Summary
Intravenous infusion of crystalloid solutions is one of the most frequently administered medications worldwide. Available crystalloid infusion solutions have a variety of compositions and have a major impact on body systems; however, administration of crystalloid fluids currently follows a “one fluid for all” approach than a patient-centered fluid prescription. Normal saline is associated with hyperchloremic metabolic acidosis, increased rates of acute kidney injury, increased hemodynamic instability and potentially mortality. Regarding balanced infusates, evidence remains less clear since most studies compared normal saline to buffered infusion solutes.; however, buffered solutes are not homogeneous. The term “buffered solutes” only refers to the concept of acid-buffering in infusion fluids but this does not necessarily imply that the solutes have similar physiological impacts. The currently available data indicate that balanced infusates might have some advantages; however, evidence still is inconclusive. Taking the available evidence together, there is no single fluid that is superior for all patients and settings, because all currently available infusates have distinct differences, advantages and disadvantages; therefore, it seems inevitable to abandon the “one fluid for all” strategy towards a more differentiated and patient-centered approach to fluid therapy in the critically ill.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Intravenous fluids are among the most commonly used medications in daily medical practice [1]. Currently available crystalloid infusion solutions have a variety of compositions and may therefore influence acid-base balance, body water content, the volume of body water compartments and plasma electrolyte composition [2] and may thus have a major impact on organ (dys)function and clinical outcome when used for fluid resuscitation purposes [3]. This may be of special importance in critically ill patients as they typically receive large quantities of intravenous fluids in order to maintain vital organ functions. The choice of crystalloid fluid may therefore profoundly impact on morbidity and mortality in affected patients [2,3,4]. A further characteristic of critically ill patients is that their vulnerability for fluid overload is high, e. g., in sepsis patients with concomitant capillary leakage. Fluid overload is not a benign occurrence, despite the general perception. For many years so the so-called goal-directed approach was a mainstay of fluid therapy, especially in patients with sepsis or septic shock [11, 12]; however, mounting evidence shows that goal-directed therapy does not result in better clinical outcomes in patients with shock [11,12,13,14] and that fluid overload is itself related to increased mortality and morbidity [15,16,17,18,19,20,21,22,23,24]. It seems therefore of paramount importance to further investigate crystalloid fluids that are used for volume replacement in the critically ill patient as well as to minimize fluid overload.
The aim of this review is to provide an overview on currently used crystalloid fluids and to investigate respective risk-benefit profiles.
Crystalloid fluids
Several types of infusates are currently available for fluid resuscitation in the critical ill. The available solutes have variable salt-acid compositions (details depicted in Table 1).
Isotonic 0.9% saline is probably the best known and studied infusion solution in clinical practice [5,6,7,8,9,10]. In recent years, several drawbacks of normal saline were identified [11,12,13,14,15,16]; however, normal saline still remains among the most commonly used infusates worldwide [2, 11, 12, 15, 17, 18]. The benefit of balanced infusion solutions may be the ability to compensate the ion fluctuations better and buffering of the physiological acid-base status when compared to normal saline [2, 4, 19]. Nevertheless, despite continuing evaluation, no superiority of one particular type of infusion fluid has so far been shown [4, 15, 20, 21].
Normal saline (0.9% NaCl)
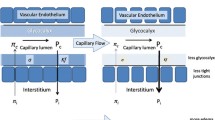
The most frequently used infusion solution worldwide is 0.9% saline [11, 12, 17, 18]. Interestingly, 0.9% saline was initially invented for physiological experiments [22], and not for infusion purposes. Isotonic saline has a sodium and chloride content of 154 mmol/l. In reality isotonic saline solution, often referred to as “physiological” saline, is thus slightly hypernatremic and markedly hyperchloremic [22]. In contrast to buffered infusions, isotonic saline does not contain potassium. Studies evaluating hypertonic saline showed a tendency to hyperchloremia and concurrent metabolic acidosis in patients receiving normal saline [11,12,13,14]. Additionally, the infusion of isotonic saline, as all non-buffered solutions, produces metabolic acidosis by plasma dilution. Through infusion of non-bicarbonate containing isotonic saline, the bicarbonate concentration of the plasma is diluted resulting in dilutional acidosis. Dilutional acidosis can either be produced by infusion of chloride-rich isotonic saline, non-buffered solutes, 5% glucose or mannitol solutions [23, 24]. Furthermore, the use of normal saline is most likely associated with an increased incidence of acute kidney injury, coagulation disturbances, hemodynamic instability and mortality [1, 12, 25, 26]. Physiological consequences of normal saline are shown in Fig. 1.
Buffered or balanced crystalloids
Balanced infusates were first introduced in 1931 by Alexis Hartmann with the aim to create an infusion solution with a reduced chloride content [27]. The search for a chloride-reduced solute was problematic; however, as electroneutrality within fluids needs to be ensured, a reduction in chloride will automatically lead to a cation excess. Chloride is the main extracellular anion of the human body; therefore, at first sight it seems an ideal anion for intravenous fluids. Nevertheless, hyperchloremia may result in triggering hyperchloremic metabolic acidosis [11,12,13,14]. The combination of bicarbonate with chloride might be a good choice regarding anions in infusion solutes and may result in a stable acid-base homeostasis and avoidance of dilutional acidosis. Nevertheless, bicarbonate has a short shelf life and is therefore unsuitable. This problem was solved by adding weak acids or so-called metabolizable anions, such as lactate, gluconate, malate or acetate to respective solutes [28, 29]. These anions are metabolized to bicarbonate leading to a more balanced acid-base homeostasis and avoidance of dilution at the same time [13, 30,31,32,33]. This may explain why they are called buffered infusates. Balanced infusion solutions contain, in contrast to isotonic saline, calcium, potassium and magnesium. One of the fears of physicians using balanced crystalloids is the respective potassium content; however, even though balanced infusates contain potassium they do not augment plasma potassium levels when compared to 0.9% saline [28, 34] and are thus not suitable for substitution of potassium. Currently, there is no evidence that balanced infusates increase plasma potassium concentrations even in patients with acute kidney injury [29]. Balanced infusates are supplemented with calcium and magnesium as cations in order to reduce sodium content and to maintain electroneutrality. Calcium-containing infusions might be problematic because of increased coagulation activity when concomitantly infused with blood products [28, 35]; however, some controversy on this topic exists.
