Abstract
This study aimed to better understand environmental heat stress and physiological heat strain indicators in lactating dairy cows. Sixteen heat stress indicators were derived using microenvironmental parameters that were measured at the surrounding of cows and at usual fixed locations in the barn by using handheld and fixed subarea sensors, respectively. Twenty high-producing Holstein–Friesian dairy cows (> 30.0 kg/day) from an intensive dairy farm were chosen to measure respiration rate (RR), vaginal temperature (VT), and body surface temperature of forehead (FT), eye (ET), and muzzle (MT). Our results show that microenvironments measured by the handheld sensor were slightly warmer and drier than those measured by the fixed subarea sensor; however, their derived heat stress indicators correlated equally well with physiological indicators. Interestingly, ambient temperature (Ta) had the highest correlations with physiological indicators and the best classification performance in recognizing actual heat strain state. Using segmented mixed models, the determined Ta thresholds for maximum FT, mean FT, RR, maximum ET, mean ET, VT, mean MT, and maximum MT were 24.1 °C, 24.2 °C, 24.4 °C, 24.6 °C, 24.6 °C, 25.3 °C, 25.4 °C, and 25.4 °C, respectively. Thus, we concluded that the fixed subarea sensor is a reliable tool for measuring cows’ microenvironments; Ta is an appropriate heat stress indicator; FT, RR, and ET are good early heat strain indicators. The results of this study could be helpful for dairy practitioners in a similar intensive setting to detect and respond to heat strain with more appropriate indicators.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Heat waves are predicted to increase in both frequency and magnitude (Ranjitkar et al. 2020), putting dairy cows at greater risk due to their high heat production but limited heat dissipation capacity (Kadzere et al. 2002). In the present study, heat stress refers to the total amount of environmental heat stressors that animals are exposed to, whereas heat strain refers to the overall animal response induced by heat stress. These two concepts are separated for better interpreting the indicators from environment and animal perspectives, respectively.
To characterize thermal environment, efforts have been made for decades to develop thermal indices (TIs), among which temperature-humidity indices (THIs) as a family of indices incorporating environmental temperature and humidity are most prevalent in both studies and practice. Recently, advanced TIs compassing more components of thermal environment have been proposed, adding wind speed (WS) and solar radiation (SR), both of relevance contributing to creating the thermal environment of the cow (Mader et al. 2010; Wang et al. 2018). Mostly, validation of TIs has been done based on their correlations with animal-based heat strain indicators (Li et al. 2009; Ji et al. 2020; Yan et al. 2020). However, results are highly inconsistent among relevant studies due to huge differences in climate, housing, management, and animal, stressing the fact that the selection of TIs should continue in specific regions for both study and production purposes.
The location of sensors greatly impacts the resulting calculation of TIs especially when weather data came from nearby weather stations (Wijffels et al. 2021). Sensors have been placed on cows to measure the closest cows’ microenvironments (Schüller and Heuwieser 2016). However, in practice, there is a trade-off between the cost and the benefits of deploying more accurate microenvironmental sensors. Microenvironmental sensors were deployed more commonly at single (center) or multiple (equally spaced) fixed locations in the barn (Li et al. 2020; Pinto et al. 2020). Due to the movement of cows, the fixed measurement is sometimes close to the cow and sometimes far away from the cow, which may affect the accuracy of the microenvironmental measurement. Therefore, it should be investigated whether the use of these fixed measurements is adequate to represent the microenvironments to which cows are exposed.
Segmented models and polynomial models are standard methods performed to fit animal-based variables as functions of environmental variables (Carabaño et al. 2016). Although polynomial models have shown better fit, segmented models are most welcomed and used due to high interpretability and sufficient goodness of fit. More importantly, TI thresholds for different animal-based heat strain indicators can help determine which animal indicator reacts first to heat stress. Growing attention has recently been paid to earlier heat abatement by using the most sensitive physiological heat strain indicators (Hoffmann et al. 2020). Body surface temperatures (BSTs) have shown to be good early indicators (Dalcin et al. 2016; Amamou et al. 2019; Wang et al. 2018), among which facial parts might be promising locations for the probability to be integrated with a cow facial recognition system (Qiao et al. 2021). Locations and thresholds, however, are still worth investigating. Thresholds developed by relevant studies are difficult to compare directly, particularly when they were determined using different animals, weather conditions, and analytic methodologies (Carabaño et al. 2016).
To explore the abovementioned questions, the present study was conducted to comprehensively evaluate both heat stress and heat strain indicators. Specifically, the objectives of this study were to (1) investigate the differences between microenvironmental parameters measured at the surrounding of cows and those measured at usual fixed locations in the barn; (2) evaluate environmental indicators’ correlations with physiological indicators and their ability in recognizing cows’ actual heat strain states; and (3) compare the sensitivity of physiological indicators to heat stress.
We hypothesized that fixed subarea sensors would be good in measuring cows’ microenvironment; heat stress indicators would perform differently in representing thermal environment; and BST of specific locations would provide an alternative in reflecting heat strain in dairy cows.
Materials and methods
Location
The present study was carried out for 20 days from May to June 2021 at an organic intensive dairy farm in Shandong, China (coordinates: 34° 50′ 37″ N, 115° 26′ 11″ E; altitude: 52 m), which belongs to a temperate continental monsoon climate. This timepoint was selected to capture the pattern of changes in physiological heat strain indicators when the weather got warmer from spring to summer and cows first began to feel thermal discomfort.
Housing, animals, and management
The dairy farm was an organic intensive system that relied totally on home-grown feed and was free of antibiotics. The study free-stall barn (15 m × 90 m, oriented along the north–south longitudinal axis) had the capacity of 122 cows, with concrete floors and no outdoor area. The double-pitched roof covered the entire area of the barn and therefore prevented most direct SR from affecting the cows indoor.
