Abstract
Background
Various training models have been developed for laparoscopic training. Inanimate models including cadavers, ex-vivo simulator, and virtual reality (VR), are less realistic and often fail to display specific events such as bleeding, bile leakage, etc. Animal models provide more realistic experience, but constraints like cost involved, anesthetic requirement, and ethical approval have limited its application. We have designed a new training ex-vivo simulator—Smagister to address these issues.
Methods
The Smagister consists of a normothermic machine perfusion platform, multivisceral organ of porcine abdominal cavity (liver, gallbladder, pancreas, stomach, intestine, kidney, uterus, bladders, etc.), high-definition display, and software system. Blood gas analysis and number of peristalsis per hour were recorded. A questionnaire was used to subjectively assess vitality of the organ cluster every hour. Three laparoscopic procedures including cholecystectomy (LC), enterotomy closure (LEC) and hepatectomy (LLR) were performed on Smagister, with demonstration of specific events for each procedure. Six experts compared the procedures with actual surgery in terms of feasibility to complete procedures and demonstration of complications.
Results
The fluctuation of perfusate glucose (6.1–8.2 mmol/L) and lactate (5.82–6.55 mmol/L) suggested metabolic function of the multivisceral organs. The mean number of peristalsis was 2.2/min. The simulated surgical view and anatomic structures closely resembled actual surgery during continuous perfusion (3.5 ± 1.0, 3.8 ± 0.8, respectively). The evaluation scores of haptic feedbacks were 3.8 ± 0.8, resembling live tissue handling. LC, LEC, and LLR were performed well on the Smagister, with clear display of the specific events. All six experts considered Smagister as a suitable training modality for both basic and advanced laparoscopic surgery.
Conclusion
The amalgamation of live animal model and ex-vivo simulation in Smagister centralizes the virtue of both modalities, expands the training field, and provides high-fidelity laparoscopic training for both novice and senior surgeons.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Laparoscopic procedures have significantly evolved and advanced in the past decades and have become the standard approach for many diseases given the reduced postoperative pain, accelerated recovery and better cosmetic results [1,2,3]. Unlike open surgery, several technical challenges such as limited tactile feedback, fulcrum effect, reliance on two-dimensional vision, and difficult hand-eye-coordination have contributed to the prolonged learning curve in acquiring laparoscopic skills [4, 5]. In addition, there is a shift in idea on the suitability of developing such skills during clinical practice owing to the ever-increasing concerns on patient safety.
Training out of the operating room (OR) has proven its positive impact on surgical training as it eliminates the ethical concerns of attaining laparoscopic skills on patients in clinical practice [6, 7]. Various models have been explored around the world with the purpose of reducing the inherent learning curve and to reproduce real-life experience for surgeons. At present, basic laparoscopic skills can be learned from a box trainer, while the acquisition of further complex procedures can be achieved using inanimate training models like cadavers, virtual reality (VR), ex-vivo simulator and live animal models. Traditionally, the use of human cadaver as training models have been implemented to improve spatial perception of surgical anatomy as it has similar tissue planes and organ consistency with that in actual surgery [8, 9]. Nevertheless, the limited availability, high cost, specialized operative facility, and funeral services for donated bodies have restricted their applicability [10]. Contrarily, the use of VR training models has gained widespread popularity in recent years due to their flexibility, and ability to incorporate various level of difficulties into practice sessions as well as simulate whole or partial operations [11, 12]. Nonetheless, they are considered expensive, low fidelity and incapable of providing haptic sensation through force feedback.
Ex-vivo simulators have become widely accepted in the field of surgery as they are in the mid-fidelity range, cost-effective, and commercially available [13, 14]. Despite benefits of ex-vivo simulators, VR and cadavers, these models demonstrate one crucial drawback: the failure to provide realistic simulation of specific events in live tissue, such as intraprocedural bleeding, bile leakage, etc.