Lactate versus acetate-buffered infusion solutions
Lactate and acetate are the most commonly used metabolizable anions in infusion solutes. Lactate and acetate-buffered crystalloids have some marked differences and advantages or disadvantages that may favor lactate over acetate-buffered crystalloids. When compared to lactate, acetate acts differently as a buffering substance in infusion solutions. Under normal circumstances, generation of bicarbonate from acetate would take approximately 15 min, as compared to 1 h for lactate [36,37,38,39,40]. Moreover, bicarbonate can be generated from acetate in almost all organs [19, 41, 42]. Lactate is a physiological intermediate, meaning it can cause acidemia and alkalemia and is metabolized at an extremely high rate in the liver but to a little extent in other organs, such as the heart or the brain [3].
The most feared side effect of acetate infusion is its vasodilatory effect; however, this issue remains controversial. While some studies reported reduced blood pressures after sodium acetate infusions [40, 43,44,45], others showed stable [46, 47] or even increased blood pressures [48]. Additionally, there is some uncertainty with respect to whether vasodilatory side effects of acetate-containing infusates would be dose-dependent [49, 50]. In general, balanced crystalloids are more often associated with alkalemic states but factors leading to alkalemia are as yet unknown. One could assume that this may vastly depend on renal, hepatological and respiratory functions but clinical trials in this area are needed.
Metabolic alkalosis was associated with acetate-containing infusates; however, infusion therapy with acetate-buffered crystalloid infusates may lead to increased bicarbonate levels [19, 21, 51, 52] but changes in pH and concurrent metabolic alkalosis seem less frequent than changes in bicarbonate after infusion of an acetate-buffered crystalloid solute due to slower metabolizing rates [19, 21]. Metabolic alkalosis may even be more detrimental than mild metabolic acidosis; however, the optimal mount of organic base has not yet been defined.
Use of lactate-buffered infusions may have additional problems: infusion of a solution containing lactate was shown to increase serum lactate levels and therefore theoretically impair use of lactate as a marker of tissue hypoxia [3, 19, 21, 53,54,55,56].
In patients suffering, e. g., from diabetes, lactate can theoretically lead to hyperglycemia via increased hepatic gluconeogenesis [36, 37, 57]. Lactate acidosis was associated with lowered levels of ionized calcium [58, 59], it is however, unclear whether this is also true for lactate-buffered infusates as the respective solutes do not induce lactic acidosis. Furthermore, it is unknown whether this effect is clinically relevant with respect to coagulation cascades. For comparison of the metabolic properties of lactate and acetate, please refer to Fig. 2.
Normal saline or balanced infusates: what is the current evidence?
Metabolic acidosis and hyperchloremia
Mounting data show that infusion of chloride-rich infusates results in metabolic acidosis in critically ill patients when compared to buffered infusates [1, 11,12,13,14,15,16, 29, 34, 60,61,62,63,64]. Many studies have shown that even moderate infusion volumes (≥2 l per 24 h) of isotonic saline induces metabolic acidosis [65,66,67,68]. Earlier, the clinical relevance of hyperchloremic metabolic acidosis was controversially discussed [69] but recently, a growing number of studies have shown that hyperchloremic metabolic acidosis is related to morbidity and increased mortality in the critically ill [10, 12, 70]. Metabolic hyperchloremic acidosis following infusion of normal saline has a marked impact on excitable tissue [71, 72] and cellular function [73]. Studies showed that in comparison to 0.9% saline, the occurrence of acidosis and electrolyte disturbances is significantly lower in patients receiving lactated Ringer’s solution [2, 16, 28, 29, 52, 53, 68, 74,75,76,77,78,79].
Renal function
In the 1980s it was first shown that infusion of hyperchloremic solutions leads to renal vasoconstriction and a fall in the glomerular filtration rate (GFR; [80, 81]). Additionally, in animals and humans, it was shown that infusion of normal saline is associated with markedly decreased renal perfusion pressures [11, 12, 61, 68]; however, with respect to patient-centered outcomes, such as occurrence of acute kidney injury (AKI) and the need for renal replacement therapy (RRT), the discussion remains controversial [5, 12, 26, 29, 62, 70, 82,83,84,85,86,87]. Whereas some trials did not observe a change in creatinine or an increased rate of AKI under chloride-rich infusates in both general intensive care unit (ICU) patients and in patients with sepsis [5, 83, 86], others demonstrated a markedly increased incidence of AKI and need for RRT under infusion of normal saline [12, 26, 29, 82, 87]. This effect remained significant after adjusting for confounders such as illness severity, operative status, mechanical ventilation, and type of admission [26, 29, 82]. A recently published meta-analysis concluded that the use of chloride-rich infusates is associated with a significant high risk to develop AKI [62].
Generally, the body of available literature varies widely with respect to methodology, total fluid volumes, AKI definition, as well as RRT initiation [5, 12, 29, 82]. For example, the recently published SPLIT trial compared the effects of normal saline to an acetate-buffered crystalloid solution on RRT did not find a difference in relation to RRT; however this trial only included low risk intensive care populations (as defined by relatively low APACHE scores) and patients only received very small fluid amounts [5]. Thus, the results of this trial may hardly be generalizable.
In conclusion, hyperchloremia and AKI after infusion of chloride-rich infusate may be dose dependent [88]. The volume of chloride-rich infusate infused as well as prior kidney function are major contributors to AKI.