Cows with milk yield above 30.0 kg/day (the average of previous 3 days), in mid-lactation (between 100 and 200 days in milk (DIM)), and normal body condition score (BCS; within 2.75–3.50) (Wildman et al. 1982) were screened out. BCS was evaluated by two trained raters with a high agreement (intraclass correlation coefficient: 0.96). Twenty-six high-producing Holstein–Friesian cows met the inclusion criteria, and 20 of them were randomly selected. Mean ± standard deviation daily milk yield, parity, DIM, and BCS of selected cows were 40.9 ± 5.1 kg/day, 2.7 ± 0.9, 150.2 ± 21.1, and 3.0 ± 0.2, respectively. The cows were milked three times daily at 0830–0910 h, 1630–1710 h, and 0000–0040 h, and were fed a total mixed ration three times daily after milked. The cows had free access to water. Cow information, including daily milk yield, DIM, and parity, was acquired from the milking system (Afimilk, Kibbutz Afikim, Israel).
Cooling measures included electronic fans (turned on when indoor temperature reaching 20 °C) and sprinklers. The former was installed at lying zone at an interval of 6 m and along feeding line at an interval of 12 m, while the latter was installed along feeding line at an interval of 2 m. Sprinklers were used since the 11th test day according to the cooling management of the farm. To eliminate the interference of evaporative cooling by sprinklers, only the observations from the first 10 test days were entered into analysis.
Physiological measurement
Respiration rate (RR), vaginal temperature (VT), and three BSTs measured at forehead (FT), eye (ET; medial canthus), and muzzle (MT) were used as physiological heat strain indicators. Two trained observers with a high agreement (intraclass correlation coefficient: 0.91) determined RR by timing 15 flank movements and converting to breaths per min (bpm). VT was measured every 5 min using a data logger (DS1922L, Maxim Integrated, San Jose, CA, USA) attached to a blank vaginal controlled internal drug release (CIDR; Pfizer Animal Health, New York, NY, USA). CIDRs were kept intravaginally for a week since the first test day. FT, ET, and MT were taken by a photographer using a portable infrared camera (VarioCAM HR, InfraTec, Dresden, Germany) with a resolution of 640 × 480 pixels. Infrared photography was performed right after RR observation. The emissivity was set to 0.98 and the distance between cows and camera was approximately 1.5 m, as per previous studies (Peng et al. 2019). The infrared images were interpreted and calibrated with IRBIS 3 Standard software (YSHY, Beijing, China).
Physiological measurement was conducted twice on each of the 10 test days (0800–1130 and 1330–1630). For each measurement, each cow was expected to be observed twice, once when standing and once when lying, contributing to 80 observations expected for each test day. For every observation, the location of the test cow was recorded for further merging with the corresponding fixed subarea sensor.
Microenvironmental measurement and thermal indices calculation
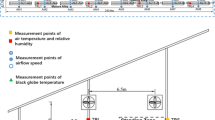
Microenvironmental parameters were simultaneously measured at the surrounding of cows and at usual fixed locations in the barn by using handheld and fixed sensors, respectively. For handheld measurement, a Kestrel 5400 heat stress tracker (Nielsen-Kellerman, Boothwyn, PA, USA) was held in hand by the photographer at a distance of 1.5 m from the cows. The environmental parameters were averaged during the process of infrared photography for every observation. For fixed measurement, environmental parameters were recorded every 10 min with Kestrel 5000 environment meters and Kestrel 5400 heat stress trackers (Nielsen-Kellerman, Boothwyn, PA, USA). Specifically, Kestrel 5000 measured ambient temperature (Ta), wet bulb temperature (Tw), dew point temperature (Tdp), relative humidity (RH), and WS, and Kestrel 5400 measured black globe temperature (Tbg) and wet bulb globe temperature (WBGT) additionally. The barn was divided into six subareas (three in the lying zone and three in the standing zone; Fig. 1). Sensors were fixed 2.2 m above the ground in each subarea to avoid being destroyed by the cows and to get as close to them as possible. Besides, SR was continuously recorded every 5 min with a weather station (H21 weather station and 3-LIB-M003 pyranometer, Onset Computer Co. Ltd., Bourne, MA, USA).
The environmental parameters were further used for calculating TIs listed in Table 1. Besides, Ta, Tbg, and WBGT were regarded as separate heat stress indicators and entered directly into the following analysis, contributing to 16 candidate heat stress indicators in total. For every observation, the derivation of heat stress indicators used microenvironmental data from two sources (i.e., the handheld sensor and the fixed subarea sensor). The fixed subarea sensors were determined by merging with the location of the test cows.
Statistical analysis
Data were summarized as mean, standard deviation, minimum, and maximum. Two-way analyses of variance (ANOVA) including interaction were performed to investigate the effect of zone (lying vs. standing) and source of sensors (handheld vs. fixed subarea) on microenvironmental parameters. A Bland–Altman plot was used to reveal the difference between handheld and fixed temperature readings (Altman and Bland 1983). To select heat stress indicator, correlation analyses between heat stress and heat strain indicators based on Pearson correlation coefficient were performed first. The classification performances of heat stress indicators were compared then using receiver operator characteristic (ROC) curves in which the actual heat strain state was determined using a RR threshold of 48 bpm and a VT threshold of 38.6 °C, respectively (Li et al. 2020). The performance in correlation and ROC analyses as well as the ease of obtaining data was considered to select the most appropriate heat stress indicator. The selected heat stress indicator was further conveyed to segmented mixed models to customize the thresholds with physiological heat strain indicators. Random intercept effect was included for cow. The model can be written as follows:
where Y is the physiological heat strain indicators, β0 is the population intercept, u0i is the random intercept effect for i-th cow, X is the selected heat stress indicator, Xbp is the breakpoint, Xk is the dummy variable, β1 is the left slope, β2 is the difference between right slope and left slope, and εij is the random residual for the j-th observation.
