, P < 0.05). When the data were analysed in each disc area, a weak correlation was identified with change in 3D disc height at the posterior (r = 0.38, P < 0.0001), L-lateral (r = 0.24, P < 0.05) and R-lateral areas (r = 0.28, P < 0.01).
Correlations between sagittal placement of cage and changes in 3D disc height and foramen diameter
The averaged sagittal placement of cage (SPC) at the superior endplate was 50.8 ± 10.1% and at the inferior endplate was 48.4 ± 10.0%.
There was no significant correlation between the SPC and changes in whole 3D disc height at both the superior and inferior endplates (Fig. 7a). However, when the data were analysed in each disc area, a significant but weak correlation was identified in the posterior area at both superior (r = 0.35, P < 0.05) and inferior (r = 0.42, P < 0.01) endplates (Fig. 7b) and in the right-lateral area at the inferior endplate (r = 0.33, P < 0.05) (Fig. 7e).
Correlation between sagittal placement of cages and change in three-dimensional disc height in all disc areas. The whole intervertebral disc (whole disc: a) was separated into five anatomical areas: posterior (b), left-lateral (L-lateral: c), anterior (d), right-lateral (R-lateral: e), nucleus pulposus (NP: f). Superior: superior endplate; inferior: inferior endplate; r: correlation efficient. *P < 0.05, **P < 0.01
There was a significant but weak correlation between the SPC at the superior endplate and the changes in both minimum (r = 0.23, P < 0.05) and maximum (r = 0.30, P < 0.01) diameter. On the other hand, the SPC at the inferior endplate was only correlated with the changes in maximum foramen diameter (r = 0.35, P < 0.01).
Correlation between change in 3D disc height and cross-sectional area of the thecal sac (CSA)
The averaged CSA increased from 75.8 ± 33.5 mm2 pre-operatively to 100.2 ± 34.9 mm2 post-operatively (P < 0.001). The averaged change in CSA was 21.6 ± 21.2 mm2, and the averaged % change was 36.1 ± 41.4%. No significant correlation was found between the change in whole 3D disc height and the CSA (r = 0.21, P = 0.24). There were also significant changes between the change of disc height in each zonal disc area and the CSA.
Discussion
Lateral lumbar interbody fusion, including XLIF, has been successfully used to treat degenerative lumbar diseases such as stenosis, degenerative spondylolisthesis and scoliosis [1]. It has been reported that LLIF provides safe and successful clinical outcomes and radiographic improvement, including the restoration of disc height and foraminal height. However, because the clinical images and measurement methods were not consistent, the radiographic changes following LLIF varied depending on the study.
Among the previous papers that quantified 2D radiographic disc height changes following XLIF (LLIF) surgery, 16 manuscripts that presented numerical data are summarized in Table 4. In most previous studies, disc height was measured on lateral plain radiographs using image analysis software commonly used in clinical practice. Although the indicator of disc height varied depending on the study, the average of anterior and posterior disc height has been most frequently used. The averaged change of disc height following XLIF was reported to range from 2.7 to 6.1 mm.
Previous studies showed that 3D measurement of disc height is independent of the 3D orientation of the vertebra and provides disc height distribution throughout the entire surface of the individual endplate using in vivo clinical CT images [18, 22, 24]. We utilized this 3D CT method in this study to measure 3D changes in disc height following LLIF surgery. The data from 3D CT measurements in our study showed that disc height changes were 2.9 mm. Our data were close to those of Malham’s [14] study in which disc height was measured by sagittal CT between the posterior borders of vertebrae.
Among the previous papers that quantified radiographic changes of foraminal height following XLIF (LLIF) surgery, ten manuscripts that presented numerical data are summarized in Table 5. The distance from the "inferior pedicle wall to the superior pedicle wall of the level below" was defined and measured as the highest foraminal height in many of the studies. A lateral plain radiograph was used in most studies for the measurement of foraminal height. A wide range of the averaged changes in foraminal height following XLIF was reported, ranging from 1.97 to 9.1 mm.
