Abstract
Duodenal biopsy is an important tool to diagnose coeliac disease (CD); however, the most reliable location of biopsy site is still questionable. Claudins (CLDNs), members of a large family of adherent junction proteins, show characteristic expression pattern in inflammatory disorders; nevertheless, CLDN expression in CD is unknown. This is a comparative study to examine the CLDN 2, 3 and 4 expressions in proximal and distal part of duodenum in children with CD and in controls. Thirty-three children with newly diagnosed CD were enrolled. Fourteen healthy children served as controls. Biopsies from proximal and distal part of duodenum were taken for routine histological analysis. Immunohistochemistry were used to detect CD3+ intraepithelial lymphocytes and CLDN 2, 3 and 4 protein expressions. Macroscopic picture, routine histology and Marsh grade depicted no differences between biopsies taken from proximal or distal part of duodenum. However, CLDN 2 expression was significantly increased in severe form of coeliac disease in bulb and in distal duodenum, and in distal part of non-severe coeliac patients, in comparison to controls. Similar association was found concerning CLDN 3 expression. Expression of CLDN 4 was similar in all groups studied. Both proximal and distal mucosal duodenal biopsies are suitable for diagnosing villous atrophy in patients with CD. Increased expressions of CLDN 2 and 3 suggest structural changes of tight junction in coeliac disease which may be, at least in part, responsible for increased permeability and proliferation observed in coeliac disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coeliac disease (CD) or gluten-sensitive enteropathy is currently considered as a unique immune-mediated disorder leading to malabsorption. It is caused by the gluten fraction of cereals in susceptible people [1, 2]. The frequency of CD is constantly growing [3–5]. Through the screening examinations, the recognition of CD is higher, and the hit to find the disease in an early stage is better. High prevalence of CD was verified by Hungarian data as well (1:85) [6]. In the past, jejunal biopsies for diagnosis of CD were taken by means of suction capsule, but nowadays, biopsies are taken by upper endoscopy from the distal part of duodenum. Endoscopy is superior to suction biopsy due to the possibility of macroscopical visualisation, and several tissue samples can be collected at one time [7, 8]. However, it is still unclear, which would be the most reliable location of biopsy site: the duodenal bulb or the duodeno-jejunal junction [9–11].
Claudins (CLDNs) play a central role in the regulation of cell–cell adhesion, cell polarity and control of paracellular ion transport [12]. CLDNs are integral components of tight junction (TJ); 24 subgroups are known [13]. Recent studies have detected changes in claudin expression during cancer development and progression [14–17], but in degenerative diseases, it was not investigated yet. Earlier studies suggest that TJ has a possible role in CD pathomechanism [18–21], but no reference on published literature is known concerning claudins [22, 23]. We proposed that claudins may have a significant role in the intestinal permeability, cell to cell adhesion and the proliferation also in patients with CD.
The aim of this present study was duplex. First, we determined which part of the duodenum (the proximal or the distal region) is better for taking biopsies. Besides macroscopical view, detailed histological and CD3 immunohistochemical analysis were applied to detect subtle differences in the two duodenal regions. Moreover, the expressions of CLDNs were characterised in the proximal and distal part of duodenum in children with CD.
Materials and methods
Biopsy samples were composed of 33 newly diagnosed children with CD (20 girls and 13 boys) at the 1st Department of Pediatrics, Semmelweis University. Thirty-three children (range 2–17 years, mean age 6.4, 20 girls, 13 boys) were enrolled (Table 1). Coeliac disease was diagnosed after gastrointestinal symptoms (weight loss, failure to thrive, chronic diarrhoea, abdominal pain and fatigue), positive coeliac serology (endomysial antibody or IgA tissue transglutaminase) levels and abnormal small bowel biopsies (partial or total villous atrophy). Fourteen healthy children (11 girls, three boys) served as controls. Samples were taken from the proximal and distal part of duodenum. Biopsies were examined and analysed at the 2nd Department of Pathology, Semmelweis University.
Tissue blocks prepared from the mucosal biopsies were analysed on the basis of the morphological and the immunohistochemistrical analysis (monoclonal CD3). The villous/crypt ratio and the numbers of lymphocytes in the intraepithelial region were determined (original Marsh scoring) [24, 25]. According to the Marsh criteria, we divided our patients in two groups: Group I with less serious damages (Marsh grade up to 2) and Group II for the most serious change with villous atrophy and increased number of lymphocytes (Marsh grade 3).
