Abstract
The human spine is influenced by mechanical loads. To our knowledge, this is the first study to assess the effect of long-term axial unloading on morphology of healthy vertebras in adults. The objective of this study is to quantify the effects of long-term relative axial unloading on thoracolumbar vertebral body height in adults. In this study, 218 vertebras on 200 plain lateral radiograms of patients with thoracolumbar vertebral body fracture, which underwent long segment instrumentation and fusion and had a relative axial unloading on five vertebrae of thoracolumbar spine were evaluated. Anterior vertebral body height (AVBH) and posterior vertebral body height (PVBH) proximal and distal to the fractured vertebrae were measured before and at least 1 year after the unloading operative procedures. AVBH of the first distal adjacent vertebrae and summative AVBHs of the first distal and proximal adjacent vertebras to the fractured vertebrae were significantly increased after 1 year of unloading, whereas the PVBH changes were not noticeable and the mean of cumulative height of two levels of proximal and distal adjacent vertebras to the fractured vertebrae did not show significant difference. Vertebral body height of lumbar was more influenced by unloading when compared with thoracic spine. Long-term relative axial unloading can affect the height of healthy vertebral bodies in adult spine.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In adult human spine, vertebras gradually increase in size and dimension from the cervical region to the lumbar region. The size of vertebral centrums increases cranio–caudally as body weight percentage increases [5]. Mechanical forces play a critical role in physiological processes such as bone formation remodeling and angiogenesis [1, 16]. Mechanical stimuli resulting from weight loading and muscle contractions play an important role in remodeling the mature bone. Skeletal unloading as a result of prolonged bed rest, paralysis, or extended periods of weightlessness (as in spaceflight) leads to the rapid loss of bone mass [17, 25, 26, 29]. The height of spine increases about 5 cm in 3 months of weightlessness condition [17]. This increase in body height is, in some part, due to the increase of intervertebral disc height [21]. However, numerous reports have shown inconsistent effects of weightlessness or unloading on longitudinal bone growth: it can inhibit bone formation [18], have no effect at all on bone size [18], or suppress bone enlargement [20, 24, 28, 30, 32]. The reported effects of excess loading, as in the case of competitive athletes, are also contradictory, showing induction or reduction of longitudinal growth, most likely due to variations in the type and rate of the physical activity [4, 15, 27]. All morphological changes due to altered mechanical loads could also be attributed to surgical side effects, particularly those related to instrumentation for spine stabilization.
Much less is known about the effect of mechanical loads on the vertebral morphology in an adult human. Hence, in this study, we assessed the effect of long-term relative axial unloading on adult vertebral body height.
Materials and methods
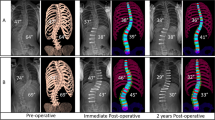
In a cross-sectional analytical study during a 15 year period, we measured 218 vertebral body heights of 200 processed radiographies (X-ray) belonging to adult patients (mean age: 32.3; range: 20–50 years) which had undergone ‘long segment instrumentation and fusion’ due to unstable thoracolumbar vertebral fracture. Patients with wedge compression or burst fracture were selected and those with other fracture types were excluded. Eleven patients were female (19.7%) and 45 were male (80.3%). The most frequently fractured vertebrae was first lumbar (N = 30) and the second was the twelfth thoracic (N = 14). In this operation, using a certain instrument (hooks and rods) on two vertebras proximal and distal to the fractured vertebrae, a simple distraction was exerted, and due to this action a relative axial unloading is applied on five vertebras. The anterior and posterior body heights of four normal vertebras (two adjacent proximal and two adjacent distal to the fractured vertebrae) were measured by two observers (intra-class correlation coefficient of 0.90).