In 2014, Senoo et al. measured foraminal height and width in vivo using 3D CT models of asymptomatic subjects to analyse the complex 3D structure of the lumbar foramen [19]. Furthermore, to validate the accuracy of 3D CT-based measurement, data using 3D CT of cadaveric lumbar spines were compared to those of direct measurement [19]. They concluded that a strong correlation was identified between both measurements, suggesting that this 3D CT model accurately reflects the 3D morphometry of the true lumbar foramen of the subjects. Our study showed that the change in 3D measured maximum foraminal diameter was 1.9 mm. This result was close to the data from Tessitore’s study [9] in which the change of foraminal height was measured using plain radiographs.
Interestingly, the results of our study showed that no significant changes in 3D disc height were identified among the different heights of LLIF cages that ranged from 8 to 11 mm. The surgeon decided the appropriate size of the cage by inserting several sizes of trial cages during surgery; hence, the insertion of the appropriate size of the cage would produce a constant increase of disc height independent of cage height.
In this 3D measurement of foraminal diameter, the minimal foraminal diameter reflects foraminal width and the maximum foraminal diameter reflects foraminal height. Therefore, increasing disc height was significantly associated with foraminal height, but not foraminal width. On the other hand, the minimum foraminal diameter was only associated with disc height changes in the posterior area. This suggests that the disc height increase in the posterior area is essential for increasing both the foraminal height and width that result in the effective decompression of the spinal nerve (root) within the foramen.
Park et al. evaluated the effect of cage position (anterior and middle groups) on the changes of disc height and foraminal area using sagittal CT or MRI following LLIF surgery; they reported that cage positioning had a significant effect on change in anterior disc height, but not posterior disc height and foraminal area [25]. Different from the results of Park’s study [25], the results of our 3D CT analysis revealed that sagittal placement of the LLIF cage at both the superior and inferior endplates was significantly, but weakly, correlated with changes in 3D disc height at the posterior disc area and maximum foraminal diameter. The disparity in results could be attributed to differences in radiographic analyses between 2 and 3D and the definition of cage position.
In this study, the SPC was also significantly correlated with change in disc height in the R-lateral area, but not in the L-lateral area. The pre-operative disc height in the R-lateral area was less than that in the L-lateral area, and the change in disc height in the R-lateral area was greater than that in the L-lateral area following LLIF cage insertion (Table 2). The asymmetry in pre-operative disc height, but not the effect of the left-side approach for LLIF, would contribute to the bilateral differences in the change in disc height in sagittal cage placement. In the case of the anterior translation of a lumbar vertebra, the SPC at the superior endplate is larger than that at the inferior endplate (Fig. 4). This suggests that the extent of anterior translation is more accurately associated with the SPC at the superior endplate than that at the inferior endplate. The authors speculated that these differences in the relative relation of cage placement in superior and inferior endplates would contribute to subtle differences in the correlation coefficient in the changes in disc height and foramen diameter following LLIF surgery.
We next evaluated whether changes in 3D disc height have a significant effect on the indirect decompression of the thecal sac following LLIF surgery by measuring the post-operative change of CSA. Our results showed that the change in 3D disc height had no significant correlation with the change in CSA. Ebata et al. [23] recently reported that the change in CSA following LLIF surgery showed a significant correlation with change in 2D disc height; however, the correlation coefficient (r = 0.2) was very low. They concluded that a decrease in flavum buckling is important for obtaining indirect thecal sac decompression.
There were some limitations to this study. Radiological changes in 3D disc height and foraminal diameter were evaluated by comparing the pre-operative CT images and those immediately following surgery. Future studies to compare CT data from long-term follow-up, including the extent of cage subsidence, will be needed to evaluate the clinical outcomes of the 3D CT analysis.
We have conducted a 3D morphometric analysis of the lumbar disc space and foramen using a 3D CT model in degenerative lumbar disease patients who underwent LLIF surgery. The presented data provide (1) the clinical reference for change in disc height and foraminal height by LLIF cage insertion, (2) the clinical evidence that the sagittal placement of a LLIF cage is associated with change in disc height and foraminal diameter, and (3) the foundation for future studies for comparison with long-term follow-up data including the extent of cage subsidence and clinical outcomes.