All statistical analyses were performed using R version 3.4.4 (https://www.R-project.org/). Two-way ANOVA were performed using ANOVA function included in “car” package. Correlation analyses were performed using the cor function. ROC curves were plotted using the “pROC” package and their areas under the curve (AUCs) were compared using roc.test function. The basic linear mixed models were fitted using lme function included in the “nlme” package, and then updated using the “segmented” package. Significance was declared at P < 0.05 and tendency was declared at 0.05 < P < 0.10.
Results
Environmental and physiological description
During the study, all temperature parameters followed a similar trajectory, whereas RH followed the opposite (Fig. 2). Consistently, Tbg was slightly higher than Ta, and their trajectories almost coincided. Only a few SR readings exceeded zero, all of which occurred during sunset and sunrise when sunlight shined inside from the side of the barn.
All microenvironmental parameters were higher in the afternoon than in the morning, except for Tdp and RH which were higher in the morning (Table 2). All physiological parameters were higher in the afternoon than in the morning except for mean and maximum ET which remained unchanged during the daytime (Table 2). When summarizing daily mean, a wide range was found for all microenvironmental and physiological parameters (Table 2).
Differences in microenvironmental parameters measured by handheld and fixed subarea sensors
There was a tendency for higher Ta measured by the handheld sensor than measured by the fixed subarea sensor (P = 0.056; Table 3), and the difference was 0.3 °C and 0.6 °C for lying and standing zones, respectively. The Tw measured by the handheld sensor was significantly lower than the Tw measured by the fixed subarea sensor (P < 0.0001), in which the difference was smaller at the lying zone than at the standing zone with a tendency of a zone by source interaction (− 0.4 °C vs. − 1.0 °C, P = 0.066). There was a zone by source interaction for Tdp (P = 0.013), with a smaller difference between handheld and fixed subarea readings obtained at the lying zone than that obtained at the standing zone (− 0.9 °C vs. − 2.1 °C). Similarly, a zone by source interaction was found for RH (P = 0.001), with a smaller difference between handheld and fixed subarea readings obtained at lying zone than that obtained at the standing zone (− 4.8% vs. − 11.3%). There was a zone by source interaction for WS (P < 0.0001), whereby the handheld sensor had lower readings than the fixed subarea sensor when measuring at lying zone (1.3 m/s vs. 2.2 m/s), but higher readings than it when measuring at the standing zone (0.9 m/s vs. 0.4 m/s). Besides, the lying zone had a higher Tbg than the standing zone (P < 0.0001).
The Bland–Altman plot comparing the difference between handheld and fixed Ta readings is shown in Fig. 3, where a date-related color gradient was added to facilitate visualizing the temporal variation of Ta during this study. In general, Ta increased as time went by. The difference at both lying and standing zones shrank as Ta increased, and the mean difference was nearly zero when Ta reached above 30 °C.
A Bland–Altman plot showing the ambient temperature difference between handheld and fixed subarea readings measured at a the lying zone and b the standing zone. The limits of agreement are shown with dashed horizontal lines above and below the horizontal solid lines indicating the mean value (95% CI of a and b: − 0.8 to 1.4 and − 0.5 to 1.7, respectively; mean difference of a and b: 0.3 and 0.6, respectively). Linear regression lines are shown in red
Correlations between heat stress and heat strain indicators
Pearson correlation coefficients between heat stress and heat strain indicators presented in Fig. 4 show that heat stress indicators derived using data from the handheld sensor had slightly higher correlations with most physiological indicators than heat stress indicators derived using data from the fixed subarea sensor. Besides, FT correlated most with heat stress indicators, followed by MT, RR, ET, and VT. For FT and ET, mean temperature yielded higher correlations with most heat stress indicators than maximum temperature. For MT, maximum temperature performed slightly better in correlations with heat stress indicators than mean temperature.
Pearson correlation coefficients between heat strain indicators and heat stress indicators derived using data from a handheld sensor and b fixed subarea sensor. RR, respiration rate (breaths per min); VT, vaginal temperature (°C); FT, forehead temperature (°C); ET, eye temperature (°C); MT, muzzle temperature (°C); THI, temperature-humidity index; THIadj, adjusted temperature-humidity index; BGHI, black globe-humidity index; CCI, comprehensive climate index; HLI, heat load index; DHLI, dairy heat load index; ETI, equivalent temperature index; ETIC, equivalent temperature index for cattle; WBGT, wet bulb globe temperature (°C); Ta, ambient temperature (°C); Tbg, black globe temperature (°C). WBGT, Ta, and Tbg were obtained directly by environmental sensors. Other thermal indices were calculated based on formulas listed in Table 1
Specifically, when using handheld sensor, six physiological indicators (RR, mean FT, maximum FT, mean ET, mean MT, and maximum MT) yielded the highest correlation with Ta (r = 0.58, 0.84, 0.81, 0.52, 0.67, 0.67, respectively, all P < 0.0001; Fig. 4a). When using fixed subarea sensor, seven physiological indicators (RR, VT, mean FT, maximum FT, mean ET, mean MT, and maximum MT) yielded the highest correlation with Ta (r = 0.57, 0.36, 0.83, 0.79, 0.50, 0.66, 0.66, respectively, all P < 0.0001; Fig. 4b).
