Abstract
Background
It is widely understood that insulin resistance (IR) critically correlates with the development of liver fibrosis in several types of chronic liver injuries. Several experiments have proved that anti-IR treatment can alleviate liver fibrosis. Sodium–glucose cotransporter 2 (SGLT2) inhibitors comprise a new class of antidiabetic agents that inhibit glucose reabsorption in the renal proximal tubules, improving IR. The aim of this study was to elucidate the effect of an SGLT2 inhibitor on the development of liver fibrosis using obese diabetic Otsuka Long-Evans Tokushima fatty (OLETF) rats and their littermate nondiabetic Long–Evans Tokushima Otsuka (LETO) rats.
Methods
Male OLETF and LETO rats were intraperitoneally injected with porcine serum twice a week for 12 weeks to augment liver fibrogenesis. Different concentrations of ipragliflozin (3 and 6 mg/kg) were orally administered during the experimental period. Serological and histological data were examined at the end of the experimental period. The direct effect of ipragliflozin on the proliferation of a human hepatic stellate cell (HSC) line, LX-2, was also evaluated in vitro.
Results
OLETF rats, but not LETO rats, received 12 weeks of porcine serum injection to induce severe fibrosis. Treatment with ipragliflozin markedly attenuated the development of liver fibrosis and expression of hepatic fibrosis markers, such as alpha smooth muscle actin, collagen 1A1, and transforming growth factor beta (TGF-β), and improved IR in a dose-dependent manner in OLETF rats. In contrast, the proliferation of LX-2 in vitro was not affected, suggesting that ipragliflozin had no significant direct effect on the proliferation of HSCs.
Conclusion
In conclusion, our dataset suggests that an SGLT2 inhibitor could alleviate the development of liver fibrosis by improving IR in naturally diabetic rats. This may provide the basis for creating new therapeutic strategies for chronic liver injuries with IR.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Liver cirrhosis arises from various etiological factors, such as hepatitis virus B or C, autoimmune hepatitis, alcohol consumption, and metabolic disorders [1–3]. Hepatocellular carcinoma commonly develops in patients with cirrhotic liver; therefore, preventing liver cirrhosis is crucial to improving the prognosis of chronic liver injuries. Recently, with the influence of metabolic syndrome, the prevalence of non-alcoholic fatty liver diseases (NAFLDs) has increased rapidly [4]. Among these NAFLDs, non-alcoholic steatohepatitis (NASH) is widely known as a progressive pathological phenotype, and an increase of NASH-related liver cirrhosis has emerged as one of the critical issues to solve [5]. NASH is widely known as a hepatic phenotype of metabolic syndrome, particularly if it is deeply associated with type 2 diabetes mellitus (DM) [6].
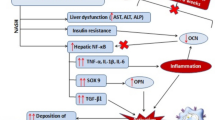
In patients with type 2 DM, insulin resistance (IR) is central to the pathogenesis of the disease [7]. Recent studies have suggested a strong relationship between type 2 DM and the development of chronic hepatitis C and NASH [8]. For example, IR is one of the major risk factors for the progression of liver fibrosis in patients with chronic hepatitis C and NASH [9–12]. In addition, rodent experiments revealed that an IR-mimicking, high-glucose, and high-insulin condition increased collagen and extracellular matrix production from activated hepatic stellate cells (HSCs), which play a central role in liver fibrogenesis [13, 14]. Therefore, particularly in NASH, several therapeutic strategies using hypoglycemic medicines have been attempted in order to reverse the development of liver fibrosis. Among these, peroxisome proliferator-activated receptor γ (PPARγ) agonist and dipeptidyl peptidase 4 (DPPIV) inhibitor are reported to be promising hypoglycemic agents that can also reverse the progression of liver disease. Both PPARγ agonist and DPPIV inhibitor exerted antifibrogenic activity in rodent NASH-mimicking models [15]. Furthermore, clinical studies elucidated that PPARγ agonist alleviated the pathological progression of patients with NASH [16]. Thus, IR-targeted therapy is a promising option for IR-associated chronic liver diseases.
