Abstract
Breeding seabirds are central-place foragers and therefore exploit food resources most intensively nearer their colonies. When nesting aggregations are close to one another density-dependent competition is likely to be high, potentially promoting foraging segregation (i.e. neighbouring colonies may segregate to search for food in different areas). However, little is known about spatial segregation in foraging behaviour between closely adjacent colonies, particularly in species that are wide-ranging foragers. Here, we tested for foraging segregation between two sub-colonies of a wide-ranging seabird, Cory’s shearwater Calonectris borealis, separated by only 2 km, on a small Island in the North Atlantic. During the 2010 chick-rearing period, 43 breeding adults of both sexes were simultaneously sampled at both sub-colonies. A GPS logger was deployed on each individual and removed after several foraging trips at sea. Blood samples (plasma and red blood cells) were collected from each tracked individual for stable isotope analysis. Results indicated partial spatial segregation between the two sub-colonies during local foraging trips (i.e. those of ≤1 day duration and 216 km from the colony) accounting for 84.2 % of all trips recorded. The location of the breeding sub-colony influenced the direction of travel of birds during local trips resulting in sub-colony-specific foraging areas. Although the oceanographic conditions associated with the foraging range of the two sub-colonies differed, no differences were found in the habitat exploited and in their estimated diets. This suggests that birds concentrated their feeding activity in patches of similar habitat and prey during the chick-rearing period.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
When seabird colonies are located near each other, potential home ranges will often overlap and this may result in the partitioning of resources between members of different colonies (Wakefield et al. 2013). As seabirds are typically not territorial at sea (except for those species that engage in piracy and aggressive interactions in search of food), intra-specific competition for food in pelagic seabirds is often difficult to demonstrate (Furness and Birkhead 1984; Lewis et al. 2001). However, it may drive segregation of foraging resources between neighbouring colonies of several seabird species (e.g. Masello et al. 2010; Wiley et al. 2012). According to Ashmole (1963), high intra-specific competition, which is frequently density-dependent, is mainly attributed to large colonies during the breeding season, which may result in prey depletion in their vicinity and longer foraging ranges to obtain food (Furness and Birkhead 1984; Lewis et al. 2001; Garthe et al. 2011). However, the Ashmole model does not consider interactions between adjacent colonies and implicitly assumes that their home ranges overlap (Furness and Birkhead 1984; Wakefield et al. 2013). On the other hand, Cairns (1989) proposed that colony locations are limited by geography and, once they are established, they should be spaced so that their home ranges do not overlap (the hinterland model); this model predicts that colony home ranges will segregate along lines of equidistance between colonies.
Breeding seabirds are central-place foragers, resulting in populations exploiting prey around their colonies (Phillips et al. 2009), which, in turn, may be limited by local oceanographic conditions such as bathymetry, sea surface temperature and chlorophyll-a concentration (Masello et al. 2010). We can expect segregation in both foraging areas and prey between geographically distant (i.e. hundreds of kilometres) breeding populations, perhaps leading to diversification of foraging tactics between populations and ultimately speciation. In a recent paper, Wakefield et al. (2013) provide strong evidence that wide-ranging seabirds in neighbouring colonies (i.e. in adjacent colonies), breeding around the British Isles, forage in largely mutually exclusive areas and that these colony-specific home ranges are determined by density-dependent competition. They proposed a multi-colony extension of Ashmole’s model, by combining predictions of models of both Ashmole and Cairns, to explain how spatial segregation might occur between neighbouring colonies (Weimerskirch 2013).
Several studies (e.g. Grémillet et al. 2004; Yamamoto et al. 2011; Wiley et al. 2012) demonstrated spatial foraging segregation between colonies in wide-ranging seabirds (maximum foraging range greater than 60 km), but these were separated by tens or hundreds of kilometres. Studies demonstrating spatial foraging segregation within seabird species breeding in adjacent colonies or sub-colonies as close as 2–2.5 km apart (i.e. at a small spatial scale) are scarce, especially concerning wide-ranging species. Wanless and Harris (1993) and Masello et al. (2010) studied South Georgia shags Phalacrocorax georgianus and three penguin species, respectively, but these species generally have smaller foraging ranges at sea (i.e. maximum foraging range typically less than 40 km). However, Waggitt et al. (2014) examined variation in foraging behaviour between closely spaced sub-colonies of a more wide-ranging seabird species, the northern gannet Morus bassanus, finding no apparent foraging segregation between sub-colonies.