Animal model closely resembles actual surgery and is by far the only model that can effectively imitate specific adverse events during laparoscopic training. Many training curriculums involve performing procedures on live animal models in order to encounter these events before progressing to active practice in the OR, especially in advanced surgery training [15, 16]. However, there are fewer opportunities to participate in training using live animal model because of higher cost, requirement of general anesthesia and need for ethical approval [17, 18], as well as the 3R approach—Reduction, Refinement and Replacement in using animals as described by Russell and Burch [19]. Therefore, it is imminently necessary to develop a training system with optimal combination of various training modalities for laparoscopic training.
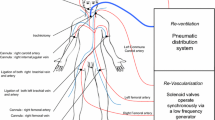
Normothermic machine perfusion (NMP) allows provision of continuous oxygen and nutrient for ex-vivo organs. Thus, NMP is becoming an area of great academic and clinical interest in organ transplantation [20]. We have successfully designed an abdominal multivisceral NMP device to improve transplantation outcomes [21, 22] and this technology can be used in organ-oriented research platform that we have introduced in a review, such as isolated organ culture and functional evaluation, and organ pharmacology and toxicology research [23]. In our present study, we incorporated this technique into laparoscopic training simulator to maintain vitality and normal physiological function of the whole ex-vivo porcine abdominal organ (Fig. 1A).
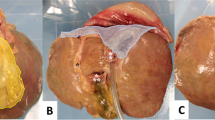
Procedures and specific events on the Smagister. A An overview of device in use by our expert; B laparoscopic cholecystectomy (LC); C bleeding during LC; D bile leakage during LC; E laparoscopic enterotomy closure (LEC); F perforation during LEC; G ischemia during LEC; H laparoscopic liver resection (LLR); I bleeding during LLR; J bleeding control during LLR
The aims of this study were to evaluate the possibility of using the live ex-vivo multivisceral training device (Smagister) which consists of whole porcine abdominal organs for laparoscopic training.
Materials and methods
Preparation of living organ laparoscopic training system
The Smagister consists of an NMP platform, multivisceral organ (liver, gallbladder, pancreas, stomach, intestine, kidney, uterus, bladder, etc.), high-definition display, and software system. The approval of Institution Review Board was waived since the porcine abdominal organ clusters were directly obtained from the slaughterhouse. Initially, arterial blood was collected from the carotid artery and mildly anticoagulated in preservation for NMP. An arterial cannula was inserted into the abdominal aorta and perfused with 3L of 4 °C normal saline, then flushed with 3L cold organ preservation solution (Table 1), while the inferior vena cava was cannulated by a venous cannula for outflow. The entire abdominal organ was removed and stored in 4 °C organ preservation solution after ligating the esophagus and anal canal. At last, the organ cluster was transported to the training center and connected to the Smagister.
NMP system
The venous cannula was connected to the organ reservoir of Smagister for venous outflow and the arterial cannula was connected to the arterial line. The ex-vivo perfusion circuit was established. The perfusate was prepared consisting of 1500 mL anticoagulated whole blood, 1000 mL gelofusine, 200 mL 5% sodium bicarbonate, 30 mL 20% calcium gluconate and 8000 units heparin. The circuit was slowly warmed up to 37 °C, and a mixture of O2 and air (25% O2) was supplied by an oxygenator. The flow and pressure of artery were recorded. The infusion pressure was adjusted to keep the inflow at approximately 1000 L/min.
Evaluation of vitality
Six experienced surgeons who have at least 5 years’ experience in performing laparoscopic surgery were invited to evaluate the Smagister. The arterial blood gas analysis, temperature of organ clusters and number of peristalsis were recorded every 60 min. Porcine hematology reference ranges were obtained from Appendix VIII: Blood analyte reference values in large animals in Clinical Biochemistry of Domestic Animals [24]. Each surgeon graded the vitality of the multivisceral porcine organ from the perspective of liver color, intestine color, and tissue elasticity per hour (Table 2).
