Abstract
Trichomonas vaginalis is an extracellular parasite that binds to the epithelium of the human urogenital tract and causes the sexually transmitted infection, trichomoniasis. In view of increased resistance to drugs belonging to the 5-nitroimidazole class, new treatment alternatives are urgently needed. In this study, eight semisynthetized triterpene derivatives were evaluated for in vitro anti-T. vaginalis activity. Ursolic acid and its derivative, 3-oxime-urs-12-en-28-oic-ursolic acid (9), presented the best anti-T. vaginalis activity when compared to other derivatives, with minimum inhibitory concentration (MIC) at 25 μM. Moreover, 9 was active against several T. vaginalis fresh clinical isolates. Hemolysis assay demonstrated that 9 presented a low hemolytic effect. Importantly, 25 μM 9 was not cytotoxic against the Vero cell lineage. Finally, we demonstrated that compound 9 acts synergistically with metronidazole against a T. vaginalis metronidazole-resistant isolate. This report reveals the high potential of the triterpenoid derivative 9 as trichomonicidal agent.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Trichomonas vaginalis is a flagellate protozoan that causes trichomoniasis, the most common non-viral sexually transmitted infection (STI). The parasite is responsible for 276 million new cases annually worldwide (WHO 2012). In symptomatic women, the most common symptoms are vaginal itching, odor, irritation and pruritus, and abdominal pain. Among men, more than 75% are asymptomatic and may not seek treatment, generating chronic inflammation (Poole and McClelland 2013). In symptomatic cases, T. vaginalis has been identified as the etiological agent of nongonococcal and nonchlamydial urethritis (Twu et al. 2014). However, currently, about 80% of cases among women and men are asymptomatic, a fact of concern since the infection turns into a silent disease. Trichomoniasis is associated with important health consequences, including predisposition to cervical and prostate cancer (Viikki et al. 2000; Sutcliffe et al. 2012), adverse pregnancy outcomes (Silver et al. 2014), and increase risk of transmission and acquisition of human immunodeficiency virus (HIV) (Kissinger and Adamski 2013).
The therapy is restricted to the class of 5-nitroimazoles, and metronidazole and tinidazole are the only drugs approved for the treatment of trichomoniasis by the Food and Drug Administration (FDA, USA). In this scenario of few options for treatment, the number of metronidazole-resistant T. vaginalis isolates has increased, reaching 2.5 to 9.6% of clinical isolates (Schwebke and Barrientes 2006). In addition, tinidazole, the alternative to metronidazole, belongs to the same class and cross resistance may be observed. Besides resistance cases, side effects such as nausea, diarrhea, and abdominal discomfort have been observed (Ali and Nozaki 2007), stimulating the search of new anti-T. vaginalis compounds.
Plants are commonly used as sources for new drug discovery. Natural products are a rich source of active compounds representing a promising alternative for the treatment of trichomoniasis (Vieira et al. 2015). Natural compounds like betulinic and ursolic acids exhibit a variety of biological activities, including anticancer, inhibition of HIV, antibacterial, anti-inflammatory, and antimicrobial potential (Bache et al. 2011). The search for triterpene derivatives is a promising approach for development of new trichomonicidal agents. In this context, the aim of this study was to evaluate the anti-T. vaginalis activity of betulinic and ursolic acid derivatives.
Materials and methods
Betulinic and ursolic acid derivative synthesis
The compounds 1–4 and 6–9 tested in this study (Table 1) were synthesized as previously described by Dalla Vechia et al. (2009). Compound 5 synthesis data are described in Supporting Information.
Culture of T. vaginalis
Trichomonas vaginalis ATCC 30236 (JH 31A#4) and fresh clinical isolates TV-LACM2R, TV-LACM5, TV-LACM6, TV-LACM11, TV-LACM15, TV-LACM22, and TV-LACM24 (from female patients) and TV-LACH4 and TV-LACH6 (from male patients) were used in this study. The fresh clinical isolates were obtained from Laboratório de Análises Clínicas e Toxicológicas, Faculdade de Farmácia UFRGS, Brazil (project approval by UFRGS Ethical Committee, number 18923). The organisms were cultured in vitro in trypticase-yeast extract-maltose (TYM) medium and pH 6.0, supplemented with 10% (v/v) heat-inactivated serum, and incubated at 37 °C (Diamond 1957). Organisms in the logarithmic phase of growth and exhibiting more than 95% of viability and normal morphology were harvested, centrifuged, and re-suspended on new TYM medium for the assays. All experiments were performed in triplicate and, at least, with three independent cultures (n = 3).