Large populations of Cory’s shearwaters Calonectris borealis breed in the Azores, a chain of nine islands aggregated into three groups (western, central and eastern) straddling the mid-Atlantic ridge over a distance of about 600 km. The islands of the Azores have relatively narrow peri-insular shelves and are surrounded by deep, oceanic waters. Corvo Island is the smallest (ca. 17 km2, Fig. 1) and holds a greater density (mean of 2,732 birds per km of coast) of breeding Cory’s shearwater than any other in the archipelago (Furness et al. 2000). Given this high density of birds nesting on Corvo, it is an appropriate site to test for between-sub-colony foraging segregation within the same colony. We tested the hypothesis that spatial and dietary segregation occur between two sub-colonies of Cory’s shearwaters separated by only 2 km during early chick-rearing when their foraging effort is highest and most highly concentrated in the vicinity of the colony (Granadeiro et al. 2000; Paiva et al. 2010a; Ceia et al. 2014). Cory’s shearwater is sexually dimorphic in body size and bill dimensions (Granadeiro 1993; Ramos et al. 2009a), suggesting possible at-sea foraging differences between sexes that could confound the sub-colony differences. Therefore, we assessed differences between males and females. Specifically, we predicted that: (1) central-place foragers use sub-colony-specific foraging areas by partitioning available space (i.e. geographical location) and marine habitats (i.e. a set of environmental variables) during the early chick-rearing period; and (2) individuals from different sub-colonies display dietary segregation throughout the breeding period (i.e. throughout incubation and chick-rearing). Our goal was to measure differences and patterns associated with spatial distributions at sea, habitat usage, dietary preferences and trophic positions to determine whether small-scale differences in breeding location could potentially cause differences between sub-colonies in foraging ecology at sea. We tested for foraging segregation between proximate seabird sub-colonies whose putative home ranges completely overlap, using a combination of movement data of individual birds, environmental variables, stable isotope analysis of the blood and more conventional direct dietary sampling. We measured stable isotope values (δ13C and δ15N) of plasma and red blood cells, reflecting diet over a few days and over the previous 3–4 weeks, respectively (Hobson and Clark 1993; Votier et al. 2010). The carbon stable isotope value (i.e. δ13C) mainly reflects the consumer’s foraging habitat while the nitrogen stable isotope value (i.e. δ15N) mainly reflects the consumer’s trophic position (Cherel et al. 2005b).
Materials and methods
Fieldwork was conducted in Corvo Island (39°40′N, 31°06′W; Fig. 1) in the Azores archipelago, Portugal, during the 2010 chick-rearing period. Forty-three breeding adults of two sub-colonies of Cory’s shearwaters separated by 2 km (Fig. 1) were sampled over 35 days, between 30 July and 2 September (inclusive). The time between capture and recapture ranged from 2 to 30 days (mean ± SD: 14.9 ± 5.5 days). At capture and recapture, birds were ringed, and body mass (to the nearest 10 g) and wing length (to the nearest 1 mm) were measured. Although sex was known for most birds, bill measurements (culmen, bill width at the gonys and at the base) were taken to determine the sex of remaining birds (using a discriminant function established by Granadeiro 1993). A GPS logger was deployed on birds tending chicks, and removed after several foraging trips (details of the devices below). Birds from the two sub-colonies were tracked simultaneously (Online Resource 1). Blood samples (0.5–1 ml from the tarsal vein) were collected from each bird on capture and recapture using 25-G needles. Stomach contents were sampled from 20 tracked individuals from both sub-colonies by water-offloading (after Wilson 1984). Deployment or retrieval of devices, collection of samples and release took 10–15 min and birds were returned immediately to their nests. Blood samples were separated into plasma and red blood cell (RBC) fractions, within 2–3 h, using a centrifuge (15 min at 2,910g). Hematocrit was recorded and samples were then stored frozen until stable isotope analyses were conducted.
Tracking data collection
Each bird was fitted with a GPS logger (CatTraq GT-120; Perthold Engineering). The total mass of the device (17 g) corresponded to 1.6–2.6 % of avian body mass which is below the 3 % of adult mass critical threshold above which deleterious effects occur on seabird species, including Cory’s shearwaters, during short-term deployments (Phillips et al. 2003; Igual et al. 2005; but see Vandenabeele et al. 2012). Each GPS logger was attached to feathers in the mantle region with Tesa® tape, and recorded the bird’s GPS coordinate (median error of <10 m) every 5 min. We tracked birds continuously from 2 to 17 days (12.7 ± 3.7 days).
Environmental data
We characterized Cory’s shearwater oceanographic habitat use using three environmental variables: bathymetry (m), August 2010 mean chlorophyll-a concentration (Chl a, mg m−3) and sea surface temperature (SST, °C) (Paiva et al. 2010b). Bathymetry data were extracted from a grid of 0.01º (approx. 1 km) from http://www.ngdc.noaa.gov/mgg/global/global.html (we selected the ETOPO1 blended product). Chl a and SST data were downloaded for a spatial resolution of 0.04º (approx. 4 km) of Aqua-MODIS mapped products from http://oceancolor.gsfc.nasa.gov/cgi/13.
Diet sampling and identification
All regurgitant samples came from breeding birds but empty stomachs were not considered in the analysis. Following Xavier et al. (2004), regurgitant samples were separated into oil and solid fractions. Each solid fraction, containing fish and cephalopods, was separated into individual dietary constituents which were counted and weighed, with each prey item identified to species level whenever possible. Fresh squid and fish (with beaks and otoliths attached, respectively) were stored frozen for stable isotope analyses.