Training procedures on Smagister
Laparoscopic cholecystectomy (LC), laparoscopic liver resection (LLR) and enterotomy closure (LEC) were performed on Smagister (Fig. 1B, E and H). Following these, specific adverse events of each surgery were initiated, including bile leakage, bleeding, perforation etc. (Table 3). The completion of procedures and manifestation of specific events for each procedure were assessed, respectively, by the six experts on a 5-point Likert scale questionnaire (Table 3). Finally, the experts were asked to compare realism of Smagister with actual surgery in terms of anatomic structure, surgical view, haptic feedback, and to quantify their impression on the value of Smagister in basic and advanced surgical training (Table 2).
Statistical analysis
The data of objective outcomes were expressed as the minimum and maximum. Data of experts’ assessment scores were expressed as the mean ± standard deviation (SD). Statistical analyses were performed using SPSS 24.0 software package (SPSS, USA).
Results
The vitality of organ cluster
NMP was achieved and stable throughout the operation. The mean artery pressure was equivalent to 60 mmHg, with the inflow approximating 1000 mL/min (Fig. 2A). Organ temperature was maintained at 36.5–37.4 °C. The mean number of peristalsis was 2.2/min and was well maintained until the end (Fig. 2B). Metabolic function was determined by blood gas analysis of arterial perfusate including pH, lactate, PO2, PCO2, base excess, HCO3−, urea, creatinine, and glucose (Fig. 2C–G). Monitoring of pH, HCO3−, and base excess demonstrated essentially normal acid base balance throughout the perfusion (Fig. 2C, E). The perfusate lactate was low at initiation of NMP due to dilution of the whole blood (2.53 mmol/L) but increased and remained stable after perfusion (5.82–6.55 mmol/L, Fig. 2C). The fluctuation in perfusate glucose suggested glucose utilization in the multivisceral organs (6.1–8.2 mmol/L, Fig. 2G). The liver color, intestine color, and tissue elasticity were well preserved until the end and all six experts considered these features identical to that of live animal (Fig. 2H,I).
Vital parameters of the Smagister including hemodynamic parameters (n = 2), peristalsis, results of blood gas analysis (n = 9), and the assessment by experts (n = 3). A Inflow pressure and volume; B number of peristalsis; C pH and lactate; D PO2 and PCO2; E HCO3− and base excess; F creatinine and urea;G glucose; H,I liver color, intestine color, and tissue elasticity assessed by experts
Evaluation of training on Smagister
Most experts considered the LC procedure on the Smagister to be mostly completed (Figs. 2B and 3B). The specific events during LC were well demonstrated (Fig. 3B), such as vessel injury (Fig. 1C) and bile leakage (Fig. 1D), etc. For LEC, the procedure and manifestation of specific events have yielded comparable results with LC (Figs. 1E,F and 3C). The LLR procedure was performed completely (Fig. 3D) and demonstration of bleeding control and bile leakage were analogous to that of real surgery (Figs. 1H–J and 3D). However, the experts perceived bleeding control to be more challenging than in actual surgery since the blood was mildly anticoagulated.
Evaluation of procedures on Smagister. A Overall evaluation of the Smagister including surgical view, anatomic structure, and haptic feedback; B–D assess completion of procedure and the manifestation of events in laparoscopic cholecystectomy (LC), laparoscopic enterotomy closure (LEC), and laparoscopic liver resection (LLR)
The experts gave positive evaluation of anatomic structure, in that the abdominal cavity has been well reproduced (3.5 ± 1.0, Fig. 3A). Since the organs were unable to be fixed by ligaments, the intraprocedural difference should be noticed. Most experts thought that adequate blood supply made the surgical view identical to actual procedures (3.8 ± 0.8, Fig. 3A). Although haptic feedback is slightly different from operations performed in the OR, usage of live tissues made the experience realistic (3.8 ± 0.8, Fig. 3A). All six experts strongly recommended that Smagister should be incorporated into laparoscopic training for both basic and advanced surgery.
