Abstract
Modulation of the actin cytoskeleton in chondrocytes has been used to prevent or reverse dedifferentiation and to enhance protein synthesis. We have hypothesized that an actin-modifying agent, staurosporine, could be used with fibrochondrocytes to increase the gene expression and synthesis of critical fibrocartilage proteins. A range of concentrations (0.1–100 nM) was applied to fibrochondrocytes in monolayer and evaluated after 24 h and after 4 days. High-dose staurosporine treatment (10–100 nM) increased cartilage oligomeric matrix protein 60– to 500-fold and aggrecan gene expression two-fold. This effective range of staurosporine was then applied to scaffoldless tissue-engineered fibrochondrocyte constructs for 4 weeks. Whereas glycosaminoglycan synthesis was not affected, collagen content doubled, from 27.6 ± 8.8 μg in the untreated constructs to 55.2 ± 12.2 μg per construct with 100 nM treatment. When analyzed for specific collagens, the 10-nM group showed a significant increase in collagen type I content, whereas collagen type II was unaffected. A concomitant dose-dependent reduction was noted in construct contraction, reflecting the actin-disrupting action of staurosporine. Thus, staurosporine increases the gene expression for important matrix proteins and can be used to enhance matrix production and reduce contraction in tissue-engineered fibrocartilage constructs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A significant challenge in fibrocartilage tissue engineering is the production of sufficient extracellular matrix (ECM). Common approaches to stimulating fibrochondrocytes to produce matrix include the use of growth factors (Imler et al. 2004; Lietman et al. 2003; Pangborn and Athanasiou 2005a; Pangborn and Athanasiou 2005b), mechanical stimulation (Deschner et al. 2005a, b; Fink et al. 2001; Imler et al. 2004; Vanderploeg et al. 2004), and gene transfection (Hidaka et al. 2002). Another strategy might be to ameliorate the changes in fibrochondrocyte phenotype that lead to the decline in ECM production.
Part of the difficulty in regulating ECM synthesis by fibrochondrocytes is that in vitro culture of the cells leads to their dedifferentiation; rates of synthesis decrease and the synthesis profile changes (Isoda and Saito 1998; Tanaka et al. 1999). Gene expression studies have demonstrated a drop in collagen II and cartilage oligomeric matrix protein (COMP) expression in fibrochondrocytes over time (Gunja and Athanasiou 2007), whereas cell matrix evaluation has found decreases in aggrecan (Verdonk et al. 2005). Increased production of α-smooth muscle actin (αSMA), leading to deleterious cell contraction and destruction of scaffold porosity in tissue-engineered constructs, is another consequence of fibrochondrocyte dedifferentiation (Mueller et al. 1999a, b). These findings in fibrochondrocytes are further supported by the larger body of evidence with chondrocytes. Declining collagen II, COMP, and aggrecan gene expression has been found in chondrocyte cultures, and again, increased αSMA synthesis is evident (Darling and Athanasiou 2005; Diaz-Romero et al. 2005; Honda et al. 2004; Kaps et al. 2006; Kinner and Spector 2001; Schnabel et al. 2002). Preventing the changes associated with dedifferentiation and helping fibrochondrocytes retain their native phenotype should aid tissue-engineering strategies.
Actin-integrin interactions play a significant role in the manifestation of dedifferentiation, including changes in cell shape, gene expression, and ECM production (Geiger et al. 2001). Indeed, studies of chondrocyte redifferentiation in the presence of actin-modifying agents have shown significant potential to prevent the deleterious effects of dedifferentiation (Borge et al. 1997; Loty et al. 2000). One particular agent, staurosporine, has been particularly effective in redifferentiating chondrocytes. Staurosporine treatment of passaged dedifferentiated chondrocytes has been found to increase collagen II expression and staining for glycosaminoglycans (GAG; Benya 1988; Borge et al. 1997). In staurosporine-treated chondrocyte-seeded matrices, the uptake of collagen and GAG precursors increases two-fold and ten-fold, respectively (Lee et al. 2003). Treatment of chondrocytes with staurosporine has also resulted in reduced synthesis of αSMA (Lee et al. 2003; Zaleskas et al. 2004).