Classification performance of heat stress indicators
The four ROC curves with the highest AUCs among 16 candidate heat stress indicators are shown in Fig. 5. When using a RR of 48 bpm (Li et at. 2020) to determine the actual heat strain state, the AUC of Tbg (0.749) was significantly higher than that of THI1 (0.725) or THI5 (0.724) (P = 0.017, 0.015, respectively; Fig. 5a). Besides, Ta had the second highest AUC (0.745) which was significantly higher than that of THI1 or THI5 (P = 0.036, 0.031, respectively; Fig. 5a). When using a VT of 38.6 °C (Li et at. 2020) to determine the actual heat strain state, the AUC of Ta (0.662) was the highest, and no significant difference was found among the AUCs of the best four ROC curves (all P > 0.05; Fig. 5b).
The receiver operator characteristic (ROC) curves of the four heat stress indicators with the highest area under the curve (AUC) on recognizing cows’ actual heat strain states based on a respiration rate and b vaginal temperature. Tbg, black globe temperature (°C); Ta, ambient temperature (°C); THI, temperature-humidity index; BGHI, black globe-humidity index. THIs and BGHI were calculated based on formulas listed in Table 1
Threshold development and comparison
The slopes of the segmented model for RR indicate that for every one unit increase in Ta below and above 24.4 °C, RR increased by 1.53 bpm and 3.37 bpm, respectively (Table 4). The VT was relatively stable with a slope of 0.02 when Ta was below 25.3 °C, representing the presence of a plateau period. Then, VT increased by 0.11 °C for every one unit increase in Ta when Ta exceeded 25.3 °C. Similarly, the left slopes for mean and maximum ET were fairly small of 0.02 and − 0.02, respectively. Mean and maximum ET began to increase by 0.17 °C and 0.12 °C, respectively, for every one unit increase in Ta when Ta exceeded 24.6 °C. For FT, a greater increase per unit Ta was witnessed for mean FT when Ta exceeded 24.2 °C (0.61 °C vs. 0.16 °C), and for maximum FT when Ta exceeded 24.1 °C (0.54 °C vs. 0.14 °C). For MT, a greater increase per unit Ta was witnessed for mean MT (0.30 °C vs. 0.10 °C) and maximum MT (0.23 °C vs. 0.11 °C) when Ta exceeded 25.4 °C.
A date-related color gradient was added to the fitted models to facilitate visualizing the temporal variation of Ta during this study (Fig. 6). In general, Ta increased as time went by, indicating that the changes in physiological indicators well captured the onset of heat strain in dairy cows. The comparison of the thresholds (Fig. 7) shows that maximum FT, mean FT, RR, maximum ET, mean ET, VT, mean MT, and maximum MT began to increase sequentially when Ta exceeded 24.1 °C, 24.2 °C, 24.4 °C, 24.6 °C, 24.6 °C, 25.3 °C, 25.4 °C, and 25.4 °C, respectively. The corresponding heat strain thresholds for maximum FT, mean FT, RR, maximum ET, mean ET, VT, mean MT, and maximum MT were 31.1 °C, 30.1 °C, 53.8 bpm, 37.5 °C, 36.7 °C, 38.7 °C, 35.1 °C, and 36.1 °C, respectively.
Observed values and predictions of segmented mixed models for a respiration rate, b vaginal temperature, c mean forehead temperature, d maximum forehead temperature, e mean eye temperature, f maximum eye temperature, g mean muzzle temperature, and h maximum muzzle temperature. 95% CI of the breakpoints are marked as a line above the x-axis
Discussion
Differences in microenvironmental parameters measured by handheld and fixed subarea sensors
The environmental parameters of different locations inside the barn are not homogeneously distributed due to the interaction of building layout and cooling measures such as ventilation and sprinkler facilities (Collier et al. 2006; Mondaca et al. 2019; Herbut 2013). Additionally, cows dissipate heat through a variety of ways, including excretion, sweating, and panting, all of which contribute to the formation of the microenvironment (Kadzere et al. 2002; Schüller and Heuwieser 2016). In the present study, the handheld sensor recorded higher Ta and lower RH compared with the fixed subarea sensor at both lying and standing zones. This tendency is consistent with Schüller and Heuwieser (2016) who found that the Ta and RH measured using mobile loggers mounted on cows were 1.56 °C higher and 1.75% lower than measured using stationary loggers, respectively. Our results could not be directly compared numerically with the results of Schüller and Heuwieser (2016) due to the differences in barn structure, cooling measures, and measurement distance. However, closer measurement in their study (30 cm to body surface) should result in a higher temperature record to some extent.
In theory, the closer the sensor is placed to the cow, the more precisely the microenvironment the cow is exposed to may be estimated. However, microenvironmental sensors worn on cows may have some problems during long-term continuous measurement, such as being easily crushed, contaminated by feed dust, lost, and difficult to calibrate regularly, resulting in inaccurate and incomplete data. Measuring the precise microenvironment around cows in the barn is impractical unless the material and cost issues are solved. On the other hand, fixed subarea measurement is more accessible and cost-effective, since the correlations with physiological indicators were not greatly reduced. Besides, due to the combined effect of the air heated by cows rising with air pressure and the sunlight radiating downward from the roof, Ta was expected to drop first and then rise from the ground to the roof (Hempel et al. 2018). Our results show that the difference in Ta between handheld and fixed (height: 2.2 m) readings was reduced to about 0.3–0.5 °C around the threshold of heat strain, and further to almost zero when Ta increased above 30 °C, making fixed subarea readings being a reliable representative of cows’ microenvironments. This also indicates that the Ta gradient became much less favorable to the cows since heat was unable to passively flow from the cow to the environment. The critical point (30 °C) is very close to the finding of Maia et al. (2005) that cattle can gain sensible heat when air temperature is close to and above 32 °C. There have been some actual cases where a series of sensors were deployed to accurately measure the microenvironmental variables of subareas and further informed the activation of local environment-controlled cooling measures (Chen and Chen 2019). By combining microenvironmental sensors with a cow perception system, local cooling equipment can be automatically turned on and off according to the presence of cows, allowing the energy to be saved and the cows to be cooled more precisely. As a result, the readings of fixed subarea sensors were used for the subsequent threshold analysis in this study.