Anti-T. vaginalis activity screening assay
The activity of eight compounds against T. vaginalis was tested in vitro in the concentration of 100 μM. The stock solution of all compounds was prepared in DMSO. For the assay, 96-microtiter plates were used. Parasites in the final cellular density of 1.0 × 105 trophozoites/ml were added and incubated with compounds at 37 °C, 5% CO2 atmosphere for 24 h. Three controls were carried out: negative control with parasites only, vehicle control (DMSO 0.62%), and positive control (100 μM metronidazole). The number of viable trophozoites was accessed by counting parasites with a hemocytometer using exclusion dye trypan blue (0.2%). The results were expressed as the percentage of living organisms compared to untreated parasites, considering motility and normal morphology.
Determination of minimum inhibitory concentration
The minimum inhibitory concentration (MIC) shows the lowest concentration of compound that is able to kill the trophozoites. MIC was determined for 9 because this compound was the only one among betulinic and ursolic acid derivatives that reduced parasite viability 100% in the screening assay. The MIC values of metronidazole were determinate for the fresh clinical T. vaginalis isolates in a previous study (Becker et al. 2015). After compound eightfold dilution, the MIC value was determined as described above. MIC was analyzed for 5 days and confirmed by the failure of non-motile parasites to grow after re-inoculation into drug-free medium.
Hemolytic assay
This assay was performed as described by Rocha et al. (2012) with some modifications. Fresh human blood was obtained from healthy volunteers. The Universidade Federal do Rio Grande do Sul Research Ethical Committee approved documents, procedures, and project under authorization CAAE 47423415.5.0000.5347. The informed consent for each participant was approved by the Institutional Review Board. The erythrocytes were washed three times with PBS 1× (pH 7.0) and re-suspended to obtain a 1.0% (v/v) erythrocytic suspension. The concentration of 9 was chosen based on the MIC. Then, erythrocytes (1.0%) were incubated with the samples at 37 °C for 1 and 24 h. Supernatant absorbance was measured at 540 nm. Results were expressed as hemolysis percentage of each test sample, comparing to 100% hemolysis that was attributed to hemolytic action of the positive control 0.1% triton X-100. This experiment was carried out in triplicate, and three independent experiments (n = 3) were performed.
Cytotoxicity against HMVII, HeLa, and Vero cells
The cancer cell lines, HMVII and HeLa, and Vero, normal cell line, were used to evaluate the cytotoxicity of 9. HMVII and Vero lines were grown in RPMI-1640 medium, while HeLa cells were maintained in DMEM; both media were supplemented with 10% fetal bovine serum (FBS) and incubated at 37 °C, 5% CO2. For the assay, 3.0 × 104 or 1.0 × 104 cells/well were seeded in 96-well microtiter plates overnight. The medium was replaced by fresh medium containing or not (control condition) 9 at 25 μM. Triton X-100 at 0.2%, final concentration, was added as the positive control and 0.6% DMSO as the vehicle control. The plates were incubated for 24 and 48 h; then, after washing with PBS, the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution (0.5 mg/ml) was added and incubated for 1 h at 37 °C. The plates were washed twice with PBS, and the insoluble purple formazan was dissolved in DMSO. The amount of reduced MTT was measured at 570 nm. The experiment was performed in triplicate with three independent culture (n = 3) for 24 and 48 h.
Synergism evaluation
The TV-LACM2R is a fresh clinical metronidazole-resistant isolate used in this assay because it presents a metronidazole MIC of 73 μM. Low-level resistance is defined as an anaerobic MIC 30–60 μM, moderate-level resistance as 60–120 μM, and high-level resistance as 235 μM or greater as previously described by Butler et al. (2010). In addition, the TV-LACM2R 9 MIC was performed. Organisms were treated with MIC and MIC/2 (12.5 and 6.25 μM, respectively), in association or not with 15 and 73 μM metronidazole and incubated for 24 h at 37 °C and 5.0% CO2. The parasite viability was determined as described in the anti-T. vaginalis activity assay. This test was used to evaluate whether the compound and metronidazole presented synergic effects against T. vaginalis.