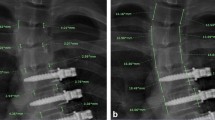
Measuring the height of vertebrae was done only based on the lateral radiograms of patients, which was done in one center with a Schimatzu X-ray set, with at least 1 year gap between two turns (preoperative and 1 year postoperative). The height of the vertebras was measured using a standard millimeter ruler to outline the AVBH and PVBH. The length of a line that was drawn between two points in the anterior upper and anterior lower corner of the vertebral body was measured as AVBH, and the length of a line that was drawn between two points in the posterior upper and posterior lower corner of the vertebral body was measured as PVBH. In order to increase the accuracy and minimize the effect of any confounding factors on the measurement of vertebral size (such as magnification, position of patients, rotation, etc.), one normal unloaded vertebra was selected as the reference vertebra. Those where there was no change observed when compared with the reference vertebrae before and after the operation were included as the final group samples (N = 56) and all cases with any subtle difference in AVBH or PVBH against the reference vertebra were excluded from the study. The minimum unloading time was at least 1 year on healthy vertebras. Paired sampled t test and ANCOVA model were used to compare the means of AVBH and PVBH. Statistical analysis was performed with SPSS® software (version 11.5).
Results
The comparison of mean values of AVBH and PVBH before and after the unloading period, for one or two adjacent vertebras proximal and distal to the fractured ones, is shown in Table 1. The AVBH of the first vertebra distal to the fractured site is significantly increased during the unloading period (before unloading: 30.11 mm, after unloading: 30.48 mm; P = 0.037). In other levels and sides, we did not find any statistically significant differences. Testing the summation of AVBH in the first vertebra proximal and distal to the site of fracture resulted in significant increase after unloading (before unloading: 28.51 mm, postop: 28.83 mm; P = 0.021); however, the similar summation for two vertebrae proximal and distal to the fracture did not show significant difference.
The ANCOVA model showed that AVBH values in the thoracic were comparable to the lumbar spine. However, we detected a marginally significant difference between the thoracic and lumbar spine regarding PVBH values before and after unloading (Table 2).
Discussion
The effect of mechanical loading on the density of mature bone has been widely studied in animal models [33] as well as in human physiological and pathological conditions [9]. However, the effect of mechanical axial unloading on the body height of mature vertebrae in adult is not clear. In the present study, we unloaded adult thoracolumbar spine for 1 year and found a significant increase in the AVBHs.
Vertebral morphometric analysis is a detailed examination of the shape and dimensions of vertebral bodies. Vertebral body changes can be detected by using either a semiquantitative or a quantitative method. The semiquantitative method is based on the classification of deformities by visual interpretation [11]. The quantitative method is based on the measurement of vertebral dimensions (height, width, or area) [2]. The implementation of standardized, conventional lateral spinal radiographs could enable a comparison of dimensions in longitudinal studies or provide reference values [3]. Using this technique, the clinician manually measures the size of the vertebra using a straight-edge ruler and actually draws on the film to outline the vertebra, selects the appropriate fiducial points, and then, with a ruler, measures the distance between the fiducial points [31]. Divergence of the X-ray beam, the focus-to-film distance, and the vertebra-to-film distance are major factors affecting the projected vertebral dimensions [3]. Although the manual technique described above is still widely used, computerized techniques have also become available. One computerized technique requires an analog lateral radiograph which is then digitized. Once digitized, the radiographic image is analyzed using a software program which provides measuring tools but still requires the clinician to manually select the fiducial points for measurement [19]. According to Cooley et al. [7] a millimetric ruler is an acceptable alternative to digital measurement devices. Regardless of the method used, distortion of the image can occur, leading to erroneous results [31]. In this study we measured vertebral body heights using a quantitative method with a millimetric ruler. The observed differences in pre- and postoperative vertebral body heights were small and were largely attributed to the limitations in the precision of the ruler.