Furthermore, the expression pattern of claudins in the duodenal mucosa was investigated by immunohistochemistry. The removed tissues were fixed in a 4% buffered formalin for 24 h, dehydrated and embedded in paraffin. The 3- to 4-μm thick sections were stained with haematoxylin–eosin. Paraffin sections were used for immunohistochemistry studies. The anti-CLDNs 2 and 4 antibodies were monoclonal; anti-CLDNs 3 antibodies were polyclonal purified from rabbit antiserum (Zymed, San Francisco, CA, USA). First, the slides were treated for 30 min in a target retrieval solution (Dako, Glostrup, Denmark) in a microwave oven, followed by incubation with the primary antibody at a 1:80 dilution at room temperature for 60 min and secondary antibody (biotinilated goat anti-rabbit) for 5 min in 41°C.
For visualisation, a standard avidin–biotin peroxidase technique (ABC system, Dako) was used with diaminobenzidine as chromogen. The sections were counterstained with haematoxylin. For each CLDN, a negative control with omission of the primary antibody was included. The reactions were carried out in a Ventana ES automatic immunostainer (Ventana Medical System Inc., Tucson, AZ, USA) using the reagents provided by the manufacturer.
The number of positive cells was calculated as follows: 10 randomly selected areas per slide were analysed using ×20 objective with 100 enterocytes counted in each field in the surface epithelium on the top of the villi. The scoring standardised for each group was as follows: 0 = 5%>; 1 = 5–20%; 2 = 20–40%; 3 = 40–60%; 4 = 60–80%; 5 = 80–100% of the cells showed positive reactions. For statistical analysis, Shapiro–Wilk test was applied to verify the normality, and the Mann–Whitney U test was used to compare the expression of individual CLDNs in the different groups. Results were considered to be statistically significant when p < 0.05. The Institutional Ethical Committee approved our study; written parental informed consent was obtained.
Results
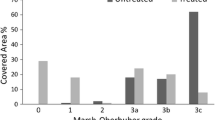
There was no significant difference between proximal and distal part of duodenum concerning macroscopical and microscopical view (Marsh scoring system) in patients with CD. Biopsies taken from proximal and distal area were shown as significant difference in the expression of CD3 positive cells compared to controls (p 1 = 0.026 and p 2 = 0.006, respectively). Increased CD3 expression was detected in biopsies from the distal region compared to the bulbus duodeni, but the difference was not significant (p = ns; Fig. 1). Similar tendency was observed in CLDN study; hence, elevated expression of CLDNs 2 and 3 in the distal duodenum of patients with CD was detected.
The percentage of CD3 positive T cells in the intraepithelial region in biopsies of coeliac patients and controls (bulbus and distal duodenum). CD3 positive cells, taken from proximal and distal area, were significantly increased in CD patients when compared with controls (*p 1 = 0.026 and #p 2 = 0.006)
In our study, we have investigated the expression of CLDNs 2, 3 and 4 in intestinal epithelial mucosa. In CD, CLDN 2 reacted in a granular pattern, outlining the cell membrane and sometimes found inside the cytopasma. CLDNs 3 and 4 in CD were localised in the suprabasal layer of the epithelium showing a honeycomb appearance (Fig. 2).
Claudin expression in coeliac disease (b, d, f) and normal duodenal epithelium (a, c, e). Claudin 2 reacted in a grandular pattern, outlining the cell membrane and sometimes found inside the cytopasma. (a ×20, b ×20, b insert ×100 imm.) Claudin 3 was localised in the suprabasal layer of the epithelium showing a honeycomb appearance. (c ×20, d ×20, d insert ×100 imm.) Expression of Claudin 4 was similar. (e ×20, f ×40, f insert ×100 imm.)
Increased CLDNs 2 and 3 expressions in the proximal and distal part of duodenum in patients with CD compared to controls was observed Moreover, in CD patients, the expression of CLDNs was more increased in the distal region. We have found a significant difference concerning CLDN 2 in the case of mild villous atrophy (Group I) in the distal region (p = 0.02) and in pronounced atrophy (Group II) in the bulb and the distal region as well (p = 0.001) in CD patients when compared with controls. CLDN 2 expression was significantly increased in the proximal and distal part of CD patients with severe atrophy (Group II) in comparison to the mild atrophy (Group I; p = 0.04 and p = 0.03, respectively; Fig. 3). Similar changes were observed with CLDN 3, too. The expression of CLDN 3 was significantly increased in proximal and distal part of duodenum in overt atrophy (Group II) as compared to controls (p = 0.01 and p = 0.001, respectively). Moreover, CLDN 3 expression was significantly increased in the proximal and distal part in CD patients with pronounced atrophy (Group II) in comparison to the mild atrophy (Group I; p = 0.008 and p = 0.004; Fig. 4). Expression of CLDN 4 was similar in all groups studied.
