Abstract
Copper is an essential element in cells; it can act as either a recipient or a donor of electrons, participating in various reactions. However, an excess of copper ions in cells is detrimental as these copper ions can generate free radicals and increase oxidative stress. In multicellular organisms, copper metabolism involves uptake, distribution, sequestration, and excretion, at both the cellular and systemic levels. Mammalian enterocytes take in bioavailable copper ions from the diet in a Ctr1-dependent manner. After incorporation, cuprous ions are delivered to ATP7A, which pumps Cu+ from enterocytes into the blood. Copper ions arrive at the liver through the portal vein and are incorporated into hepatocytes by Ctr1. Then, Cu+ can be secreted into the bile or the blood via the Atox1/ATP7B/ceruloplasmin route. In the bloodstream, this micronutrient can reach peripheral tissues and is again incorporated by Ctr1. In peripheral tissue cells, cuprous ions are either sequestrated by molecules such as metallothioneins or targeted to utilization pathways by chaperons such as Atox1, Cox17, and CCS. Copper metabolism must be tightly controlled in order to achieve homeostasis and avoid disorders. A hereditary or acquired copper unbalance, including deficiency, overload, or misdistribution, may cause or aggravate certain diseases such as Menkes disease, Wilson disease, neurodegenerative diseases, anemia, metabolic syndrome, cardiovascular diseases, and cancer. A full understanding of copper metabolism and its roles in diseases underlies the identification of novel effective therapies for such diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As a transition metal, copper (Cu) plays several key roles in the human body. These various roles are due to its two oxidation states, Cu(I)/Cu+ (cuprous ion) and Cu(II)/Cu2+ (cupric ion), which endow it with the ability to act as either a recipient or a donor of electrons. Cu+ prefers to bind to the thiol group in cysteine or the thioether group in methionine, while Cu2+ exhibits a high affinity for the secondary carboxyl group in aspartic/glutamic acid or the imidazole nitrogen group in histidine [37]. As a result, copper ions readily form complexes with biomolecules containing these amino acid residues. It has been reported that Cu atoms are involved in a wide spectrum of proteins, such as copper/zinc superoxide dismutase (Cu/Zn SOD or SOD1), cytochrome c oxidase (CcO), mitogen-activated protein kinase MEK1 [10, 149], and cAMP-degrading phosphodiesterase PDE3B [79]. In these proteins, copper ions participate in diverse biochemical reactions (especially redox reactions) via donation or accepting of electrons [30], and maintain specific protein structures by coordinating with the abovementioned groups. Thus, although copper is much less abundant in organisms compared with iron, it is indispensable in cell metabolism, especially in the mitochondrial respiratory chain.
However, excess intracellular free copper ions are potentially toxic [30]. During the switch between Cu(I) and Cu(II) states, electron transfer results in the generation of reactive oxygen species (ROS), including superoxide anion (O2−), nitric oxide (NO−), hydroxyl radical (OH−), and hydrogen peroxide (H2O2), through Fenton reactions [151]. ROS can damage organic molecules, including proteins, nucleic acids, and lipids. ROS can also interfere with the synthesis of iron-sulfur clusters, which have pivotal roles in a myriad of essential enzymes. Additionally, superfluous copper ions may displace other metals from their cognate ligands in metalloproteins, resulting in improper protein conformations or impairment of enzymatic activities [37].
Given that both an excess and a lack of copper ions are pernicious, copper homeostasis must be maintained in living organisms. Otherwise, a copper imbalance may lead to various kinds of disorders. The following sections aim to provide an updated summary of the literature on copper metabolism and diseases related to copper dyshomeostasis.
Copper metabolism in mammals
In multicellular animals, copper ions undergo uptake, distribution, and efflux. Each event takes place at both the cellular and systemic levels. Over the last several decades, the mechanisms underlying these pathways have been intensively studied and several molecules involved in these processes have been verified in mouse models (shown in Table 1). A schematic diagram of copper metabolism in mammals at the cellular and molecular levels is shown in Fig. 1.
Schematic diagram of copper metabolism at the cellular and molecular levels in mammals. Bioavailable copper ions are primarily transported by Ctr1. Then, they are reduced by reductases such as Steap 2, 3, 4, and DCYTB. Cu2+ can be incorporated by DMT1. After incorporation, cuprous ions undergo utilization and detoxification pathways: the chaperons CCS, Cox17, and Atox1 target Cu+ to SOD1, CcO, and Golgi bodies, while superfluous cuprous ions are sequestrated by metallothioneins (MT1 and MT2). Cuproproteins (CuPrs) assembled in the Golgi compartment are secreted out of the cell or sorted to specific organelles. In enterocytes or hepatocytes, ATP7A and ATP7B relocate to the plasma membrane to pump Cu+ into the blood or bile, respectively. Elevated copper levels stimulate expression of MT1 and MT2, mediated by the transcription factors MTF1 and Nrf2, to scavenge the excessive Cu+
Uptake of copper ions
Humans and other mammals obtain copper from their diet. This micronutrient is present in high concentrations in foods such as liver, crustaceans, red meat, milk, chocolate, mushrooms, nuts, and beans [108]. According to the US Food and Nutrition Board, a human adult should consume 0.9 mg of copper per day [15]. It has been reported that the median intake of dietary copper in adults falls between 1.0 and 1.6 mg daily [9]. Thus, most adults obtain adequate copper from their daily diet. Pregnant women, infants, and children should consume more copper through their diet [108].
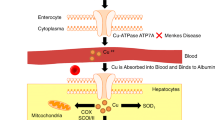
In the mammalian digestive tract, copper in consumed food is taken up by enterocytes. The extracellular copper ions are in the form of Cu(II), which can be directly incorporated by DMT1 (divalent metal transporter 1). However, these cupric ions cannot be used directly by cells [131]. It was conventionally assumed that all types of cells, including enterocytes, share the same copper incorporation pathway: 4 reductases; DCYTB; and Steap 2, 3, and 4 are responsible for reducing Cu2+ into Cu+ on the surface of mammalian cells [112, 160]; then, the cuprous ions are incorporated by Ctr1 (copper transporter 1) in a high-affinity manner [34]. It has been widely accepted that Ctr1-dependent copper incorporation is the primary means of incorporation of cupric ions in peripheral tissues. However, the process for enterocytes is more complicated. It has been reported that Ctr1 is located at the apical membrane of enterocytes [109, 110]. This is consistent with the assumption that Ctr1 might mediate uptake of dietary copper from intestinal lumen. Confusingly, other studies have shown that Ctr1 is located at both the apical and blastolateral membranes, as well as inside enterocytes [166]. A further investigation revealed that copper absorption from the intestinal lumen is largely independent of Ctr1, because inactivation of intestinal Ctr1 barely impaired accumulation of copper in enterocytes [110]. This indicates that copper transportation across the apical membrane is mainly an uncharacterized low-affinity process, which might be mediated by transporters such as DMT1 or driven by endocytosis [96]. It is likely that Ctr1 is required to release dietary copper from subapical vesicles for further utilization; however, the underlying mechanism remains unclear. Another possibility is that only cuprous ions transported by Ctr1 from intestinal lumen are bioavailable [54, 109]. Thus, in the absence of Ctr1, copper ions absorbed via other pathways are unavailable to the organism because they are sequestrated in subapical vesicles [116]. This has been confirmed by the observation that enterocyte-specific Ctr1 knockout mice exhibit severe copper deficiency in peripheral tissues and die about 3 weeks post-birth; these effects can be rescued by intraperitoneal copper injection [109, 110]. On the other hand, systemic knockout of Ctr1 leads to embryonic lethality [80, 85], while cardiac-specific Ctr1 knockout produces cardiac hypertrophy and a decrease in copper accumulation in cardiac tissue [77]. These genetic models confirm the importance of Ctr1 in both central and peripheral copper uptake (Table 1).
Distribution and sequestration of copper ions
After incorporation, some copper ions will be targeted to different cuproproteins (CuPrs) via three utilization pathways: cytosolic, mitochondrial, and Golgi routes [37]. In cytosol, CCS (copper chaperone for Sod1) mediates Cu+ loading and activation of superoxide dismutase 1 (Sod1). This role of CCS has been verified in CCS knockout (KO) mice (Table 1) [101, 159]. On the other hand, KO mouse models have demonstrated the key roles of CCS and Sod1 in ROS scavenging in germline cells and neurons (Table 1) [101, 118, 159]. A recent study suggested that CCS acquires copper from Ctr1 and passes it to Sod1 via formation of a Ctr1-CCS-Sod1 complex, which can be broken with complete activation of Sod1 [135]. Besides CCS, the chaperons Cox17 and Atox1 can also escort Cu+ from Ctr1 [37]. Cox17 shuttles between the cytoplasm and mitochondrial lumen, delivering cytosolic cuprous ions to Sco1 or Sco2, both of which are located on the inner membrane of the mitochondrion. They pass copper atoms to the copper A (CuA) site of Cox2 [20]. Cox17 also delivers copper from the cytoplasm to Cox11, which acts as a copper donor to the copper B (CuB) site of Cox1 [20]. Cox1 and Cox2 are two subunits of CcO, which is an essential copper-dependent enzyme in the oxidative phosphorylation process [20]. It has been reported that Cox17-mediated chaperoning of copper is also crucial to CcO biogenesis [20, 152]. Atox1 passes copper to P1B-type ATPases, including ATP7A (ATPase copper-transporting α) and ATP7B (ATPase copper-transporting β) [58]. These ATPases are located on the trans-Golgi network (TGN) or plasma membrane in different circumstances, and pump Cu+ from Atox1 to the other side of the membrane [27]. ATP7A and ATP7B transporters on the plasma membrane are pivotal in the systemic distribution of copper ions (detailed below), while those on the TGN membrane contribute to loading of copper to various CuPrs, such as Sod3 (superoxide dismutase 3) and ceruloplasmin (CP). Then, these CuPrs assembled with Cu+ are sorted to specific organelles or secreted out of the cell [27].