Stable isotope analyses (SIA)
For each bird blood sampled, we analysed δ13C (‰) and δ15N (‰) in the RBC fraction from initial capture and the plasma fraction from recapture (difference of between 2 and 30 days) to minimize dietary overlap as represented by samples (Votier et al. 2010; Ceia et al. 2012). δ13C (‰) and δ15N (‰) of fresh prey items obtained from regurgitants were also analysed to allow interpretation of isotopic values of blood fractions through construction of mixing models (see data analysis below).
Samples were freeze-dried and homogenized prior to SIA. Because high lipid concentrations in plasma and in soft tissues of prey items can result in depleted δ13C values, lipids were removed using successive rinses in a 2:1 chloroform–methanol solution (Cherel et al. 2005b). Nitrogen and carbon isotope values were determined via Finnigan conflo II interface to a Thermo Delta V S mass spectrometer coupled to a Flash EA1112 Series elemental analyser, following Ceia et al. (2014). Isotope values are expressed as: δ13C and δ15N = [(R sample/R standard) − 1] × 1,000, where R = 13C/12C and 15N/14N, respectively. The standards for carbon and nitrogen isotope values were Vienna PeeDee Belemnite (V-PDB) and atmospheric N2 (AIR), respectively. Replicate measurements of internal laboratory standards (acetanilide STD: Thermo scientific—PN 338 36700, C and N contents of 71.09 and 10.36 %, respectively) indicate precision <0.2 ‰ for both δ13C and δ15N.
Data analysis
We limited our detailed analysis to trips ≤1 day duration and ≤216 km from the colony (i.e. local trips). The number and frequency of local trips were sufficiently robust to test our hypothesis that birds from the two sub-colonies use sub-colony-specific foraging areas by partitioning available space and oceanographic habitats. Therefore, the subsequent analyses of spatial foraging segregation were only performed for local trips.
The non-parametric fixed kernel density (FKD) estimator was used to calculate the 25, 50 and 75 % density contour areas, and respective areas (km2), of each local trip using the adehabitat package (h = 0.03º, grid = 500; Calenge 2006) in R 2.14.0 (R Development Core Team 2011). GPS loca tions at the colony were excluded from analyses and we only used foraging-point kernels, based on the areas where birds were presumed to be foraging (by trip sinuosity index ≥3; adapted from Grémillet et al. 2004). The degree of overlap in the estimated home ranges of individuals from different sub-colonies was calculated based on the 25, 50 and 75 % FKD overlap for each bird to assess space-use shared between birds, using the function “kerneloverlap” of the adehabitat package (meth = “VI”; Calenge 2006) in R 2.14.0. Zones of area-restricted search (ARS) where prey capture is more predictable (i.e. areas in which the tracking data suggest the foraging bird increases its search effort by increasing its turning rate and reducing its speed; Weimerskirch et al. 2007) were estimated for each trip by applying first-passage time (FPT) analysis (see Fauchald and Tveraa 2003) and using the adehabitat package (Calenge 2006) in R 2.14.0. Usually, positions which correspond to the bird sitting on water result in small-scale ARS zones (i.e.<100 m diameter), increasing the variance in FPT and reducing the ability to detect larger-scale ARS zones (Weimerskirch et al. 2007). To address this problem, we removed bouts on the water (i.e. when speed was <9 km h−1) and interpolated locations to obtain a distance interval of 1 km for FPT analysis (Pinaud 2008). Following Pinaud (2008), FPT analysis was performed in two steps to detect: (1) large-scale ARS zones, for which we ran the analysis on the whole path, estimating the FPT every 25 km for a radius r from 1 to 300 km; and (2) small spatial scale events, for which we ran the FPT analysis again every 1 km for r varying between 1 and 50 km. The plot representing variance in log (FPT) as a function of r allowed us to identify the ARS scales by peaks in the variance. In this calculation, FPT was log-transformed to make the variance independent of the magnitude of the mean FPT (after Fauchald and Tveraa 2003). The centroid of geographic position (i.e. latitude and longitude) in each ARS zone was calculated for each trip and used to assess spatial differences between sub-colonies in the zones exploited. Given the geometry of the two sub-colonies (i.e. sub-colony A on east side and sub-colony B on west side), we asked whether breeding site had an effect on departure direction of birds on each foraging trip. Following Robson et al. (2004), the bearing (in degrees) of the most distant location recorded during each trip from each sub-colony was used to test the null hypothesis that there would be no difference in the direction of foraging trips of birds from different sub-colonies. This question was tested using a Watson-Williams test for circular data using the circular package (Agostinelli and Lund 2011) in R 2.14.0. One trip for each bird was randomly selected to control for pseudoreplication, because more than one foraging trip per individual was recorded (7.2 ± 3.4 trips per bird). Mean values of environmental variables inside the kernel contours and ARS zones of each trip were extracted using ArcGIS 9.2. With the exception of circular variables, behavioural indices were modelled using mixed-effects ANOVA with sex, sub-colony and environmental variables treated as fixed effects and the individual as a random effect.