In recent years, a sodium–glucose cotransporter-2 (SGLT2) inhibitor that inhibits glucose reabsorption through SGLT2 blockade, has been proposed as a promising new agent for type 2 DM treatment [17]. SGLT2 is expressed on the apical side of proximal tubular cells [18], where it reabsorbs approximately 90 % of the urine glucose in the proximal tubules [19]. This agent can lower the blood glucose level by promoting urinary glucose excretion [20]. Ipragliflozin is the first selective SGLT2 inhibitor to be approved in Japan [21]. Several clinical trials have reported the benefit of this agent in relation to blood glucose control used as both monotherapy and in combination therapy with other hypoglycemic medicines [22–24]. Another rodent experiment suggested that ipragliflozin can also improve IR through pancreatic β-cell protection [25]. Recently, ipragliflozin has been reported to prevent the development of liver fibrosis in choline-deficient, amino acid defined (CDAA) diet-treated rats, which express NASH-mimicking hepatic phenotypes [26]. According to this report, an SGLT2 inhibitor could also be a promising hypoglycemic agent, like a PPARγ agonist and DPPIV inhibitor. However, there is a critical issue based on a previous report that treatment with the CDAA diet did not exert severe IR [27, 28].
Therefore, in this study, we evaluated whether ipragliflozin can inhibit the progression of liver fibrosis using obese diabetic rats as a model of liver fibrosis with severe IR both in vivo and in vitro.
Materials and methods
Animal treatment
Male Otsuka Long–Evans Tokushima fatty (OLETF) rats and Long–Evans Tokushima Otsuka (LETO) rats were supplied by Otsuka Pharmaceutical Co. (Tokushima, Japan) [29]. Ipragliflozin was supplied by Astellas Pharma Co. (Japan). At the age of 8 weeks, the OLETF and LETO rats were each divided into four groups: G1–G4 for OLETF and G5–G8 for LETO rats. Each group consisted of six rats. Porcine serum (1 ml/kg) was intraperitoneally injected in all rats, except in G1 and G5 rats that served as negative controls, twice a week for 12 weeks; the same amount of saline was injected in negative control rats. Through drinking water, G3 and G7 rats were treated with low-dose ipragliflozin (3 mg/kg/day), while G4 and G8 rats were treated with high-dose ipragliflozin (6 mg/kg/day). G1, G2, G5, and G6 rats were given normal drinking water (control treatment). At the end of this experiment, the rats were anesthetized, their abdominal cavities were opened, blood samples were drawn via aortic puncture, and livers were harvested for histological evaluation. Serum biological markers were measured by routine laboratory methods. IR was evaluated with the homeostasis model assessment of insulin resistance (HOMA-R) and the quantitative insulin sensitivity check index (QUICKI), as described previously [30]. All animal procedures were performed according to a standard protocol and in accordance with the standard recommendations for the proper care and use of laboratory animals.
Immunohistochemical staining and semi-quantification
The liver sections were routinely stained with sirius red (SR) to detect the development of liver fibrosis. Immunohistochemical staining of α-smooth muscle actin (α-SMA) (Dako, Kyoto, Japan) was performed as described previously [31–33]. Semi-quantitative analyses of liver fibrosis were conducted using Adobe Photoshop software.
Real-time polymerase chain reaction (PCR)
Hepatic mRNA expression levels of transforming growth factor-β (TGF-β), alpha-1 type I collagen (collagen 1A1) and alpha smooth muscle actin (αSMA) were evaluated by real-time PCR, as described previously [33]. In brief, liver tissues were immediately flash-frozen for RNA extraction. In vitro, human LX-2 HSCs were plated at a density of 5 × 105 cells/ml on uncoated plastic dishes under three different conditions for 24 h. The first group was treated with low-glucose DMEM (100 mg/dl). The second group was treated under IR-mimicking conditions with high-glucose DMEM (280 mg/dl) plus insulin (200 nM). The last group was incubated under the IR-mimicking conditions plus ipragliflozin (10 µM). After incubation, cells were harvested and RNA was extracted for real-time PCR, as described previously [33]. Real-time PCR was performed with the ABI Prism 7700 sequence detection system (PE Applied Biosystems, Foster City, CA, USA) according to the manufacturer’s manual. Relative quantification of gene expression was performed using glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as an internal control. Threshold cycles and the standard curve method were used to calculate the relative amount of target RNA. Each rat primer was as follows: TGF-β, forward ATACGCCTGAGTGGCTGTCTT, reverse ATACGCTGAGTGGCTGTCTT; collagen 1A1, forward TGCTGCCTTTTCTGTTCCTT, reverse AAGGTGCTGGGTAGGGAAGT; αSMA, forward ACTGGGACGACATGGAAAAG, reverse CATCTCCAGAGTCCAGCACA; and GAPDH, forward AGACAGCCGCATCTTCTTGT, reverse CTTGCCGTGGGTAGAGTCAT. Human primers were as follows: TGF-β, forward GGGACTATCCACCTGCAAGA, reverse CCTCCTTGGCGTAGTAGTCG; and collagen 1A1, forward CCAAATCTGTCTCCCCAGAA, reverse TCAAAAACGAAGGGGAGATG.