The present study provides more information by considering the difference between lying and standing zones. No zone by source interaction was found for Ta and Tbg. However, a large spatial variation of RH was found between lying and standing zones, in which the variation was greater within the standing zone. This effect further resulted in the trending or significant interaction of other humidity-related parameters (i.e., Tw and Tdp). These results are consistent with a previous study conducted in two naturally ventilated dairy barns in which the observed spatial deviations were reported to be as high as approximately ± 20% (Hempel et al. 2018). Therefore, it is preferable to measure the lying and standing zones separately, and a single or too few sensors might be insufficient to account for these variations especially for studies focusing on the relationship between environment and animals.
Selection of heat stress indicators
In the present study, Ta was selected as the most appropriate heat stress indicator since it had the highest correlations with most physiological heat strain indicators, the best classification performance in recognizing the actual heat strain state, and the easiest access. This is not the first study that reported Ta as an appropriate heat stress indicator for dairy cows. Similar results have been demonstrated in temperate climates (Kovács et al. 2018; Li et al. 2020; Tian et al. 2021), Mediterranean climates (Davison et al. 2020), tropical climates (Kabuga 1992; Mbuthia et al. 2021), and subtropical climates (Dikmen and Hansen 2009; Ji et al. 2020).
In this study, cows were kept in an area with a typical temperate continental monsoon climate. This kind of climate is characterized by the lack of transition throughout late spring and early summer, leaving animals with no time to adapt and suffering from acute heat strain (Carabaño et al. 2016). Therefore, this study focused on the sudden heat stress event happened in May to capture the pattern of changes in physiological heat strain indicators when cows first experienced thermal discomfort during the cycle of hot and cold climate in the same year. Our results show a huge environmental variation beginning with a sudden increase in Ta accompanied by a decrease in RH, whereas THI did not change much in the early days of this study. Therefore, Ta was dominant and RH did not play an important role in the thermal environment during this study, which might lead to Ta alone being more effective as a heat stress indicator. However, TIs encompassing RH (e.g., THI) are expected to be superior at characterizing the severity of heat stress during humid months when RH has a greater impact (Bohmanova et al. 2007).
Although more complicated TIs taking into account SR and WS have shown good performances in predicting heat strain indicators in cattle (Li et al. 2009; Wang et al. 2018; Yan et al. 2020; Hammami et al. 2013), they performed poorly in this study. This is mainly because WS and SR are less dominant in indoor settings (Li et al. 2020; Gorczyca and Gebremedhin 2020). Although electronic fans were used to facilitate air flows in this study, WS averaged 1.2 m/s only, much lower than the mean WS (5.16 m/s) where the CCI was originally developed (Mader et al. 2010). Besides, SR was zero for the majority of the time due to the protection of the double-pitched roof. The close readings of Ta and Tbg also indicate a minor effect of SR in heating indoor air temperature. Although Ta outperformed in the present study, TIs incorporating more components of thermal environment would be far more useful in grazing pastures.
Sensitivity of physiological indicators to heat stress
In this study, all physiological indicators had significant correlations with heat stress indicators, indicating good performance in reflecting the true response of cows to heat stress. Notably, ET and VT showed lower correlation coefficients with heat stress indicators compared with FT, MT, and RR, which is consistent with Peng et al. (2019). This could be due to the presence of the plateau in which ET and VT stayed relatively stable within thermoneutral conditions. To some extent, ET is in good proximity to core body temperature in terms of correlation with thermal environment, and thus could be used as an external sensor to predict core body temperature.
The responding sequence of heat strain indicators has been reported by previous studies. Mean FT was found to increase at lower THI compared with RT (71.4 vs. 74.1) (Peng et al. 2019). A lower THI threshold was determined in RR than RT in 139 lactating Holstein–Friesian cows (65 vs. 70) (Pinto et al. 2020). Similarly, RR increased at lower BGHI thresholds than RT in the study by Dalcin et al. (2016) who used 38 lactating cows from different genetic groups. More recently, RR and RT thresholds were compared in 826 lactating Holstein–Friesian cows taking into account various animal-related factors (Yan et al. 2021). Their results showed that the THI thresholds for RR were consistently lower than those for RT. The temporal difference between RR and RT was determined as an hour in a climate-controlled chamber (Gaughan et al. 2000). All of this evidence indicates that increased core body temperature should be regarded as the outcome of insufficient thermoregulation instead of an early indicator.
In the present study, the responding sequence of heat strain indicators has been confirmed. By comparing Ta thresholds, FT was found to be most sensitive to heat stress, followed by RR, ET, VT, and MT. In general, peripheral body temperature, respiratory dynamics, and core body temperature sequentially react to the external stress caused by thermal environment (Shu et al. 2021). Interestingly, MT was found to have the highest Ta threshold (25.4 °C), even higher than VT (25.3 °C). We speculate that this higher threshold might be due to the combined effect of the ambient environment and the hot air exhaled from the cows’ lungs. As a result, MT may tend to react to changes in the environment in a similar way that the core body temperature does. This result also points out that not all BSTs are good indicators of heat strain when earlier thresholds are expected.
The use of an early indicator allows for early detection of heat strain in dairy cows, and the corresponding threshold can support early treatment by informing the use of cooling measures. However, the selection of heat strain indicator should consider the actual situation in field. For instance, cows’ surfaces are always wet when sprinklers or soakers are used during heat stress seasons, rendering most BSTs (e.g., FT and MT) useless as heat strain indicators. The ET, on the other hand, is less likely to be affected by water, but had higher Ta thresholds compared with RR. Collectively, RR is expected to be the most appropriate heat strain indicator because of its high correlation with heat stress indicators, earlier threshold, and good feasibility.