Mixing models were used to estimate the relative proportion of different dietary sources. We adopted a Bayesian multi-source stable isotope mixing model (stable isotope analyses in R: SIAR, function “siarsolomcmcv4”; Parnell et al. 2010) to estimate contributions of each prey type to the diet of each individual. All possible combinations of each source contribution were examined using both isotope values (δ13C and δ15N) from RBC on capture and plasma from recapture (corresponding to incubation and chick-rearing periods, respectively) for each bird, and the mean and standard deviation of each of the three prey types collected from the regurgitant samples. To the best of our knowledge, no studies have assessed isotopic discrimination factors between food and blood for Cory’s shearwaters or other procellariform species; hence, we used the mean values of isotopic discrimination factors between marine prey and blood of four seabird species available from the literature: 0.3 and 2.3 ‰ enrichment for carbon and nitrogen, respectively (Bearhop et al. 2002; Cherel et al. 2005a; Ramos et al. 2009b). Due to potential differences in isotopic discrimination factors among species, a standard deviation of ±0.5 ‰ was assumed (Votier et al. 2010; Ceia et al. 2012; Pedro et al. 2013).
We used the standard residuals of the relationship between body mass (mean between capture and recapture) and wing length (F 1,39 = 11.5, r = 0.48, P = 0.002) to estimate the body condition of 41 sampled birds to assess whether it differed between adults from the two sub-colonies. In addition, the variation in the hematocrit (% of RBC in total blood volume on recapture − % RBC in total blood volume on capture) was calculated as a proxy of the individuals’ current health condition status (reviewed in Fair et al. 2007).
All data were tested for normality and homoscedasticity; in order to meet normality, foraging area (25, 50 and 75 % FKD) and Chl a were log10-transformed, bearings (degrees) were transformed into radians and proportions were arcsine square-root transformed, with exception of those obtained from SIAR mixing model. Values are presented as mean ± SD unless otherwise stated.
Results
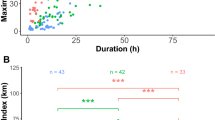
We obtained a total of 368 foraging trips from 43 birds from the two sub-colonies (A: 23, and B: 20 birds). The frequencies of trip duration for the entire dataset showed a clear unimodal distribution during the study period, during which 310 (84.2 %) were ≤1 day made by 42 individuals and 58 (15.8 %) were >1 day made by 32 individuals (Fig. 2). Local foraging trips were confined to the colony surroundings over a maximum distance of 216 km (62 ± 39 km).
Spatial segregation and sub-colony-specific foraging areas
First-passage time analyses indicated that birds from the two sub-colonies foraged at significantly different longitudes with therefore distinctly different ARS zones during local trips, but no significant differences were found in their latitudinal positions (Table 1). The overlap between birds from different sub-colonies during local trips was 16.2 ± 4.2, 10.5 ± 3.0 and 6.0 ± 2.1 % at 75, 50 and 25 % fixed kernel densities, respectively. Overall, during local trips, birds from sub-colonies on respective east (A) and west (B) sides of the island foraged predominantly on that side of the island (Fig. 3). We found that birds from the two sub-colonies travelled in different directions to forage during local trips; the bearing from the colony to the most distant location differed significantly between sub-colonies [A: mean 84.4º (circular variance = 38.0º); B: mean 251.2º (circular variance = 52.5º); Watson–William test: F 1,40 = 8.5, P = 0.006], suggesting birds from different sub-colonies favoured different foraging areas. Differences were also found in the size of foraging areas during local trips with sub-colony A members foraging over larger areas at 75, 50 and 25 % FKDs (Table 1). No differences were found in the spatial distribution and in the size of foraging areas between males and females during local trips (Table 1).
Spatial distribution of Cory’s shearwaters Calonectris borealis breeding in two sub-colonies (A: upper panel; B: lower panel) on Corvo Island during the 2010 chick-rearing period and foraging locally (i.e. for ≤1 day duration over 216 km from the colony). Dashed line indicates the bound of equidistance between the two sub-colonies. Dark, medium and light shades represent 25, 50 and 75 % kernel densities, respectively. White triangle indicates the location of Corvo Island
Oceanographic habitat
Fixed kernel densities (i.e. 75, 50 and 25 %) indicated that environmental variables (i.e. SST, Chl a and bathymetry) within the foraging areas of birds from the two sub-colonies during local trips diverged significantly (Table 2). Overall, birds from sub-colony B foraged over shallower and warmer waters with greater Chl a than birds from sub-colony A. However, there were no significant differences in the environmental variables in ARS zones estimated by FPT analyses (Table 2). No significant between-sex differences were found in the oceanographic conditions experienced during local trips, estimated by FKDs (75, 50 and 25 %) or FPT analyses, except in sea surface temperature at 75 % FKD (Table 2).