Water-soluble tetrazolium salt (WST)-1 assay and cell counting
To evaluate the direct effect of ipragliflozin on human HSC lines, we compared cell proliferation with or without ipragliflozin treatment. The human HSC line LX-2 was seeded on uncoated plastic dishes at a density of 5 × 104 cells/ml. After overnight culture, the cells were treated with different concentrations of ipragliflozin (0, 0.1, 1, 5, 10, 20, and 50 µM, respectively) for 24 h. As a positive control, cells were treated with platelet-derived growth factor-BB (PDGF-BB; 10 ng/ml, R&D Systems, Minneapolis, MN, USA). Cell proliferation was measured by WST-1 assay according to the manufacturer’s manual.
Next, LX-2 HSCs were seeded at a density of 5 × 105 cells/ml on uncoated plastic dishes under five different conditions: normal glucose (100 mg/dl), high glucose (280 mg/dl), normal glucose plus insulin (200 nM), high glucose plus insulin, and high glucose plus insulin plus ipragliflozin (10 µM). These glucose concentrations were compared with the serological data for OLETF and LETO rats, as previously reported [34]. After incubation for 48 h, cells were harvested and the number of cells per group was counted with a cell counter (Waken B Tech Co. Ltd., Kyoto, Japan).
Western blotting for IRS1–PI3K–Akt signaling in vitro
To examine whether SGLT2-I had a direct inhibitory effect on insulin signaling in activated HSCs, western blotting was performed using a standard protocol. LX-2 cells were seeded on uncoated plastic dishes under the following three conditions for 24 h: normal glucose (100 mg/dl), high glucose (280 mg/dl) plus insulin (200 nM), and high glucose plus insulin plus SGLT2-I (10 µM). After incubation, cells were harvested and whole cell lysate was prepared before being subjected to SDS-PAGE (ATTO, Tokyo, Japan). Western blot analysis was performed with rabbit monoclonal anti-IRS1, anti-Akt, anti-phospho Akt, anti-β-actin antibody (Cell Signaling Technology, Danvers, MA, USA), and mouse monoclonal anti-phospho-IRS1 antibody (Abcam, Cambridge, UK).
Statistical analysis
Results are expressed as the mean ± SD and analyzed using an analysis of variance test. A p value of less than 0.05 was considered statistically significant.
Results
Ipragliflozin ameliorated IR in diabetic OLETF rats but not in nondiabetic LETO rats
Physical, serological, and urinary data for all experimental groups of diabetic OLETF rats (G1–4) and nondiabetic LETO rats (G5–8) are shown in Fig. 1 and Table 1. The final body weight of OLETF rats was significantly higher than that of LETO rats, as also observed in previous work [34]. Rats receiving the porcine serum injection exhibited a loss of body weight. Treatment with ipragliflozin lowered the body weight of OLETF rats that received that porcine serum injection in a dose-dependent manner, but not the body weight of the LETO rats (Fig. 1a). Liver weight in the OLETF groups was altered by neither porcine serum injection nor ipragliflozin. In the LETO groups, liver weight was lowered by porcine serum injection, but not by treatment with ipragliflozin (Fig. 1b).
According to the serological data (Table 1), for each group, the serum albumin (Alb) level was similar in the OLETF and LETO rats. Both serum total bilirubin (T-Bil) and alanine aminotransferase (ALT) levels were increased by porcine serum injection in OLETF rats but not in LETO rats. Treatment with ipragliflozin did not alter the levels of these serum markers.