In this study, the earliest manifestation of heat strain was determined by comparing the Ta thresholds. Ideally, this should be done by looking at not only the relationship with environmental indicators, but also the time when the physiological indicators cross the thresholds. Further studies are required which measure these heat strain indicators continuously to confirm the responding sequence of heat strain indicators found in this study.
Thresholds for heat strain in dairy cows
The milk production and the determined Ta thresholds in this study were both higher than recent studies (Li et al. 2020; Ji et al. 2020), which is contrary to the consensus that high-producing dairy cows are more thermally sensitive (Collier et al. 2012). Therefore, the negative effect of increased milk yield on heat tolerance might be confounded or modified by other factors, probably the organic strategies (i.e., home-grown feed and free of antibiotics). Organic dairy farms have shown to be more tolerant to heat stress compared with traditional intensive farms due to the better adaptability of animals to environment (Blanco-Penedo et al. 2020). However, to know the effect of the conducted organic strategies, further studies are required which compare the thermal tolerance of herds reared under different strategies after controlling productivity, parity, DIM, and other influencing factors.
From a methodological point of view, there may be two reasons to explain the lower thresholds determined in some relevant studies. On the one hand, there is a spatial difference among relative studies in terms of their locations of measurement points. As discussed above, when using usual fixed measurements within a certain height, the greater horizontal distance between sensor and cow might result in lower measurement and threshold of Ta, especially when only a single fixed sensor is used (Hempel et al. 2018). On the other hand, the temporal pattern of heat stress and heat strain is often ignored. Some studies did not observe physiological indicators from the beginning of heat stress season but began their measurements from severely stressed months until the temperature dropped sufficiently (Peng et al. 2019; Yan et al. 2021). Lots of evidence has shown that dairy cows have carryover effects of heat strain in autumn (Bertocchi et al. 2014; Becker et al. 2020), and cows acclimate to having a higher physiological level after long-term exposure to heat stress (Amamou et al. 2019). Therefore, it takes longer for cows to recover to their previous physiological and production level relative to the time it takes for thermal environment to relieve naturally, particularly in temperate climates without a gradual transition between summer and autumn. As a result, such datasets lacking data at the beginning of heat stress season will tend to have a lower environmental threshold to some extent. The timepoint in this study was early enough and met the upward trend of thermal environment to capture the physiological pattern when heat strain first began in cows.
Since different animals began to feel thermal discomfort at different environmental conditions, the classification performance of environmental indicators in recognizing heat strain states was limited in this study. Thus, comparing the thresholds on physiological side might reveal some information for further development of heat strain detection using animal-based indicators. For BST, the determined thresholds for mean and maximum FT were 30.1 °C and 31.1 °C, respectively, which are consistent with a recent study using an identical infrared camera (30.1 °C and 30.3 °C, respectively) (Peng et al. 2019). For RR, the determined threshold was 53.8 bpm, which is very close to the study of Collier et al. (2012) where the onset of heat strain was determined at a RR of 60 bpm. For VT, our threshold (38.7 °C) is higher than a recent study using Holstein heifers (38.32 °C) (Tian et al. 2021). However, heifers should have higher body temperatures relative to lactating cows (Sartori et al. 2002). Therefore, we speculate that this difference might be due to different insertion depths or sensor accuracy (Burfeind et al. 2010).
Conclusion
In conclusion, the fixed subarea sensor is a reliable tool for measuring cows’ microenvironments, and Ta seems to be an appropriate heat stress indicator in early summer when heat strain first begins in dairy cows. On the other hand, FT and RR, as well as ET, are good early indicators of heat strain in dairy cows. In practice, the use of heat strain indicators should consider the actual situation, and RR would be most appropriate in the settings equipped with evaporative cooling devices. The results of this study could be helpful to dairy practitioners in a similar organic intensive setting, allowing them to use more appropriate indicators to better detect and respond to heat strain in dairy cows.
References
Altman DG, Bland JM (1983) Measurement in medicine: the analysis of method comparison studies. J R Stat Soc Ser D (Stat) 32:307–317. https://doi.org/10.2307/2987937
Amamou H, Beckers Y, Mahouachi M, Hammami H (2019) Thermotolerance indicators related to production and physiological responses to heat stress of holstein cows. J Therm Biol 82:90–98. https://doi.org/10.1016/j.jtherbio.2019.03.016
Baeta FC, Meador NF, Shanklin MD, Johnson HD (1987) Equivalent temperature index at temperatures above the thermoneutral for lactating dairy cows. American Society of Agricultural Engineers, St. Joseph, p 874015
Becker CA, Collier RJ, Stone AE (2020) Invited review: physiological and behavioral effects of heat stress in dairy cows. J Dairy Sci 103:6751–6770. https://doi.org/10.3168/jds.2019-17929
Bertocchi L, Vitali A, Lacetera N, Nardone A, Varisco G, Bernabucci U (2014) Seasonal variations in the composition of Holstein cow’s milk and temperature–humidity index relationship. Animal 8:667–674. https://doi.org/10.1017/S1751731114000032
Bianca W (1962) Relative importance of dry- and wet-bulb temperatures in causing heat stress in cattle. Nature 195:251–252. https://doi.org/10.1038/195251a0
Blanco-Penedo I, Velarde A, Kipling RP, Ruete A (2020) Modeling heat stress under organic dairy farming conditions in warm temperate climates within the Mediterranean basin. Clim Change 162:1269–1285. https://doi.org/10.1007/s10584-020-02818-y