Diet and stable isotope analysis
Diet samples were obtained by stomach irrigation from 18 birds during the chick-rearing period. These mainly comprised fish (98.5 % by mass and 90.7 % by numerical frequency) and a small amount of cephalopods (1.5 % by mass and 9.3 % by numerical frequency). The three prey species identified in flushed material were boarfish Capros aper, blue jack mackerel Trachurus picturatus and cock-eyed squid Histioteuthis sp.
Mean δ13C and δ15N were greater in the cephalopod species (i.e. Histioteuthis sp.) followed by the two fish species, T. picturatus and C. aper, respectively (Fig. 4). δ13C and δ15N in the plasma and RBC fractions of blood from 43 birds were greater than in their prey. There were no differences in the mean δ13C and δ15N of Cory’s shearwaters plasma and RBC fractions of blood between sub-colonies, sexes and sub-colony by sex (Online Resource 2).
The SIAR mixing model based on plasma and RBC fractions, and prey isotope values, indicated that C. aper was the largest component in the diet of birds during the chick-rearing and the incubation periods (37.7 and 40.1 %, respectively) followed by T. picturatus (33.0 and 32.4 %, respectively) and Histioteuthis sp. (29.3 and 27.5 %, respectively). No significant differences were found in the proportions of the three prey items in the diet of birds estimated by the model based on isotope values of the plasma fraction (representing the chick-rearing period) in statistical comparisons between sub-colonies, sexes and sub-colony by sex (Online Resource 3).
Body condition
Body condition varied between −1.81 and 1.85 g in all birds. Birds from different sub-colonies did not differ significantly in body condition and nor was there a significant sub-colony by sex interaction (Online Resource 4). However, males were in higher body condition than females (F 1,37 = 8.4, P = 0.006). There was no effect of sub-colony, sex or sub-colony by sex on the hematocrit variation, varying overall between −0.09 and 0.05 % (Online Resource 4).
Discussion
We used Cory’s shearwater as a model species that is a wide-ranging top predator to compare the foraging distributions and the trophic ecology of breeding birds from two different sub-colonies on a North Atlantic island. Our analysis of foraging trips was limited to local trips that accounted for 84.2 % of all trips recorded. Therefore, our conclusions are restricted to segregation in the vicinity of the colony (i.e. within approx. 200 km) and should not be extrapolated to a larger scale. The sub-colonies were much closer to each other (i.e. 2 km) than the maximum foraging range of the species during local trips (216 km; mean: 62 ± 39 km). Therefore, we expected a large overlap in the foraging areas and diets of birds from different sub-colonies.
Kernel and first-passage time analyses indicated that birds from different sub-colonies selected different areas in which to forage during local trips. However, spatial segregation was only partial, not absolute as predicted by Cairns (1989). Several studies have demonstrated partial or complete spatial foraging segregation for seabirds, such as in black-browed albatrosses Thalassarche melanophris (Huin 2002; Granadeiro et al. 2011), cape gannets Morus capensis (Grémillet et al. 2004), northern gannets (Wakefield et al. 2013), Hawaiian petrels Pterodroma sandwichensis (Wiley et al. 2012) and streaked shearwaters Calonectris leucomelas (Yamamoto et al. 2011). Wanless and Harris (1993) and Masello et al. (2010) found partial and/or complete spatial segregation in foraging seabirds from different colonies that were only 2–2.5 km apart. Wanless and Harris (1993) attributed inter-colony differences in foraging areas and diets of South Georgia shags during the breeding season to highly localized differences in feeding conditions, given the extremely limited foraging range of most individuals (ca. 1 km from the colony). On the other hand, Masello et al. (2010) attributed foraging segregation among colonies of three penguin species to intra- and inter-specific competition, and also to reductions in predation risk from South American fur seals Arctocephalus australis that were present on the island.
Our observations that birds from the two sub-colonies travelled in different directions to forage during local trips suggest that specific foraging areas are determined by breeding location (i.e. sub-colony membership). Although the potential foraging areas overlapped, our results demonstrated that birds preferred areas adjacent to their own sub-colony rather than the neighbouring sub-colony in accordance with Cairns (1989) and Wakefield et al. (2013). Robson et al. (2004) studied northern fur seals Callorhinus ursinus, and suggested that a directional bias in foraging behaviour could be explained by individuals repeatedly initiating foraging trips on a bearing consistent with the general orientation of the breeding site. This directional tendency may be reinforced by public information transfer between foragers (Ward and Zahavi 1973). This results in cultural evolution and divergence between colonies and may allow active segregation between members of neighbouring colonies (Wakefield et al. 2013; Weimerskirch 2013). On Corvo Island, there were dispersed birds breeding along the cliffs, but these sub-colonies are inaccessible. This results in biases to our sampling effort towards the south-east- and south-west-located sub-colonies on Corvo Island. However, the two studied sub-colonies are separated by only 2 km and isolated from each other by rocky outcrops; hence, birds from one sub-colony were visually isolated from the other while at the nest.