References
Kwon B, Kim DH (2016) Lateral lumbar interbody fusion: indications, outcomes, and complications. J Am Acad Orthop Surg 24:96–105. https://doi.org/10.5435/JAAOS-D-14-00208
Alimi M, Hofstetter CP, Cong GT, Tsiouris AJ, James AR, Paulo D, Elowitz E, Hartl R (2014) Radiological and clinical outcomes following extreme lateral interbody fusion. J Neurosurg Spine 20:623–635. https://doi.org/10.3171/2014.1.SPINE13569
Oliveira L, Marchi L, Coutinho E, Pimenta L (2010) A radiographic assessment of the ability of the extreme lateral interbody fusion procedure to indirectly decompress the neural elements. Spine (Phila Pa 1976) 35:S331-337. https://doi.org/10.1097/BRS.0b013e3182022db0
Jeremy Goh KM, Liow MHL, Xu S, Yeo W, Ling ZM, Soh CCR, Tan SB, Guo CM (2019) Reduction in foraminal height after lateral access surgery does not affect quality of life: a 2-year outcome study on lateral lumbar interbody fusion. J Orthop Surg (Hong Kong) 27:2309499019829336. https://doi.org/10.1177/2309499019829336
Louie PK, Varthi AG, Narain AS, Lei V, Bohl DD, Shifflett GD, Phillips FM (2018) Stand-alone lateral lumbar interbody fusion for the treatment of symptomatic adjacent segment degeneration following previous lumbar fusion. Spine J 18:2025–2032. https://doi.org/10.1016/j.spinee.2018.04.008
Kono Y, Gen H, Sakuma Y, Koshika Y (2018) Comparison of clinical and radiologic results of mini-open transforaminal lumbar interbody fusion and extreme lateral interbody fusion indirect decompression for degenerative lumbar spondylolisthesis. Asian Spine J 12:356–364. https://doi.org/10.4184/asj.2018.12.2.356
Alimi M, Lang G, Navarro-Ramirez R, Perrech M, Berlin C, Hofstetter CP, Moriguchi Y, Elowitz E, Hartl R (2018) The impact of cage dimensions, positioning, and side of approach in extreme lateral interbody fusion. Clin Spine Surg 31:E42–E49. https://doi.org/10.1097/BSD.0000000000000507
Yen CP, Beckman JM, Vivas AC, Bach K, Uribe JS (2017) Effects of intradiscal vacuum phenomenon on surgical outcome of lateral interbody fusion for degenerative lumbar disease. J Neurosurg Spine 26:419–425. https://doi.org/10.3171/2016.8.SPINE16421
Tessitore E, Molliqaj G, Schaller K, Gautschi OP (2017) Extreme lateral interbody fusion (XLIF): a single-center clinical and radiological follow-up study of 20 patients. J Clin Neurosci 36:76–79. https://doi.org/10.1016/j.jocn.2016.10.001
Isaacs RE, Sembrano JN, Tohmeh AG, Group SDS (2016) Two-year comparative outcomes of MIS lateral and MIS transforaminal interbody fusion in the treatment of degenerative spondylolisthesis: Part II: radiographic findings. Spine (Phila Pa 1976) 8:S133-144. https://doi.org/10.1097/BRS.0000000000001472
Sembrano JN, Yson SC, Horazdovsky RD, Santos ER, Polly DW Jr (2015) Radiographic comparison of lateral lumbar interbody fusion versus traditional fusion approaches: analysis of sagittal contour change. Int J Spine Surg 9:16. https://doi.org/10.14444/2016
Alimi M, Hofstetter CP, Tsiouris AJ, Elowitz E, Hartl R (2015) Extreme lateral interbody fusion for unilateral symptomatic vertical foraminal stenosis. Eur Spine J 24(Suppl 3):346–352. https://doi.org/10.1007/s00586-015-3940-z
Tohmeh AG, Khorsand D, Watson B, Zielinski X (2014) Radiographical and clinical evaluation of extreme lateral interbody fusion: effects of cage size and instrumentation type with a minimum of 1-year follow-up. Spine (Phila Pa 1976) 39:E1582-1591. https://doi.org/10.1097/BRS.0000000000000645
Malham GM, Parker RM, Goss B, Blecher CM, Ballok ZE (2014) Indirect foraminal decompression is independent of metabolically active facet arthropathy in extreme lateral interbody fusion. Spine (Phila Pa 1976) 39:E1303-1310. https://doi.org/10.1097/BRS.0000000000000551