Bohmanova J, Misztal I, Cole JB (2007) Temperature-humidity indices as indicators of milk production losses due to heat stress. J Dairy Sci 90:1947–1956. https://doi.org/10.3168/jds.2006-513
Buffington DE, Collazo-Arocho A, Canton GH, Pitt D, Thatcher WW, Collier RJ (1981) Black globe-humidity index (BGHI) as comfort equation for dairy cows. Trans ASAE 24:711–0714. https://doi.org/10.13031/2013.34325
Burfeind O, Von Keyserlingk MAG, Weary DM, Veira DM, Heuwieser W (2010) Short communication: repeatability of measures of rectal temperature in dairy cows. J Dairy Sci 93:624–627. https://doi.org/10.3168/jds.2009-2689
Carabaño MJ, Logar B, Bormann J, Minet J, Vanrobays ML, Díaz C, Tychon B, Gengler N, Hammami H (2016) Modeling heat stress under different environmental conditions. J Dairy Sci 99:3798–3814. https://doi.org/10.3168/jds.2015-10212
Chen C-S, Chen W-C (2019) Research and development of automatic monitoring system for livestock farms. Appl Sci 9:1132. https://doi.org/10.3390/app9061132
Collier RJ, Dahl GE, VanBaale MJ (2006) Major advances associated with environmental effects on dairy cattle. J Dairy Sci 89:1244–1253. https://doi.org/10.3168/jds.s0022-0302(06)72193-2
Collier RJ, Laun WH, Rungruang S, Zimbleman RB (2012) quantifying heat stress and its impact on metabolism and performance. In: Florida Ruminant Nutrition Symposium, Gainesville, FL, USA, 31 January–1 February 2012 2012. University of Florida, Gainesville, FL, USA, pp 74–83
Dalcin VC, Fischer V, Daltro DdS, Alfonzo EPM, Stumpf MT, Kolling GJ, Silva MVGBd, McManus C (2016) Physiological parameters for thermal stress in dairy cattle. Rev Bras Zootec 45:458–465. https://doi.org/10.1590/S1806-92902016000800006
Davison C, Michie C, Hamilton A, Tachtatzis C, Andonovic I, Gilroy M (2020) Detecting heat stress in dairy cattle using neck-mounted activity collars. Agriculture 10:210. https://doi.org/10.3390/agriculture10060210
Dikmen S, Hansen PJ (2009) Is the temperature-humidity index the best indicator of heat stress in lactating dairy cows in a subtropical environment? J Dairy Sci 92:109–116. https://doi.org/10.3168/jds.2008-1370
Gaughan JB, Holt SM, Hahn GL, Mader TL, Eigenberg R (2000) Respiration rate—is it a good measure of heat stress in cattle? Asian-Aust J Anim Sci 13:329–332
Gaughan JB, Mader TL, Holt SM, Lisle A (2008) A new heat load index for feedlot cattle. J Anim Sci 86:226–234. https://doi.org/10.2527/jas.2007-0305
Gorczyca MT, Gebremedhin KG (2020) Ranking of environmental heat stressors for dairy cows using machine learning algorithms. Comput Electron Agric 168:105124. https://doi.org/10.1016/j.compag.2019.105124
Hammami H, Bormann J, M’Hamdi N, Montaldo HH, Gengler N (2013) Evaluation of heat stress effects on production traits and somatic cell score of Holsteins in a temperate environment. J Dairy Sci 96:1844–1855. https://doi.org/10.3168/jds.2012-5947
Hempel S, König M, Menz C, Janke D, Amon B, Banhazi TM, Estellés F, Amon T (2018) Uncertainty in the measurement of indoor temperature and humidity in naturally ventilated dairy buildings as influenced by measurement technique and data variability. Biosys Eng 166:58–75. https://doi.org/10.1016/j.biosystemseng.2017.11.004
Herbut P (2013) Temperature, humidity and air movement variations inside a free-stall barn during heavy frost. Ann Anim Sci 13:587–596. https://doi.org/10.2478/aoas-2013-0025
Hoffmann G, Herbut P, Pinto S, Heinicke J, Kuhla B, Amon T (2020) Animal-related, non-invasive indicators for determining heat stress in dairy cows. Biosys Eng 199:83–96. https://doi.org/10.1016/j.biosystemseng.2019.10.017
Ji B, Banhazi T, Ghahramani A, Bowtell L, Wang C, Li B (2020) Modelling of heat stress in a robotic dairy farm. Part 1: thermal comfort indices as the indicators of production loss. Biosys Eng 199:27–42. https://doi.org/10.1016/j.biosystemseng.2019.11.004
Kabuga JD (1992) The influence of thermal conditions on rectal temperature, respiration rate and pulse rate of lactating Holstein-Friesian cows in the humid tropics. Int J Biometeorol 36:146–150. https://doi.org/10.1007/BF01224817
Kadzere CT, Murphy MR, Silanikove N, Maltz E (2002) Heat stress in lactating dairy cows: a review. Livest Prod Sci 77:59–91. https://doi.org/10.1016/S0301-6226(01)00330-X
Kovács L, Kezer FL, Ruff F, Jurkovich V, Szenci O (2018) Assessment of heat stress in 7-week old dairy calves with non-invasive physiological parameters in different thermal environments. PLoS ONE 13:e0200622. https://doi.org/10.1371/journal.pone.0200622
Lees JC, Lees AM, Gaughan JB (2018) Developing a heat load index for lactating dairy cows. Anim Prod Sci 58:1387. https://doi.org/10.1071/AN17776
Li S, Gebremedhin KG, Lee C, Collier R (2009) Evaluation of thermal stress indices for cattle. ASABE Annual International Meeting, Reno, p 096003
Li G, Chen S, Chen J, Peng D, Gu X (2020) Predicting rectal temperature and respiration rate responses in lactating dairy cows exposed to heat stress. J Dairy Sci 103:5466–5484. https://doi.org/10.3168/jds.2019-16411
Mader T, Davis M, Brown-Brandl T (2006) Environmental factors influencing heat stress in feedlot cattle. J Anim Sci 84:712–719. https://doi.org/10.2527/2006.843712x
Mader TL, Johnson LJ, Gaughan JB (2010) A comprehensive index for assessing environmental stress in animals. J Anim Sci 88:2153–2165. https://doi.org/10.2527/jas.2009-2586
Maia ASC, daSilva RG, Battiston Loureiro CM (2005) Sensible and latent heat loss from the body surface of Holstein cows in a tropical environment. Int J Biometeorol 50:17–22. https://doi.org/10.1007/s00484-005-0267-1
Mbuthia JM, Mayer M, Reinsch N (2021) Modeling heat stress effects on dairy cattle milk production in a tropical environment using test-day records and random regression models. Animal 15:100222–100222. https://doi.org/10.1016/j.animal.2021.100222
Mondaca MR, Choi CY, Cook NB (2019) Understanding microenvironments within tunnel-ventilated dairy cow freestall facilities: examination using computational fluid dynamics and experimental validation. Biosys Eng 183:70–84. https://doi.org/10.1016/j.biosystemseng.2019.04.014
NRC (1971) A guide to environmental research on animals. National Academy Press, Washington, D.C.