In diabetic OLETF rats (G1, G2), as expected, significantly higher plasma glucose and plasma insulin levels than those in nondiabetic LETO rats (G5, G6) were observed (Table 1). Porcine serum injection increased the plasma glucose and insulin levels in LETO rats, but not in diabetic OLETF rats. Treatment with ipragliflozin significantly increased the excretion of urinary glucose in a dose-dependent manner in both rats. Urinary glucose level in OLETF rats was higher in diabetic OLETF rats than in LETO rats in the indicated groups. Ipragliflozin significantly lowered plasma glucose and insulin levels in a dose-dependent manner (G2 vs. G3 and G2 vs. G4). However, Ipragliflozin only lowered these in LETO rats in the high-dose group (G6 vs. G7 and G6 vs. G8). Similarly, treatment with ipragliflozin significantly improved the HOMA-R and QUICKI score in a dose-dependent manner, indicating that ipragliflozin alleviated IR in diabetic OLETF rats. In LETO rats, this finding was observed only in the high-dose ipragliflozin group (G6 vs. G7, G6 vs. G8), indicating that IR states were much less affected in non-diabetic LETO rats compared with OLETF rats in this experimental model.
Ipragliflozin ameliorated porcine-serum-induced liver fibrosis in diabetic OLETF rats
Histological findings were evaluated after a 12-week treatment with porcine serum and ipragliflozin. Liver fibrosis significantly developed in diabetic OLETF rats treated with porcine serum (G2) but not in nondiabetic LETO rats in each treatment group (G5–G8). Porcine serum-induced hepatic fibrosis was ameliorated by ipragliflozin treatment in a dose-dependent manner (Fig. 2). Activated HSCs indicated by immunohistochemistry of αSMA were significantly increased in diabetic OLETF rats treated with porcine serum (G2), and treatment with ipragliflozin decreased αSMA-positive activated HSCs in parallel with the liver fibrosis level (Fig. 3a–d, e).
Representative sirius red staining photomicrographs of the development of liver fibrosis in OLETF rats and LETO rats (original magnification, ×40): a–h G1 to G8, respectively. i Semi-quantitative analysis of the development of liver fibrosis by an image analyzer system. Data represent the mean ± SD values. *p < 0.05, indicating a significant difference between groups
a–e Representative αSMA immunohistochemistry in OLETF rats (original magnification, ×40): a–d representative images of each treatment group (a–d, G1–G4, respectively); e semi-quantitative analysis of the αSMA-positive HSCs. f–h Hepatic mRNA expression of αSMA (f), collagen 1A1 (g), and TGF-β (h) in OLETF rats. Data represent mean ± SD values. *p < 0.05, indicating a significant difference between groups
Expression levels of representative liver fibrogenic genes (i.e., αSMA, collagen 1A1, and TGF-β) were examined in each rat group by RT-PCR (Fig. 3f–h). A significant elevation in each gene expression level indicating the development of liver fibrosis was observed in diabetic OLETF rats after porcine serum injection (G2) compared with that in control rats (G1). Reflecting on the microscopic findings, treatment with ipragliflozin alleviated porcine-serum-induced elevation of these markers of fibrosis in a dose-dependent manner. Levels of hepatic fibrogenesis markers did not change in LETO rats, regardless of treatment (data not shown). This experimental model indicated that liver fibrosis—which was observed exclusively in diabetic OLETF rats—may be augmented by the presence of IR, and that ipragliflozin successfully ameliorated liver fibrosis in those rats.
Ipragliflozin did not alter proliferation of HSCs in vitro
As shown in Fig. 4a, ipragliflozin did not inhibit the proliferation of LX-2 in clinically compatible doses. Neither SGLT1 nor SGLT2 RNA was expressed in LX-2 cells (data not shown). High glucose or insulin levels increased the proliferation of HSCs, and the combination of both further increased proliferation. In contrast, treatment with ipragliflozin did not inhibit the proliferation of LX-2 under IR-mimicking conditions (Fig. 4b, c). Also, the expression of collagen 1A1 and TGF-β did not change in LX-2 cells under each condition, indicating that ipragliflozin does not exert direct effects on fibrosis production in HSCs (Fig. 4d, e). Moreover, the IR-mimicking condition promoted the phosphorylation of both IRS1 and Akt, indicating that this upregulated the activation of IRS–PI3K–Akt signaling. However, treatment with ipragliflozin did not alter this signaling pathway (Fig. 4f). These results suggested that the antifibrotic effect of ipragliflozin was based on the amelioration of IR, not on a direct effect on HSCs.