Caputo AM, Michael KW, Chapman TM, Jennings JM, Hubbard EW, Isaacs RE, Brown CR (2013) Extreme lateral interbody fusion for the treatment of adult degenerative scoliosis. J Clin Neurosci 20:1558–1563. https://doi.org/10.1016/j.jocn.2012.12.024
Le TV, Vivas AC, Dakwar E, Baaj AA, Uribe JS (2012) The effect of the retroperitoneal transpsoas minimally invasive lateral interbody fusion on segmental and regional lumbar lordosis. ScientificWorldJournal 2012:516706. https://doi.org/10.1100/2012/516706
Kepler CK, Sharma AK, Huang RC, Meredith DS, Girardi FP, Cammisa FP Jr, Sama AA (2012) Indirect foraminal decompression after lateral transpsoas interbody fusion. J Neurosurg Spine 16:329–333. https://doi.org/10.3171/2012.1.SPINE11528
Espinoza Orias AA, Mammoser NM, Triano JJ, An HS, Andersson GB, Inoue N (2016) Effects of axial torsion on disc height distribution: an in vivo study. J Manipulative Physiol Ther 39:294–303. https://doi.org/10.1016/j.jmpt.2016.03.002
Senoo I, Espinoza Orias AA, An HS, Andersson GB, Park DK, Triano JJ, Inoue N (2014) In vivo 3-dimensional morphometric analysis of the lumbar foramen in healthy subjects. Spine (Phila Pa 1976) 39:E929-935. https://doi.org/10.1097/BRS.0000000000000399
Ochia RS, Inoue N, Renner SM, Lorenz EP, Lim TH, Andersson GB, An HS (2006) Three-dimensional in vivo measurement of lumbar spine segmental motion. Spine (Phila Pa 1976) 31:2073–2078. https://doi.org/10.1097/01.brs.0000231435.55842.9e
Ochia RS, Inoue N, Takatori R, Andersson GB, An HS (2007) In vivo measurements of lumbar segmental motion during axial rotation in asymptomatic and chronic low back pain male subjects. Spine (Phila Pa 1976) 32:1394–1399. https://doi.org/10.1097/BRS.0b013e318060122b
Watanabe S, Inoue N, Yamaguchi T, Hirano Y, Espinoza Orias AA, Nishida S, Hirose Y, Mizuno J (2012) Three-dimensional kinematic analysis of the cervical spine after anterior cervical decompression and fusion at an adjacent level: a preliminary report. Eur Spine J 21:946–955. https://doi.org/10.1007/s00586-011-2090-1
Ebata S, Ohba T, Haro H (2018) Adequate cage placement for a satisfactory outcome after lumbar lateral interbody fusion with MRI and CT analysis. Spine Surg Relat Res 2:53–59. https://doi.org/10.22603/ssrr.2017-0037
Crane BP, An HS, Ochia RS, Cornin S, Chen K, Andersson GB, Inoue N (2006) In vivo measurement of changes in lumbar intervertebral disc height distribution during torsion. In: 52nd Annual Meeting of the Orthopedic Research Society. Chicago. p. 1217
Park SJ, Lee CS, Chung SS, Kang SS, Park HJ, Kim SH (2017) The Ideal cage position for achieving both indirect neural decompression and segmental angle restoration in lateral lumbar interbody fusion (LLIF). Clin Spine Surg 30:E784–E790. https://doi.org/10.1097/BSD.0000000000000406
Fujibayashi S, Hynes RA, Otsuki B, Kimura H, Takemoto M, Matsuda S (2015) Effect of indirect neural decompression through oblique lateral interbody fusion for degenerative lumbar disease. Spine (Phila Pa 1976) 40:E175-182. https://doi.org/10.1097/BRS.0000000000000703
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Availability of data and material
The data are available via reasonable request from the corresponding author.
Ethics approval
Ethics were approved by the institutional review boards of Mie University Hospital (IRB reference number: 3110).
Informed consent
Informed consent was obtained in the form of opt-out on the website.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akeda, K., Cheng, K., Abarado, E. et al. Three-dimensional computed tomographic evaluation of lateral lumbar interbody fusion: morphometric change of intervertebral structure. Eur Spine J 30, 1355–1364 (2021). https://doi.org/10.1007/s00586-021-06776-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-021-06776-6