Abstract
Purpose
To describe the foveal avascular zone (FAZ) and vessel density (VD) in the superficial and deep capillary plexus in children with a history of prematurity on optical coherence tomography angiography (OCTA) and their correlation with gestational age (GA) and birth weight (BW).
Methods
We enrolled 81 preterm- and eight term-born children in this prospective observational study. The Optovue RTVue AVANTI (Optovue Inc., Fremont, CA) was used to procure the OCTA images. The 3 × 3 mm scan protocol centered on the fovea and the central 1 mm of the grid along with the FAZ of the superficial capillary plexus (SCP) and deep capillary plexus (DCP) was acquired.
Results
The mean SCP-VD was comparable between the preterms and term controls (p = 0.315) in the central fovea (1-mm grid). However, the SCP-VD of the 3-mm grid was lower in the preterms born without ROP, with type 1 ROP, and with type 2 ROP (47.61, 47.90, and 48.82 respectively) compared to that in the term group (51.38; p = 0.031). The FAZ in the SCP (p = 0.003) and DCP (p = 0.003) was significantly smaller in the preterms compared to that in the controls. Based on the GA sub-analysis, the FAZ was significantly smaller in the SCP and DCP of preterms born < 31 weeks and > 31 weeks GA (p < 0.000, p < 0.035, respectively). Based on the BW, the difference between the FAZ in the SCP (p = 0.002) and DCP (p = 0.003) was significant. There was no association between the visual acuity and FAZ.
Conclusion
Optical coherence tomography angiography findings in this study show an altered foveal morphology and vascularity in preterms with and without ROP.

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Optical coherence tomography angiography (OCTA) is a non-invasive, dye less, and depth-resolved technique to study the superficial and deep retinal capillary plexus as well as the choroidal circulation. It overcomes the invasive nature of fundus fluorescein angiography (FFA) and indocyanine green angiography (ICGA) by using motion contrast imaging. The automated and manual segmentation helps in analyzing the vasculature and pathology of that particular layer in greater detail [1]. On OCTA, the superficial capillary plexus (SCP) includes vasculature from the nerve fiber layer and ganglion cell layer, while the deep capillary plexus (DCP) includes both the intermediate and deep inner retinal vasculature [2].
In adults, OCTA has been extensively used to study the characteristics of retinal and choroidal circulation in various vascular diseases. In diabetic retinopathy, it can delineate retinal neovascularization, areas of capillary non-perfusion, and the foveal avascular zone (FAZ), besides being able to quantifying the flow index [3]. It is a useful non-invasive tool in age-related macular degeneration to monitor functional and morphologic characteristic and response to treatment [4]. In retinal vein occlusion, OCTA helps in determining the perfusion status of the macula and thus prognosticate [5].
Owing to its non-invasive procedure and shorter acquisition time, OCTA has contributed to our understanding of the retinal and choroidal vasculature in the pediatric population [6]. The main limitation is the requirement of a steady and precise fixation for several seconds to acquire good-quality images, which is a challenge in younger children. Our group reported the first use of OCTA in an infant wherein we non-invasively mapped the extent of neovascularization in a case of aggressive posterior retinopathy of prematurity and documented its regression after treatment [7]. There have been other reports in infants and children since then, evaluating the FAZ, foveal pit characteristics, and the degree of macular developmental arrest in preterm infants [8]. Retinopathy of prematurity (ROP), a leading cause of childhood blindness due to the abnormal development of retinal vessels, provides an opportunity to correlate vascular development with foveal topography and visual acuity in children. In the current study, we evaluate the differences in vascular features on OCTA in children with a history of preterm birth based on gestational age (GA) and the severity of ROP.