Staurosporine is known to be a protein-kinase C (PKC) inhibitor, specifically preventing ATP from binding the kinase. Recent work in hepatocytes has linked the action of PKC with the f-actin cytoskeleton, specifically through an oxidative stress mechanism. Thus, one of the effects of oxidative stress, actin disorganization, has been found to be mediated by a PKC pathway and can be prevented via the use of staurosporine (Perez et al. 2006). Evidence also suggests that staurosporine reduces stress fiber formation and cell contraction via effects on actin polymerization and mysosin contraction (Lee et al. 2003; Sakurada et al. 1998; Wakatsuki et al. 2003). Staurosporine and other actin-modifying agents are believed to have their beneficial effects on chondrocytes through these changes in the cytoskeleton. Overall, staurosporine has been shown to have several important effects at the protein level that may be harnessed for successful tissue engineering of tissue with fibrochondrocytes.
By preventing changes associated with fibrochondrocyte culture (a dedifferentiation-like process) at the gene level, we hypothesize that staurosporine treatment might result in greater ECM production in tissue-engineered constructs. In this study, we first examine a range of staurosporine treatments on short- and long-term gene expression changes in monolayer fibrochondrocytes. Next, the effective concentration range, identified in the gene expression studies, has been applied to tissue-engineered constructs to examine changes in ECM synthesis. In this study, we take a scaffoldless approach, termed self-assembly (Hu and Athanasiou 2006), to examine staurosporine treatments for fibrocartilage tissue engineering. Self-assembly has been used successfully in the past to create neofibrocartilage constructs and, moreover, avoids the challenges of scaffold-based studies (Hoben et al. 2007).
Materials and methods
Cell culture
Fibrochondrocytes were harvested from the inner 2/3 of the medial meniscus of 1-week-old male calves (Research 87, Boston, Mass.) less than 36 h after slaughter. Cells were isolated by overnight digestion in culture medium with 0.2% type II collagenase (Worthington, Lakewood, N.J.) in an incubator at 37°C, with 10% carbon dioxide. Culture medium was composed of DMEM with 4.5 g/l L-glucose and L-glutamine (Gibco, Grand Island, N.Y.), 10% fetal bovine serum (FBS; Biowhittaker, Walkersville, Md.), 1% fungizone, 1% penicillin/streptomycin, 1% non-essential amino acids, 0.4 mM proline, 10 mM HEPES, and 50 μg/ml L-ascorbic acid. Cells were frozen at –80°C in culture medium supplemented with 20% FBS and 10% dimethylsufoxide (DMSO) until sufficient cells were collected.
Staurosporine preparation
Staurosporine (Axxora, San Diego, Calif.) was dissolved in DMSO to a concentration of 1 mM and then further diluted in phosphate-buffered saline (PBS; Sigma, St. Louis, Mo.) to 2 μM. This solution was then sterile-filtered, and further dilutions to the working concentrations were made in culture medium.
Actin staining
Tissue-culture-treated coverslips (24×30 mm; Nalgene Nunc International, Naperville, Ill.) were seeded with 2×105 cells and treated with 0.1, 1.0, 10, and 100 nM staurosporine in culture medium for 24 h. Cells were then fixed in 3.7% paraformaldehyde and digested with 0.1% Triton-X (Fisher Scientific) for 5 min. The samples were then stained with 1:40 Alexa Fluor-647 phalloidin in PBS (Molecular Probes, Eugene, Ore.) for 90 min, followed by 1 mM Hoechst dye (Molecular Probes) for 7 min. Slides were then viewed on an Axioplan 2 microscope (Carl Zeiss, Oberkochen, Germany). Images were acquired and analyzed by using Metamorph 4.15 (Universal Imaging, Downington, Pa.).