Fluid overload
Volume overload is a considerable problem in the critically ill. Lowell et al. observed that 40% of patients admitted to a surgical ICU had an excessive increase in total body water of more than 10% from preoperative weight [89]. In patients with sepsis, extracellular volume overload exceeded 10 l after 2 days of resuscitation, resulting in approximately 3 weeks until the respective fluid was excreted [90]. Generally, an increase in postoperative complications and adverse outcomes was associated with administration of excess sodium and water in the perioperative period by promoting interstitial edema [14, 91,92,93,94,95,96,97,98]. It was also shown that restrictive fluid and salt infusion was associated with significantly fewer cardiopulmonary events [14, 99, 100], increased bowel motility [100, 101], improved wound and surgical site healing [14, 99, 100] and reduced hospital stay [100, 101].
Several studies showed that normal saline (due to its high sodium content) may result in more fluid overload [102, 103]. Lindner et al. showed that normal saline use in the ICU is one of the main reasons for a positive sodium balance and hypernatremic hyperosmolarity in the critically ill [103]. In addition, it was shown that normal saline takes significantly longer to be eliminated via urine than other infusion solutes; however, use of hypertonic saline has resulted in decreased perioperative weight gain, negative fluid balance, and increased diuresis after major surgical procedures [104,105,106,107]. Fluid overload may not be uniquely a result of normal saline, it may also occur following infusion of balanced infusates and in cases of decreased renal function. Nevertheless, whether the type of crystalloid fluid influences the incidence rate and extent of volume overload needs to be investigated.
Cardiovascular function
Recent evidence suggests that the type of crystalloid infusate used for fluid resuscitation may impact on baseline blood pressures, cardiac performance, and need for catecholamines support. In 2014, Kellum et al. performed a rodent study with experimental sepsis where they found significantly decreased mean arterial pressures in the normal saline group [61]. Another experimental study showed that infusion of normal saline and ensuing metabolic acidosis may induce a time dependent decrease in mean arterial blood pressures, cardiac index, and cardiac work in rodents with abdominal sepsis [108]. In humans, use of normal saline was significantly associated with increased catecholamine needs and worsened hemodynamics [34, 109, 110]. A recently published prospective randomized-controlled double blind clinical trial in patients undergoing major abdominal surgery showed that patients in the normal saline group more often needed vasoactive agents in a higher dose [110]. In addition, in this trial patients receiving saline had worsening hemodynamics with ongoing time, which was associated to the amount of fluid infused and higher doses of vasoactive medication [110].
The underlying physiological changes related to increased hemodynamic instability in patients receiving normal saline when compared to acetate-buffered crystalloid still remain elusive. Several potential contributors are discussed: first, metabolic acidosis (attributed to normal saline) reduces cardiac activity [71, 72], as well as vascular tone [111,112,113] and inhibits endogenous catecholamine production [114]. This was also seen in the KATECHOL trial [110]. Second, acetate may influence cardiovascular function. In 1978 Liang and Lowenstein infused acetate and pyruvate into anesthetized dogs to assess the impact on the circulation [115]. They found that increased acetate levels were associated with a significant increase in cardiac output [115]. Even though myocardial oxygen consumption increased during acetate infusion, the decrease in myocardial oxygen extraction and the increase in coronary sinus blood oxygen saturation suggest that an active coronary vasodilation takes place that does not result in increased cardiac work [115]. Acetate infusion also increases blood flow to the gastrointestinal tract, the renal system, intercostal muscles, and the diaphragm [115]. Another experimental study found similar results in healthy volunteers [48] and three other studies showed a positive effect on cardiac output with acetate-rich infusates, while peripheral vascular resistance was decreased [116,117,118]. Several other studies also reported a declined blood pressures after acetate infusion [40, 43,44,45]. In summary, use of acetate-buffered solutions could be harmful to patients with decreased myocardial reserve as evidence points to the fact that it increases myocardial contractility and oxygen consumption.
An overview on the risks and benefits of acetate on the cardiocirculatory system is shown in Table 2.
So far, no study has compared the effects of a lactate-based to an acetate-based crystalloid infusate on the cardiovascular system. Whether there is a difference between acetate-buffered and lactate-buffered crystalloids with respect to hemodynamic stability in the critically ill is currently under investigation [88].
Inflammation and coagulation
Inflammatory makers were found to be increased in animals receiving chloride-rich infusates in experimental sepsis [28, 61, 119, 120] and in trauma [121]. In humans, use of normal saline was associated with increased neutrophil activation [122, 123], effects on coagulation cascades [121, 124, 125] and increased transfusion needs [12].
Mortality
Whether use of normal saline is associated with increased mortality is controversially discussed. In a large observational study, Shaw et al. detected increased mortality rates in patients undergoing major abdominal surgery when treated with normal saline [12]. In fact, use of normal saline resulted in an approximately 50% absolute increase in mortality (2.9% versus 5.6% mortality) when compared to buffered crystalloids [12]. Additionally, patients treated with normal saline had higher rates of postoperative infections, blood transfusions and AKI [12].
Several other large-scale studies in various settings of critically ill patients found increased mortality rates for patients treated with normal saline, when compared to chloride-depleted infusates. This effect remained significant after adjusting for important confounders [10, 12, 63, 64, 126]; however, the results in the published literature remain controversial. A recent meta-analysis failed to confirm an association between chloride-rich infusions and mortality [62]. In addition, several large recently published randomized controlled trial, such as the SPLIT and LICRA trials did not find any difference in mortality between the groups [5, 29, 62, 86]. In a further large trial (SALT) a composite outcome of death, dialysis and persistent renal failure was not significantly different between the saline and the balanced infusion group in the general ICU population [8]. Studies investigating mortality with respect to crystalloid choice are depicted in Table 3.
A critical appraisals: what fluid should we choose in the critically ill?
Currently available crystalloid infusion solutions have multiple physiological effects; however, it seems important to remember that infusion solutes are among the most frequently used medications prescribed. In contrast to the usual choice of medications, the current approach to fluid therapy is mostly not patient-centered and generally follows a “one fluid for all” principle. Medical doctors are most often not aware of the importance of the fluids administered and knowledge on risks and benefits is most often sparse; however, the effects of infusion solutions on organ functions are not negligable, especially in the postoperative setting when patients have already received large volumes before being admitted to an ICU. In addition, with declined use of colloid solutions [127,128,129,130,131,132], choice of correct crystalloid solutes may even become more important.