Methods
This cross-sectional, observational study was approved by the Institutional Ethics Committee and the Institute Research Board and adhered to the tenets of the Declaration of Helsinki. We obtained a written informed consent from the parents or a legal guardian. We conducted the study between January 2016 and December 2017 and enrolled Asian Indian children between the age of four and eight who were previously enrolled in the Karnataka Internet Assisted Diagnosis of Retinopathy of Prematurity (KIDROP) tele-ROP program [9,10,11]. We collected demographic information such as GA, birth weight (BW), and clinical history, including the corrected distance visual acuity (CDVA) from the KIDROP medical records. Age-matched controls with a history of full-term birth and a normal ocular examination were enrolled. All participants included in the study had a CDVA better than 20/30. Only one eye of each study child, either with better vision or better image quality, was included in the study. In participants with comparable vision and image quality, the study eye was selected randomly. Eyes with poor fixation due to either low vision or poor cooperation were excluded from the study. We also excluded participants who had hereditary retinochoroidal disease, media opacities that precluded imaging, vitreomacular interface disorders, or previous intraocular surgery.
All participants underwent a detailed anterior segment examination and dilated fundus evaluation with an indirect ophthalmoscope. The OCTA images were acquired with the participant in an upright position without any anesthesia using the RTVue XR Avanti AngioVue OCTA (Optovue Inc., Fermont, CA). The 3 × 3 mm scan protocol centered on the fovea was acquired for each study eye. Automated OCTA segmentation was done using the in-built AngioVue module. The segmentation of the SCP en face image was performed with an inner offset of 3 mm from the internal limiting membrane to the outer boundary set at 16 mm from the inner plexiform layer. The module segmented the DCP en face image from an inner boundary of 16 mm from the inner plexiform layer to an outer boundary of 69 mm from the inner plexiform layer. The grid-based vascular density (VD) of the SCP (SCP-VD) and DCP (DCP-VD) of the 3 × 3 mm and central 1 × 1 mm of the grid was evaluated using the automated quantification by the in-built RTVue XR Avanti AngioVue software (Fig. 1). The VD was quantified as a percentage. The FAZ was considered present if a distinct avascular zone was present without any vessel crossing the center. The FAZ was quantified both automatically by the machine using the flow measure software module and manually by an independent investigator.
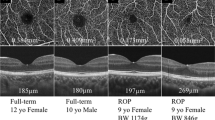
Optical coherence tomography angiography scans of controls and preterm children with historical type 1 and type 2 retinopathy of prematurity. The top panel depicts the superficial capillary plexus with the white star denoting the foveal avascular zone (FAZ) and the arrow indicating the central FAZ vessel crossing. The lower panel depicts the deep capillary plexus of the same eye
For statistical analysis, study groups were divided based on the history of prematurity and ROP: term as controls, cases as preterm with no ROP, preterm with type 1, and preterm with type 2 ROP. They were divided based on gestational age into GA < 31 weeks, GA > 31 weeks, and GA > 37 weeks (controls). There were three groups based on birth weight into BW < 1500 g, BW > 1500 (less than 2800 g), and BW > 2800 g (controls).
Statistical analysis
All analyses were performed using the IBM SPSS ver 25 (IBM Corp, Chicago, USA) and JMP Pro 13.1 (SAS, Cary, NC). The distribution of the data was assessed using the Shapiro-Wilk test. The mean difference between the groups with continuous variables was analyzed using one-way ANOVA, and post hoc analysis was performed using the Bonferroni correction, and p < 0.05 was considered significant. Pearson’s chi-squared test was used to analyze categorical variables. To test the agreement between the automatically quantified FAZ and the manual quantification, we used the inter-class correlation coefficient and Bland-Altman plots.
Results
We included 81 eyes from 81 preterm children and eight eyes from eight children born at term for this study. The demographic data for both cohorts are outlined in Table 1. The mean GA and BW were significantly different between the preterm and term age groups, as expected. There was no significant difference in the age at imaging between the preterm and term groups (6.8 ± 1.5 versus 7.8 ± 1.5; p = 0.10). There was a good agreement (ICC = 0.89, CI = 0.76-0.94) between the automated and manual measurements performed by the investigator (Fig. 2).