In enterocytes, a large proportion of absorbed cuprous ions are secreted into circulation to reach other tissues. This process is dependent on the abovementioned Atox1/ATPase routes. ATP7A is widely expressed and is located on the TGN membrane in most cases [27]. Unlike in other cell types, in enterocytes, a large amount of ATP7A molecules relocate to the basolateral membrane. These P-type ATPases on the plasma membrane also receive cuprous ions from Atox1 like they do on the TGN membrane, and mediate the exodus of copper from enterocytes into the bloodstream [73]. Genetic mouse models have demonstrated that deficiency of the Atox1/ATP7A route causes defects in Cu distribution, leading to pathological changes in several organs (Table 1) [50, 57, 64, 88, 119]. Albumins and free amino acids in the blood associate to the copper ions secreted from enterocytes, and these can be absorbed by Ctr1 upon arrival at peripheral tissues/organs [143].
Under physiological conditions, a healthy adult body contains about 80 mg of copper in total, with the concentrations being highest in the eyes, heart, liver, and brain [51]. Among these organs, the liver is an important organ that stores copper from the portal vein. Surplus cuprous ions in hepatocytes bind to metallothioneins (MTs) in the cytosol or are stored within the lysosome as a reservoir [8]. ATP7A is normally expressed in many tissues, except for the liver, where it is replaced by its paralogue, ATP7B [37]. One study reported that cardiac-specific KO of Ctr1 leads to a dramatic increase in expression of ATP7A in the mouse liver [77].
A large proportion of copper in hepatocytes is targeted to CP via the Atox1/ATP7B/Golgi route. Then, CP is secreted into the blood and oxidizes Fe(II) into Fe(III) to facilitate its absorption and mobilization [18, 32, 61]. It has been reported that the normal serum copper concentration is approximately 109 μg/100 mL, with 90% bound to CP and the remaining cuprous ions associated with albumins or free amino acids [51].
Since free copper ions have the potential to generate ROS in cells, the excess intracellular Cu+ must be sequestered by molecules such as MTs and glutathione (GSH). MTs are a family of low molecular weight proteins encoded by four genes (MT1, MT2, MT3, and MT4), whose expression can be induced by increased metal ions, such as copper and zinc. MTs contain 61–68 amino acid residues, among which, 20 cysteine residues are distributed in two globular domains, α and β clusters [70]. Thiol groups in cysteins endow MTs, especially MT1 and MT2, with the capacity to chelate a large portion of Cu+ incorporated by Ctr1 to avoid cellular toxicity [13].
GSH is a tripeptide containing glutamate, cysteine, and glycine residues. It is synthesized by two enzymes, γ-glutamylcysteine sythetase and GSH synthase, instead of by ribosomes [7, 95]. Like MTs, GSH can buffer excess copper via its association with this ion. This tripeptide is also crucial to intracellular distribution of copper, as GSH is likely the first acceptor of copper after its entry into the cell, following which, Cu+ is either delivered to the chaperones or sequestered by MTs [42, 99, 135]. On the other hand, GSH plays a key role in maintaining the redox condition of cysteines in the N-terminus of ATP7A and ATP7B, which is important for the exportation of copper ions by ATPases [134].
Excretion of copper ions
Copper ions can be secreted out of cells via several pathways (shown in Fig. 1). In all cell types, various CuPrs are loaded with Cu+ by ATP7A or ATP7B at the TGN; they are then secreted out of cells. In enterocytes, incorporated Cu+ can be directly pumped into the blood by ATP7A residing at the blastolateral membrane; these copper ions can be delivered to the liver and absorbed by hepatocytes in a Ctr1-dependent manner [73]. In hepatocytes, CP is synthesized and assembled with Cu+ at the TGN; these holo-CPs can then be released into the blood along with the copper ions bound on them, which can, in turn, be recycled by other tissues via blood circulation [90]. In response to elevated copper levels, ATP7B in hepatocytes also translocates from the Golgi apparatus to the lysosome and imports copper into its lumen. Then, lysosomal ATP7B collaborates with p62 to trigger exocytosis, releasing excess copper into the bile; this is then excreted out of the body [117]. Hepatic ATP7B on the apical membrane can also directly pump copper ions into bile to be recycled or excreted via the digestive tract [5]. The importance of ATP7B in copper excretion has been verified in mouse models generated by either homologous recombination or random mutation [11, 24, 65, 81, 145].
Copper homeostasis in mammals
In mammals, homeostasis of a nutrient consists of two aspects: cellular homeostasis and systemic distribution. Copper homeostasis in mammalian cells is achieved through the regulation of genes involved in copper influx and detoxification. The abundance of the mammalian Ctr1 protein at the plasma membrane is negatively regulated by the cellular copper concentration according to a feedback mechanism: Ctr1 is removed from the cell surface and undergoes internalization or endocytosis-dependent degradation in response to elevated copper levels [106, 115]; the internalized Ctr1 can also be recycled back to the plasma membrane when the extracellular copper concentration is reduced [43, 126]. This regulation may explain the observation that varying dosages of dietary copper affect the copper incorporation efficiency of enterocytes: copper depletion increases the fraction of copper that is absorbed, with little drained out of the digestive tract, while chronic dietary consumption of copper significantly impairs the absorption efficiency, regardless of the high level of overall copper in the intestine [116]. On the other hand, it has been reported that high concentrations of cellular copper ions can enhance transcription of metallothionein genes (MT1 and MT2) to scavenge the excess and toxic copper ions. This transactivation is mediated by the transcription factor MTF1, which is indirectly regulated by copper. Furthermore, data from the Tohru Fukai group revealed that mammalian Atox1 not only acts as a copper chaperon but also acts as a copper-dependent transcription factor. The researchers found that copper stimulates nuclear translocation of Atox1, which then binds to promotors of genes including cyclin D1, NADPH oxidase organizer p47phox, and SOD3 [16, 67, 75, 113]. Whether this transcription factor function of Atox1 is involved in copper homeostasis in mammals remains unclear. There also is evidence suggesting that the MT1 gene contains four functional metal response elements (MREs) and that the transcription factor Nrf2 is crucial to MRE-mediated transcription of MT1 in response to increased copper [136]. Under low cellular concentrations of copper and other metal ions, the metal-free MTs, termed apo-MTs, are highly vulnerable to proteolytic processes, resulting in a low level of these chelators [137].
A large body of studies demonstrates that mammals have evolved an exquisite ability to sense and retain copper in the organs; however, the underlying mechanisms are still not clearly understood. In early studies, Levenson CW measured copper turnover by monitoring two naturally occurring stable isotopes of this metal (63Cu and 65Cu) [86, 87]. They found that copper conservation in organs could be induced in response to dietary copper restriction. This conservation was highly organ specific: copper conservation in organs such as the brain and heart was very efficient, with little loss of copper, while conservation in the liver was induced only after the loss of a significant amount of copper [86, 87].
It is well-established that liver and hepatic ATP7B are crucial to systemic homeostasis of copper. As mentioned above, copper ions incorporated by enterocyts are delivered to the liver via the blood, and are then secreted out of hepatocytes through different ATP7B-dependent routes: at basal copper concentrations, hepatic ATP7B resides at the TGN and loads Cu+ to CP, which mediates copper recycling through the circulation system [90, 163]; high concentrations of hepatic copper induce ATP7B translocation to either the apical surface or lysosome, where copper excretion occurs via bile [5, 117]. In the cardiac-specific Ctr1 knockout mouse model mentioned above, expression of ATP7A was strongly increased in the liver and intestine [77]. These data suggest that demand for copper in peripheral tissues can be sensed via an as-yet unknown mechanism, which then upregulates expression of ATP7A in both the liver and intestine to promote copper distribution.
Roles of copper in human diseases
Due to the dual role of copper ions, either insufficiency or superfluity of this metal may cause various diseases (listed in Table 2). As discussed above, ATP7A and ATP7B play key roles in copper distribution in the human body. Genetic inactivation of ATP7A leads to a syndrome characterized by severe systemic copper deficiency, known as Menkes disease (MD), while mutations of ATP7B impair the ability of the body to excrete copper into bile, causing accumulation of this metal in the body and a disease known as Wilson disease (WD). Perturbations in copper homeostasis due to ATP7A/B mutations manifest as a variety of disorders, including neuropathy, anemia, and cardiovascular diseases. These pathological manifestations of MD and WD offer clues to understanding the physiological consequences of copper deficiency, overload, and misdistribution.
Menkes disease and copper deficiency disorders
ATP7A is vital for pumping copper from enterocytes to the blood. Inactivation of this transporter results in disturbed copper acquisition and systemic copper deficiency, giving rise to Menkes disease (MD). The ATP7A gene, located at Xq13.3, encodes a transmembrane peptide with 1500 amino acid residues. To date, more than 370 different mutations of ATP7A have been identified as implicated in MD [53, 147]. Classical MD and its milder allelic variant, occipital horn syndrome (OHS), are characterized by progressive cerebral and cerebellar neurodegeneration, fair skin with kinky hair, and connective tissue disturbances [73]. In some individuals with MD, intervention with supplemental copper can attenuate disease progression and relieve most of the symptoms, highlighting the essential role of copper in humans [147].
Copper deficiency results in decreased activities of multiple enzymes, and this is believed to underlie the neuropathic manifestations of MD. Dopamine-β-hydroxylase (DBH) is a copper-dependent enzyme responsible for converting dopamine to norepinephrine; it is a crucial neurotransmitter in norepinephrinergic neurons. Reduced DBH activity contributes to the abnormal neurochemical patterns observed in the plasma and cerebrospinal fluid (CSF) of MD patients [47, 73, 74, 164]. Peptidyl-α-amidating monooxygenase (PAM) is another copper-requiring enzyme. Like DBH, PAM is metallated at the TGN. PAM is essential in neurogenesis and neuronal survival via mediation of the maturation of a variety of neuroendocrine peptides such as corticotropin-releasing hormone, neuropeptide Y, and pituitary adenylyl cyclase–activating polypeptide [33, 59]. In addition, copper deficiency in MD may also result in decreased activities of other cuproenzymes, including CcO, SOD1, and tyrosinase, which might also contribute to the neuropathology observed in this disorder [71, 73]. It also has been reported that synaptic release of copper might competitively inhibit N-methyl-d-aspartate receptors (NMDAR) in a neuroprotective fashion, while dysfunction of ATP7A and copper deficiency might lead to prolonged, potentially deleterious NMDAR activation, inducing degenerative loss of neurons in MD patients [73, 128].