The currently available data indicate that balanced infusates might have some advantages but evidence is still inconclusive. Normal saline is still among the most widely used iv fluids. In fact, media reports reveal that as many as 740 units of 0.9% saline are used each minute in the USA. [133].
Current evidence shows that normal saline can be used safely when some caution towards its negative side effects is applied and it is only used in patients requiring smaller fluid volumes with reduced illness severity scores. With respect to balanced infusates, the evidence remains less clear as most studies compare normal saline to a buffered infusion solute. Studies comparing the different buffered solutes are much less common but it seems important that buffered solutes should not be regarded as a homogeneous group. The term “buffered solutes” refers to the concept of acid-buffering in infusion fluids but does not imply that respective solutes have similar physiological impact. Nevertheless, different buffered solutes should be characterized more clearly in the future. Taking the available evidence together there is no single fluid that is superior for all patients and settings, because all of the currently available infusates have distinct differences, advantages and disadvantages; therefore, it seems inevitable to abandon the “one fluid for all” strategy towards a more differentiated and patient-centered approach to fluid therapy in critical illness.
References
Pfortmueller CA, Fleischmann E. Acetate-buffered crystalloid fluids: current knowledge, a systematic review. J Crit Care. 2016;35:96–104.
Noritomi DT, Pereira AJ, Bugano DD, Rehder PS, Silva E. Impact of Plasma-Lyte pH 7.4 on acid-base status and hemodynamics in a model of controlled hemorrhagic shock. Clinics. 2011;66(11):1969–74.
Hofmann-Kiefer KF, Chappell D, Kammerer T, et al. Influence of an acetate- and a lactate-based balanced infusion solution on acid base physiology and hemodynamics: an observational pilot study. Eur J Med Res. 2012;17:21.
Zadak Z, Hyspler R, Hronek M, Ticha A. The energetic and metabolic effect of Ringerfundin (B. Braun) infusion and comparison with Plasma-Lyte (Baxter) in healthy volunteers. Acta Medica (Hradec Kralove). 2010;53(3):131–7.
Young P, Bailey M, Beasley R, et al. Effect of a buffered crystalloid solution vs saline on acute kidney injury among patients in the intensive care unit: the SPLIT randomized clinical trial. JAMA. 2015;314(16):1701–10.
Hafizah M, Liu CY, Ooi JS. Normal saline versus balanced-salt solution as intravenous fluid therapy during neurosurgery: Effects on acid-base balance and electrolytes. J Neurosurg Sci. 2015;61(3):263–270. https://doi.org/10.23736/S0390-5616.16.03221-5. Epub 2015 Apr 9.
Yung M, Letton G, Keeley S. Controlled trial of Hartmann’s solution versus 0.9% saline for diabetic ketoacidosis. J Paediatr Child Health. 2017;53(1):12–7.
Semler MW, Wanderer JP, Ehrenfeld JM, et al. Balanced Crystalloids versus saline in the intensive care unit: the SALT randomized trial. Am J Respir Crit Care Med. 2017;195(10):1362. https://doi.org/10.1164/rccm.201607-1345OC.
Semler MW, Self WH, Wang L, et al. Balanced crystalloids versus saline in the intensive care unit: study protocol for a cluster-randomized, multiple-crossover trial. Trials. 2017;18(1):129.
Raghunathan K, Shaw A, Nathanson B, et al. Association between the choice of IV crystalloid and in-hospital mortality among critically ill adults with sepsis*. Crit Care Med. 2014;42(7):1585–91.
Lobo DN. Intravenous 0.9% saline and general surgical patients: a problem, not a solution. Ann Surg. 2012;255(5):830–2.
Shaw AD, Bagshaw SM, Goldstein SL, et al. Major complications, mortality, and resource utilization after open abdominal surgery: 0.9% saline compared to Plasma-Lyte. Ann Surg. 2012;255(5):821–9.
Gattinoni L, Carlesso E. Supporting hemodynamics: what should we target? What treatments should we use? Crit Care. 2013;17(Suppl 1):4.
Chappell D, Jacob M, Hofmann-Kiefer K, Conzen P, Rehm M. A rational approach to perioperative fluid management. Anesthesiology. 2008;109(4):723–40.
Story DA, Lees L, Weinberg L, et al. Cognitive changes after saline or plasmalyte infusion in healthy volunteers: a multiple blinded, randomized, cross-over trial. Anesthesiology. 2013;119(3):569–75.
Hahn RG, Nyberg Isacson M, Fagerstrom T, Rosvall J, Nyman CR. Isotonic saline in elderly men: an open-labelled controlled infusion study of electrolyte balance, urine flow and kidney function. Anaesthesia. 2016;71:155. https://doi.org/10.1111/anae.13301.
Cecconi M, Corredor C, Arulkumaran N, et al. Clinical review: goal-directed therapy-what is the evidence in surgical patients? The effect on different risk groups. Crit Care. 2013;17(2):209.
O’Malley CM, Frumento RJ, Bennett-Guerrero E. Intravenous fluid therapy in renal transplant recipients: results of a US survey. Transplant Proc. 2002;34(8):3142–5.
Rohrig R, Wegewitz C, Lendemans S, Petrat F, de Groot H. Superiority of acetate compared with lactate in a rodent model of severe hemorrhagic shock. J Surg Res. 2014;186(1):338–45.
Voigtsberger S, Urner M, Hasler M, et al. Modulation of early inflammatory response by different balanced and non-balanced colloids and crystalloids in a rodent model of endotoxemia. PLoS ONE. 2014;9(4):e93863.
Keibl C, Sipos W, Ponschab M, Schlimp CJ. Blood biochemical changes in pigs after infusion with acetate-buffered or lactate-buffered crystalloid solutions. Lab Anim (NY). 2015;44(7):268–73.
Awad S, Allison SP, Lobo DN. The history of 0.9% saline. Clin Nutr. 2008;27(2):179–88.
Asano S, Kato E, Yamauchi M, Ozawa Y, Iwasa M. The mechanism of acidosis caused by infusion of saline solution. Lancet. 1966;1(7449):1245–6.