Quantitative mRNA analysis
Fibrochondrocytes from a single animal were seeded in 6-well plates at 2×105 cells/well and allowed to grow until confluent. The experiment was repeated with cells from five separate animals. Cells were then treated with 0.1, 1.0, 10, and 100 nM staurosporine in culture medium with medium changes every day. Cells were collected and lysed from monolayer culture with Trizol reagent (Invitrogen, Carlsbad, Calif.) after 24 h and after 4 days of culture. RNA was then isolated from the lysate by the addition of chloroform, centrifugation to collect the RNA layer, and further purification with isopropanol and DNase-I (Stratagene, La Jolla, Calif.) treatment to remove any contaminating DNA. RNA was then quantified on a fluorospectrometer (NanoDrop, Wilmington, Del.). Samples of 200 ng RNA were reverse-transcribed to cDNA. For the reverse transcription reaction, RNA samples were mixed with 1 mM dNTPs, 1 mM random hexamers, and Stratagene Stratascript RT enzyme (Stratagene) for 90 min at 37°C. After cDNA synthesis, real-time polymerase chain reaction (PCR) for glyceraldehydes-3-phosphate dehydrogenase (GAPDH), collagen I, collagen II, aggrecan, and COMP was performed in a Rotor-gene 3000 real-time PCR machine (Corbett Research, Sydney, Australia). Primers and probe sequences were used as previously described (Darling and Athanasiou 2005; Shieh and Athanasiou 2007). Relative gene abundance for the gene of interest (RAGOI) was calculated by using the number of take-off cycles (Ct) and reaction efficiency (EGOI) and normalized to GAPDH, a housekeeping gene.
Three-dimensional construct formation
Cylindrical 2% agarose (Sigma) molds with a 5-mm diameter were produced in a 48-well plate, as previously described (Hoben et al. 2007). Bovine fibrochondrocytes were harvested as described above, and cells from 10 animals were pooled to obtain sufficient cells. Constructs consisted of 5.5×106 cells allowed to self-assemble in the mold. Control constructs were given culture medium, and experimental groups received 10 nM, 50 nM, or 100 nM staurosporine-supplemented culture medium. Constructs were given daily complete media changes of 500 μl. Construct dimensions, while still in culture, were measured by using Image J software (free access at http://rsb.info.nih.gov/ij). Construct micrographs were imported to Image J, and measurements were scaled in the software to calculate the construct area by using internal standards.
Construct histology and immunohistochemistry
Samples were frozen and sectioned at 12 μm. Safranin-O and fast green were used to examine GAG distribution, and picrosirius red staining was used to analyze collagen distribution. Immunohistochemical analysis was performed by fixation of the sections in chilled acetone, rehydration, treatment with 3% H2O2 in methanol, and blocking with horse serum. The primary antibody, mouse anti-human anti-αSMA monoclonal antibody (Sigma), was diluted 1:200 in PBS and applied for 1 h. Visualization by using a secondary biotinylated antibody, the ABC reagent, and diaminobenzidine (DAB) was performed with the Vectastain kit (Vector Laboratories, Burlingame, Calif.), and counterstaining with Harris’s hematoxylin. Sections of meniscal fibrocartilage and aorta tissue were run as positive controls, whereas samples stained without application of the primary antibody were used as negative controls.