The oceanographic habitat used by birds in the current study was significantly different between sub-colonies during local trips. Surprisingly, however, differences detected by kernel analyses (i.e. explored areas) were not detected by first-passage time analyses (i.e. zones of area-restricted search), which correspond to the environment where the probability of prey capture should be higher. These results suggest that birds from both sub-colonies searched various wide areas for food but focussed their feeding in small concentrated patches of shared oceanographic characteristics. Top predators appear to forage at locations where prey accumulate due to specific physical processes and/or due to patchiness in primary or secondary productivity (e.g. oceanographic fronts, currents, eddies, seamounts or upwelling zones) (Bost et al. 2009; Paiva et al. 2010c). Ultimately, such locations could share similar characteristics, especially at a smaller scale (Wakefield et al. 2009).
Despite specificity of foraging areas according to sub-colony membership, which resulted in partial spatial segregation during the local trips, no differences were detected between sub-colonies in the estimated diet and in the stable isotope values of plasma and RBC fractions of blood. This strongly suggests no differences in the habitat exploited by birds in the zones of area-restricted search during chick-rearing.
In summary, we observed that Cory’s shearwaters breeding in close sub-colonies on Corvo Island showed a partial spatial segregation during chick-rearing when foraging locally. No such differences were found between males and females. Birds tracked simultaneously from the sub-colonies travelled in different directions according to their sub-colony origins during the local trips, resulting in specific and distinct foraging areas. Birds preferred areas adjacent to their own sub-colony with a bearing consistent with the general orientation of their breeding site. Our results suggest that birds from both sub-colonies foraged in areas with different oceanographic conditions during local trips, but concentrated their feeding activity in patches of similar habitat. Consequently, individuals from both sub-colonies did not display dietary segregation during early chick-rearing and no differences in body condition or hematocrit status were found when comparing birds from different sub-colonies.
References
Agostinelli C, Lund U (2011) R package ‘circular’: circular statistics (version 0.4–3). https://r-forge.r-project.org/projects/circular/. Accessed 05 June 2013
Ashmole NP (1963) The regulation of numbers of tropical and oceanic birds. Ibis 103:458–473. doi:10.1111/j.1474-919X.1963.tb06766.x
Bearhop S, Waldron S, Votier SC, Furness RW (2002) Factors that influence assimilation rates and fractionation of nitrogen and carbon stable isotopes in avian blood and feathers. Physiol Biochem Zool 75:451–458. doi:10.1086/342800
Bost CA, Cotté C, Bailleul F, Cherel Y, Charrassin JB, Guinet C, Ainley DG, Weimerskirch H (2009) The importance of oceanographic fronts to marine birds and mammals of the southern oceans. J Mar Syst 78:363–376. doi:10.1016/j.jmarsys.2008.11.022
Cairns DK (1989) The regulation of seabird colony size: a hinterland model. Am Nat 134:141–146
Calenge C (2006) The package “adehabitat” for the R software: a tool for the analysis of space and habitat use by animals. Ecol Modell 197:516–519. doi:10.1016/j.ecolmodel.2006.03.017
Ceia FR, Phillips RA, Ramos JA, Cherel Y, Vieira RP, Richard P, Xavier JC (2012) Short- and long-term consistency in the foraging niche of wandering albatrosses. Mar Biol 159:1581–1591. doi:10.1007/s00227-012-1946-1
Ceia FR, Paiva VH, Garthe S, Marques JC, Ramos JA (2014) Can variations in the spatial distribution at sea and isotopic niche width be associated with consistency in the isotopic niche of a pelagic seabird species? Mar Biol 161:1861–1872. doi:10.1007/s00227-014-2468-9
Cherel Y, Hobson KA, Hassani S (2005a) Isotopic discrimination between food and blood and feathers of captive penguins: implications for dietary studies in the wild. Physiol Biochem Zool 78:106–115. doi:10.1086/425202
Cherel Y, Hobson KA, Weimerskirch H (2005b) Using stable isotopes to study resource acquisition and allocation in procellariiform seabirds. Oecologia 145:533–540. doi:10.1007/s00442-005-0156-7
Fair J, Whitaker S, Pearson B (2007) Sources of variation in haematocrit in birds. Ibis 149:535–552. doi:10.1111/j.1474-919X.2007.00680.x