a Cell proliferation (WST-1) assay of LX-2 cells treated with different concentrations of ipragliflozin in vitro. There was no significant difference between the treated group and the untreated group (negative control). b Cell proliferation assay. c Number of LX-2 cells treated under normal glucose conditions, IR-mimicking conditions, and IR-mimicking conditions plus ipragliflozin. The IR-mimicking condition increased the proliferation of HSCs, but ipragliflozin did not. d, e Expression levels of gene markers for fibrosis in LX-2 cells treated with normal glucose, high glucose plus insulin, and high glucose plus insulin plus ipragliflozin. The mRNA expression levels for TGF-β (d) and collagen 1A1 (e) were not changed by any condition, indicating that IR and SGLT2-I had no influence on the fibrotic ability of HSCs. f The effects of IR and ipragliflozin on the IRS1–PI3K–Akt signaling pathway were investigated by western blotting. IR was shown to upregulate the phosphorylation of IRS-1 and Akt, while ipragliflozin did not. Data represent the mean ± SD values. *p < 0.05, indicating a significant difference between groups
Discussion
In the current study, we found that the antidiabetic SGLT2 inhibitor ipragliflozin successfully ameliorated liver fibrosis in a diabetic rat model. It is widely recognized that IR is a common pathological condition that underlies several chronic liver diseases such as chronic hepatitis C and NASH [11, 12]. Several reports indicate that IR is associated with various events involved in the development of chronic liver diseases, including hepatic inflammation, liver fibrosis, and hepatic carcinogenesis [34]. Among these, the role of IR in the development of liver fibrosis has been widely examined. Both hyperglycemia and hyperinsulinemia, the main manifestations of IR, directly augment the proliferation of HSCs and enhance the production of connective tissue growth factor and extracellular matrix [34]. We also noted that a combination of hyperglycemia-mimicking and hyperinsulinemia-mimicking conditions cumulatively increased the proliferation of human HSC line LX-2 compared with that observed for each condition separately [34]. In addition, it has been reported that insulin receptor is upregulated by activated HSCs [14]. Among several signaling pathways involved in the IR-induced development of liver fibrosis, insulin receptor substrate (IRS) 1/2 plays a central role [35]. The activation of the phosphatidylinositol 3-kinase (PI3K)–Akt pathway followed by the tyrosine phosphorylation of IRS1/2 is crucial to the proliferation of HSCs [35]. In the current study, we also confirmed that high glucose and insulin states accelerate cell proliferation among HSCs through the activation of the IRS1–PI3K–Akt pathway. Therefore, there is a strong and direct interaction of the presence of IR with the development of liver fibrosis in chronic liver disease.
In our experimental rodent liver fibrosis model, the immune response against porcine serum antigens activated HSCs via hepatic macrophages and led to fibrogenesis [36]. Of note, nondiabetic littermate LETO rats did not exhibit the development of fibrosis despite repeated fibrogenic stimulus by porcine serum injection. This finding may provide evidence that IR is a potent promoter of liver fibrosis following a particular initiating condition for liver fibrogenesis.
In this experimental model, we found that treatment with ipragliflozin successfully alleviated IR states in OLETF rats injected with porcine serum, resulting in amelioration of the development of liver fibrosis. Ipragliflozin is categorized as an SGLT2 inhibitor which inhibits urine glucose reabsorption from the proximal tubular cells [21]. We showed that ipragliflozin could lower plasma glucose and insulin levels, especially in diabetic OLETF rats, resulting in improved IR. In contrast, injection with porcine serum into nondiabetic LETO rats caused increasing plasma insulin levels and exacerbated IR; however, liver fibrosis did not occur in this group. According to a previous report, these observations may have resulted from high postprandial glucose and insulin levels caused by insulin hyposecretion in OLETF rats [29].
The expression of SGLT2, a sodium–glucose cotransporter, is limited in the renal proximal tubules, but not in the liver. Because of this localization, we postulated that an SGLT2 inhibitor would have no direct effect on HSC. In this study, we showed that ipragliflozin could not inhibit LX-2 proliferation induced under IR-mimicking conditions. In addition, ipragliflozin altered neither the gene expression of fibrotic markers (i.e., TGF-β and collagen 1A1) nor the IRS1–PI3K–Akt signaling induced by insulin in vitro. These data suggest that the SGLT2 inhibitor does not have a direct effect on liver fibrogenesis, but that it might inhibit hepatic fibrosis indirectly by improving systemic IR.