Statistical analysis
Student’s t test was chosen for comparisons between two groups. The results are expressed as the mean ± SEM of at least three individual experiments. P < 0.05 was considered a statistically significant difference. Analyses were performed using Statistical Package for the Social Sciences (SPSS) software v.14.
Results
Anti-T. vaginalis screening of synthetic derivatives
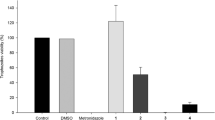
In order to test the anti-T. vaginalis activity of betulinic and ursolic acids and their derivatives (Table 1), a screening was conducted at 100 μM. Betulinic acid and its derivatives, 2–5, induced a slight reduction of the parasite viability. Derivatives 7 and 8 also demonstrated low activity against T. vaginalis, reducing about 15% of the parasite viability. The best anti-T. vaginalis activity was demonstrated by ursolic acid and 9, which reduced parasite viability by 100% (Fig. 1). Taking into account the complete abolishment of T. vaginalis viability, the MIC value of 9 was determined as 25 μM (Fig. 2). This concentration was used in the following experiments.
Anti-T. vaginalis screening of semi-synthesized derivatives of betulinic and ursolic acids against ATCC 30236 isolate at 100 μM. Data were expressed as mean ± SD compared to control (considering trophozoite viability 100%) of three different experiments (parasite suspensions) performed in triplicate
Activity of 9 against fresh clinical T. vaginalis isolates
Compound 9 was tested against T. vaginalis fresh clinical isolates TV-LACM2R, TV-LACM5, TV-LACM6, TV-LACM11, TV-LACM15, TV-LACM22, TV-LACM24, TV-LACH4, and TV-LACH6 (Table 2). All isolates were highly sensitive to 9 at MIC, including TV-LACM2R, a fresh clinical isolate resistant to metronidazole, with abolishment of viable parasites. Considering that TV-LACM2R is a resistant isolate, a serial dilution was performed in order to determine the MIC value specifically for this isolate. The results showed the MIC value in the concentration of 12.5 μM that was subsequently used in the synergism test.
Cytotoxicity
Figure 3 shows the cytotoxicity of 9 against HMVII and HeLa cancer cell lines and the Vero normal cell line with Triton X-100 as positive control. After 24 or 48 h of incubation, 9 at 25 μM reduced cancer cell lines viability about 90%. Conversely and importantly, the compound 9 presented very low cytotoxicity against Vero cells, normal cell line, reducing about only 15% of cell viability.
Effect of 9 on the viability of HMVII, HeLa, and Vero cells. Cells were exposed to 25 μM 9 for 24 and 48 h; Triton X-100 was used as positive control; and control means only cells in medium without exposure to 9. Data represent mean ± SD compared to control (only cells) of three different experiments performed in triplicate
Hemolysis
In order to investigate whether the mechanism of killing by 9 involves membrane damage, a hemolytic assay was performed (Gauthier et al. 2009). After 1 h of incubation, the compound 9, at 25 μM, did not induce lysis of the erythrocytes. However, 9 induced some erythrocyte lysis (25%) after 24 h of incubation (data not shown).
Synergism
The effect of 9 on the viability of the T. vaginalis TV-LACM2R isolate, resistant to metronidazole, was performed. Compound 9 completely abolished the TV-LACM2R viability and the MIC was 12.5 μM (Fig. 4a). In order to investigate the synergic effect of 9 and metronidazole, the trophozoites were treated with a low concentration of 15 μM metronidazole and 6.25 μM compound 9 (MIC/2). When 15 μM metronidazole was incubated alone, the viability of the parasites was reduced about 10%. However, when 15 μM metronidazole and 6.25 μM 9 were associated, a strong anti-T. vaginalis activity was observed, demonstrating the synergic effect of 9 and metronidazole (Fig. 4b).