Discussion
This study presents a novel innovation using live multivisceral organ in an ex-vivo simulator for laparoscopic surgery education. The entire porcine abdominal organ and anatomic structure have been well reproduced in the simulator with the organs continually perfused and kept alive under normal physiologic conditions using the NMP technique. Therefore, specific events of each procedure were successfully demonstrated including intraprocedural hemorrhage, bile leakage and ischemia which were unattainable in other training models like human cadaver, simulators, or VR. In addition, in comparison to animal models, Smagister negates the issues surrounding ethics, has no logistic demands for managing animals and is easy to transport. Concisely, the Smagister focuses on the merits of both animal models and ex-vivo simulators, while disregarding their individual shortcoming. For this reason, it facilitates the implementation of laparoscopic training curriculum based on the device—Smagister in any hospital.
The most prominent feature of Smagister is the ability to maintain viability of the multivisceral organs throughout the simulation. Porcine blood was used for blood circulation in line with the NMP technique to supply oxygen and nutrient for the organ clusters. Blood gas analysis performed during the procedures indicated pH, lactate, PO2, PCO2, base excess, HCO3−, urea, creatinine, and glucose that were maintained near physiological limits for 5 h. Besides, peristalsis, liver and intestinal color, and tissue elasticity were well preserved during perfusion. Thus, these parameters substantiate a state of stable physiological function and tissue property until the end of the investigation. As we demonstrated, the accurate display of fatal intraoperative events can contribute towards resolving concerns surrounding patient safety as surgeons can encounter these during the simulated training and be equipped prior to embarking onto their clinical practice.
Several systematic reviews on laparoscopic training curriculums found that most participants preferred the use of live animal models in training modules as other modalities cannot realistically reproduce the events [15, 16]. However, the constraints rendered by animal-based training curriculums pose a great challenge in organizing such modules. Various efforts have been made to improve the experience of ex-vivo simulators. Liu et al. [25] developed a laparoscopic training system with ex-vivo porcine liver for hepatobiliary surgery. A continuous machine perfusion and red fluid was used to simulate virtual blood circulation in order to facilitate demonstration of intraprocedural bleeding. Lzawa et al. [14] used a fluid pump to mimic circulation in an ex-vivo model and appeared to achieve similar effect in live tissue training. However, there were several drawbacks as the loss of organ vitality made it impossible to display other intraprocedural events, preserve tissue properties, while the usage of a single organ failed to incorporate other adjoining structures. In our study, aside from acquisition of surgical skills, trainees are also trained in mental endurance, as a crucial aspect in surgery. Detailed observation and prompt measures to fix the specific events initiated during simulation serves to further ingrain the scenarios and technique involved to optimize patient outcome in clinical practice.
Another important advantage is the applicability of Smagister in both current and future advanced laparoscopic training. Recently, the rapid expansion and acceptance of laparoscopic surgery by surgeons and patients alike have tremendously increased the demand of laparoscopic training for advanced surgeries. Unlike novice surgeons, most senior surgical trainees tend to prefer high-fidelity live animal models or ex-vivo simulators rather than VR [16, 26]. The need for more advanced laparoscopic skills is parallel to the fact that senior trainees encounter more complex surgeries [27]. For example, LLR requires surgeons to be trained in managing hemostasis during parenchymal transection, and this can only be replicated in animal models. Therefore, the reduced application of animal models and existing limitation in ex-vivo simulators including lack of anatomic plane, adjoining structures and normal physiological functions present an immense challenge in meeting the growing demands of advanced training [9, 28].