It is worth noting that a clinically comparable low dose (3–6 mg/kg/day) of ipragliflozin was sufficient to ameliorate the development of liver fibrosis. In recent years, SGLT2 inhibitors have been proposed as promising agents for the treatment of type 2 DM [21]. Several therapeutic attempts to use other antidiabetic agents against the development of liver fibrosis have focused on IR and demonstrated certain effects [15, 16]. We have previously reported that DPPIV inhibitor suppressed HSC proliferation and fibrogenic gene expression through the inhibition of ERK1/2, p38, and Smad2/3 phosphorylation [33]. In addition, synthetic PPARγ ligands and thiazolidinediones (TZD) reduced extracellular matrix deposition and HSC activation both in vitro and in vivo [37]. Our findings support previous evidence that antidiabetic treatment may contribute to preventing the progression of chronic liver disease in diabetes.
There are several limitations of the current experiment. First, our rat liver fibrosis model did not exhibit steatosis. As is commonly known, chronic liver diseases with coexistent IR will probably present with steatosis [38]. In addition, there is considerable evidence of an interaction between IR and steatosis [38]. Both IR and steatosis are related to and may enhance hepatic inflammation and fibrosis [39]. In this experiment, however, neither OLETF nor LETO rats had hepatic steatosis or steatosis-generated reactive oxidative stress (data not shown). Therefore, our rodent model lacks these interaction effects on the pathogenesis of liver fibrosis. Second, unlike other antidiabetic agents, ipragliflozin did not have a direct effect on HSCs; the antifibrotic effect of ipragliflozin was exclusively attributed to the alleviation of IR in our experimental model. However, on the basis of clinical evidence, SGLT2 inhibitors were reported to reduce the body weight of patients with type 2 DM [40]. Similar to those reports, the body weights of obese diabetic OLETF rats were reduced by ipragliflozin in our current experiment. Because reducing excess body weight is the most effective way to reduce IR [41, 42], SGLT2 inhibitors are expected to exert additional indirect ameliorating effects on liver fibrosis via the reduction of body weight. Further experiments using different rodent models are required.
In conclusion, low-dose ipragliflozin—a clinically available antidiabetic SGLT2 inhibitor—has been shown, for the first time, to successfully ameliorate porcine-serum-induced liver fibrosis in diabetic OLETF rats. Because SGLT2 inhibitors are already known to be clinically useful for type 2 DM, and are safe and widely used, this drug may represent a potential therapeutic strategy for liver fibrosis.
Abbreviations
- NASH:
-
Non-alcoholic steatohepatitis
- IR:
-
Insulin resistance
- SGLT2:
-
Sodium–glucose cotransporter 2
- OLETF:
-
Otsuka Long–Evans Tokushima fatty
- LETO:
-
Long–Evans Tokushima Otsuka
- HSC:
-
Hepatic stellate cell
- NAFLD:
-
Non-alcoholic fatty liver disease
- DM:
-
Diabetes mellitus
- PPARγ:
-
Peroxisome proliferator-activated receptor gamma
- DPPIV:
-
Dipeptidyl peptidase IV
- DPPIV-I:
-
Dipeptidyl peptidase IV inhibitor
- CDAA:
-
Choline-deficient l-amino acid-defined
- QUICKI:
-
Quantitative insulin sensitivity check index
- SR:
-
Sirius red
- TGF-β:
-
Transforming growth factor beta
- αSMA:
-
Alpha smooth muscle actin
- RT-PCR:
-
Real-time polymerase chain reaction
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- Alb:
-
Albumin
- T-Bil:
-
Total bilirubin
- ALT:
-
Alanine aminotransferase
- IRS:
-
Insulin receptor substrate
- PI3K:
-
Phosphatidylinositol 3-kinase
- TZD:
-
Thiazolidinediones
References
Friedman SL. Liver fibrosis—from bench to bedside. J Hepatol. 2003;38(Suppl 1):S38–53.
Friedman SL. Mechanisms of hepatic fibrogenesis. Gastroenterology. 2008;134(6):1655–69.
Iwaisako K, Taura K, Koyama Y, et al. Strategies to detect hepatic myofibroblasts in liver cirrhosis of different etiologies. Curr Pathobiol Rep. 2014;2(4):209–15.
Angulo P. Nonalcoholic fatty liver disease. N Engl J Med. 2002;346(16):1221–31.