CLDN 2 expression in the proximal (bulbus duodeni) and distal region of the duodenum in coeliac patients and controls (Group 1 mild, Group 2 severe villous atrophy)/*p < 0.05 in CD with mild villous damage (Group 1) in the distal region and with serious damage (Group 2) in the bulb and distal region as compared to controls, #p < 0.05 in CD with severe villous atrophy as compared to Group 1 in the proximal and distal part of duodenum
CLDN 3 expression in the proximal (bulbus duodeni) and distal region of the duodenum in coeliac patients and controls. (Group 1: mild, Group 2: severe villous atrophy)/*p < 0.05 in CD with serious damage (Group 2) in the bulb and distal region as compared to controls, #p < 0.05 in CD with severe villous atrophy as compared to Group 1 in the proximal and distal part of duodenum
Discussion
Our results demonstrated that biopsies taken from proximal or distal part of duodenum are reliable for diagnosing CD. Nevertheless, histological scoring grade (Marsh) and the percentage of CD3 positive T cells showed slightly more severity in the distal part of duodenum. Moreover, CLDNs 2 and 3 expressions were also increased in the distal part of duodenum in comparison to the proximal part in patients with CD. These results may demonstrate that there is a slightly more serious damage in the mucosa of the distal duodenum in patients with CD. However, in everyday practice, both regions are suitable to diagnose villous atrophy in CD.
Earlier studies found a 95% concordance in the biopsies taken from the proximal duodenum and jejunum in adult population with CD [9]. Differences were found in three cases only in which there was a minor change in the proximal region (the possibility of latent CD) and more serious atrophy in the distal region. Ravelli et al. found that the total villous atrophy was significantly more frequently occurring in the distal area (distal duodenum, proximal jejunum), but when compared with the biopsies taken from one single patient, the differences were not significant [10]. As a result of their investigation, it is enough to take biopsy from one place for a trustworthy diagnosis, and this place must not necessarily be the farthest intestinal region.
As our results were compared with former investigations, we found the same results. For the correct diagnosis, it is not necessary to take the biopsy from the most distal part of the duodenum; nevertheless, the more severe damage was seen in the distal area. The difference was not significant; however, it can be important in the earlier stage of CD.
Claudins are the main components of TJ. CLDNs play an important role in the regulation of cell–cell adhesion and are responsible for maintaining cell polarity cellular arrangement, adhesion and paracellular ion transport of different tissues [26, 27]. As the damage of the cell–cell adhesion is an important role in the carcinogenesis, several recent studies have detected changes in CLDN expression during tumour diseases [28, 29]. All CLDNs were found in carcinomas, and their expression was tumour-specific [30–32]. Interestingly, in non-epithelial tumours (e.g. lymphoma), there was no expression of CLDN at all, or it was detected only in the cytoplasm. Győrffy et al. showed that the expression of CLDN 2 was increased in adenocarcinoma compared to Barrett oesophagus, while CLDN 3 and 4 were increased in both diseases compared to the foveolar epithelial cells [14].
Resnick et al. found that decreased presence of CLDN 3 was detected in normal gastric mucosa, where increased expression of CLDN 3 was associated with gastric adenocarcinomas [33]. In this paper, moderate membranous staining for CLDN 3 in intestinal metaplasia but not in normal gastric epithelium was shown similarly to our finding on duodenal mucosa. Japanese paper showed similar result, decreased CLDN 3 expression in normal gastric mucosa [34]. In a recent study, a significant increase of claudins 1 and 7 in premalignant cervical lesions and invasive cancer compared with normal cervical epithelia was shown. Moreover, CLDNs 3 and 4 were elevated in endometrial cancer in comparison to normal endometrium [35].
The possible role of CLDNs was also investigated in inflammatory bowel disease (IBD) [36, 37]. CLDN 2 expression was increased in IBD, while the expressions of CLDNs 3 and 4 were decreased in colonic epithelium [36]. This finding is contrast to our result (increased expression of CLDN 3 in CD patients); nevertheless, in human intestine, there is functional differences concerning macromolecular permeability in duodenal mucosa in comparison to colonic mucosa. Based on this knowledge, different expression of CLDN 3 in duodenal and colonic mucosa may be conceivable. It seems that proximal intestinal mucosa (gastric, duodenal) has different CLDN profile when compared with colonic mucosa. Further studies are necessary to analyse the simultaneous CLDNs expression in different part of human intestine of healthy individuals and of patients with coeliac disease or with IBD.