Shires GT, Holman J. Dilution acidosis. Ann Intern Med. 1948;28(3):557–9.
Potura ELG, Biesenbach P, Funk G‑C, Reiterer C, Schwarz C, Druml W, Fleischmann E. An acetate-buffered balanced crystalloid versus 0.9% saline in patients with end-stage renal disease undergoing cadaveric renal transplantation: a prospective randomized controlled trial. Anesth Analg. 2015;120(1):123. https://doi.org/10.1213/ANE.0000000000000419.
Marttinen M, Wilkman E, Petaja L, et al. Association of plasma chloride values with acute kidney injury in the critically ill—a prospective observational study. Acta Anaesthesiol Scand. 2016;60(6):790–9.
Lee JA. Sydney Ringer (1834–1910) and Alexis Hartmann (1898–1964). Anaesthesia. 1981;36(12):1115–21.
Zhou F, Peng ZY, Bishop JV, et al. Effects of fluid resuscitation with 0.9% saline versus a balanced electrolyte solution on acute kidney injury in a rat model of sepsis. Crit Care Med. 2014;42(4):e270–e8.
Yunos NM, Bellomo R, Hegarty C, et al. Association between a chloride-liberal vs chloride-restrictive intravenous fluid administration strategy and kidney injury in critically ill adults. JAMA. 2012;308(15):1566–72.
Gattinoni L, Carlesso E, Maiocchi G, Polli F, Cadringher P. Dilutional acidosis: where do the protons come from? Intensive Care Med. 2009;35(12):2033–43.
Carlesso E, Maiocchi G, Tallarini F, et al. The rule regulating pH changes during crystalloid infusion. Intensive Care Med. 2011;37(3):461–8.
Langer T, Carlesso E, Protti A, et al. In vivo conditioning of acid-base equilibrium by crystalloid solutions: an experimental study on pigs. Intensive Care Med. 2012;38(4):686–93.
Morgan TJ, Venkatesh B, Hall J. Crystalloid strong ion difference determines metabolic acid-base change during acute normovolaemic haemodilution. Intensive Care Med. 2004;30(7):1432–7.
Potura E, Lindner G, Biesenbach P, et al. An acetate-buffered balanced crystalloid versus 0.9% saline in patients with end-stage renal disease undergoing cadaveric renal transplantation: a prospective randomized controlled trial. Anesth Analg. 2015;120(1):123–9.
Morgan TJ. The ideal crystalloid—what is ‘balanced’? Curr Opin Crit Care. 2013;19(4):299–307.
Akanji AO, Bruce MA, Frayn KN. Effect of acetate infusion on energy expenditure and substrate oxidation rates in non-diabetic and diabetic subjects. Eur J Clin Nutr. 1989;43(2):107–15.
Akanji AO, Hockaday TD. Acetate tolerance and the kinetics of acetate utilization in diabetic and nondiabetic subjects. Am J Clin Nutr. 1990;51(1):112–8.
Hamada T, Yamamoto M, Nakamaru K, et al. The pharmacokinetics of D‑lactate, L‑lactate and acetate in humans. Masui. 1997;46(2):229–36.
Davidson WD, Rorke SJ, Guo LS, Morin RJ. Comparison of acetate-1-14 C metabolism in uremic and nonuremic dogs. Am J Clin Nutr. 1978;31(10):1897–902.
Kirkendol PL, Starrs J, Gonzalez FM. The effects of acetate, lactate, succinate and gluconate on plasma pH and electrolytes in dogs. Trans Am Soc Artif Intern Organs. 1980;26:323–7.
Kuze S, Ito Y, Miyahara T. Expiration of radioactive carbon dioxide by rats after administration of isotopic lactate and acetate. Acta Med Biol. 1986;34:93–102.
Knowles SE, Jarrett IG, Filsell OH, Ballard FJ. Production and utilization of acetate in mammals. Biochem J. 1974;142(2):401–11.
Iseki K, Onoyama K, Maeda T, et al. Comparison of hemodynamics induced by conventional acetate hemodialysis, bicarbonate hemodialysis and ultrafiltration. Clin Nephrol. 1980;14(6):294–8.
Mansell MA, Nunan TO, Laker MF, Boon NA, Wing AJ. Incidence and significance of rising blood acetate levels during hemodialysis. Clin Nephrol. 1979;12(1):22–5.
Holbert RD, Pearson JE, Gonzalez FM. Effect of sodium acetate infusion on renal function in the dog. Arch Int Pharmacodyn Ther. 1976;219(2):211–22.
Liang CS, Lowenstein JM. Metabolic control of the circulation. Effects of acetate and pyruvate. J Clin Invest. 1978;62(5):1029–38.
Kveim M, Nesbakken R. Acetate metabolizing capacity in man. J Oslo City Hosp. 1980;30:101–4.
Nitenberg A, Huyghebaert MF, Blanchet F, Amiel C. Analysis of increased myocardial contractility during sodium acetate infusion in humans. Kidney Int. 1984;26(5):744–51.
Tolchin N, Roberts JL, Hayashi J, Lewis EJ. Metabolic consequences of high mass-transfer hemodialysis. Kidney Int. 1977;11(5):366–78.
Neavyn MJ, Boyer EW, Bird SB, Babu KM. Sodium acetate as a replacement for sodium bicarbonate in medical toxicology: a review. J Med Toxicol. 2013;9(3):250–4.
Conahan ST, Dupre A, Giaimo ME, et al. Resuscitation fluid composition and myocardial performance during burn shock. Circ Shock. 1987;23(1):37–49.
Hasman H, Cinar O, Uzun A, et al. A randomized clinical trial comparing the effect of rapidly infused crystalloids on acid-base status in dehydrated patients in the emergency department. Int J Med Sci. 2012;9(1):59–64.
Hadimioglu N, Saadawy I, Saglam T, Ertug Z, Dinckan A. The effect of different crystalloid solutions on acid-base balance and early kidney function after kidney transplantation. Anesth Analg. 2008;107(1):264–9.