Construct quantitative biochemistry
Samples were lyophilized for 48 h and digested in 125 μg/ml papain (Sigma) for 18 h at 60°C. For each biochemical analysis, four to six constructs were analyzed. Cell number was determined by using the Picogreen Cell Proliferation Assay Kit (Molecular Probes). A hydroxyproline assay (Reddy and Enwemeka 1996) was performed to gauge total collagen by using bovine collagen standards (Biocolor, Newtonabbey, Northern Ireland). Sulfated GAG was measured with the Blyscan GAG Assay Kit (Biocolor). Samples for enzyme-linked immunosorbent assay (ELISA) were digested in papain at 4°C for 4 days and then a 1/10 volume of elastase (Sigma) solution in 10× TRIS-buffered saline was added to achieve a concentration of 0.1 mg/ml elastase. Samples were allowed to digest for an additional 48 h. The amount of collagen I amounts was quantified by using an indirect ELISA, and that for collagen II was determined by using a capture ELISA. For the indirect ELISA, 96-well plates were coated with standards and samples and incubated overnight at 4°C. The wells were blocked with bovine serum albumin for 2 h and then exposed to a primary antibody to collagen I (US Biological, Swampscott, Mass.) for 1 h. A secondary antibody (US Biological) was then exposed to the plate for 1 h, after which the results were visualized at 450 nm by using TMB as a liquid substrate. Collagen II was quantified by using a capture ELISA kit (Chondrex, Redmond, Wash.). Plates were coated in capture antibody solution overnight at 4°C, and then samples and standards were applied. Protein was visualized with TMB reagent following reaction between the biotinylated secondary antibody (Chondrex) and streptavidin-peroxidase. Between each incubation step in the ELISA, plates were washed in PBS with 0.05% Tween-20 (Sigma).
Statistics
All data were compiled as mean±SD. A one-factor analysis of variance (ANOVA) was used when comparing means for quantitative biochemistry. A two-factor ANOVA was used to analyze construct contraction by using time and staurosporine dose as factors. If an analysis showed a significant difference (P < 0.05), a Tukey’s post hoc analysis was performed to compare sample sets. A paired (by animal) Student’s t-test was used to determine significance between a treatment group and the no-dose group for gene expression.
Results
Monolayer staining
Untreated cells appeared well-spread with multiple cytoplasmic extensions, and fluorescence microscopy revealed dense actin staining, especially around the cell periphery (Fig. 1a). In contrast, the cells treated with staurosporine showed faint diffuse actin staining throughout the cell body (Fig. 1b).
Phalloidin actin staining (red) of monolayer cells after 24 h on tissue culture plastic (blue nuclear counterstaining). a Untreated cells show dense stress bundles and localization of staining at the cell periphery. b Following 100 nM staurosporine treatment, a clear decrease can be seen in the intensity of actin staining, with diminished abundance at the cell periphery. Bar 10 μm
Quantitative gene expression
Animal-to-animal variation was significant for all of the genes measured, with the exception of COMP. To account for this, a paired Student’s t-test was employed; abundance was normalized to GAPDH and scaled relative to the untreated controls at the respective time point (Fig. 2). At 24 h of treatment, a significant increase was seen in aggrecan and COMP with high-dose staurosporine, together with a dose-dependent decreasing trend in collagen I expression (P = 0.097). After 4 days, high-dose staurosporine led to increased COMP expression compared with the no-dose control, and although not significant, 100 nM staurosporine showed an increase in collagen II expression.
Quantitative gene expression in fibrochondrocytes at 24 h and 4 days of treatment with staurosporine (y-axis mRNA abundance relative to GAPDH expression normalized to untreated fibrochondrocytes at the same time point). Because of the increases over orders of magnitude, cartilage oligomeric matrix protein (COMP) was normalized to the log abundance (note that the control value is set at 0). *P < 0.05, **P < 0.10; significantly different when compared with the no-dose control
Tissue-engineered constructs
The tissue-engineered constructs coalesced within 24 h in the agarose wells and, after 4 weeks of culture, grew to wet weights ranging from 2.18 mg to 2.46 mg (Table 1). All tissue-engineered constructs had an initial disc diameter of 5 mm, and the untreated constructs showed continuous contraction to 3.72 ± 1.09 mm, whereas the 100-nM-treated constructs only contracted to 4.27 ± 0.60 mm (Fig. 3). Two-factor ANOVA demonstrated both dose and time to be significant factors. Histological examination showed intense staining for collagen and a minimal appearance of GAGs (data not shown). Staining for αSMA revealed an accumulation of actin near the edges of the construct, whereas staining was less intense following staurosporine treatment, especially with the 50 nM dose (Fig. 4).