a MIC determination of 9 against TV-LACM2R metronidazole-resistant isolate. b Synergic effect of 9 and metronidazole in MIC and MIC/2 of the compound. The asterisk (*) shows that metronidazole and 9 association is statistically different from 15 μM MTZ treatment. Data represent mean ± SD compared to control (only trophozoites) of three different experiments (parasite suspensions) performed in triplicate
Discussion
Ursolic acid is widely found in certain medicinal herbs and it is the main component of the peel of fruits such as apples, pears, olives, prunes, cranberries, and figs. This compound exhibits many biological effects including anticancer activity and antimicrobial potential (Chen et al. 2015). Ursolic acid is a well-known triterpenoid that has been reported to possess a wide range of biological activities, including antimicrobial, antitumor, antiviral, antiprotozoal, antioxidant, and anti-inflammatory activities (Innocente et al. 2014; Silva et al. 2013; Gu et al. 2015). Among these activities, it showed significant anti-protozoa effects against Plasmodium falciparum, Toxoplasma gondii, Trypanosoma cruzi, and Leishmania sp. (Jesus et al. 2015). Studies indicate that the activity of ursolic acid and its derivatives was also markedly influenced by their structural properties (Gauthier et al. 2009; Chen et al. 2015; Mazumder et al. 2013).
In our study, we tested a semisynthetic ursolic acid derivative, 9, against the pathogen T. vaginalis and checked the cytotoxicity against three cell lines, HMVII and HeLa, two cancer lines, and against a normal cell, Vero line. Compound 9 was cytotoxic against both cancer lines, and these results agreed with previous studies that demonstrate the anticancer activity of ursolic acid and its derivatives (Hua et al. 2015). On the other hand, against Vero, normal cell, 9 presented a slight cytotoxicity, and once again, the result agreed with an earlier study (Mahlo et al. 2013). Derivative 9 deserves a considerable attention, because this compound presented a potent activity against metronidazole-sensitive and metronidazole-resistant T. vaginalis isolates and low cytotoxicity against mammalian normal cells, demonstrating the potential of 9 for trichomoniasis treatment. In addition, this compound presented a low hemolytic effect. This result indicates that 9 probably does not produce damage in the parasite membrane; however, more studies are needed to elucidate the mechanism of action of this active ursolic acid derivative.
Trichomoniasis is the most prevalent non-viral STI and the treatment is based on metronidazole and tinidazole. Although the cure rate is high, failures are observed to be mainly associated with drug resistance (Schwebke and Barrientes 2006). Natural and synthetic compounds play an important role in this field. Many studies show that triterpenoid derivatives have good activity against microorganisms (Silva et al. 2015; Innocente et al. 2014; Taketa et al. 2004); among these compounds, betulinic and ursolic acids display great relevance. In this study, we showed anti-T. vaginalis activity of different synthetic derivatives from betulinic and ursolic acids. The most relevant anti-T. vaginalis activity was demonstrated by compound 9 with MIC of 25 μM. Although data from the literature demonstrate that derivatives from different structures present different biological activities (Gauthier et al. 2009; Liu 2005; Wang and Fang 2009), in this study, we demonstrated the strong anti-T. vaginalis activity produced by compound 9. Moreover, as shown in Fig. 4b, when 15 μM metronidazole and 6.25 μM 9 were tested, a significant reduction of parasite viability was observed, demonstrating that 9 is able to potentiate metronidazole action against a resistant isolate.
Conclusion
Trichomonas vaginalis causes an infection that may turn into a serious public health problem when treatment failure occurs. The increasing resistance to traditional treatments with metronidazole leads us to search for other therapies and a large field of study is natural products, including the synthetic derivatives of natural products. The semisynthetic triterpene derivative 9 demonstrated potent anti-T. vaginalis activity against ATCC and fresh clinical isolates. Importantly, 9 was active against metronidazole-resistant isolate and, when tested in association with metronidazole, 9 potentiated anti-T. vaginalis activity against the resistant isolate. Besides, this compound was not cytotoxic to normal mammalian cell line. Further studies are necessary to provide details regarding the mechanism of death of 9 anti-trichomonad activity. Altogether, the results presented in this study demonstrated that 9 is a promising alternative to treat trichomoniasis.