Strickland et al. [27] developed an augmented reality simulator which can simulate bleeding control using red fluid during LLR, but they were unable to overcome these shortages and mimic coagulation. In this study, Smagister replicated normal physiological functions, vitality and anatomic structure to ensure a realistic surgical view. In addition, the tissue elasticity in our device produced high-fidelity haptic feedback. Given these, both transection and bleeding control during LLR could be demonstrated simultaneously. Since Smagister contains the entire porcine abdominal organ cluster, we envisage this simulator to be equally useful in other advanced laparoscopic surgery training and to support future innovative operations, such as gastric and splenic procedures (Fig. 4), as well as pancreatic surgery since it has yet to be published across literature.
The exact costs involved in this study was not calculated, however, estimates can be made. The main cost incurred is limited to specimen fee ($60 for each porcine abdominal organ cluster), rental cost of Smagister ($500) and cost of consumable items, like self-made organ preservation solution ($50 for 3L). The set-up time required was 20 min which involves connecting the organ cluster to Smagister, circuit warmup, and arrangement of organs. In this training curriculum, organ arrangement can be performed by trainees in order to help them familiarize with the anatomic structures. In terms of time taken for procurement and preservation of organs, two skilled workers can obtain ten sets of porcine organ cluster in 3 h. The organs procured can be preserved in organ preservation solution for 24 h.
Despite the potential advantages of Smagister and the contributions of our study, there are some limitations. First, we have not recruited surgical trainees to evaluate the effectiveness of Smagister as a training modality and make direct comparisons with other models. Second, anatomical features of the live tissue cannot be reproduced completely in our model, such as diaphragmatic movement and ligament fixation. Third, Smagister lacks the ability to allow identification of tumor edge in certain procedures like tumor resection. Fourth, it should be noted that animal organs cannot resemble human anatomy in its entirety which is necessary in advanced surgery. Thus, additional video-based program should be introduced to identify anatomical landmarks in human.
In summary, the application of Smagister aims to improve laparoscopic surgical skills and strengthen mental endurance to help novice and senior surgeons alike climb the inherent learning curve in a more realistic environment as offered in animal models, without the restrictions of exorbitant expenses and ethical issues. The vitality of multivisceral organ in an ex-vivo simulator was successfully established using NMP, demonstrating the amalgamation of live animal model and ex-vivo simulation. Most importantly, trainees can improve their ability to deal with unexpected events intraoperatively using Smagister, as it is currently unattainable in other inanimate training model like human cadaver, VR, and ex-vivo simulator. Our novel device—Smagister is a suitable platform for laparoscopic training in current advanced surgeries and has a promising role in future innovations in the surgical arena. Further prospective studies should be done to provide a robust evaluation of trainees’ skill acquisition in comparison with other training models and develop a corresponding assessment system.
References
Bang JY, Arnoletti JP, Holt BA, Sutton B, Hasan MK, Navaneethan U, Feranec N, Wilcox CM, Tharian B, Hawes RH, Varadarajulu S (2019) An endoscopic transluminal approach, compared with minimally invasive surgery, reduces complications and costs for patients with necrotizing pancreatitis. Gastroenterology 156:1027–1040.e1023