Immunohistochemical staining for αSMA. Constructs were treated with 0 nM (a), 10 nM (b), 50 nM (c), 100 nM (d) staurosporine. Brown staining indicates positive protein staining. The untreated construct shows intense staining along the borders of the construct, whereas staining is lighter and more diffuse in the treated constructs. The trend in increasing construct diameter suggests that regulation of αSMA by staurosporine could have benefits in the retention of construct dimensions. Bar 100 μm
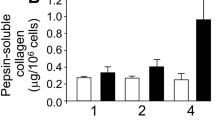
Quantitative biochemistry demonstrated significant changes in the total quantity and proportions of the different collagens (Fig. 5). High-dose staurosporine treatment led to a 100% increase in total collagen per construct from 27.6 ± 8.8 μg to 55.2 ± 12.2 μg per construct. When analyzed for specific collagens, the 10-nM group exhibited a significant increase over the control and higher doses for collagen type I, whereas collagen type II was similar for all groups. No change was found in the number of cells per construct or the GAG quantity with staurosporine treatment (Table 1).
Total and specific collagen content of the constructs. a Total collagen showed a significant increase with the highest dose of staurosporine. Bars not connected by the same letter are significantly different, P < 0.05. b Collagen I content was greatest in the 10-nM-treated constructs. *P < 0.05. c Collagen II content did not vary significantly in the constructs
Discussion
The unique agent employed in this two-phase study, staurosporine, functions both as a PKC inhibitor and, by a separate undefined mechanism, as an actin-disrupting agent (Mobley et al. 1994). The modulation of actin structure with agents ranging from cytochalasin D to latrunculin has shown marked changes in protein production by various cell types including fibroblasts, mesenchymal stem cells, and especially, chondrocytes (Gerecht et al. 2007; Loty et al. 2000; Newman and Watt 1988; Pirttiniemi and Kantomaa 1998; Zhang et al. 2006). The critical role of actin stress fibers in the transformation of fibrochondrocytes to αSMA–containing myofibroblasts (Kambic et al. 2000; Mueller et al. 1999a, b) prompted the evaluation of staurosporine treatment in this study. First, monolayer fibrochondrocytes were treated with staurosporine and examined for changes in actin appearance and gene expression. Next, the effective doses of staurosporine identified in the first phase were applied to tissue-engineered constructs.
In monolayer culture of fibrochondrocytes, staurosporine clearly disrupted the actin bundles and significantly increased aggrecan and COMP gene expression; it also stimulated an increasing trend in collagen II gene expression. These results are in agreement with our hypothesis that staurosporine prevents a dedifferentiation-like process in fibrochondrocytes, because prior work with chondrocytes and fibrochondrocytes has shown decreases in both aggrecan and COMP expression over time as these cells dedifferentiate in two-dimensional culture (Darling and Athanasiou 2005; Gunja and Athanasiou 2007). The large increases in COMP expression in this study are particularly interesting, as COMP has been identified as a sensitive marker of chondrocyte dedifferentiation (Zaucke et al. 2001). Since collagen I gene expression actually increases with dedifferentiation, the trends seen in this work reflect the role of staurosporine as an inhibitor of this process (Darling and Athanasiou 2005; Gunja and Athanasiou 2007). Overall, these promising gene expression results indicate that concentrations from 10 to 100 nM would be most effective in ameliorating the changes in fibrochondrocyte characteristics seen in culture.
The concentration range identified in the monolayer study was then applied to tissue-engineered constructs. Most remarkably, the high-dose treatment resulted in a doubling of the collagen content. This increase in collagen may be related to the increased COMP gene expression, as COMP has been found to be integral in collagen fibril organization (Rosenberg et al. 1998; Thur et al. 2001). Additionally, we have observed a dose-dependent inhibition in construct contraction. These results suggest that the modulation of actin structures in fibrochondrocytes not only changes the gene expression and protein synthesis profiles, but may also, given the large increase in collagen production, produce significant benefits for tissue-engineering strategies.