References
Ali V, Nozaki T (2007) Current therapeutics, their problems, and sulfur-containing-amino-acid metabolism as a novel target against infections by “amitochondriate” protozoan parasites. Clin Microbiol Rev 20:164–187. https://doi.org/10.1128/CMR.00019-06
Bache M, Zschornak MP, Passin S, Kessler J, Wichmann H, Kappler M, Paschke R, Kaluderovic GN, Kommera H, Taubert H, Vordermark D (2011) Increased betulinic acid induced cytotoxicity and radiosensitivity in glioma cells under hypoxic conditions. Radiat Oncol 6:111. https://doi.org/10.1186/1748-717X-6-111
Becker DL, Santos O, Frasson AP, Rigo GV, Macedo AJ, Tasca T (2015) High rates of double-stranded RNA viruses and Mycoplasma hominis in Trichomonas vaginalis clinical isolates in South Brazil. Infect Genet Evol 34:181–187. https://doi.org/10.1016/j.meegid.2015.07.005
Butler SE, Augostini P, Secor WE (2010) Mycoplasma hominis infection of Trichomonas vaginalis is not associated with metronidazole-resistant trichomoniasis in clinical isolates from the United States. Parasitol Res 107:1023–1027. https://doi.org/10.1007/s00436-010-1975-y
Chen H, Gao Y, Wang A, Zhou X, Zheng Y, Zhou J (2015) Evolution in medicinal chemistry of ursolic acid derivatives as anticancer agents. Eur J Med Chem 92:648–655. https://doi.org/10.1016/j.ejmech.2015.01.031
Dalla Vechia L, Gnoatto SCB, Gosmann G (2009) Derivados oleananos e ursanos e sua importância na descoberta de novos fármacos com atividade antitumoral, anti-inflamatória e antioxidante. Quim Nova 32:1245–1252
Diamond LS (1957) The establishment of various trichomonads of animals and man in axenic cultures. J Parasitol 43:488–490
Gauthier C, Legault J, Girard-Lalancette K, Mshvildadze V, Pichette A (2009) Haemolytic activity, cytotoxicity and membrane cell permeabilization of semi-synthetic and natural lupane- and oleanane-type saponins. Bioorg Med Chem 17:2002–2008. https://doi.org/10.1016/j.bmc.2009.01.022
Gu W, Hao Y, Zhang G, Wang SF, Miao TT, Zhang KP (2015) Synthesis, in vitro antimicrobial and cytotoxic activities of new carbazole derivatives of ursolic acid. Bioorg Med Chem Lett 25:554–557. https://doi.org/10.1016/j.bmcl.2014.12.021
Hua SX, Huang RZ, Ye MY, Pan YM, Yao GY, Zhang Y, Wang HS (2015) Design, synthesis and in vitro evaluation of novel ursolic acid derivatives as potential anticancer agents. Eur J Med Chem 95:435–452. https://doi.org/10.1016/j.ejmech.2015.03.051
Innocente AM, Vieira PB, Frasson AP, Casanova BB, Gosmann G, Gnoatto SC, Tasca T (2014) Anti-Trichomonas vaginalis activity from triterpenoid derivatives. Parasitol Res 113:2933–2940. https://doi.org/10.1007/s00436-014-3955-0
Jesus JA, Lago JH, Laurenti MD, Yamamoto ES, Passero LF (2015) Antimicrobial activity of oleanolic and ursolic acids: an update. Evid Based Complement Alternat Med 2015:620472. https://doi.org/10.1155/2015/620472
Kissinger P, Adamski A (2013) Trichomoniasis and HIV interactions: a review. Sex Transm Infect 89:426–433. https://doi.org/10.1136/sextrans-2012-051005
Liu J (2005) Oleanolic acid and ursolic acid: research perspectives. J Ethnopharmacol 100:92–94. https://doi.org/10.1016/j.jep.2005.05.024
Mahlo SM, McGaw LJ, Eloff JN (2013) Antifungal activity and cytotoxicity of isolated compounds from leaves of Breonadia salicina. J Ethnopharmacol 148:909–913. https://doi.org/10.1016/j.jep.2013.05.041
Mazumder K, Tanaka K, Fukase K (2013) Cytotoxic activity of ursolic acid derivatives obtained by isolation and oxidative derivatization. Molecules 18:8929–8944. https://doi.org/10.3390/molecules18088929
Poole DN, McClelland RS (2013) Global epidemiology of Trichomonas vaginalis. Sex Transm Infect 89:418–422. https://doi.org/10.1136/sextrans-2013-051075