Peng D, Chen S, Li G, Chen J, Wang J, Gu X (2019) Infrared thermography measured body surface temperature and its relationship with rectal temperature in dairy cows under different temperature-humidity indexes. Int J Biometeorol 63:327–336. https://doi.org/10.1007/s00484-018-01666-x
Pinto S, Hoffmann G, Ammon C, Amon T (2020) Critical THI thresholds based on the physiological parameters of lactating dairy cows. J Therm Biol 88. https://doi.org/10.1016/j.jtherbio.2020.102523
Qiao Y, Kong H, Clark C, Lomax S, Su D, Eiffert S, Sukkarieh S (2021) Intelligent perception for cattle monitoring: a review for cattle identification, body condition score evaluation, and weight estimation. Comput Electron Agric 185:106143. https://doi.org/10.1016/j.compag.2021.106143
Ranjitkar S, Bu D, Van Wijk M, Ma Y, Ma L, Zhao L, Shi J, Liu C, Xu J (2020) Will heat stress take its toll on milk production in China? Clim Change 161:637–652. https://doi.org/10.1007/s10584-020-02688-4
Sartori R, Sartor-Bergfelt R, Mertens SA, Guenther JN, Parrish JJ, Wiltbank MC (2002) Fertilization and early embryonic development in heifers and lactating cows in summer and lactating and dry cows in winter. J Dairy Sci 85:2803–2812. https://doi.org/10.3168/jds.S0022-0302(02)74367-1
Schüller LK, Heuwieser W (2016) Measurement of heat stress conditions at cow level and comparison to climate conditions at stationary locations inside a dairy barn. J Dairy Res 83:305–311. https://doi.org/10.1017/S0022029916000388
Shu H, Wang W, Guo L, Bindelle J (2021) Recent advances on early detection of heat strain in dairy cows using animal-based indicators: a review. Animals 11:980. https://doi.org/10.3390/ani11040980
Tian H, Liu J, Chen X, Li S, Li X, Mengal K, Lu Y, Wang D (2021) Effects of ambient temperature and humidity on body temperature and activity of heifers, and a novel idea of heat stress monitoring. Anim Prod Sci 61:1584–1591. https://doi.org/10.1071/AN20156
Wang X, Gao H, Gebremedhin KG, Bjerg BS, Van Os J, Tucker CB, Zhang G (2018) A predictive model of equivalent temperature index for dairy cattle (ETIC). J Therm Biol 76:165–170. https://doi.org/10.1016/j.jtherbio.2018.07.013
Wijffels G, Sullivan M, Gaughan J (2021) Methods to quantify heat stress in ruminants: current status and future prospects. Methods 186:3–13. https://doi.org/10.1016/j.ymeth.2020.09.004
Wildman EE, Jones GM, Wagner PE, Boman RL, Troutt HF, Lesch TN (1982) A dairy cow body condition scoring system and its relationship to selected production characteristics. J Dairy Sci 65:495–501. https://doi.org/10.3168/jds.S0022-0302(82)82223-6
Yan G, Li H, Zhao W, Shi Z (2020) Evaluation of thermal indices based on their relationships with some physiological responses of housed lactating cows under heat stress. Int J Biometeorol 64:2077–2091. https://doi.org/10.1007/s00484-020-01999-6
Yan G, Liu K, Hao Z, Shi Z, Li H (2021) The effects of cow-related factors on rectal temperature, respiration rate, and temperature-humidity index thresholds for lactating cows exposed to heat stress. J Therm Biol 100:103041. https://doi.org/10.1016/j.jtherbio.2021.103041
Yousef MK (1985) Stress physiology: definition and terminology. CRC Press, Boca Raton
Acknowledgements
The authors are grateful for the assistance received from Yinxiang dairy farm in Shandong, China.
Funding
This work was supported by Major Science and Technology Program of Inner Mongolia Autonomous Region, grant number 2020ZD0004; Key Research and Development Plan of Hebei Province, grant number 20327202D; and Key Research and Development Plan of Hebei Province, grant number 20326602D.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The experimental protocols were approved by the Experimental Animal Care and Committee of Institute of Animal Sciences, Chinese Academy of Agricultural Sciences (approval number IAS2021-220).
Conflict of interest
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Shu, H., Guo, L., Bindelle, J. et al. Evaluation of environmental and physiological indicators in lactating dairy cows exposed to heat stress. Int J Biometeorol 66, 1219–1232 (2022). https://doi.org/10.1007/s00484-022-02270-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00484-022-02270-w