OHS is a less severe form of MD, and manifests as abnormal connective tissues. These connective tissue defects are likely due to impairment in lysyl oxidase (LOX) activity. LOX is a copper-requiring enzyme that normally deaminates lysine and hydroxylysine at the first step of collagen crosslink formation. In MD patients, reduced plasma copper levels and defective transportation seem to directly decrease LOX enzyme activity, and indirectly downregulate mRNA levels of both LOX and its substrate tropoelastin [25, 44]. As a result, defects in collagen and elastin may lead to connective tissue abnormalities, kinky hair, and abnormal vasculopathy such as vascular tortuosity and peripheral aneurysms, which are observed in MD and OHS [25, 46, 107].
Mutations in ATP7A can also cause X-linked Spinal Muscular Atrophy type 3 (SMAX3), a distal hereditary motor neuropathy [76]. In SMAX3, missense mutations induce subtle defects in ATP7A intracellular trafficking, resulting in preferential accumulation at the plasma membrane [163]. By specifically deleting the ATP7A gene within motor neurons, Hodgkinson VL et al. generated a SMAX3 mouse model, named the ATP7AMN/Y mouse [64]. This model perfectly mimics the symptoms of SMAX3 patients; although no overt signs of systemic copper deficiency were observed in this model, the ATP7AMN/Y mice exhibited an age-dependent overall reduction in copper concentration in the spinal cord and a concomitant increase in copper content in motor neurons, as compared with wild-type mice (Table 1) [64]. These findings further confirm the importance of ATP7A in copper distribution and neuronal functions.
Besides ATP7A, the CcO assembly gene SCO2 has also been identified to cause severe inherited disease in humans. SCO2 encodes a copper chaperon located on the mitochondrial inner membrane. As mentioned above, Sco2 passes cuprous ions from Cox17 to Cox2 in mitochondrion and facilitates the formation of holoenzymes of CcO. SCO2 mutations cause CcO activity deficiency in several tissues, particularly in skeletal and heart muscles, leading to neonatal death in humans [114, 144].
In addition to the abovementioned hereditary causes of copper disturbance, multiple acquisition-related factors may result in copper deficiency and consequential disorders in humans. Poverty, malnutrition, anorexia, vegetarianism, malabsorption, long-term diarrhea, and parenteral hyperalimentation may lead to inadequate intake of copper [39]. Gestation and lactation in women, as well as the infant and child developmental period, increases the demand for copper, making these groups vulnerable to copper deficiency [108]. Acquired copper deficiency in these cases often leads to serum copper and CP concentrations below the normal range, with symptoms including progressive lower extremity paresthesias, dysesthesias, optic neuropathy, other neurological dysfunctions, weakness, fatigue, and pancytopenia [9, 19, 162]. The neurological symptoms can be explained by the abovementioned functions of CuPrs in the neural system, while the hematological manifestations may be due to the crucial role of copper in CP synthesis and hematopoiesis. Since copper-containing enzymes such as hephestin and CP are essential in iron transportation, copper deficiency always produces iron deficiency and anemia [108]. Copper deficiency anemia, as well as decreased activity of CcO, may account for weakness and fatigue in these patients. On the other hand, it has been reported that dietary copper can affect lipid storage [35], and inadequate copper intake can lead to lipid accumulation, causing non-alcoholic fatty liver disease (NAFLD) [2]. This is likely due to copper-mediated inhibition of the cAMP-degrading phosphodiesterase PDE3B: cuprous ion directly binds to PDE3B and inhibits its enzymatic activity; copper deficiency leads to derepression of PDE3B and a reduction in cellular cAMP level, causing a decrease in cAMP-dependent lipolysis in adipocytes [79]. Copper is also thought to be involved in atherosclerosis via its impact on the metabolism of low-density lipoprotein (LDL) and cholesterol [83, 142]. However, whether these pathways are involved in metabolic syndrome remains unknown. Moreover, it has been reported that deletion of extracellular SOD3, whose activity is dependent on copper loading at the TGN via the ATP7A/Atox1 axis, increases O2− production and induces hypertension in response to angiotensin II stimuli [92, 93]. Based on data from animal models and clinical research, copper deficiency has been proposed as a risk factor for atherosclerosis and coronary heart disease, although the mechanisms are unknown [78, 82]. Taken together, these findings indicate that acquired copper deficiency can result in neurological, hematological, cardiovascular, and metabolic disorders via varying mechanisms.
For both congenital and acquired copper deficiency, the most effective treatment is copper supplementation. For patients with copper deficiency as a result of inadequate intake of copper, a diet containing sufficient copper is required for recovery [9, 162]. However, since patients with ATP7A mutations exhibit defective copper delivery from the intestine to peripheral tissues, the only way to reverse this deficiency is to administer intravenous copper. Given that copper-histidine is naturally present in serum and is quantitatively important in copper transport, this compound is currently the most preferred treatment agent. However, administration of copper-histidine for more than 3 years is not desirable due to the expected nephrotoxicity [72, 74, 148]. Recently, Ogata et al. reported a case of a 29-year-old MD patient who tolerated long-term intravenous therapy with copper chloride for more than 20 years without major adverse effects [111]. The relationship between his survival into adulthood and this unconventional treatment regimen remains controversial [111].
Wilson disease and copper overload disorders
ATP7B is a paralogue of ATP7A. It encodes an intracellular copper transporter in hepatocytes that is located on the Golgi apparatus or canalicular membrane, depending on the cellular cuprous level. Under low concentrations of hepatocellular copper, ATP7B loads cuprous ions onto CP at the Golgi complex; when high levels of copper ions are stored in the liver, ATP7B promotes exodus of them via bile [5, 117]. Since first identified as the gene responsible for WD in 1993, more than 500 mutations of ATP7B have been implicated in the disease. In WD, copper accumulates and causes toxicity in the liver due to inactivation of ATP7B and the consequential defect in biliary excretion of copper [62, 103]. As a result, patients may suffer from hepatitis, liver failure, and cirrhosis [105]. Inactivation or deletion of ATP7B in mice also produces WD symptoms [81, 145].
Since ATP7B is inactivated in WD patients, excess hepatic copper ions overflow into the blood, rather than into the bile, with secondary pathological copper accumulation in other tissues, particularly the brain. The toxic copper can produce neurodegeneration, manifesting as neurological symptoms and psychiatric disturbances [29, 62, 103]. Dysfunction of ATP7B may also result in hemolytic anemia. In WD patients, increased non-CP-bound copper in the blood can loosely bind to albumin and attack erythrocyte plasma membranes. This causes coombs-negative hemolysis, and finally, a hemolytic crisis, resulting in anemia [139].
There is also evidence suggesting the involvement of copper in tumor formation and progression. It is well-known that copper levels are elevated in a variety of malignancies, including both solid tumors and blood cancers. The high levels of copper concentrated in cancer cells are essential in tumorigenesis, angiogenesis, and metastasis [38, 55, 66]. Stephen Howell and his research group revealed that copper acquired from Ctr1 is required to increase phosphorylation of ERK1/2 or AKT via FGF-, PDGF-, and EGF-induced MAPK signaling; importantly, ERK1/2 and AKT play major roles in organismal development and tumorigenesis [146]. Dennis Thiele and Christopher Counter and their research groups reported that copper taken up by Ctr1 can bind to MEK1 kinase, which in turn phosphorylates and activates ERK1/2 kinases, mediating tumorigenesis driven by the V600E mutant of BRAF kinase [10, 149]. In addition, copper directly binds to multiple factors such as FGF1 and its receptor, as well as to angiogenin, to promote angiogenesis via enhancing the release of these factors or the affinity between the ligands and receptors [60, 84, 97, 150]. It has also been reported that copper activates hypoxia-inducible factor-1α (HIF1α) via CCS, and consequentially, induces expression of VEGF, which can mediate angiogenesis [69]. As Atox1 can transactivate cyclin D1 in a copper-dependent manner [67], this likely contributes to cancer cell proliferation and angiogenesis. Moreover, copper may promote tumor progression by elevating diverse cuproenzymes including SOD1, CcO, DBH, LOX, and CP [60, 94, 150].
As mentioned above, copper deficiency and the copper-dependent enzyme SOD3 can contribute to cardiovascular diseases. Intriguingly, several researchers have found that excess copper can also induce atherosclerosis and hypertension. It has been reported that hypoxia increases Cu fluxes by upregulating Ctr1 and ATP7A in pulmonary arteries and pulmonary arterial smooth muscle cells. The increased copper levels elevate LOX activity and cross-linking of extracellular matrix (ECM), eventually leading to an increase in pulmonary arterial stiffness and development of pulmonary hypertension [43, 167]. On the other hand, copper can promote neointimal thickening and vascular remodeling after vascular injury through directly binding to IL-1α and FGF1 to facilitate their release [97]. Transactivation of p47phox and SOD3 mediated by copper and Atox1 may also promote neovascularization and hypertension [16, 113]. Copper can also promote atherogenesis by elevating the expression or activity of proteins involved in LDL uptake and oxidation, as well as those involved in cholesterol biosynthesis [83, 142]. In addition, plasma homocysteine interacts with copper to enhance hydrogen peroxide, leading to peripheral vascular disease and ischemic heart disease [68, 98, 138]. Taken together, these findings indicate that excess copper promotes cardiovascular diseases via multiple mechanisms.