Prior work with chondrocytes has shown the benefit of staurosporine as manifest by the recurrence of collagen II, the predominant collagen of hyaline cartilage, after passaging (Benya and Padilla 1986; Borge et al. 1997; Lee et al. 2003). Similarly, in this study, we have seen an increasing trend in collagen II gene expression at the 4-day time-point. The reduction in αSMA and a concomitant reduction in construct contraction have also been significant findings in chondrocyte studies by Lee et al. (2003) and Zaleskas et al. (2004); however, in our study, the inhibition in construct contraction is less prominent. A potential reason for this discrepancy is the absence of an exogenous scaffold in this study. The lack of a scaffold to resist cell-mediated contraction may have prevented the anti-contraction properties of staurosporine being as evident in our results. A disadvantage of staurosporine treatment of chondrocytes is its potential toxicity at concentrations approaching 1 μM, with 100 nM concentrations resulting in 35% cell loss in monolayer cultures after 8 h (Mukherjee et al. 2001). In contrast, no cell loss has been seen in the staurosporine-treated constructs versus controls in our study, suggesting that the three-dimensional configuration may have prevented staurosporine-mediated apoptosis. Thus, the increases in collagen II expression and decreased construct contraction correlates well with prior work on chondrocytes, although we have not observed the cell losses seen in previous chondrocyte studies.
This is the first analysis of the effects of staurosporine applied to fibrochondrocytes and, as these cells are distinct from chondrocytes, their increased ECM production in response to this unique stimulus merits further study. Additionally, the mechanical properties of the new ECM must be evaluated in future studies as another step toward the use of this treatment in tissue-engineering strategies. The evidence that staurosporine can modulate construct contraction is promising for fibrocartilage tissue engineering and will be the focus of future work.
References
Benya PD (1988) Modulation and reexpression of the chondrocyte phenotype; mediation by cell shape and microfilament modification. Pathol Immunopathol Res 7:51–54
Benya PD, Padilla SR (1986) Modulation of the rabbit chondrocyte phenotype by retinoic acid terminates type II collagen synthesis without inducing type I collagen: the modulated phenotype differs from that produced by subculture. Dev Biol 118:296–305
Borge L, Lemare F, Demignot S, Adolphe M (1997) Restoration of the differentiated functions of serially passaged chondrocytes using staurosporine. In Vitro Cell Dev Biol Anim 33:703–709
Darling EM, Athanasiou KA (2005) Rapid phenotypic changes in passaged articular chondrocyte subpopulations. J Orthop Res 23:425–432
Deschner J, Rath-Deschner B, Agarwal S (2005a) Regulation of matrix metalloproteinase expression by dynamic tensile strain in rat fibrochondrocytes. Osteoarthritis Cartilage 14:264–272
Deschner J, Wypasek E, Ferretti M, Rath B, Anghelina M, Agarwal S (2005b) Regulation of RANKL by biomechanical loading in fibrochondrocytes of meniscus. J Biomech 39:1796–1803
Diaz-Romero J, Gaillard JP, Grogan SP, Nesic D, Trub T, Mainil-Varlet P (2005) Immunophenotypic analysis of human articular chondrocytes: changes in surface markers associated with cell expansion in monolayer culture. J Cell Physiol 202:731–742
Fink C, Fermor B, Weinberg JB, Pisetsky DS, Misukonis MA, Guilak F (2001) The effect of dynamic mechanical compression on nitric oxide production in the meniscus. Osteoarthritis Cartilage 9:481–487