Tolman KG, Fonseca V, Dalpiaz A, et al. Spectrum of liver disease in type 2 diabetes and management of patients with diabetes and liver disease. Diabetes Care. 2007;30(3):734–43.
Bugianesi E, McCullough AJ, Marchesini G. Insulin resistance: a metabolic pathway to chronic liver disease. Hepatology. 2005;42(5):987–1000.
Kahn CR. Insulin resistance, insulin insensitivity, and insulin unresponsiveness: a necessary distinction. Metabolism. 1978;27(12 Suppl 2):1893–902.
Gastaldelli A, Cusi K, Pettiti M, et al. Relationship between hepatic/visceral fat and hepatic insulin resistance in nondiabetic and type 2 diabetic subjects. Gastroenterology. 2007;133(2):496–506.
Mason AL, Lau JY, Hoang N, et al. Association of diabetes mellitus and chronic hepatitis C virus infection. Hepatology. 1999;29(2):328–33.
Shintani Y, Fujie H, Miyoshi H, et al. Hepatitis C virus infection and diabetes: direct involvement of the virus in the development of insulin resistance. Gastroenterology. 2004;126(3):840–8.
Allison ME, Wreghitt T, Palmer CR, et al. Evidence for a link between hepatitis C virus infection and diabetes mellitus in a cirrhotic population. J Hepatol. 1994;21(6):1135–9.
Chitturi S, Abeygunasekera S, Farrell GC, et al. NASH and insulin resistance: insulin hypersecretion and specific association with the insulin resistance syndrome. Hepatology. 2002;35(2):373–9.
Sugimoto R, Enjoji M, Kohjima M, et al. High glucose stimulates hepatic stellate cells to proliferate and to produce collagen through free radical production and activation ofmitogen-activated protein kinase. Liver Int. 2005;25(5):1018–26.
Svegliati-Baroni G, Ridolfi F, Di Sario A, et al. Insulin and insulin-like growth factor-1 stimulate proliferation and type I collagen accumulation by human hepatic stellate cells: differential effects on signal transduction pathways. Hepatology. 1999;29(6):1743–51.
Van Wagner LB, Rinella ME. The role of insulin-sensitizing agents in the treatment of nonalcoholic steatohepatitis. Therap Adv Gastroenterol. 2011;4(4):249–63.
Caldwell SH, Hespenheide EE, Redick JA, et al. A pilot study of a thiazolidinedione, troglitazone, in nonalcoholic steatohepatitis. Am J Gastroenterol. 2001;96(2):519–25.
Gerich JE. Role of the kidney in normal glucose homeostasis and in the hyperglycaemia of diabetes mellitus: therapeutic implications. Diabet Med. 2010;27(2):136–42.
Mather A, Pollock C. Glucose handling by the kidney. Kidney Int Suppl. 2011;120:S1–6.
Mather A, Pollock C. Renal glucose transporters: novel targets for hyperglycemia management. Nat Rev Nephrol. 2010;6(5):307–11.
Kanai Y, Lee WS, You G, et al. The human kidney low affinity Na+/glucose cotransporter SGLT2. Delineation of the major renal reabsorptive mechanism for d-glucose. J Clin Invest. 1994;93(1):397–404.
Kurosaki E, Ogasawara H. Ipragliflozin and other sodium glucose cotransporter-2 (SGLT2) inhibitors in the treatment of type 2 diabetes: preclinical and clinical data. Pharmacol Ther. 2013;139(1):51–9.
Kashiwagi A, Kazuta K, Goto K, et al. Ipragliflozin in combination with metformin for the treatment of Japanese patients with type 2 diabetes: ILLUMINATE, a randomized, double-blind, placebo-controlled study. Diabetes Obes Metab. 2015;17(3):304–8.
Watanabe Y, Nakayama K, Taniuchi N, et al. Beneficial effects of canagliflozin in combination with pioglitazone on insulin sensitivity in rodent models of obese type 2 diabetes. PLoS One. 2015;10(1):e0116851.
Vickers SP, Cheetham SC, Headland KR, et al. Combination of the sodium-glucose cotransporter-2 inhibitor empagliflozin with orlistat or sibutramine further improves the body-weight reduction and glucose homeostasis of obese rats fed a cafeteria diet. Diabetes Metab Syndr Obes. 2014;1(7):265–75.