WD can be successfully treated with oral agents, including copper chelators and zinc preparations [29, 91, 124]. Patients with hepatic presentations are more often treated by chelators, including d-penicillamine and trientine or tetrathiomolybdate. Excess Cu ions can bind to these chelators and are excreted from the body via the kidneys. As a result, urinary excretion of non-CP-bound copper is promoted [154, 165]. Unfortunately, chelator therapy, especially d-penicillamine, is associated with numerous side effects. Early sensitivity reactions, characterized by fever and cutaneous eruptions, lymphadenopathy, neutropenia, or thrombocytopenia, and proteinuria, may occur during the first 1–3 weeks. Late reactions include nephrotoxicity, usually heralded by proteinuria or the appearance of other cellular elements in the urine [6, 31, 104, 132, 155]. Liver transplantation is often performed in WD patients with acute liver failure [124]. For pre-symptomatic and neurological patients, zinc therapies such as zinc sulfate and zinc acetate seem to be preferred [158]. With increased zinc incorporated in enterocytes, expression of MTs can be readily induced; MTs have a greater affinity for copper than for zinc. Once bound, the copper is not pumped into the circulation but is lost into the fecal contents [26, 63, 124, 127]. Gastrointestinal reactions and hepatic deterioration are relatively common side effects of zinc therapies [100, 158].
Copper misdistribution and diseases
As discussed above, both MD and WD may present with neurodegeneration, highlighting the complex roles of copper in neuropathies. A considerable body of literature has demonstrated a correlation between neurodegenerative diseases and copper misdistribution in the neural system. As mentioned above, the copper content of motor neurons is increased while that of the spinal cord is decreased in the SMAX3 mouse model [64]. In Parkinson’s disease (PD), copper levels are decreased in the degenerating substantia nigra pars compacta, while Lewy bodies are enriched in this metal because it co-aggregates with α-synuclein (α-Syn) in neurons [21, 45]. In the brains of Alzheimer’s disease (AD) patients, copper accumulates in senile plaques composed of amyloid-β (Aβ) peptides as well as in neurofibrillary tangles (NFTs), the latter of which is composed of hyperphosphorylated Tau proteins. However, some important brain regions, such as the neocortex, are deficient of copper [1, 21, 49, 120, 129]. It has also been reported that CCS delivers copper to BACE1, which is a crucial aspartic protease involved in the generation of Aβ; this highlights the involvement of CCS-mediated copper delivery in AD [3]. It is believed that Aβ and α-Syn can sequestrate copper ions and form aggregates, reducing the available copper in some parts of the brain [4, 56]. Copper has also been found to be sequestrated by Huntington, mutant SOD1, and Prion, which are involved in Huntington’s disease (HD), amyotrophic lateral sclerosis (ALS), and Prion disease (PrD), respectively [41, 125, 153, 156, 157, 161]. In some cases, sequestration of copper ions by these culprit proteins might cause or aggravate these neurodegenerative disorders via synergistic generation of toxic free radicals [12, 133, 151]. However, not all pathogenic characteristics of these diseases can be explained by toxic free radicals. For example, meta-analysis has indicated that copper is significantly reduced in the AD brain [129]. Furthermore, a study in Drosophila showed that ROS do not mediate the copper effects in HD [161]. In these scenarios, a decrease in copper ions available for cuproproteins such as DBH, PAM, CcO, NMDAR, and tyrosinase may contribute to the development of these neuropathies, like it does in MD-induced neurodegeneration [33, 47, 59, 71, 73, 74, 128, 153].
Pharmacological intervention for copper distribution can ameliorate the progression of neurodegenerative diseases. Lipophilic chelators like clioquinol and its derivative PBT2 can reduce amyloid burden and improve cognitive performance in AD animal models and patients [17, 52, 122, 130]. Data from us and others have demonstrated that clioquinol and its analogues function as metal-protein attenuating compounds (MPACs), thereby redistributing copper and other metal ions from enriched to depleted brain regions. This redistribution may, at least partially, account for the treatment effects of these MPACs in AD patients and animals [89, 102, 122, 130]. Moreover, copper redistribution or delivery with MPACs can also mitigate cognitive decline and improve memory or movement in mouse models of other neurodegenerative disorders, including ALS and PD [21, 28, 123]. These findings further support the assertion that misdistribution of copper plays crucial roles in the progression of neurodegenerative diseases.
Copper misdistribution also occurs in diabetes. On the one hand, hepatic and renal copper levels are increased in streptozotocin (STZ)-induced diabetes rat models [36]. Furthermore, copper chelation therapy, with drugs such as trientine, has been found to mitigate various pathogenic states of diabetes, suggesting a key role of excess copper in this disorder [14, 22, 23, 48]. However, the underlying mechanism remains to be determined. Furthermore, it was recently reported that metformin, a first-line drug for the treatment of type II diabetes, may act by chelating mitochondrial copper, resulting in alterations to cellular energy metabolism [40, 121]. On the other hand, CuPrs such as SOD3 may lack available Cu in diabetes: insulin deficiency or defects in the insulin/Akt2 pathway cause downregulation of ATP7A in the vessels in multiple diabetes mouse models, including STZ-induced mice; the downregulation of ATP7A impairs metallization/activation of SOD3 with copper at TGN, and in turn, enhances vascular O2− production, which induces endothelial dysfunction in diabetes [43, 140, 141]. These findings suggest that an imbalance in copper distribution contributes to diabetes.
Conclusions
Copper levels are tightly controlled through either cellular or systemic homeostasis. The mechanism of copper metabolism in mammals is shown in Fig. 1. First, the influx of extracellular copper ions is primarily mediated by the high-affinity transporter Ctr1. In enterocytes, further detailed understanding of copper influx is still needed, but it is widely accepted that Ctr1 in the apical membrane mediates absorption of most available cuprous ions from the intestinal lumen. After incorporation, most copper ions are sequestrated by MTs or stored within lysosomes, while only a small fraction of them are delivered to different apparatuses. The copper utilization pathways include the CCS-SOD1 route, the Atox1-ATP7A/B-CuPrs pathway, and the Cox17-Sco1/2-CcO route. Cells maintain copper homeostasis via regulation of genes involved in incorporation, detoxification, and utilization pathways. In mammalian enterocytes and hepatocytes, ATP7A and ATP7B, respectively, relocate to the basolateral and canalicular membrane to pump copper ions into the blood or bile. Copper ions pumped into the blood by basolateral ATP7A from enterocytes can be transported all over the body, while those released into the bile, mediated by ATP7B, are flushed out of the body. The importance of components of copper transportation has been verified in several mouse models (summarized in Table 1).
Copper deficiency can bring about various diseases (listed in Table 2). In MD, patients generally exhibit copper deficiency due to a loss of function of ATP7A. As a result, CuPrs involved in the production of neurotransmitters/neuropeptides, collagen/elastin, and ATP are impaired, leading to neurodegeneration, connective tissue abnormalities, and other pathologies. Mutation of SCO2 can lead to neonatal death due to defects in mitochondria. Copper deficiency caused by acquired factors can also bring about metabolic syndrome by disturbing lipolysis, and can lead to cardiovascular diseases by impairing free radical scavenging. Since copper is required for iron transportation, copper deficiency also leads to anemia.
On the other hand, excess copper is toxic and can also give rise to several disorders. Inactivation of ATP7B can cause WD, with copper accumulation in the liver and brain. Toxicity produced by excess copper in these organs leads to hepatitis, neurodegeneration, and hemolytic anemia. In cancer cells, copper levels are elevated to play a role in proliferation, angiogenesis, and metastasis via modulation of diverse cuproenzymes and signaling pathways. Unlike copper deficiency, excess copper promotes atherosclerosis and hypertension through multiple mechanisms, including by increasing ECM linking and arterial stiffness, neointimal thickening and vascular remodeling, and metabolism of LDL and cholesterol.
In neurodegenerative diseases and diabetes, copper misdistribution seems to occur, and this contributes to the progression of these disorders. Copper ions are sequestrated by several proteins, including Aβ, Tau, α-synuclein, Huntington, mutant SOD1, and Prion, and they tend to accumulate in several inclusion bodies. Binding to copper increases the potency of these proteins to generate ROS, which may play a key role in neurodegeneration. On the other hand, copper deficiency in other CuPrs or regions might exacerbate neuropathies. Although the total level of hepatic and renal copper is increased in animal models of diabetes, vascular SOD3 cannot obtain enough cuprous ions to scavenge free radicals, which aggravates this disease.
Thus, the available evidence indicates that copper is a key nutrient with two opposing functions. Mammals and humans have evolved delicate systems to maintain proper levels of copper ions, both at the cellular and systemic level. Both deficiency and excess of this metal can cause, or produce deterioration in, several kinds of disorders in humans. Copper supplementation or chelation has been widely adopted to treat diseases caused by copper dishomeostasis, especially MD and WD. The use of MPACs like clioquinol and PBT2 is also a promising therapeutic strategy for neurodegenerative diseases; these MPACs act to redistribute copper in the brain. Clarification of the roles and mechanisms of copper under normal physiological conditions and in pathological events will contribute to the identification of novel and effective methods for maintaining copper homeostasis, and these could act as potential therapies for copper-related diseases.