Much of what is known about axial spinal unloading is based on experiments addressing the rehabilitation [12, 22]. In these studies, the spine was unloaded for a short time and measurement was done mainly on intervertebral disc height rather than on vertebral body height. It is known that increased compression slows growth and that decreased compression or distraction accelerates it, according to principles attributed to Hueter and Volkmann [28]. This rule is true for growing bones whereas we studied mature vertebral bone in adults; hence we cannot justify the increase in vertebral body height based on this rule and it is not also justifiable to explain the insignificant increase of other unloaded vertebral bodies. The process of bone elongation occurs via the development of cartilage rather than bone (endochondral ossification) [6, 26]. The cartilage (produced by the chondrocytes) is calcified and degraded by the invasion of osteoclasts, and a typical osseous tissue is formed in the epiphyseal growth plate [10]. The rate of bone elongation is regulated by the rate of chondrocyte proliferation and differentiation on one side of the growth plate and by blood vessel penetration and cartilage resorption at the border of the plate and at the metaphyseal bone on the opposite side [1, 8, 14]. It could not be concluded that axial unloading promoted endochondral ossification and chondrocyte proliferation and differentiation in adult spine. Cells in vivo are routinely exposed to a wide variety of stimuli, originating from chemical, mechanical, and electromagnetic sources. When challenged with a relevant stimulus for an extended period, cells undergo a process of desensitization, or adaptation, that enables them to reduce the stimulatory effects and, consequently, maintain environmental homeostasis. Bone cells are capable of sensing and responding to mechanical forces, but mechanosensitivity begins to decline soon after the stimulus is initiated [23]. These facts can explain the reduced long-term effects of mechanical unloading on vertebral body height. Whether the effects of unloading excreted before fusion hardening (about 3 months postoperative) or continue after, to our knowledge, is not clear. All our patients underwent posterior long segment instrumentation and fusion. It could be expected that posterior vertebral body was more influenced than anterior, but there was no significant increase in PVBHs. Perhaps posterior vertebral body is reinforced by pedicles and is more resistant to mechanical stimuli. Hiwatashi and co-workers [13] also reported an increase in AVBH, not in PVBH, after vertebroplasty. However, we are not aware of any studies defining the mechanism of increase in AVBH.
Interestingly, PVBH in lumbar was more influenced by unloading than in thoracic spine (marginally significant differences). To our knowledge, at the present time, we are unable to explain this difference in behavior.
The clinical relevance of the present study is as follows: one of the side-effects of spinal instrumentation can be vertebral morphological changes due to altered mechanical loads; performing spinal fusion is not equal to stiffness and rigidity: fused vertebras may also change in size owing to altered loads.
Conclusion
This study introduces the concept of biomechanical and morphological changes in the adult spine. Obviously, understanding all aspects of this concept needs a more comprehensive study regarding imaging and the biomechanical, histological and morphological profiles; only then will the true clinical relevance of this study be clear.
Comprehensive experimental studies with a specially designed instrument capable of exerting a constant tensile force on vertebras is recommended for the evaluation of all changes in the adult spine following long-term unloading.
References
Bianco P, Cancedda FD, Riminucci M, Cancedda R (1998) Bone formation via cartilage models: the “borderline” chondrocyte. Matrix Biol 17:185–192
Black DM, Cummings SR, Stone K, Hudes E, Palermo L, Steiger P (1991) A new approach to defining normal vertebral dimensions. J Bone Miner Res 6(8):883–892
Bodegom JW, Kuiper JW, Rijn RR et al (1998) Vertebral dimensions: influence of x-ray technique and patient size on measurements. Calcif Tissue Int 62:214–218
Bourrin S, Genty C, Palle S, Gharib C, Alexandre C (1994) Adverse effects of strenuous exercise: a densitometric and histomorphometric study in the rat. J Appl Physiol 76:1999–2005