Takahara M, Shiraiwa T, Matsuoka TA, et al. Ameliorated pancreatic β cell dysfunction in type 2 diabetic patients treated with a sodium-glucose cotransporter 2 inhibitor ipragliflozin. Endocr J. 2015;62(1):77–86.
Hayashizaki-Someya Y, Kurosaki E, Takasu T, et al. Ipragliflozin, an SGLT2 inhibitor, exhibits a prophylactic effect on hepatic steatosis and fibrosis induced by choline-deficient l-amino acid-defined diet in rats. Eur J Pharmacol. 2015;5(754):19–24.
Kodama Y, Kisseleva T, Iwaisako K, et al. c-Jun N-terminal kinase-1 from hematopoietic cells mediates progression from hepatic steatosis to steatohepatitis and fibrosis in mice. Gastroenterology. 2009;137(4):1467–77.
Miura K, Kodama Y, Inokuchi S, et al. Toll-like receptor 9 promotes steatohepatitis by induction of interleukin-1beta in mice. Gastroenterology. 2010;139(1):323–34.
Sato T, Asahi Y, Toide K, et al. Insulin resistance in skeletal muscle of the male Otsuka Long-Evans Tokushima fatty rat, a new model of NIDDM. Diabetologia. 1995;38(9):1033–41.
Katz A, Nambi SS, Mather K, et al. Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivity in humans. J Clin Endocrinol Metab. 2000;85(7):2402–10.
Yoshiji H, Kuriyama S, Yoshii J, et al. Angiotensin-II type 1 receptor interaction is a major regulator for liver fibrosis development in rats. Hepatology. 2001;34(4 Pt 1):745–50.
Yoshiji H, Kuriyama S, Hicklin DJ, et al. KDR/Flk-1 is a major regulator of vascular endothelial growth factor-induced tumor development and angiogenesis in murine hepatocellular carcinoma cells. Hepatology. 1999;30(5):1179–86.
Kaji K, Yoshiji H, Ikenaka Y, et al. Dipeptidyl peptidase-4 inhibitor attenuates hepatic fibrosis via suppression of activated hepatic stellate cell in rats. J Gastroenterol. 2014;49:481–91.
Kaji K, Yoshiji H, Kitade M, et al. Impact of insulin resistance on the progression of chronic liver diseases. Int J Mol Med. 2008;22(6):801–43.
Spector SA, Olson ET, Gumbs AA, et al. Human insulin receptor and insulin signaling proteins in hepatic disease. J Surg Res. 1999;83(1):32–5.
Bhunchet E, Eishi Y, Wake K. Contribution of immune response to the hepatic fibrosis induced by porcine serum. Hepatology. 1996;4:811–7.
Galli A, Ceni E, Crabb DW, et al. Antidiabetic thiazolidinediones inhibit invasiveness of pancreatic cancer cells via PPAR-gamma independent mechanisms. Gut. 2004;53(11):1688–97.
Ota T, Takamura T, Kurita S, et al. Insulin resistance accelerates a dietary rat model of nonalcoholic steatohepatitis. Gastroenterology. 2007;132(1):282–93.
Peverill W, Powell LW, Skoien R. Evolving concepts in the pathogenesis of NASH: beyond steatosis and inflammation. Int J Mol Sci. 2014;15(5):8591–638.
Suzuki M, Takeda M, et al. Tofogliflozin, a sodium/glucose cotransporter 2 inhibitor, attenuates body weight gain and fat accumulation in diabetic and obese animal models. Nutr Diabetes. 2014;7(4):e125.
Oliveira AG, Carvalho BM, Tobar N, et al. Physical exercise reduces circulating lipopolysaccharide and TLR4 activation and improves insulin signaling in tissues of DIO rats. Diabetes. 2011;60(3):784–96.
Gao X, Yan D, Zhao Y, et al. Moderate calorie restriction to achieve normal weight reverses β-cell dysfunction in diet-induced obese mice: involvement of autophagy. Nutr Metab (Lond). 2015;6(12):34.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Nishimura, N., Kitade, M., Noguchi, R. et al. Ipragliflozin, a sodium–glucose cotransporter 2 inhibitor, ameliorates the development of liver fibrosis in diabetic Otsuka Long–Evans Tokushima fatty rats. J Gastroenterol 51, 1141–1149 (2016). https://doi.org/10.1007/s00535-016-1200-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-016-1200-6