van Dijk AH, Wennmacker SZ, de Reuver PR, Latenstein CSS, Buyne O, Donkervoort SC, Eijsbouts QAJ, Heisterkamp J, Hof KI, Janssen J, Nieuwenhuijs VB, Schaap HM, Steenvoorde P, Stockmann H, Boerma D, Westert GP, Drenth JPH, Dijkgraaf MGW, Boermeester MA, van Laarhoven C (2019) Restrictive strategy versus usual care for cholecystectomy in patients with gallstones and abdominal pain (SECURE): a multicentre, randomised, parallel-arm, non-inferiority trial. Lancet (Lond, Engl) 393:2322–2330
Vennix S, Musters GD, Mulder IM, Swank HA, Consten EC, Belgers EH, van Geloven AA, Gerhards MF, Govaert MJ, van Grevenstein WM, Hoofwijk AG, Kruyt PM, Nienhuijs SW, Boermeester MA, Vermeulen J, van Dieren S, Lange JF, Bemelman WA (2015) Laparoscopic peritoneal lavage or sigmoidectomy for perforated diverticulitis with purulent peritonitis: a multicentre, parallel-group, randomised, open-label trial. Lancet (Lond, Engl) 386:1269–1277
Crothers IR, Gallagher AG, McClure N, James DT, McGuigan J (1999) Experienced laparoscopic surgeons are automated to the "fulcrum effect": an ergonomic demonstration. Endoscopy 31:365–369
FuchsWeizman N, Maurer R, Einarsson JI, Vitonis AF, Cohen SL (2015) Survey on barriers to adoption of laparoscopic surgery. J surg educ 72:985–994
Kassab E, Tun JK, Arora S, King D, Ahmed K, Miskovic D, Cope A, Vadhwana B, Bello F, Sevdalis N, Kneebone R (2011) "Blowing up the barriers" in surgical training: exploring and validating the concept of distributed simulation. Ann Surg 254:1059–1065
Mackenzie H, Cuming T, Miskovic D, Wyles SM, Langsford L, Anderson J, Thomas-Gibson S, Valori R, Hanna GB, Coleman MG, Francis N (2015) Design, delivery, and validation of a trainer curriculum for the national laparoscopic colorectal training program in England. Ann Surg 261:149–156
Giger U, Fresard I, Hafliger A, Bergmann M, Krahenbuhl L (2008) Laparoscopic training on thiel human cadavers: a model to teach advanced laparoscopic procedures. Surg Endosc 22:901–906
Wyles SM, Miskovic D, Ni Z, Acheson AG, Maxwell-Armstrong C, Longman R, Cecil T, Coleman MG, Horgan AF, Hanna GB (2011) Analysis of laboratory-based laparoscopic colorectal surgery workshops within the english national training programme. Surg Endosc 25:1559–1566
LeBlanc F, Champagne BJ, Augestad KM, Neary PC, Senagore AJ, Ellis CN, Delaney CP (2010) A comparison of human cadaver and augmented reality simulator models for straight laparoscopic colorectal skills acquisition training. J Am Coll Surg 211:250–255
Cohen J, Cohen SA, Vora KC, Xue X, Burdick JS, Bank S, Bini EJ, Bodenheimer H, Cerulli M, Gerdes H, Greenwald D, Gress F, Grosman I, Hawes R, Mullin G, Schnoll-Sussman F, Starpoli A, Stevens P, Tenner S, Villanueva G (2006) Multicenter, randomized, controlled trial of virtual-reality simulator training in acquisition of competency in colonoscopy. Gastrointest Endosc 64:361–368
Orzech N, Palter VN, Reznick RK, Aggarwal R, Grantcharov TP (2012) A comparison of 2 ex vivo training curricula for advanced laparoscopic skills: a randomized controlled trial. Ann Surg 255:833–839
Gromski MA, Ahn W, Matthes K, De S (2016) Pre-clinical training for new notes procedures: from ex-vivo models to virtual reality simulators. Gastrointest Endosc Clin N Am 26:401–412
Izawa Y, Hishikawa S, Muronoi T, Yamashita K, Maruyama H, Suzukawa M, Lefor AK (2016) Ex-vivo and live animal models are equally effective training for the management of a penetrating cardiac injury. World J Emerg Surg 11:45
BeyerBerjot L, Palter V, Grantcharov T, Aggarwal R (2014) Advanced training in laparoscopic abdominal surgery: a systematic review. Surgery 156:676–688