Rocha TD, Vieira PB, Gnoatto SC, Tasca T, Gosmann G (2012) Anti-Trichomonas vaginalis activity of saponins from Quillaja, Passiflora, and Ilex species. Parasitol Res 110:2551–2556. https://doi.org/10.1007/s00436-011-2798-1
Schwebke JR, Barrientes FJ (2006) Prevalence of Trichomonas vaginalis isolates with resistance to metronidazole and tinidazole. Antimicrob Agents Chemother 50:4209–4210. https://doi.org/10.1128/aac.00814-06
Silva GN, Maria NR, Schuck DC, Cruz LN, de Moraes MS, Nakabashi M, Graebin C, Gosmann G, Garcia CR, Gnoatto SC (2013) Two series of new semisynthetic triterpene derivatives: differences in anti-malarial activity, cytotoxicity and mechanism of action. Malar J 12:89. https://doi.org/10.1186/1475-2875-12-89
Silva GN, Schuck DC, Cruz LN, Moraes MS, Nakabashi M, Gosmann G, Garcia CR, Gnoatto SC (2015) Investigation of antimalarial activity, cytotoxicity and action mechanism of piperazine derivatives of betulinic acid. Tropical Med Int Health 20:29–39. https://doi.org/10.1111/tmi.12395
Silver BJ, Guy RJ, Kaldor JM, Jamil MS, Rumbold AR (2014) Trichomonas vaginalis as a cause of perinatal morbidity: a systematic review and meta-analysis. Sex Transm Dis 41:369–376. https://doi.org/10.1097/olq.0000000000000134
Sutcliffe S, Neace C, Magnuson NS, Reeves R, Alderete JF (2012) Trichomonosis, a common curable STI, and prostate carcinogenesis—a proposed molecular mechanism. PLoS Pathog 8:e1002801. https://doi.org/10.1371/journal.ppat.1002801
Taketa ATC, Gnoatto SCB, Gosmann G, Pires VS, Schenkel EP, Guillaume D (2004) Triterpenoids from Brazilian Ilex species and their in vitro antitrypanosomal activity. J Nat Prod 67:1697–1700. https://doi.org/10.1021/np040059+
Twu O, Dessi D, Vu A, Mercer F, Stevens GC, de Miguel N, Rappelli P, Cocco AR, Clubb RT, Fiori PL, Johnson PJ (2014) Trichomonas vaginalis homolog of macrophage migration inhibitory factor induces prostate cell growth, invasiveness, and inflammatory responses. PNAS 111:8179–8184. https://doi.org/10.1073/pnas.1321884111
Vieira PB, Giordani RB, Macedo AJ, Tasca T (2015) Natural and synthetic compound anti-Trichomonas vaginalis: an update review. Parasitol Res 114:1249–1261. https://doi.org/10.1007/s00436-015-4340-3
Viikki M, Pukkala E, Nieminen P, Hakama M (2000) Gynaecological infections as risk determinants of subsequent cervical neoplasia. Acta Oncol 39:71–75
Wang SR, Fang WS (2009) Pentacyclic triterpenoids and their saponins with apoptosis-inducing activity. Curr Top Med Chem 9:1581–1596
World Health Organization (2012) Global incidence and prevalence of selected curable sexually transmitted infections—2008. World Health Organization, Geneva. https://doi.org/10.1016/s0968-8080(12)40660-7
Acknowledgements
The authors thank LRNANO/CNANO for the NMR analysis.
Funding
This work received financial support from REDE NANOBIOTEC-CAPES/Brazil (grant 23038.019022/2009-68), CNPq/Brazil (grant 474930/2012-2), and INCT-IF. Patrícia B. Vieira receives scholarship from CAPES; Tiana Tasca received researcher fellowship (grant 307447/2014-6) from CNPq.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The Universidade Federal do Rio Grande do Sul Research Ethical Committee approved documents, procedures, and project under authorization CAAE 47423415.5.0000.5347 and 18923. The informed consent for each participant was approved by the Institutional Review Board.
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(DOC 26 kb)
Rights and permissions
About this article
Cite this article
Bitencourt, F.G., de Brum Vieira, P., Meirelles, L.C. et al. Anti-Trichomonas vaginalis activity of ursolic acid derivative: a promising alternative. Parasitol Res 117, 1573–1580 (2018). https://doi.org/10.1007/s00436-018-5839-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-018-5839-1