Fauchald P, Tveraa T (2003) Using first-passage time in the analysis of area-restricted search and habitat selection. Ecology 84:282–288
Furness RW, Birkhead TR (1984) Seabird colony distributions suggest competition for food supplies during the breeding season. Nature 311:655–656. doi:10.1038/311655a0
Furness RW, Hilton G, Monteiro LR (2000) Influences of coastal habitat characteristics on the distribution of Cory’s Shearwaters Calonectris diomedea in the Azores archipelago. Bird Study 47:257–265. doi:10.1080/00063650009461185
Garthe S, Montevecchi WA, Davoren GK (2011) Inter-annual changes in prey fields trigger different foraging tactics in a large marine predator. Limnol Oceanogr 56:802–812. doi:10.4319/lo.2011.56.3.0802
Granadeiro JP (1993) Variation in measurements of Cory’s shearwater between populations and sexing by discriminant analysis. Ringing Migr 14:103–112. doi:10.1080/03078698.1993.9674051
Granadeiro JP, Bolton M, Silva MC, Nunes M, Furness RW (2000) Responses of breeding Cory’s shearwater Calonectris diomedea to experimental manipulation of chick condition. Behav Ecol 11:274–281. doi:10.1093/beheco/11.3.274
Granadeiro JP, Phillips RA, Brickle P, Catry P (2011) Albatrosses following fishing vessels: how badly hooked are they on an easy meal? PLoS ONE 6:e17467. doi:10.1371/journal.pone.0017467
Grémillet D, Dell’Omo G, Ryan PG, Peters G, Ropert-Coudert Y, Weeks SJ (2004) Offshore diplomacy or how seabirds mitigate intra-specific competition: a case study based on GPS tracking of Cape gannets from neighbouring colonies. Mar Ecol Prog Ser 268:265–279. doi:10.3354/meps268265
Hobson KA, Clark RG (1993) Turnover of d13C in cellular and plasma reactions of blood: implications for nondestructive sampling in avian dietary studies. Auk 110:638–641
Huin N (2002) Foraging distribution of the black-browed albatross, Thalassarche melanophris, breeding in the Falkland Islands. Aquat Conserv Mar Freshw Ecosyst 12:89–99. doi:10.1002/aqc.479
Igual JM, Forero MG, Tavecchia G, Gonzalez-Solis J, Martinez-Abrain A, Hobson KA, Ruiz X, Oro D (2005) Short-term effects of data-loggers on Cory’s shearwater (Calonectris diomedea). Mar Biol 146:619–624. doi:10.1007/s00227-004-1461-0
Lewis S, Sherratt TN, Hamer KC, Wanless S (2001) Evidence of intra-specific competition for food in a pelagic seabird. Nature 412:816–819. doi:10.1038/35090566
Masello JF, Mundry R, Poisbleau M, Demongin L, Voigt CC, Wikelski M, Quillfeldt P (2010) Diving seabirds share foraging space and time within and among species. Ecosphere 1:1–20. doi:10.1890/ES10-00103.1
Paiva VH, Geraldes P, Ramírez I, Meirinho A, Garthe S, Ramos JA (2010a) Foraging plasticity in a pelagic seabird species along a marine productivity gradient. Mar Ecol Prog Ser 398:259–274. doi:10.3354/meps08319
Paiva VH, Geraldes P, Ramírez I, Garthe S, Ramos JA (2010b) How area restricted search of a pelagic seabird changes while performing a dual foraging strategy. Oikos 119:1423–1434. doi:10.1111/j.1600-0706.2010.18294.x
Paiva VH, Geraldes P, Ramírez I, Meirinho A, Garthe S, Ramos JA (2010c) Oceanographic characteristics of areas used by Cory’s shearwaters during short and long foraging trips in the North Atlantic. Mar Biol 157:1385–1399. doi:10.1007/s00227-010-1417-5
Parnell AC, Inger R, Bearhop S, Jackson AL (2010) Source partitioning using stable isotopes: coping with too much variation. PLoS ONE 5:e9672. doi:10.1371/journal.pone.0009672
Pedro PI, Ramos JA, Neves VC, Paiva VH (2013) Past and present trophic position and decadal changes in diet of yellow-legged gull in the Azores archipelago, NE Atlantic. Eur J Wildl Res 59:833–845. doi:10.1007/s10344-013-0737-4
Phillips RA, Xavier JC, Croxall JP (2003) Effects of satellite transmitters on albatrosses and petrels. Auk 120:1082–1090. doi:10.1642/0004-8038
Phillips R, Wakefield E, Croxall J, Fukuda A, Higuchi H (2009) Albatross foraging behaviour: no evidence for dual foraging, and limited support for anticipatory regulation of provisioning at South Georgia. Mar Ecol Prog Ser 391:279–292. doi:10.3354/meps08028
Pinaud D (2008) Quantifying search effort of moving animals at several spatial scales using first-passage time analysis: effect of the structure of environment and tracking systems. J Appl Ecol 45:91–99. doi:10.1111/j.1365-2664.2007.01370.x
Ramos JA, Granadeiro JP, Phillips RA, Catry P (2009a) Flight morphology and foraging behavior of male and female Cory’s shearwaters. Condor 111:424–432. doi:10.1525/cond.2009.090008