References
Acevedo K, Masaldan S, Opazo CM, Bush AI (2019) Redox active metals in neurodegenerative diseases. J Biol Inorg Chem 24:1141–1157
Aigner E, Strasser M, Haufe H, Sonnweber T, Hohla F, Stadlmayr A, Solioz M, Tilg H, Patsch W, Weiss G, Stickel F, Datz C (2010) A role for low hepatic copper concentrations in nonalcoholic fatty liver disease. Am J Gastroenterol 105:1978–1985
Angeletti B, Waldron KJ, Freeman KB, Bawagan H, Hussain I, Miller CC, Lau KF, Tennant ME, Dennison C, Robinson NJ, Dingwall C (2005) BACE1 cytoplasmic domain interacts with the copper chaperone for superoxide dismutase-1 and binds copper. J Biol Chem 280:17930–17937
Atwood CS, Scarpa RC, Huang X, Moir RD, Jones WD, Fairlie DP, Tanzi RE, Bush AI (2000) Characterization of copper interactions with alzheimer amyloid beta peptides: identification of an attomolar-affinity copper binding site on amyloid beta1-42. J Neurochem 75:1219–1233
Barry AN, Shinde U, Lutsenko S (2010) Structural organization of human Cu-transporting ATPases: learning from building blocks. J Biol Inorg Chem 15:47–59
Becuwe C, Dalle S, Ronger-Savle S, Skowron F, Balme B, Kanitakis J, Thomas L (2005) Elastosis perforans serpiginosa associated with pseudo-pseudoxanthoma elasticum during treatment of Wilson’s disease with penicillamine. Dermatology 210:60–63
Bhattacharjee A, Chakraborty K, Shukla A (2017) Cellular copper homeostasis: current concepts on its interplay with glutathione homeostasis and its implication in physiology and human diseases. Metallomics 9:1376–1388
Blaby-Haas CE, Merchant SS (2014) Lysosome-related organelles as mediators of metal homeostasis. J Biol Chem 289:28129–28136
Boullata J, Muthukumaran G, Piarulli A, Labarre J, Compher C (2017) Oral copper absorption in men with morbid obesity. J Trace Elem Med Biol 44:146–150
Brady DC, Crowe MS, Turski ML, Hobbs GA, Yao X, Chaikuad A, Knapp S, Xiao K, Campbell SL, Thiele DJ, Counter CM (2014) Copper is required for oncogenic BRAF signalling and tumorigenesis. Nature 509:492–496
Buiakova OI, Xu J, Lutsenko S, Zeitlin S, Das K, Das S, Ross BM, Mekios C, Scheinberg IH, Gilliam TC (1999) Null mutation of the murine ATP7B (Wilson disease) gene results in intracellular copper accumulation and late-onset hepatic nodular transformation. Hum Mol Genet 8:1665–1671
Bush AI, Curtain CC (2008) Twenty years of metallo-neurobiology: where to now? Eur Biophys J 37:241–245
Calvo J, Jung H, Meloni G (2017) Copper metallothioneins. IUBMB Life 69:236–245. https://doi.org/10.1002/iub.1618
Cameron NE, Cotter MA (1995) Neurovascular dysfunction in diabetic rats. Potential contribution of autoxidation and free radicals examined using transition metal chelating agents. J Clin Invest 96:1159–1163
Chambers A, Krewski D, Birkett N, Plunkett L, Hertzberg R, Danzeisen R, Aggett PJ, Starr TB, Baker S, Dourson M, Jones P, Keen CL, Meek B, Schoeny R, Slob W (2010) An exposure-response curve for copper excess and deficiency. J Toxicol Environ Health B Crit Rev 13:546–578
Chen GF, Sudhahar V, Youn SW, Das A, Cho J, Kamiya T, Urao N, McKinney RD, Surenkhuu B, Hamakubo T, Iwanari H, Li S, Christman JW, Shantikumar S, Angelini GD, Emanueli C, Ushio-Fukai M, Fukai T (2015) Copper transport protein antioxidant-1 promotes inflammatory neovascularization via chaperone and transcription factor function. Sci Rep 5:14780
Cherny RA, Atwood CS, Xilinas ME, Gray DN, Jones WD, McLean CA, Barnham KJ, Volitakis I, Fraser FW, Kim Y, Huang X, Goldstein LE, Moir RD, Lim JT, Beyreuther K, Zheng H, Tanzi RE, Masters CL, Bush AI (2001) Treatment with a copper-zinc chelator markedly and rapidly inhibits beta-amyloid accumulation in Alzheimer’s disease transgenic mice. Neuron 30:665–676
Cherukuri S, Tripoulas NA, Nurko S, Fox PL (2004) Anemia and impaired stress-induced erythropoiesis in aceruloplasminemic mice. Blood Cells Mol Dis 33:346–355
Choi EH, Strum W (2010) Hypocupremia-related myeloneuropathy following gastrojejunal bypass surgery. Ann Nutr Metab 57:190–192
Chojnacka M, Gornicka A, Oeljeklaus S, Warscheid B, Chacinska A (2015) Cox17 Protein is an auxiliary factor involved in the control of the mitochondrial contact site and cristae organizing system. J Biol Chem 290:15304–15312
Choo XY, Liddell JR, Huuskonen MT, Grubman A, Moujalled D, Roberts J, Kysenius K, Patten L, Quek H, Oikari LE, Duncan C, James SA, McInnes LE, Hayne DJ, Donnelly PS, Pollari E, Vahatalo S, Lejavova K, Kettunen MI, Malm T, Koistinaho J, White AR, Kanninen KM (2018) Cu(II)(atsm) attenuates neuroinflammation. Front Neurosci 12:668
Cooper GJ, Phillips AR, Choong SY, Leonard BL, Crossman DJ, Brunton DH, Saafi L, Dissanayake AM, Cowan BR, Young AA, Occleshaw CJ, Chan YK, Leahy FE, Keogh GF, Gamble GD, Allen GR, Pope AJ, Boyd PD, Poppitt SD, Borg TK, Doughty RN, Baker JR (2004) Regeneration of the heart in diabetes by selective copper chelation. Diabetes 53:2501–2508
Cooper GJ, Young AA, Gamble GD, Occleshaw CJ, Dissanayake AM, Cowan BR, Brunton DH, Baker JR, Phillips AR, Frampton CM, Poppitt SD, Doughty RN (2009) A copper(II)-selective chelator ameliorates left-ventricular hypertrophy in type 2 diabetic patients: a randomised placebo-controlled study. Diabetologia 52:715–722
Coronado V, Nanji M, Cox DW (2001) The Jackson toxic milk mouse as a model for copper loading. Mamm Genome 12:793–795
Cosimo QC, Daniela L, Elsa B, Carlo DV, Giuseppe F (2011) Kinky hair, kinky vessels, and bladder diverticula in Menkes disease. J Neuroimaging 21:e114–e116
Cousins RJ (1985) Absorption, transport, and hepatic metabolism of copper and zinc: special reference to metallothionein and ceruloplasmin. Physiol Rev 65:238–309
Cox DW, Moore SD (2002) Copper transporting P-type ATPases and human disease. J Bioenerg Biomembr 34:333–338
Crouch PJ, Hung LW, Adlard PA, Cortes M, Lal V, Filiz G, Perez KA, Nurjono M, Caragounis A, Du T, Laughton K, Volitakis I, Bush AI, Li QX, Masters CL, Cappai R, Cherny RA, Donnelly PS, White AR, Barnham KJ (2009) Increasing Cu bioavailability inhibits Abeta oligomers and tau phosphorylation. Proc Natl Acad Sci U S A 106:381–386
Czlonkowska A, Litwin T, Dusek P, Ferenci P, Lutsenko S, Medici V, Rybakowski JK, Weiss KH, Schilsky ML (2018) Wilson disease. Nat Rev Dis Primers 4:21
De Freitas J, Wintz H, Kim JH, Poynton H, Fox T, Vulpe C (2003) Yeast, a model organism for iron and copper metabolism studies. Biometals 16:185–197
Deutscher J, Kiess W, Scheerschmidt G, Willgerodt H (1999) Potential hepatotoxicity of penicillamine treatment in three patients with Wilson’s disease. J Pediatr Gastroenterol Nutr 29:628. https://doi.org/10.1097/00005176-199911000-00031
Eid C, Hemadi M, Ha-Duong NT, El Hage Chahine JM (2014) Iron uptake and transfer from ceruloplasmin to transferrin. Biochim Biophys Acta 1840:1771–1781
Eipper BA, Stoffers DA, Mains RE (1992) The biosynthesis of neuropeptides: peptide alpha-amidation. Annu Rev Neurosci 15:57–85
Eisses JF, Chi Y, Kaplan JH (2005) Stable plasma membrane levels of hCTR1 mediate cellular copper uptake. J Biol Chem 280:9635–9639
Engle TE (2011) Copper and lipid metabolism in beef cattle: a review. J Anim Sci 89:591–596
Failla ML, Kiser RA (1981) Altered tissue content and cytosol distribution of trace metals in experimental diabetes. J Nutr 111:1900–1909
Festa RA, Thiele DJ (2011) Copper: an essential metal in biology. Curr Biol 21:R877–R883
Finney L, Mandava S, Ursos L, Zhang W, Rodi D, Vogt S, Legnini D, Maser J, Ikpatt F, Olopade OI, Glesne D (2007) X-ray fluorescence microscopy reveals large-scale relocalization and extracellular translocation of cellular copper during angiogenesis. Proc Natl Acad Sci U S A 104:2247–2252
Fleming CR, Hodges RE, Hurley LS (1976) A prospective study of serum copper and zinc levels in patients receiving total parenteral nutrition. Am J Clin Nutr 29:70–77
Foretz M, Guigas B, Viollet B (2019) Understanding the glucoregulatory mechanisms of metformin in type 2 diabetes mellitus. Nat Rev Endocrinol 15:569–589
Fox JH, Kama JA, Lieberman G, Chopra R, Dorsey K, Chopra V, Volitakis I, Cherny RA, Bush AI, Hersch S (2007) Mechanisms of copper ion mediated Huntington’s disease progression. PLoS One 2:e334
Freedman JH, Ciriolo MR, Peisach J (1989) The role of glutathione in copper metabolism and toxicity. J Biol Chem 264:5598–5605
Fukai T, Ushio-Fukai M, Kaplan JH (2018) Copper transporters and copper chaperones: roles in cardiovascular physiology and disease. Am J Phys Cell Phys 315:C186–C201. https://doi.org/10.1152/ajpcell.00132.2018