Geiger B, Bershadsky A, Pankov R, Yamada KM (2001) Transmembrane crosstalk between the extracellular matrix—cytoskeleton crosstalk. Nat Rev Mol Cell Biol 2:793–805
Gerecht S, Bettinger CJ, Zhang Z, Borenstein JT, Vunjak-Novakovic G, Langer R (2007) The effect of actin disrupting agents on contact guidance of human embryonic stem cells. Biomaterials 28:4068–4077
Gunja NJ, Athanasiou K (2007) Passage and reversal effects on gene expression of bovine meniscal fibrochondrocytes. Arthritis Res Ther 9:R93
Hidaka C, Ibarra C, Hannafin JA, Torzilli PA, Quitoriano M, Jen SS, Warren RF, Crystal RG (2002) Formation of vascularized meniscal tissue by combining gene therapy with tissue engineering. Tissue Eng 8:93–105
Hoben G, Hu J, James R, Athanasiou KA (2007) Self-assembly of fibrochondrocytes and chondrocytes for tissue engineering of the knee meniscus. Tissue Eng 13:939–946
Honda MJ, Yada T, Ueda M, Kimata K (2004) Cartilage formation by serial passaged cultured chondrocytes in a new scaffold: hybrid 75:25 poly(L-lactide-epsilon-caprolactone) sponge. J Oral Maxillofac Surg 62:1510–1516
Hu JC, Athanasiou KA (2006) A self-assembling process in articular cartilage tissue engineering. Tissue Eng 12:969–979
Imler SM, Doshi AN, Levenston ME (2004) Combined effects of growth factors and static mechanical compression on meniscus explant biosynthesis. Osteoarthritis Cartilage 12:736–744
Isoda K, Saito S (1998) In vitro and in vivo fibrochondrocyte growth behavior in fibrin gel: an immunohistochemical study in the rabbit. Am J Knee Surg 11:209–216
Kambic HE, Futani H, McDevitt CA (2000) Cell, matrix changes and alpha-smooth muscle actin expression in repair of the canine meniscus. Wound Repair Regen 8:554–561
Kaps C, Frauenschuh S, Endres M, Ringe J, Haisch A, Lauber J, Buer J, Krenn V, Haupl T, Burmester GR, Sittinger M (2006) Gene expression profiling of human articular cartilage grafts generated by tissue engineering. Biomaterials 27:3617–3630
Kinner B, Spector M (2001) Smooth muscle actin expression by human articular chondrocytes and their contraction of a collagen-glycosaminoglycan matrix in vitro. J Orthop Res 19:233–241
Lee CR, Grodzinsky AJ, Spector M (2003) Modulation of the contractile and biosynthetic activity of chondrocytes seeded in collagen-glycosaminoglycan matrices. Tissue Eng 9:27–36
Lietman SA, Hobbs W, Inoue N, Reddi AH (2003) Effects of selected growth factors on porcine meniscus in chemically defined medium. Orthopedics 26:799–803
Loty S, Foll C, Forest N, Sautier JM (2000) Association of enhanced expression of gap junctions with in vitro chondrogenic differentiation of rat nasal septal cartilage-released cells following their dedifferentiation and redifferentiation. Arch Oral Biol 45:843–856
Mobley PL, Hedberg K, Bonin L, Chen B, Griffith OH (1994) Decreased phosphorylation of four 20-kDa proteins precedes staurosporine-induced disruption of the actin/myosin cytoskeleton in rat astrocytes. Exp Cell Res 214:55–66
Mueller SM, Schneider TO, Shortkroff S, Breinan HA, Spector M (1999a) Alpha-smooth muscle actin and contractile behavior of bovine meniscus cells seeded in type I and type II collagen-GAG matrices. J Biomed Mater Res 45:157–166
Mueller SM, Shortkroff S, Schneider TO, Breinan HA, Yannas IV, Spector M (1999b) Meniscus cells seeded in type I and type II collagen-GAG matrices in vitro. Biomaterials 20:701–709
Mukherjee P, Rachita C, Aisen PS, Pasinetti GM (2001) Non-steroidal anti-inflammatory drugs protect against chondrocyte apoptotic death. Clin Exp Rheumatol 19:S7–11
Newman P, Watt FM (1988) Influence of cytochalasin D-induced changes in cell shape on proteoglycan synthesis by cultured articular chondrocytes. Exp Cell Res 178:199–210