Increased permeability and altered cell to cell junctions in CD may correlate with the expression of TJ proteins such as CLDNs. However, the expression of CLDNs in coeliac disease was not investigated yet. Fasano et al. found that zonuline induced a pathological aggregation of tight junctions and increased the permeability of intestinal mucosa of untreated CD patients [23]. Further, studies showed that chronic gliadin exposure caused downregulation of both zonula occludens-1 (ZO-1) and occludin gene expression and activated zonulin signalling irrespective of the genetic expression of autoimmunity, leading to increased intestinal permeability [22]. Sander et al. described that the gliadin almost immediately changed the barrier function of the intestinal mucosa caused a reorganisation of actin filaments and altered expression of the TJ proteins (e.g. occludin, ZO-1, E-cadherin and claudins). Claudins 1, 3 and 4 each showed differences in their distribution and expression. After gliadin treatment of cells (human intestinal enterocytes), claudins 3 and 4 were decreased in plasma membrane staining and were increased in punctate cytosolic staining. Furthermore, the transepithelial resistance was decreased, and permeability was increased [19]. As a result of our study, there is a reference to the structural change in the TJ due to an increased CLDNs 2 and 3 expressions. In addition, our results demonstrated that the expressions of CLDNs 2 and 3 were increased in the mucosa in children with untreated CD when compared to controls. Furthermore, the expression of CLDNs and the grade of the atrophy were associated.
First and last, our study suggests that both proximal and distal part of duodenum are also reliable regions of taking biopsy sample to prove the villous atrophy in CD. However, using more sensitive methods, the distal part of duodenum depicted earlier signs of mucosal deterioration. Therefore, to diagnose slight changes of mucosal abnormality, the distal duodenum seems to be better location for intestinal biopsies. The increased expressions of CLDNs 2 and 3 suggest the structural damage of the TJs, which play a central role in the increased permeability and modified cell to cell adhesion and proliferation.
References
Dicke WK (1950) Coeliakie. MD Thesis, Utrecht
Marsh MN (1992) Gluten, major histocompatibility complex, and the small intestine. A molecular and immunbiologic approach to the spectrum of gluten sensitivity (‘coeliac sprue’). Gastroenterology 102:330–354
van Heel DA, West J (2006) Recent advances in coeliac disease. Gut 55:1037–1046
Green PH, Cellier C (2007) Celiac disease. N Engl J Med 357:1731–1743
Armstrong MJ, Robins GG, Howdle PD (2009) Recent advances in coeliac disease. Curr Opin Gastroenterol 25:100–109
Korponay-Szabó IR, Kovács JB, Czinner A et al (1999) High prevalence of silent celiac disease in preschool children screened with IgA/IgG antiendomysium antibodies. J Pediatr Gastroenterol Nutr 28:26–30
O'Loughlin EV, Dutt S, Kamath R et al (2007) Prospective peer-review audit of paediatric upper gastrointestinal endoscopy. J Paediatr Child Health 43:551–554
Tursi A, Brandimarte G, Giorgetti GM (2002) Endoscopic features of celiac disease in adults and their correlation with age, histological damage, and clinical form of the disease. Endoscopy 34:787–792
Thijs WJ, van Baarlen J, Kleibeuker JH et al (2004) Duodenal versus jejunal biopsies in suspected celiac disease. Endoscopy 36:993–996
Ravelli A, Bolognini S, Gambarotti M et al (2005) Variability of histologic lesions in relation to biopsy site in gluten-sensitive enteropathy. Am J Gastroenterol 100:177–185
Meijer JW, Wahab PJ, Mulder CJ (2003) Small intestinal biopsies in celiac disease: duodenal or jejunal? Virchows Arch 442:124–128
Kinugasa T, Sakaguchi T, Gu X et al (2000) Claudins regulate the intestinal barrier in response to immune mediators. Gastroenterology 118:1001–1011
González-Mariscal L, Betanzos A, Nava P et al (2003) Tight junction proteins. Prog Biophys Mol Biol 81:1–44