Gacheru S, McGee C, Uriu-Hare JY, Kosonen T, Packman S, Tinker D, Krawetz SA, Reiser K, Keen CL, Rucker RB (1993) Expression and accumulation of lysyl oxidase, elastin, and type I procollagen in human Menkes and mottled mouse fibroblasts. Arch Biochem Biophys 301:325–329
Genoud S, Roberts BR, Gunn AP, Halliday GM, Lewis SJG, Ball HJ, Hare DJ, Double KL (2017) Subcellular compartmentalisation of copper, iron, manganese, and zinc in the Parkinson’s disease brain. Metallomics 9:1447–1455
Godwin SC, Shawker T, Chang B, Kaler SG (2006) Brachial artery aneurysms in Menkes disease. J Pediatr 149:412–415
Goldstein DS, Holmes CS, Kaler SG (2009) Relative efficiencies of plasma catechol levels and ratios for neonatal diagnosis of menkes disease. Neurochem Res 34:1464–1468
Gong D, Lu J, Chen X, Reddy S, Crossman DJ, Glyn-Jones S, Choong YS, Kennedy J, Barry B, Zhang S, Chan YK, Ruggiero K, Phillips AR, Cooper GJ (2008) A copper(II)-selective chelator ameliorates diabetes-evoked renal fibrosis and albuminuria, and suppresses pathogenic TGF-beta activation in the kidneys of rats used as a model of diabetes. Diabetologia 51:1741–1751
Graham SF, Nasaruddin MB, Carey M, Holscher C, McGuinness B, Kehoe PG, Love S, Passmore P, Elliott CT, Meharg AA, Green BD (2014) Age-associated changes of brain copper, iron, and zinc in Alzheimer’s disease and dementia with Lewy bodies. J Alzheimers Dis 42:1407–1413
Grimes A, Hearn CJ, Lockhart P, Newgreen DF, Mercer JF (1997) Molecular basis of the brindled mouse mutant (Mo(br)): a murine model of Menkes disease. Hum Mol Genet 6:1037–1042
Grochowski C, Blicharska E, Baj J, Mierzwinska A, Brzozowska K, Forma A, Maciejewski R (2019) Serum iron, magnesium, copper, and manganese levels in alcoholism: a systematic review. Molecules 24
Grossi C, Francese S, Casini A, Rosi MC, Luccarini I, Fiorentini A, Gabbiani C, Messori L, Moneti G, Casamenti F (2009) Clioquinol decreases amyloid-beta burden and reduces working memory impairment in a transgenic mouse model of Alzheimer’s disease. J Alzheimers Dis 17:423–440
Gualandi F, Sette E, Fortunato F, Bigoni S, De Grandis D, Scotton C, Selvatici R, Neri M, Incensi A, Liguori R, Storbeck M, Karakaya M, Simioni V, Squarzoni S, Timmerman V, Wirth B, Donadio V, Tugnoli V, Ferlini A (2019) Report of a novel ATP7A mutation causing distal motor neuropathy. Neuromuscul Disord 29:776–785
Gupta A, Lutsenko S (2009) Human copper transporters: mechanism, role in human diseases and therapeutic potential. Future Med Chem 1:1125–1142
Gupte A, Mumper RJ (2009) Elevated copper and oxidative stress in cancer cells as a target for cancer treatment. Cancer Treat Rev 35:32–46
Ha C, Ryu J, Park CB (2007) Metal ions differentially influence the aggregation and deposition of Alzheimer’s beta-amyloid on a solid template. Biochemistry 46:6118–6125
Haddad MR, Patel KD, Sullivan PH, Goldstein DS, Murphy KM, Centeno JA, Kaler SG (2014) Molecular and biochemical characterization of Mottled-dappled, an embryonic lethal Menkes disease mouse model. Mol Genet Metab 113:294–300
Hamza I, Faisst A, Prohaska J, Chen J, Gruss P, Gitlin JD (2001) The metallochaperone Atox1 plays a critical role in perinatal copper homeostasis. Proc Natl Acad Sci U S A 98:6848–6852
Hansel DE, May V, Eipper BA, Ronnett GV (2001) Pituitary adenylyl cyclase-activating peptides and alpha-amidation in olfactory neurogenesis and neuronal survival in vitro. J Neurosci 21:4625–4636
Harris ED (2004) A requirement for copper in angiogenesis. Nutr Rev 62:60–64. https://doi.org/10.1111/j.1753-4887.2004.tb00025.x
Harris ZL, Durley AP, Man TK, Gitlin JD (1999) Targeted gene disruption reveals an essential role for ceruloplasmin in cellular iron efflux. Proc Natl Acad Sci U S A 96:10812–10817
Hermann W (2019) Classification and differential diagnosis of Wilson’s disease. Ann Transl Med 7:S63
Hill GM, Brewer GJ, Prasad AS, Hydrick CR, Hartmann DE (1987) Treatment of Wilson’s disease with zinc. I. Oral zinc therapy regimens. Hepatology 7:522–528
Hodgkinson VL, Dale JM, Garcia ML, Weisman GA, Lee J, Gitlin JD, Petris MJ (2015) X-linked spinal muscular atrophy in mice caused by autonomous loss of ATP7A in the motor neuron. J Pathol 236:241–250
Huster D, Finegold MJ, Morgan CT, Burkhead JL, Nixon R, Vanderwerf SM, Gilliam CT, Lutsenko S (2006) Consequences of copper accumulation in the livers of the Atp7b-/- (Wilson disease gene) knockout mice. Am J Pathol 168:423–434
Ishida S, Andreux P, Poitry-Yamate C, Auwerx J, Hanahan D (2013) Bioavailable copper modulates oxidative phosphorylation and growth of tumors. Proc Natl Acad Sci U S A 110:19507–19512. https://doi.org/10.1073/pnas.1318431110
Itoh S, Kim HW, Nakagawa O, Ozumi K, Lessner SM, Aoki H, Akram K, McKinney RD, Ushio-Fukai M, Fukai T (2008) Novel role of antioxidant-1 (Atox1) as a copper-dependent transcription factor involved in cell proliferation. J Biol Chem 283:9157–9167
Jeremy JY, Shukla N, Angelini GD, Day A, Wan IY, Talpahewa SP, Ascione R (2002) Sustained increases of plasma homocysteine, copper, and serum ceruloplasmin after coronary artery bypass grafting. Ann Thorac Surg 74:1553–1557. https://doi.org/10.1016/s0003-4975(02)03807-9
Jiang Y, Reynolds C, Xiao C, Feng W, Zhou Z, Rodriguez W, Tyagi SC, Eaton JW, Saari JT, Kang YJ (2007) Dietary copper supplementation reverses hypertrophic cardiomyopathy induced by chronic pressure overload in mice. J Exp Med 204:657–666. https://doi.org/10.1084/jem.20061943
Juarez-Rebollar D, Rios C, Nava-Ruiz C, Mendez-Armenta M (2017) Metallothionein in brain disorders. Oxidative Med Cell Longev 2017:5828056
Kaler SG (1994) Menkes disease. Adv Pediatr Infect Dis 41:263–304
Kaler SG (1996) Menkes disease mutations and response to early copper histidine treatment. Nat Genet 13:21–22
Kaler SG (2011) ATP7A-related copper transport diseases-emerging concepts and future trends. Nat Rev Neurol 7:15–29
Kaler SG, Holmes CS, Goldstein DS, Tang J, Godwin SC, Donsante A, Liew CJ, Sato S, Patronas N (2008) Neonatal diagnosis and treatment of Menkes disease. N Engl J Med 358:605–614
Kamiya T, Takeuchi K, Fukudome S, Hara H, Adachi T (2018) Copper chaperone antioxidant-1, Atox-1, is involved in the induction of SOD3 in THP-1 cells. Biometals 31:61–68. https://doi.org/10.1007/s10534-017-0067-1
Kennerson ML, Nicholson GA, Kaler SG, Kowalski B, Mercer JF, Tang J, Llanos RM, Chu S, Takata RI, Speck-Martins CE, Baets J, Almeida-Souza L, Fischer D, Timmerman V, Taylor PE, Scherer SS, Ferguson TA, Bird TD, De Jonghe P, Feely SM, Shy ME, Garbern JY (2010) Missense mutations in the copper transporter gene ATP7A cause X-linked distal hereditary motor neuropathy. Am J Hum Genet 86:343–352
Kim BE, Turski ML, Nose Y, Casad M, Rockman HA, Thiele DJ (2010) Cardiac copper deficiency activates a systemic signaling mechanism that communicates with the copper acquisition and storage organs. Cell Metab 11:353–363
Klevay LM (2000) Cardiovascular disease from copper deficiency—a history. J Nutr 130:489S–492S. https://doi.org/10.1093/jn/130.2.489S
Krishnamoorthy L, Cotruvo JA Jr, Chan J, Kaluarachchi H, Muchenditsi A, Pendyala VS, Jia S, Aron AT, Ackerman CM, Wal MN, Guan T, Smaga LP, Farhi SL, New EJ, Lutsenko S, Chang CJ (2016) Copper regulates cyclic-AMP-dependent lipolysis. Nat Chem Biol 12:586–592
Kuo YM, Zhou B, Cosco D, Gitschier J (2001) The copper transporter CTR1 provides an essential function in mammalian embryonic development. Proc Natl Acad Sci U S A 98:6836–6841
La Fontaine S, Theophilos MB, Firth SD, Gould R, Parton RG, Mercer JF (2001) Effect of the toxic milk mutation (tx) on the function and intracellular localization of Wnd, the murine homologue of the Wilson copper ATPase. Hum Mol Genet 10:361–370
Lamb DJ, Avades TY, Ferns GA (2001) Biphasic modulation of atherosclerosis induced by graded dietary copper supplementation in the cholesterol-fed rabbit. Int J Exp Pathol 82:287–294. https://doi.org/10.1046/j.1365-2613.2001.00200.x
Lamb DJ, Mitchinson MJ, Leake DS (1995) Transition metal ions within human atherosclerotic lesions can catalyse the oxidation of low density lipoprotein by macrophages. FEBS Lett 374:12–16. https://doi.org/10.1016/0014-5793(95)01068-p
Landriscina M, Bagala C, Mandinova A, Soldi R, Micucci I, Bellum S, Prudovsky I, Maciag T (2001) Copper induces the assembly of a multiprotein aggregate implicated in the release of fibroblast growth factor 1 in response to stress. J Biol Chem 276:25549–25557. https://doi.org/10.1074/jbc.M102925200
Lee J, Prohaska JR, Thiele DJ (2001) Essential role for mammalian copper transporter Ctr1 in copper homeostasis and embryonic development. Proc Natl Acad Sci U S A 98:6842–6847