Breggs AM, Greig AM, Wark JD et al (2004) A review of anatomical and mechanical factors affecting vertebral body integrity. Int J Med Sci 1(3):170–180
Cancedda R, Descalzi Cancedda F, Castagnola P (1995) Chondrocyte differentiation. Int Rev Cytol 159:265–358
Cooley JR, Danielson CD, Schultz GD et al (2001) Posterior disk displacement: morphologic assessment and measurement reliability-lumbar spine. J Manip Physiol Ther 24(5):317–326
De Crombrugghe B, Lefebvre V, Nakashima K (2001) Regulatory mechanisms in the pathways of cartilage and bone formation. Curr Opin Cell Biol 13:721–727
Duncan RL (1995) Transduction of mechanical strain in bone. ASGSB Bull 8:49–62
Gerber HP, Ferrara N (2000) Angiogenesis and bone growth. Trends Cardiovasc Med 10:223–228
Genant HK, Wu CY, van Kuijk C, Nevitt MC (1993) Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res 8(9):1137–1148
Hilse MA, Erdman AG, Iaizzo PA (2003) Axial unloading therapy device for cervical spinerehabilitation. J Med Eng Technol 27(5):207–217
Hiwatashi A, Moritani T, Numaguchi Y et al (2003) Increase in vertebral body height after vertebroplasty. Am J Neuroradiol 24:185–189
Hunziker EB (1994) Mechanism of longitudinal bone growth and its regulation by growth plate chondrocytes. Microsc Res Techol 28:505–519
Li KC, Zernicke RF, Barnard RJ, Li AF (1991) Differential response of rat limb bones to strenuous exercise. J Appl Physiol 70:554–560
Mao JJ, Nah HD (2004) Growth and development: hereditary and mechanical modulations. Am J Orthod Dentofacial Orthop 125:676–689
Moore TP, Thornton WE (1987) Space shuttle inflight and postflight fluid shifts measured by leg volume changes. Aviat Space Environ Med 58:A91–A96
Morey ER, Baylink DJ (1978) Inhibition of bone formation during space flight. Science 201:1138–1141
Nelson D, Peterson E, Tilley B et al (1990) Measurement of vertebral area on spine X-rays in osteoporosis: reliability of digitizing techniques. J Bone Miner Res 5(7):707–715
Ohashi N, Robling AG, Burr DB, Turner CH (2002) The effects of dynamic axial loading on the rat growth plate. J Bone Miner Res 17:284–292
Paajanen H, Lehto I, Alanen A, Erkintalo M, Komu M (1994) Diurnal fluid changes of lumbar discs measured indirectly by magnetic resonance imaging. J Orthop Res 12:509–514
Podein RJ, Iaizzo PA (1998) Applied forces and associated physiologic responses induced by axial spinal unloading with the LTX 3000 Lumbar Rehabilitation System. Arch Phys Med Rehabil 79(5):505–513
Robling AG, Burr DB, Turner CH (2001) Recovery periods restore mechanosensitivity to dynamically loaded bone. J Exp Biol 204:3389–3399
Robling AG, Duijvelaar KM, Geevers JV, Ohashi N, Turner CH (2001) Modulation of appositional and longitudinal bone growth in the rat ulna by applied static and dynamic force. Bone 29:105–113
Sibonga JD, Zhang M, Evans GL, Westerlind KC, Cavolina JM, Morey-Holton E, Turner RT (2000) Effects of spaceflight and simulated weightlessness on longitudinal bone growth. Bone 27:535–540
Sibonga JD, Zhang M, Ritman EL, Turner RT (2000) Restoration of bone mass in the severely osteopenic senescent rat. J Gerontol A Biol Sci Med Sci 55:71–78
Steinberg ME, Trueta J (1981) Effects of activity on bone growth and development in the rat. Clin Orthop 156:52–60
Stokes IA, Mente PL, Iatridis JC, Farnum CE, Aronsson DD (2002) Enlargement of growth plate chondrocytes modulated by sustained mechanical loading. J Bone Joint Surg Am 84:1842–1848
Turner RT (1995) Effects of short-term spaceflight and recombinant human growth hormone (rhGH) on bone growth in young rats. Aviat Space Environ Med 66:763–769
Turner CH (1998) Three rules for bone adaptation to mechanical stimuli. Bone 23:399–407
Smith-Bindman R, Cummings SR, Steger P, Geneta HK et al (1991) A comparison of morphometric definitions of vertebral fracture. J Bone Miner Res 6(7):25–34
Stokes LAF, Aronssen DD, Urban JPC (1994) Biomechanical factors influencing progression of angular skeletal deformity during growth. Eur J Exp Musculoskelet Res 5:51–60
Stokes IA, Spence H, Aronsson DD, Kilmer N (1996) Mechanical modulation of vertebral body growth. Implications for scoliosis progression. Spine 21:1162–1167
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kermani, H.R., Soroush, Z. Effect of long-term axial spinal unloading on vertebral body height in adult thoracolumbar spine. Eur Spine J 17, 965–969 (2008). https://doi.org/10.1007/s00586-008-0669-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-008-0669-y