Palter VN, Orzech N, Aggarwal R, Okrainec A, Grantcharov TP (2010) Resident perceptions of advanced laparoscopic skills training. Surg Endosc 24:2830–2834
Blackburn SC, Griffin SJ (2014) Role of simulation in training the next generation of endoscopists. World J Gastrointest Endosc 6:234–239
Hammoud MM, Nuthalapaty FS, Goepfert AR, Casey PM, Emmons S, Espey EL, Kaczmarczyk JM, Katz NT, Neutens JJ, Peskin EG (2008) To the point: medical education review of the role of simulators in surgical training. Am J Obstet Gynecol 199:338–343
Russell WMS, Burch RL (1959) The principles of humane experimental technique. Methuen & Co Ltd, London
Ceresa CDL, Nasralla D, Knight S, Friend PJ (2017) Cold storage or normothermic perfusion for liver transplantation: probable application and indications. Curr Opin Organ Transplant 22:300–305
He X, Ji F, Zhang Z, Tang Y, Yang L, Huang S, Li W, Su Q, Xiong W, Zhu Z, Wang L, Lv L, Yao J, Zhang L, Zhang L, Guo Z (2018) Combined liver-kidney perfusion enhances protective effects of normothermic perfusion on liver grafts from donation after cardiac death. Liver Transplant 24:67–79
Zhao Q, Huang S, Wang D, Zhang Z, Wu L, Yang L, Ma Y, Ji F, Tang Y, Wang L, Zhu Z, Zhu Y, Xiong W, Chen M, Han M, Zhou J, Hu A, Wang G, Jiao X, Zhu X, Ju W, Guo Z, He X (2018) Does ischemia free liver procurement under normothermic perfusion benefit the outcome of liver transplantation? Ann Transplant 23:258–267
Zhao Q, Nie Y, Guo Z, He X (2019) The future of organ-oriented research and treatment. Hepatobiliary Surg Nutr 8:502–505
Kaneko JJ (2013) Clinical Biochemistry of domestic animals. Academic Press, Cambridge
Liu W, Zheng X, Wu R, Jin Y, Kong S, Li J, Lu J, Yang H, Xu X, Lv Y, Zhang X (2018) Novel laparoscopic training system with continuously perfused ex-vivo porcine liver for hepatobiliary surgery. Surg Endosc 32:743–750
Grober ED, Hamstra SJ, Wanzel KR, Reznick RK, Matsumoto ED, Sidhu RS, Jarvi KA (2004) The educational impact of bench model fidelity on the acquisition of technical skill: the use of clinically relevant outcome measures. Ann Surg 240:374–381
Strickland A, Fairhurst K, Lauder C, Hewett P, Maddern G (2011) Development of an ex vivo simulated training model for laparoscopic liver resection. Surg Endosc 25:1677–1682
Bittner JGT, Coverdill JE, Imam T, Deladisma AM, Edwards MA, Mellinger JD (2008) Do increased training requirements in gastrointestinal endoscopy and advanced laparoscopy necessitate a paradigm shift? A survey of program directors in surgery. J Surg Educ 65:418–430
Acknowledgements
The authors thank the experts for participating in the trial and the staff in Singularity Medicine company for their assistance with setting up the Smagister simulator.
Funding
This study was supported by the National Natural Science Foundation of China (81970564, 81471583 and 81570587), the Guangdong Provincial Key Laboratory Construction Projection on Organ Donation and Transplant Immunology (2013A061401007 and 2017B030314018), Guangdong Provincial Natural Science Funds for Major Basic Science Culture Project (2015A030308010), Guangdong Provincial Natural Science Funds for Distinguished Young Scholars (2015A030306025), Special Support Program for Training High Level Talents in Guangdong Province (2015TQ01R168) and Science and Technology Program of Guangzhou (201704020150).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosures
Xiaobo Wang, Kunsong Zhang, Wenjie Hu, Ming Kuang, Serene Teo, Zhiyong Guo, Qiang Zhao and Xiaoshun He have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, X., Zhang, K., Hu, W. et al. A new platform for laparoscopic training: initial evaluation of the ex-vivo live multivisceral training device. Surg Endosc 35, 374–382 (2021). https://doi.org/10.1007/s00464-020-07411-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-020-07411-z