Shin WJ, Kim YK, Bang JY, et al. Lactate and liver function tests after living donor right hepatectomy: a comparison of solutions with and without lactate. Acta Anaesthesiol Scand. 2011;55(5):558–64.
Gille J, Klezcewski B, Malcharek M, et al. Safety of resuscitation with Ringer’s acetate solution in severe burn (VolTRAB)—an observational trial. Burns. 2014;40(5):871–80.
Coran AG, Ballantine TV, Horwitz DL, Herman CM. The effect of crystalloid resuscitation in hemorrhagic shock on acid-base balance: a comparison between normal saline and Ringer’s lactate solutions. Surgery. 1971;69(6):874–80.
Harper PV Jr., Neal WB Jr., Hlavacek GR. Acetate utilization in the dog. Metabolism. 1953;2(1):62–8.
Zander R. Association between plasma ionized calcium and lactate concentration. Intensive Care Med. 1993;19(6):362–3.
Cooper DJ, Walley KR, Dodek PM, Rosenberg F, Russell JA. Plasma ionized calcium and blood lactate concentrations are inversely associated in human lactic acidosis. Intensive Care Med. 1992;18(5):286–9.
Nadeem A, Salahuddin N, El Hazmi A, et al. Chloride-liberal fluids are associated with acute kidney injury after liver transplantation. Crit Care. 2014;18(6):625.
Kellum JA, Song M, Venkataraman R. Effects of hyperchloremic acidosis on arterial pressure and circulating inflammatory molecules in experimental sepsis. Chest. 2004;125(1):243–8.
Krajewski ML, Raghunathan K, Paluszkiewicz SM, Schermer CR, Shaw AD. Meta-analysis of high- versus low-chloride content in perioperative and critical care fluid resuscitation. Br J Surg. 2015;102(1):24–36.
Shaw AD, Raghunathan K, Peyerl FW, et al. Association between intravenous chloride load during resuscitation and in-hospital mortality among patients with SIRS. Intensive Care Med. 2014;40(12):1897–905.
Shaw AD, Schermer CR, Lobo DN, et al. Impact of intravenous fluid composition on outcomes in patients with systemic inflammatory response syndrome. Crit Care. 2015;19:334.
Reid F, Lobo DN, Williams RN, Rowlands BJ, Allison SP. (Ab)normal saline and physiological Hartmann’s solution: a randomized double-blind crossover study. Clin Sci. 2003;104(1):17–24.
Lobo DN, Stanga Z, Aloysius MM, et al. Effect of volume loading with 1 liter intravenous infusions of 0.9% saline, 4% succinylated gelatine (Gelofusine) and 6% hydroxyethyl starch (Voluven) on blood volume and endocrine responses: a randomized, three-way crossover study in healthy volunteers. Crit Care Med. 2010;38(2):464–70.
Mann DV, Robinson MK, Rounds JD, et al. Superiority of blood over saline resuscitation from hemorrhagic shock: a 31 P magnetic resonance spectroscopy study. Ann Surg. 1997;226(5):653–61.
Chowdhury AH, Cox EF, Francis ST, Lobo DN. A randomized, controlled, double-blind crossover study on the effects of 2‑L infusions of 0.9% saline and plasma-lyte(R) 148 on renal blood flow velocity and renal cortical tissue perfusion in healthy volunteers. Ann Surg. 2012;256(1):18–24.
Gunnerson KJ, Saul M, He S, Kellum JA. Lactate versus non-lactate metabolic acidosis: a retrospective outcome evaluation of critically ill patients. Crit Care. 2006;10(1):R22.
McCluskey SA, Karkouti K, Wijeysundera D, et al. Hyperchloremia after noncardiac surgery is independently associated with increased morbidity and mortality: a propensity-matched cohort study. Anesth Analg. 2013;117(2):412–21.
Haunstetter A, Schulze Icking B, Backs J, Kruger C, Haass M. Differential effects of acidosis, high potassium concentrations, and metabolic inhibition on noradrenaline release and its presynaptic muscarinic regulation. Pharmacol Res. 2002;45(3):221–8.
Seyfarth M, Feng Y, Hagl S, et al. Effect of myocardial ischemia on stimulation-evoked noradrenaline release. Modulated neurotransmission in rat, guinea pig, and human cardiac tissue. Circ Res. 1993;73(3):496–502.
Pedoto A, Caruso JE, Nandi J, et al. Acidosis stimulates nitric oxide production and lung damage in rats. Am J Respir Crit Care Med. 1999;159(2):397–402.
Young JB, Utter GH, Schermer CR, et al. Saline versus Plasma-Lyte A in initial resuscitation of trauma patients: a randomized trial. Ann Surg. 2014;259(2):255–62.
Almac E, Aksu U, Bezemer R, et al. The acute effects of acetate-balanced colloid and crystalloid resuscitation on renal oxygenation in a rat model of hemorrhagic shock. Resuscitation. 2012;83(9):1166–72.
McFarlane C, Lee A. A comparison of plasmalyte 148 and 0.9% saline for intra-operative fluid replacement. Anaesthesia. 1994;49(9):779–81.
Kim SY, Huh KH, Lee JR, et al. Comparison of the effects of normal saline versus plasmalyte on acid-base balance during living donor kidney transplantation using the Stewart and base excess methods. Transplant Proc. 2013;45(6):2191–6.
Schick MA, Isbary JT, Stueber T, et al. Effects of crystalloids and colloids on liver and intestine microcirculation and function in cecal ligation and puncture induced septic rodents. BMC Gastroenterol. 2012;12:179.
Song JW, Shim JK, Kim NY, Jang J, Kwak YL. The effect of 0.9% saline versus plasmalyte on coagulation in patients undergoing lumbar spinal surgery; a randomized controlled trial. Int J Surg. 2015;20:128–34.
Wilcox CS. Regulation of renal blood flow by plasma chloride. J Clin Invest. 1983;71(3):726–35.
Wilcox CS, Peart WS. Release of renin and angiotensin II into plasma and lymph during hyperchloremia. Am J Physiol. 1987;253(4 Pt 2):F734–F41.