Ramos R, Ramírez F, Sanpera C, Jover L, Xavier R (2009b) Feeding ecology of yellow-legged gulls Larus michahellis in the western Mediterranean: a comparative assessment using conventional and isotopic methods. Mar Ecol Prog Ser 377:289–297. doi:10.3354/meps07792
R Development Core Team (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Robson BW, Goebel ME, Baker JD, Ream RR, Loughlin TR, Francis RC, Antonelis GA, Costa DP (2004) Separation of foraging habitat among breeding sites of a colonial marine predator, the northern fur seal (Callorhinus ursinus). Can J Zool 82:20–29. doi:10.1139/Z03-208
Vandenabeele SP, Shepard EL, Grogan A, Wilson RP (2012) When three per cent may not be three per cent; device-equipped seabirds experience variable flight constraints. Mar Biol 159:1–14. doi:10.1007/s00227-011-1784-6
Votier SC, Bearhop S, Witt MJ, Inger R, Thompson D, Newton J (2010) Individual responses of seabirds to commercial fisheries revealed using GPS tracking, stable isotopes and vessel monitoring systems. J Appl Ecol 47:487–497. doi:10.1111/j.1365-2664.2010.01790.x
Waggitt J, Briffa M, Grecian W, Newton J, Patrick SC, Stauss C, Votier SC (2014) Testing for sub-colony variation in seabird foraging behaviour: ecological and methodological consequences for understanding colonial living. Mar Ecol Prog Ser 498:275–285. doi:10.3354/meps10628
Wakefield E, Phillips R, Matthiopoulos J (2009) Quantifying habitat use and preferences of pelagic seabirds using individual movement data: a review. Mar Ecol Prog Ser 391:165–182. doi:10.3354/meps08203
Wakefield ED, Bodey TW, Bearhop S, Blackburn J, Colhoun K, Davies R, Dwyer RG, Green JA, Grémillet D, Jackson AL, Jessopp MJ, Kane A, Langston RHW, Lescroël A, Murray S, Le Nuz M, Patrick SC, Péron C, Soanes LM, Wanless S, Votier SC, Hamer KC (2013) Space partitioning without territoriality in gannets. Science 341:68–70. doi:10.1126/science.1236077
Wanless S, Harris MP (1993) Use of mutually exclusive foraging areas by adjacent colonies of blue-eyed shags (Phalacrocorax atriceps) at South Georgia. Colon Waterbirds 16:176–182
Ward P, Zahavi A (1973) The importance of certain assemblages of birds as “information-centres” for food-finding. Ibis 115:517–534. doi:10.1111/j.1474-919X.1973.tb01990.x
Weimerskirch H (2013) Seabirds-individuals in colonies. Science 341:35–36. doi:10.1126/science.1240316
Weimerskirch H, Pinaud D, Pawlowski F, Bost CA (2007) Does prey capture induce area-restricted search? A fine-scale study using GPS in a marine predator, the wandering albatross. Am Nat 170:734–743. doi:10.1086/522059
Wiley AE, Welch AJ, Ostrom PH, James HF, Ainley DG, Duvall F, Holmes N, Hu D, Judge S, Penniman J, Swindle KA (2012) Foraging segregation and genetic divergence between geographically proximate colonies of a highly mobile seabird. Oecologia 168:119–130. doi:10.1007/s00442-011-2085-y
Wilson RP (1984) An improved stomach pump for penquins and other seabirds. J Ornithol 55:109–112
Xavier JC, Trathan PN, Croxall JP, Wood AG, Podesta G, Rodhouse PG (2004) Foraging ecology and interactions with fisheries of wandering albatrosses (Diomedea exulans) breeding at South Georgia. Fish Oceanogr 13:324–344. doi:10.1111/j.1365-2419.2004.00298.x
Yamamoto T, Takahashi A, Oka N, Iida T, Katsumata N, Sato K, Trathan P (2011) Foraging areas of streaked shearwaters in relation to seasonal changes in the marine environment of the Northwestern Pacific: inter-colony and sex-related differences. Mar Ecol Prog Ser 424:191–204. doi:10.3354/meps08973
Acknowledgments
This research was co-sponsored by the Foundation for Science and Technology (Portugal) and the European Social Fund (POPH, EU) through a PhD grant to Filipe R. Ceia (SFRH/BD/64558/2009). The authors would like to thank the support given by the LIFE project “Safe Islands for Seabirds” (LIFE07 NAT/P/000649). We thank P. Geraldes and C. Silva for help in the field, J. Xavier for help in identification of prey items, C. Docal for conducting stable isotope analyses and J. Reynolds for helpful corrections and comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Peggy Ostrom.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ceia, F.R., Paiva, V.H., Ceia, R.S. et al. Spatial foraging segregation by close neighbours in a wide-ranging seabird. Oecologia 177, 431–440 (2015). https://doi.org/10.1007/s00442-014-3109-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-014-3109-1