Levenson CW (1998) Mechanisms of copper conservation in organs. Am J Clin Nutr 67:978S–981S
Levenson CW, Janghorbani M (1994) Long-term measurement of organ copper turnover in rats by continuous feeding of a stable isotope. Anal Biochem 221:243–249
Levinson B, Packman S, Gitschier J (1997) Deletion of the promoter region in the Atp7a gene of the mottled dappled mouse. Nat Genet 16:224–225
Li C, Wang J, Zhou B (2010) The metal chelating and chaperoning effects of clioquinol: insights from yeast studies. J Alzheimers Dis 21:1249–1262
Linder MC (2016) Ceruloplasmin and other copper binding components of blood plasma and their functions: an update. Metallomics 8:887–905
Litwin T, Dziezyc K, Czlonkowska A (2019) Wilson disease-treatment perspectives. Ann Transl Med 7:S68
Lob HE, Marvar PJ, Guzik TJ, Sharma S, McCann LA, Weyand C, Gordon FJ, Harrison DG (2010) Induction of hypertension and peripheral inflammation by reduction of extracellular superoxide dismutase in the central nervous system. Hypertension 55:277–283, 276p following 283. https://doi.org/10.1161/HYPERTENSIONAHA.109.142646
Lob HE, Vinh A, Li L, Blinder Y, Offermanns S, Harrison DG (2011) Role of vascular extracellular superoxide dismutase in hypertension. Hypertension 58:232–239. https://doi.org/10.1161/HYPERTENSIONAHA.111.172718
Lowndes SA, Harris AL (2005) The role of copper in tumour angiogenesis. J Mammary Gland Biol Neoplasia 10:299–310. https://doi.org/10.1007/s10911-006-9003-7
Lu SC (2009) Regulation of glutathione synthesis. Mol Asp Med 30:42–59
Malinouski M, Hasan NM, Zhang Y, Seravalli J, Lin J, Avanesov A, Lutsenko S, Gladyshev VN (2014) Genome-wide RNAi ionomics screen reveals new genes and regulation of human trace element metabolism. Nat Commun 5:3301. https://doi.org/10.1038/ncomms4301
Mandinova A, Soldi R, Graziani I, Bagala C, Bellum S, Landriscina M, Tarantini F, Prudovsky I, Maciag T (2003) S100A13 mediates the copper-dependent stress-induced release of IL-1alpha from both human U937 and murine NIH 3 T3 cells. J Cell Sci 116:2687–2696. https://doi.org/10.1242/jcs.00471
Mansoor MA, Bergmark C, Haswell SJ, Savage IF, Evans PH, Berge RK, Svardal AM, Kristensen O (2000) Correlation between plasma total homocysteine and copper in patients with peripheral vascular disease. Clin Chem 46:385–391
Maryon EB, Molloy SA, Kaplan JH (2013) Cellular glutathione plays a key role in copper uptake mediated by human copper transporter 1. Am J Phys Cell Phys 304:C768–C779
Maselbas W, Czlonkowska A, Litwin T, Niewada M (2019) Persistence with treatment for Wilson disease: a retrospective study. BMC Neurol 19:278. https://doi.org/10.1186/s12883-019-1502-4
Matzuk MM, Dionne L, Guo Q, Kumar TR, Lebovitz RM (1998) Ovarian function in superoxide dismutase 1 and 2 knockout mice. Endocrinology 139:4008–4011
McInerney MP, Pan Y, Volitakis I, Bush AI, Short JL, Nicolazzo JA (2019) The effects of clioquinol on P-glycoprotein expression and biometal distribution in the mouse brain microvasculature. J Pharm Sci 108:2247–2255
Medici V, LaSalle JM (2019) Genetics and epigenetic factors of Wilson disease. Ann Transl Med 7:S58
Medici V, Trevisan CP, D’Inca R, Barollo M, Zancan L, Fagiuoli S, Martines D, Irato P, Sturniolo GC (2006) Diagnosis and management of Wilson’s disease: results of a single center experience. J Clin Gastroenterol 40:936–941
Mohr I, Weiss KH (2019) Biochemical markers for the diagnosis and monitoring of Wilson disease. Clin Biochem Rev 40:59–77
Molloy SA, Kaplan JH (2009) Copper-dependent recycling of hCTR1, the human high affinity copper transporter. J Biol Chem 284:29704–29713
Murakami H, Kodama H, Nemoto N (2002) Abnormality of vascular elastic fibers in the macular mouse and a patient with Menkes’ disease: ultrastructural and immunohistochemical study. Med Electron Microsc 35:24–30
Myint ZW, Oo TH, Thein KZ, Tun AM, Saeed H (2018) Copper deficiency anemia: review article. Ann Hematol 97:1527–1534
Nose Y, Kim BE, Thiele DJ (2006) Ctr1 drives intestinal copper absorption and is essential for growth, iron metabolism, and neonatal cardiac function. Cell Metab 4:235–244
Nose Y, Wood LK, Kim BE, Prohaska JR, Fry RS, Spears JW, Thiele DJ (2010) Ctr1 is an apical copper transporter in mammalian intestinal epithelial cells in vivo that is controlled at the level of protein stability. J Biol Chem 285:32385–32392
Ogata R, Chong PF, Maeda K, Imagi T, Nakamura R, Kawamura N, Kira R (2019) Long surviving classical Menkes disease treated with weekly intravenous copper therapy. J Trace Elem Med Biol 54:172–174
Ohgami RS, Campagna DR, McDonald A, Fleming MD (2006) The Steap proteins are metalloreductases. Blood 108:1388–1394
Ozumi K, Sudhahar V, Kim HW, Chen GF, Kohno T, Finney L, Vogt S, McKinney RD, Ushio-Fukai M, Fukai T (2012) Role of copper transport protein antioxidant 1 in angiotensin II-induced hypertension: a key regulator of extracellular superoxide dismutase. Hypertension 60:476–486
Papadopoulou LC, Sue CM, Davidson MM, Tanji K, Nishino I, Sadlock JE, Krishna S, Walker W, Selby J, Glerum DM, Coster RV, Lyon G, Scalais E, Lebel R, Kaplan P, Shanske S, De Vivo DC, Bonilla E, Hirano M, DiMauro S, Schon EA (1999) Fatal infantile cardioencephalomyopathy with COX deficiency and mutations in SCO2, a COX assembly gene. Nat Genet 23:333–337
Petris MJ, Smith K, Lee J, Thiele DJ (2003) Copper-stimulated endocytosis and degradation of the human copper transporter, hCtr1. J Biol Chem 278:9639–9646
Pierson H, Yang H, Lutsenko S (2019) Copper transport and disease: what can we learn from organoids? Annu Rev Nutr 39:75–94
Polishchuk EV, Concilli M, Iacobacci S, Chesi G, Pastore N, Piccolo P, Paladino S, Baldantoni D, van ISC, Chan J, Chang CJ, Amoresano A, Pane F, Pucci P, Tarallo A, Parenti G, Brunetti-Pierri N, Settembre C, Ballabio A, Polishchuk RS (2014) Wilson disease protein ATP7B utilizes lysosomal exocytosis to maintain copper homeostasis. Dev Cell 29:686–700
Reaume AG, Elliott JL, Hoffman EK, Kowall NW, Ferrante RJ, Siwek DF, Wilcox HM, Flood DG, Beal MF, Brown RH Jr, Scott RW, Snider WD (1996) Motor neurons in Cu/Zn superoxide dismutase-deficient mice develop normally but exhibit enhanced cell death after axonal injury. Nat Genet 13:43–47
Reed V, Boyd Y (1997) Mutation analysis provides additional proof that mottled is the mouse homologue of Menkes’ disease. Hum Mol Genet 6:417–423
Rembach A, Doecke JD, Roberts BR, Watt AD, Faux NG, Volitakis I, Pertile KK, Rumble RL, Trounson BO, Fowler CJ, Wilson W, Ellis KA, Martins RN, Rowe CC, Villemagne VL, Ames D, Masters CL, group Ar, Bush AI (2013) Longitudinal analysis of serum copper and ceruloplasmin in Alzheimer’s disease. J Alzheimers Dis 34:171–182
Repiscak P, Erhardt S, Rena G, Paterson MJ (2014) Biomolecular mode of action of metformin in relation to its copper binding properties. Biochemistry 53:787–795
Ritchie CW, Bush AI, Mackinnon A, Macfarlane S, Mastwyk M, MacGregor L, Kiers L, Cherny R, Li QX, Tammer A, Carrington D, Mavros C, Volitakis I, Xilinas M, Ames D, Davis S, Beyreuther K, Tanzi RE, Masters CL (2003) Metal-protein attenuation with iodochlorhydroxyquin (clioquinol) targeting Abeta amyloid deposition and toxicity in Alzheimer disease: a pilot phase 2 clinical trial. Arch Neurol 60:1685–1691
Roberts BR, Lim NK, McAllum EJ, Donnelly PS, Hare DJ, Doble PA, Turner BJ, Price KA, Lim SC, Paterson BM, Hickey JL, Rhoads TW, Williams JR, Kanninen KM, Hung LW, Liddell JR, Grubman A, Monty JF, Llanos RM, Kramer DR, Mercer JF, Bush AI, Masters CL, Duce JA, Li QX, Beckman JS, Barnham KJ, White AR, Crouch PJ (2014) Oral treatment with Cu(II)(atsm) increases mutant SOD1 in vivo but protects motor neurons and improves the phenotype of a transgenic mouse model of amyotrophic lateral sclerosis. J Neurosci 34:8021–8031
Roberts EA, Schilsky ML (2008) Diagnosis and treatment of Wilson disease: an update. Hepatology 47:2089–2111
Roos PM, Vesterberg O, Nordberg M (2006) Metals in motor neuron diseases. Exp Biol Med (Maywood) 231:1481–1487
Savir Y, Martynov A, Springer M (2017) Achieving global perfect homeostasis through transporter regulation. PLoS Comput Biol 13:e1005458. https://doi.org/10.1371/journal.pcbi.1005458
Schilsky ML, Blank RR, Czaja MJ, Zern MA, Scheinberg IH, Stockert RJ, Sternlieb I (1989) Hepatocellular copper toxicity and its attenuation by zinc. J Clin Invest 84:1562–1568
Schlief ML, West T, Craig AM, Holtzman DM, Gitlin JD (2006) Role of the Menkes copper-transporting ATPase in NMDA receptor-mediated neuronal toxicity. Proc Natl Acad Sci U S A 103:14919–14924