Pangborn CA, Athanasiou KA (2005a) Effects of growth factors on meniscal fibrochondrocytes. Tissue Eng 11:1141–1148
Pangborn CA, Athanasiou KA (2005b) Growth factors and fibrochondrocytes in scaffolds. J Orthop Res 23:1184–1190
Perez LM, Milkiewicz P, Ahmed-Choudhury J, Elias E, Ochoa JE, Sanchez Pozzi EJ, Coleman R, Roma MG (2006) Oxidative stress induces actin-cytoskeletal and tight-junctional alterations in hepatocytes by a Ca2+-dependent, PKC-mediated mechanism: protective effect of PKA. Free Radic Biol Med 40:2005–2017
Pirttiniemi P, Kantomaa T (1998) Effect of cytochalasin D on articular cartilage cell phenotype and shape in long-term organ culture. Eur J Orthod 20:491–499
Reddy GK, Enwemeka CS (1996) A simplified method for the analysis of hydroxyproline in biological tissues. Clin Biochem 29:225–229
Rosenberg K, Olsson H, Morgelin M, Heinegard D (1998) Cartilage oligomeric matrix protein shows high affinity zinc-dependent interaction with triple helical collagen. J Biol Chem 273:20397–20403
Sakurada K, Seto M, Sasaki Y (1998) Dynamics of myosin light chain phosphorylation at Ser19 and Thr18/Ser19 in smooth muscle cells in culture. Am J Physiol 274:C1563–C1572
Schnabel M, Marlovits S, Eckhoff G, Fichtel I, Gotzen L, Vecsei V, Schlegel J (2002) Dedifferentiation-associated changes in morphology and gene expression in primary human articular chondrocytes in cell culture. Osteoarthritis Cartilage 10:62–70
Shieh AC, Athanasiou KA (2007) Dynamic compression of single cells. Osteoarthritis Cartilage 15:328–334
Tanaka T, Fujii K, Kumagae Y (1999) Comparison of biochemical characteristics of cultured fibrochondrocytes isolated from the inner and outer regions of human meniscus. Knee Surg Sports Traumatol Arthrosc 7:75–80
Thur J, Rosenberg K, Nitsche DP, Pihlajamaa T, Ala-Kokko L, Heinegard D, Paulsson M, Maurer P (2001) Mutations in cartilage oligomeric matrix protein causing pseudoachondroplasia and multiple epiphyseal dysplasia affect binding of calcium and collagen I, II, and IX. J Biol Chem 276:6083–6092
Vanderploeg EJ, Imler SM, Brodkin KR, Garcia AJ, Levenston ME (2004) Oscillatory tension differentially modulates matrix metabolism and cytoskeletal organization in chondrocytes and fibrochondrocytes. J Biomech 37:1941–1952
Verdonk PC, Forsyth RG, Wang J, Almqvist KF, Verdonk R, Veys EM, Verbruggen G (2005) Characterisation of human knee meniscus cell phenotype. Osteoarthritis Cartilage 13:548–560
Wakatsuki T, Wysolmerski RB, Elson EL (2003) Mechanics of cell spreading: role of myosin II. J Cell Sci 116:1617–1625
Zaleskas JM, Kinner B, Freyman TM, Yannas IV, Gibson LJ, Spector M (2004) Contractile forces generated by articular chondrocytes in collagen-glycosaminoglycan matrices. Biomaterials 25:1299–1308
Zaucke F, Dinser R, Maurer P, Paulsson M (2001) Cartilage oligomeric matrix protein (COMP) and collagen IX are sensitive markers for the differentiation state of articular primary chondrocytes. Biochem J 358:17–24
Zhang Z, Messana J, Hwang NS, Elisseeff JH (2006) Reorganization of actin filaments enhances chondrogenic differentiation of cells derived from murine embryonic stem cells. Biochem Biophys Res Commun 348:421–427
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors gratefully acknowledge NIAMS R01 AR 47839–2 for funding this work, and the Hertz Foundation for their support of G. Hoben.
Rights and permissions
About this article
Cite this article
Hoben, G.M., Athanasiou, K.A. Use of staurosporine, an actin-modifying agent, to enhance fibrochondrocyte matrix gene expression and synthesis. Cell Tissue Res 334, 469–476 (2008). https://doi.org/10.1007/s00441-008-0705-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-008-0705-6