Győrffy H, Holczbauer A, Nagy P et al (2005) Claudin expression in Barrett's esophagus and adenocarcinoma. Virchows Arch 447:961–968
Aung PP, Mitani Y, Sanada Y et al (2006) Differential expression of claudin-2 in normal human tissues and gastrointestinal carcinomas. Virchows Arch 448:428–434
Kinugasa T, Huo Q, Higashi D et al (2007) Selective up-regulation of claudin-1 and claudin-2 in colorectal cancer. Anticancer Res 27:3729–3734
Soini Y, Tommola S, Helin H et al (2006) Claudins 1, 3, 4 and 5 in gastric carcinoma, loss of claudin expression associates with the diffuse subtype. Virchows Arch 448:52–58
Utech M, Brüwer M, Nusrat A (2006) Tight junctions and cell-cell interactions. Methods Mol Biol 341:185–195
Sander GR, Cummins AG, Henshall T et al (2005) Rapid disruption of intestinal barrier function by gliadin involves altered expression of apical junctional proteins. FEBS Lett 579:4851–4855
Visser J, Rozing J, Sapone A et al (2009) Tight junctions, intestinal permeability, and autoimmunity: celiac disease and type 1 diabetes paradigms. Ann N Y Acad Sci 1165:195–205
Guandalini S, Setty M (2008) Celiac disease. Curr Opin Gastroenterol 24:707–712
Drago S et al (2006) Gliadin, zonulin and gut permeability: effects on celiac and non-celiac intestinal mucosa and intestinal cell lines. Scand J Gastroenterol 41:408–419
Fasano A et al (2000) Zonulin, a newly discovered modulator of intestinal permeability, and its expression in coeliac disease. Lancet 355:1518–1519
Green A, Rostami B, Marsh C (2005) Diagnosis of coeliac disease. Gastroenterology 19:389–400
Oberhuber G, Granditsch G, Vogelsang H (1999) The histopathology of coeliac disease: time for a standardized report scheme for pathologists. Eur J Gastroenerol Hepatol 11:1185–1194
Furuse M (2006) Molecular mechanism behind the barrier function of tight junctions. Seikagaku 78:601–608
Tsukita S, Furuse M (2000) The structure and function of claudins, cell adhesion molecules at tight junctions. Ann N Y Acad Sci 915:129–135
Morin PJ (2005) Claudin proteins in human cancer: promising new targets for diagnosis and therapy. Cancer Res 65:9603–9606
Swisshelm K, Macek R, Kubbies M (2005) Role of claudins in tumorigenesis. Adv Drug Deliv Rev 57:919–928
Soini Y (2005) Expression of claudins 1, 2, 3, 4, 5 and 7 in various types of tumours. Histopathology 46:551–560
Kulka J, Tökés AM (2005) Claudin expression in breast tumors. Human Pathol 36:859
Tokés AM, Kulka J, Paku S et al (2005) Claudin-1, -3 and -4 proteins and mRNA expression in benign and malignant breast lesions: a research study. Breast Cancer Res 7:R296–R305
Resnick MB et al (2005) Claudin expression in gastric adenocarcinomas: a tissue microarray study with prognostic correlation. Human Pathol 36:886–892
Matsuda Y et al (2007) Gastric and intestinal claudin expression at the invasive front of gastric carcinoma. Cancer Sci 98:1014–1019
Szabó I et al (2009) Claudins as diagnostic and prognostic markers in gynecological cancer. Histol Histopathol 24:1607–1615
Prasad S, Mingrino R, Kaukinen K (2005) Inflammatory processes have differential effects on claudins 2, 3 and 4 in colonic epithelial cells. Lab Invest 85:1139–1162
Mankertz J, Schulzke JD (2007) Altered permeability in inflammatory bowel disease: pathophysiology and clinical implications. Curr Opin Gastroenterol 23:379–383
Acknowledgements
This work was supported by grants NKFP-1A/0023/2002 from Hungarian Ministry of Health, National Research Development Projects; OTKA T 049559 from the National Science Research Foundation and ETT-077/2003 from the Hungarian Ministry of Health.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dorottya Nagy Szakál and Hajnalka Győrffy have equally contributed to this work.
Rights and permissions
About this article
Cite this article
Nagy Szakál, D., Győrffy, H., Arató, A. et al. Mucosal expression of claudins 2, 3 and 4 in proximal and distal part of duodenum in children with coeliac disease. Virchows Arch 456, 245–250 (2010). https://doi.org/10.1007/s00428-009-0879-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-009-0879-7