Schrag M, Mueller C, Oyoyo U, Smith MA, Kirsch WM (2011) Iron, zinc and copper in the Alzheimer’s disease brain: a quantitative meta-analysis. Some insight on the influence of citation bias on scientific opinion. Prog Neurobiol 94:296–306
Sedjahtera A, Gunawan L, Bray L, Hung LW, Parsons J, Okamura N, Villemagne VL, Yanai K, Liu XM, Chan J, Bush AI, Finkelstein DI, Barnham KJ, Cherny RA, Adlard PA (2018) Targeting metals rescues the phenotype in an animal model of tauopathy. Metallomics 10:1339–1347
Shawki A, Anthony SR, Nose Y, Engevik MA, Niespodzany EJ, Barrientos T, Ohrvik H, Worrell RT, Thiele DJ, Mackenzie B (2015) Intestinal DMT1 is critical for iron absorption in the mouse but is not required for the absorption of copper or manganese. Am J Physiol Gastrointest Liver Physiol 309:G635–G647
Shiono Y, Wakusawa S, Hayashi H, Takikawa T, Yano M, Okada T, Mabuchi H, Kono S, Miyajima H (2001) Iron accumulation in the liver of male patients with Wilson’s disease. Am J Gastroenterol 96:3147–3151
Simunkova M, Alwasel SH, Alhazza IM, Jomova K, Kollar V, Rusko M, Valko M (2019) Management of oxidative stress and other pathologies in Alzheimer’s disease. Arch Toxicol 93:2491–2513
Singleton WC, McInnes KT, Cater MA, Winnall WR, McKirdy R, Yu Y, Taylor PE, Ke BX, Richardson DR, Mercer JF, La Fontaine S (2010) Role of glutaredoxin1 and glutathione in regulating the activity of the copper-transporting P-type ATPases, ATP7A and ATP7B. J Biol Chem 285:27111–27121
Skopp A, Boyd SD, Ullrich MS, Liu L, Winkler DD (2019) Copper-zinc superoxide dismutase (Sod1) activation terminates interaction between its copper chaperone (Ccs) and the cytosolic metal-binding domain of the copper importer Ctr1. Biometals 32:695–705. https://doi.org/10.1007/s10534-019-00206-3
Song MO, Mattie MD, Lee CH, Freedman JH (2014) The role of Nrf1 and Nrf2 in the regulation of copper-responsive transcription. Exp Cell Res 322:39–50
Stankovic RK, Chung RS, Penkowa M (2007) Metallothioneins I and II: neuroprotective significance during CNS pathology. Int J Biochem Cell Biol 39:484–489
Starkebaum G, Harlan JM (1986) Endothelial cell injury due to copper-catalyzed hydrogen peroxide generation from homocysteine. J Clin Invest 77:1370–1376. https://doi.org/10.1172/JCI112442
Stremmel W, Merle U, Weiskirchen R (2019) Clinical features of Wilson disease. Ann Transl Med 7:S61
Sudhahar V, Okur MN, Bagi Z, O’Bryan JP, Hay N, Makino A, Patel VS, Phillips SA, Stepp D, Ushio-Fukai M, Fukai T (2018) Akt2 (Protein Kinase B Beta) stabilizes ATP7A, a copper transporter for extracellular superoxide dismutase, in vascular smooth muscle: novel mechanism to limit endothelial dysfunction in type 2 diabetes mellitus. Arterioscler Thromb Vasc Biol 38:529–541
Sudhahar V, Urao N, Oshikawa J, McKinney RD, Llanos RM, Mercer JF, Ushio-Fukai M, Fukai T (2013) Copper transporter ATP7A protects against endothelial dysfunction in type 1 diabetic mice by regulating extracellular superoxide dismutase. Diabetes 62:3839–3850
Svensson PA, Englund MC, Markstrom E, Ohlsson BG, Jernas M, Billig H, Torgerson JS, Wiklund O, Carlsson LM, Carlsson B (2003) Copper induces the expression of cholesterogenic genes in human macrophages. Atherosclerosis 169:71–76. https://doi.org/10.1016/s0021-9150(03)00145-x
Tanaka YK, Ogra Y (2019) Evaluation of copper metabolism in neonatal rats by speciation analysis using liquid chromatography hyphenated to ICP mass spectrometry. Metallomics 11:1679–1686
Tay SK, Shanske S, Kaplan P, DiMauro S (2004) Association of mutations in SCO2, a cytochrome c oxidase assembly gene, with early fetal lethality. Arch Neurol 61:950–952
Theophilos MB, Cox DW, Mercer JF (1996) The toxic milk mouse is a murine model of Wilson disease. Hum Mol Genet 5:1619–1624
Tsai CY, Finley JC, Ali SS, Patel HH, Howell SB (2012) Copper influx transporter 1 is required for FGF, PDGF and EGF-induced MAPK signaling. Biochem Pharmacol 84:1007–1013
Tumer Z (2013) An overview and update of ATP7A mutations leading to Menkes disease and occipital horn syndrome. Hum Mutat 34:417–429
Tumer Z, Horn N, Tonnesen T, Christodoulou J, Clarke JT, Sarkar B (1996) Early copper-histidine treatment for Menkes disease. Nat Genet 12:11–13
Turski ML, Brady DC, Kim HJ, Kim BE, Nose Y, Counter CM, Winge DR, Thiele DJ (2012) A novel role for copper in Ras/mitogen-activated protein kinase signaling. Mol Cell Biol 32:1284–1295
Urso E, Maffia M (2015) Behind the link between copper and angiogenesis: established mechanisms and an overview on the role of vascular copper transport systems. J Vasc Res 52:172–196. https://doi.org/10.1159/000438485
Valko M, Morris H, Cronin MT (2005) Metals, toxicity and oxidative stress. Curr Med Chem 12:1161–1208
Vanisova M, Burska D, Krizova J, Danhelovska T, Dosoudilova Z, Zeman J, Stiburek L, Hansikova H (2019) Stable COX17 Downregulation leads to alterations in mitochondrial ultrastructure, decreased copper content and impaired cytochrome c oxidase biogenesis in HEK293 cells. Folia Biol (Praha) 65:181–187
Waggoner DJ, Bartnikas TB, Gitlin JD (1999) The role of copper in neurodegenerative disease. Neurobiol Dis 6:221–230
Walshe JM (1973) Copper chelation in patients with Wilson’s disease. A comparison of penicillamine and triethylene tetramine dihydrochloride. Q J Med 42:441–452
Walshe JM (1989) Wilson’s disease presenting with features of hepatic dysfunction: a clinical analysis of eighty-seven patients. Q J Med 70:253–263
Watanabe S, Nagano S, Duce J, Kiaei M, Li QX, Tucker SM, Tiwari A, Brown RH Jr, Beal MF, Hayward LJ, Culotta VC, Yoshihara S, Sakoda S, Bush AI (2007) Increased affinity for copper mediated by cysteine 111 in forms of mutant superoxide dismutase 1 linked to amyotrophic lateral sclerosis. Free Radic Biol Med 42:1534–1542
Watts JC, Balachandran A, Westaway D (2006) The expanding universe of prion diseases. PLoS Pathog 2:e26
Wiernicka A, Janczyk W, Dadalski M, Avsar Y, Schmidt H, Socha P (2013) Gastrointestinal side effects in children with Wilson’s disease treated with zinc sulphate. World J Gastroenterol 19:4356–4362
Wong PC, Waggoner D, Subramaniam JR, Tessarollo L, Bartnikas TB, Culotta VC, Price DL, Rothstein J, Gitlin JD (2000) Copper chaperone for superoxide dismutase is essential to activate mammalian Cu/Zn superoxide dismutase. Proc Natl Acad Sci U S A 97:2886–2891
Wyman S, Simpson RJ, McKie AT, Sharp PA (2008) Dcytb (Cybrd1) functions as both a ferric and a cupric reductase in vitro. FEBS Lett 582:1901–1906
Xiao G, Fan Q, Wang X, Zhou B (2013) Huntington disease arises from a combinatory toxicity of polyglutamine and copper binding. Proc Natl Acad Sci U S A 110:14995–15000
Yarandi SS, Griffith DP, Sharma R, Mohan A, Zhao VM, Ziegler TR (2014) Optic neuropathy, myelopathy, anemia, and neutropenia caused by acquired copper deficiency after gastric bypass surgery. J Clin Gastroenterol 48:862–865
Yi L, Kaler S (2014) ATP7A trafficking and mechanisms underlying the distal motor neuropathy induced by mutations in ATP7A. Ann N Y Acad Sci 1314:49–54
Yi L, Kaler SG (2018) Interaction between the AAA ATPase p97/VCP and a concealed UBX domain in the copper transporter ATP7A is associated with motor neuron degeneration. J Biol Chem 293:7606–7617
Zhou X, Xiao X, Li XH, Qin HL, Pu XY, Chen DB, Wu C, Feng L, Liang XL (2020) A study of susceptibility-weighted imaging in patients with Wilson disease during the treatment of metal chelator. J Neurol
Zimnicka AM, Maryon EB, Kaplan JH (2007) Human copper transporter hCTR1 mediates basolateral uptake of copper into enterocytes: implications for copper homeostasis. J Biol Chem 282:26471–26480
Zimnicka AM, Tang H, Guo Q, Kuhr FK, Oh MJ, Wan J, Chen J, Smith KA, Fraidenburg DR, Choudhury MS, Levitan I, Machado RF, Kaplan JH, Yuan JX (2014) Upregulated copper transporters in hypoxia-induced pulmonary hypertension. PLoS One 9:e90544. https://doi.org/10.1371/journal.pone.0090544
Funding
This work was supported by National Natural Science Foundation of China (91749121 to Li), the Outstanding Young and Middle-aged Scientific and Technological Innovation Team Program of Colleges and Universities in Hubei Province (T201819 to Jiang), and the Fundamental Research Funds for the Central Universities (SCU2019D013).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, J., Jiang, Y., Shi, H. et al. The molecular mechanisms of copper metabolism and its roles in human diseases. Pflugers Arch - Eur J Physiol 472, 1415–1429 (2020). https://doi.org/10.1007/s00424-020-02412-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-020-02412-2