Yunos NM, Bellomo R, Glassford N, et al. Chloride-liberal vs. chloride-restrictive intravenous fluid administration and acute kidney injury: an extended analysis. Intensive Care Med. 2015;41(2):257–64.
Guirgis FW, Williams DJ, Hale M, et al. The relationship of intravenous fluid chloride content to kidney function in patients with severe sepsis or septic shock. Am J Emerg Med. 2015;33(3):439–43.
Shao M, Li G, Sarvottam K, et al. Dyschloremia is a risk factor for the development of acute kidney injury in critically ill patients. PLoS ONE. 2016;11(8):e160322.
Suetrong B, Pisitsak C, Boyd JH, Russell JA, Walley KR. Hyperchloremia and moderate increase in serum chloride are associated with acute kidney injury in severe sepsis and septic shock patients. Crit Care. 2016;20(1):315.
McIlroy D, Murphy D, Kasza J, et al. Effects of restricting perioperative use of intravenous chloride on kidney injury in patients undergoing cardiac surgery: the LICRA pragmatic controlled clinical trial. Intensive Care Med. 2017;43(6):795–806.
Sadan O, Singbartl K, Kandiah PA, Martin KS, Samuels OB. Hyperchloremia is associated with acute kidney injury in patients with subarachnoid hemorrhage. Crit Care Med. 2017;45(8):1382. https://doi.org/10.1097/CCM.0000000000002497.
Bellomo R, Kellum JA, Ronco C, et al. Acute kidney injury in sepsis. Intensive Care Med. 2017;43(6):816–28.
Lowell JA, Schifferdecker C, Driscoll DF, Benotti PN, Bistrian BR. Postoperative fluid overload: not a benign problem. Crit Care Med. 1990;18(7):728–33.
Cheng AT, Plank LD, Hill GL. Prolonged overexpansion of extracellular water in elderly patients with sepsis. Arch Surg. 1998;133(7):745–51.
Prowle JR, Chua HR, Bagshaw SM, Bellomo R. Clinical review: volume of fluid resuscitation and the incidence of acute kidney injury—a systematic review. Crit Care. 2012;16(4):230.
Prowle JR, Kirwan CJ, Bellomo R. Fluid management for the prevention and attenuation of acute kidney injury. Nat Rev Nephrol. 2014;10(1):37–47.
Boyd JH, Forbes J, Nakada TA, Walley KR, Russell JA. Fluid resuscitation in septic shock: a positive fluid balance and elevated central venous pressure are associated with increased mortality. Crit Care Med. 2011;39(2):259–65.
De Pietri L, Montalti R, Begliomini B. Anaesthetic perioperative management of patients with pancreatic cancer. World J Gastroenterol. 2014;20(9):2304–20.
Tambyraja AL, Sengupta F, MacGregor AB, Bartolo DC, Fearon KC. Patterns and clinical outcomes associated with routine intravenous sodium and fluid administration after colorectal resection. World J Surg. 2004;28(10):1046–51. Discussion 1051-2. Epub 2004 Sep 29.
Gil MJ, Franch G, Guirao X, et al. Response of severely malnourished patients to preoperative parenteral nutrition: a randomized clinical trial of water and sodium restriction. Nutrition. 1997;13(1):26–31.
Arieff AI. Fatal postoperative pulmonary edema: pathogenesis and literature review. Chest. 1999;115(5):1371–7.
Alsous F, Khamiees M, DeGirolamo A, Amoateng-Adjepong Y, Manthous CA. Negative fluid balance predicts survival in patients with septic shock: a retrospective pilot study. Chest. 2000;117(6):1749–54.
Brandstrup B, Tonnesen H, Beier-Holgersen R, et al. Effects of intravenous fluid restriction on postoperative complications: comparison of two perioperative fluid regimens: a randomized assessor-blinded multicenter trial. Ann Surg. 2003;238(5):641–8.
Nisanevich V, Felsenstein I, Almogy G, et al. Effect of intraoperative fluid management on outcome after intraabdominal surgery. Anesthesiology. 2005;103(1):25–32.
Lobo DN, Bostock KA, Neal KR, et al. Effect of salt and water balance on recovery of gastrointestinal function after elective colonic resection: a randomised controlled trial. Lancet. 2002;359(9320):1812–8.
Bihari S, Ou J, Holt AW, Bersten AD. Inadvertent sodium loading in critically ill patients. Crit Care Resusc. 2012;14(1):33–7.
Lindner G, Funk GC, Schwarz C, et al. Hypernatremia in the critically ill is an independent risk factor for mortality. Am J Kidney Dis. 2007;50(6):952–7.
Jensen JM, Mose FH, Bech JN, Nielsen S, Pedersen EB. Effect of volume expansion with hypertonic- and isotonic saline and isotonic glucose on sodium and water transport in the principal cells in the kidney. BMC Nephrol. 2013;14:202.
Jarvela K, Koobi T, Kauppinen P, Kaukinen S. Effects of hypertonic 75 mg/ml (7.5%) saline on extracellular water volume when used for preloading before spinal anaesthesia. Acta Anaesthesiol Scand. 2001;45(6):776–81.
Lavu H, Sell NM, Carter TI, et al. The HYSLAR trial: a prospective randomized controlled trial of the use of a restrictive fluid regimen with 3% hypertonic saline versus lactated Ringers in patients undergoing pancreaticoduodenectomy. Ann Surg. 2014;260(3):445–53. discussion 53–5.
Cross JS, Gruber DP, Burchard KW, et al. Hypertonic saline fluid therapy following surgery: a prospective study. J Trauma. 1989;29(6):817–25. discussion 25–6.
Orbegozo D, Su F, Santacruz C, et al. Effects of different crystalloid solutions on Hemodynamics, peripheral perfusion, and the microcirculation in experimental abdominal sepsis. Anesthesiology. 2016;125(4):744–54.
Pfortmueller C, Funk GC, Potura E, et al. Acetate-buffered crystalloid infusate versus infusion of 0.9% saline and hemodynamic stability in patients undergoing renal transplantation : prospective, randomized, controlled trial. Wien Klin Wochenschr. 2017;129(17):598. https://doi.org/10.1007/s00508-017-1180-4.
Pfortmueller CA, Funk G‑C, Reiterer C, Schrott A, Zotti O, Kabon B, Fleischmann E, Lindner G. Normal saline versus a balanced crystalloid for goal-directed perioperative fluid therapy in major abdominal surgery: a double-blind randomised controlled study. Br J Anesth. 2018;120(2):274–83.
Wray S. Smooth muscle intracellular pH: measurement, regulation, and function. Am J Physiol. 1988;254(2 Pt 1):C213–C25.
Daugherty RM Jr., Scott JB, Dabney JM, Haddy FJ. Local effects of O2 and CO2 on limb, renal, and coronary vascular resistances. Am J Physiol. 1967;213(5):1102–10.
Haddy FJ, Scott JB. Metabolically linked vasoactive chemicals in local regulation of blood flow. Physiol Rev. 1968;48(4):688–707.
Le Tulzo Y, Shenkar R, Kaneko D, et al. Hemorrhage increases cytokine expression in lung mononuclear cells in mice: involvement of catecholamines in nuclear factor-kappaB regulation and cytokine expression. J Clin Invest. 1997;99(7):1516–24.
Chang-Seng LLJ. Metabolic control of the circulation: effects of acetat and pyruvate. J Clin Investig. 1978;62:1029–38.
Ward RA, Wathen RL, Harding GB, Thompson LC. Comparative metabolic effects of acetate and dichloroacetate infusion in the anesthetized dog. Metabolism. 1985;34(7):680–7.
Burnier P, Tappy L, Jequier E, Schneeberger D, Chiolero R. Metabolic and respiratory effects of infused sodium acetate in healthy human subjects. Am J Physiol. 1992;263(6 Pt 2):R1271–R6.
Suokas A, Kupari M, Heikkila J, Lindros K, Ylikahri R. Acute cardiovascular and metabolic effects of acetate in men. Alcohol Clin Exp Res. 1988;12(1):52–8.
Kellum JA, Song M, Almasri E. Hyperchloremic acidosis increases circulating inflammatory molecules in experimental sepsis. Chest. 2006;130(4):962–7.
Soussi S, Ferry A, Chaussard M, Legrand M. Chloride toxicity in critically ill patients: what’s the evidence? Anaesth Crit Care Pain Med. 2017;36(2):125–30.
Phillips CR, Vinecore K, Hagg DS, et al. Resuscitation of haemorrhagic shock with normal saline vs. lactated Ringer’s: effects on oxygenation, extravascular lung water and haemodynamics. Crit Care. 2009;13(2):R30.
Khan R, Kirschenbaum LA, Larow C, Astiz ME. The effect of resuscitation fluids on neutrophil-endothelial cell interactions in septic shock. Shock. 2011;36(5):440–4.
Rhee P, Wang D, Ruff P, et al. Human neutrophil activation and increased adhesion by various resuscitation fluids. Crit Care Med. 2000;28(1):74–8.
Kiraly LN, Differding JA, Enomoto TM, et al. Resuscitation with normal saline (NS) vs. lactated ringers (LR) modulates hypercoagulability and leads to increased blood loss in an uncontrolled hemorrhagic shock swine model. J Trauma. 2006;61(1):57–64. discussion–5.
Todd SR, Malinoski D, Muller PJ, Schreiber MA. Lactated Ringer’s is superior to normal saline in the resuscitation of uncontrolled hemorrhagic shock. J Trauma. 2007;62(3):636–9.
Raghunathan K, Bonavia A, Nathanson BH, et al. Association between initial fluid choice and subsequent in-hospital mortality during the resuscitation of adults with septic shock. Anesthesiology. 2015;123(6):1385–93.
Sponholz C, Schelenz C, Reinhart K, Schirmer U, Stehr SN. Catecholamine and volume therapy for cardiac surgery in Germany—results from a postal survey. PLoS ONE. 2014;9(8):e103996.
Bayer O, Schwarzkopf D, Doenst T, et al. Perioperative fluid therapy with tetrastarch and gelatin in cardiac surgery—a prospective sequential analysis*. Crit Care Med. 2013;41(11):2532–42.
Hans GA, Ledoux D, Roediger L, et al. The effect of intraoperative 6% balanced hydroxyethyl starch (130/0.4) during cardiac surgery on transfusion requirements. J Cardiothorac Vasc Anesth. 2015;29(2):328–32.
Verheij J, van Lingen A, Raijmakers PG, et al. Effect of fluid loading with saline or colloids on pulmonary permeability, oedema and lung injury score after cardiac and major vascular surgery. Br J Anaesth. 2006;96(1):21–30.
Jacob M, Fellahi JL, Chappell D, Kurz A. The impact of hydroxyethyl starches in cardiac surgery: a meta-analysis. Crit Care. 2014;18(6):656.
Magder S, Potter BJ, Varennes BD, Doucette S, Fergusson D. Fluids after cardiac surgery: a pilot study of the use of colloids versus crystalloids. Crit Care Med. 2010;38(11):2117–24.
Mehta Y, Arora D. Newer methods of cardiac output monitoring. World J Cardiol. 2014;6(9):1022–9.
Funding
No external funding war received for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
C.A. Pfortmueller, B. Kabon, J.C. Schefold, and E. Fleischmann declare that they have no competing interests.
Additional information
Authors’ contributions
Carmen A. Pfortmueller: designed the strategy, performed the literature review, drafted the manuscript. Barbara Kabon: revision for important intellectual content. Joerg C. Schefold: revision for important intellectual content. Edith Fleischmann: revision for important intellectual content
Rights and permissions
About this article
Cite this article
Pfortmueller, C.A., Kabon, B., Schefold, J.C. et al. Crystalloid fluid choice in the critically ill. Wien Klin Wochenschr 130, 273–282 (2018). https://doi.org/10.1007/s00508-018-1327-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00508-018-1327-y