The preterm cohort included 48% without ROP, 26% with type 2 ROP, and 26% with type 1 ROP. There was no association between the visual acuity and SCP, DCP, and the FAZ. The mean SCP-VD was comparable between the preterms and controls (p = 0.315) in the central fovea (1-mm grid). However, in the 3-mm grid, the mean SCP-VD was lower in the preterm without ROP, preterm with type 1 ROP, and preterm with type 2 ROP (47.61, 47.90, and 48.82, respectively) compared to that in the term cohort (51.38; p = 0.031). A post hoc analysis showed that this difference was significant between preterm with no ROP and term infants (p = 0.03). The DCP-VD was higher in preterm without ROP, preterm with type 1 ROP, and preterm with type 2 ROP (33.39, 35.28, 37.41, respectively) compared to that in the term group (30.12; p = 0.045) in the central fovea (1-mm grid) but a post hoc analysis revealed no significant difference between the subgroups. The mean DCP-VD was comparable between the preterms and term cohorts (p = 0.534) in the 3-mm grid.
The FAZ was significantly smaller in the preterms compared to that in the controls at both SCP and DCP levels (p = 0.003; Table 2). Post hoc analysis showed that the difference between preterms with treated ROP and term children (p = 0.014) and preterms with untreated and term children (p = 0.002) at the SCP level was significant. Post hoc analysis at the DCP level showed that the difference between controls and preterms with no ROP (p = 0.035), preterms with untreated ROP (p = 0.001), and preterms with treated ROP (p = 0.031), respectively, was significant.
Correlation with the GA
The VD in the SCP (R2 = 0.1; p = 0.001) and DCP (R2 = 0.15; p = 0.002) was significantly higher in the 1-mm grid in preterms compared to that in those born at term (Table 3). For the 3 mm zone, it was significantly lower (p = 0.009) at the level of the SCP and comparable (p = 0.967) at the DCP. The FAZ was significantly smaller at the level of SCP and DCP in preterms, when compared to that in those born at term (p < 0.001 for both comparisons). Post hoc analysis of the preterms divided into GA < 31 weeks and GA > 31 weeks and compared to term (> 37 weeks) showed that the difference was consistent for SCP-VD at both 1 and 3 mm, DCP-VD at 1 mm, and FAZ at SCP and DCP.
Correlation with the BW
We noted a significant difference between the BW and FAZ at SCP (R2 = 0.10; p = 0.002) and DCP (R2 = 0.12; p = 0.003). We further analyzed the difference in the BW groups < 1500 g, > 1500 g, and > 2800 g and found a significant difference in SCP-VD at 3 mm and FAZ at SCP and DCP, as shown in Table 4. The SCP-VD at 3 mm was found to be significantly different among the BW groups < 1500 g, > 1500 g, and > 2800 g (48.1, 47.8, and 51.4, respectively; p = 0.001). The SCP-VD at 1 mm and DCP-VD at 3 mm and at 1 mm were found to be comparable among the different groups. We also found a significant difference between the BW and FAZ at the SCP (p = 0.002) and DCP (p = 0.003).
Discussion
Prematurity has been reported to affect the development of the individual retinal layers and vasculature. A non-invasive method to study foveal vasculature parameters, including the FAZ, SCP-VD, and DCP-VD, is the OCTA. Hitherto, it was unknown if the central 1 mm versus the 3 mm zone behave differently in preterm infants with no ROP, any stage ROP, or treated ROP. Superficial and deep capillary plexus in the 1 and 3 mm may demonstrate different characteristics since fovealization occurs postnatally in premature infants. It may, therefore, be intuitive to expect that the vascular insults of ROP or laser therapy or both could impact the vascular parameters in the foveal region. In the current study, we found that the VD was “less” in the 3 mm zone in the SCP of preterms without ROP (47.61) compared to that of term infants (51.38) and was “more” in the 1 mm zone of the DCP of preterms (with and without ROP) compared to that of term infants. The FAZ was “smaller” in preterms compared to that in term infants at both the SCP and DCP levels and was significantly smaller in those with ROP compared to that in term infants in the SCP. At the DCP level in all preterms and term infants, the FAZ was significantly smaller.
Balasubramanian et al. [12] in a cross-sectional study of 32 eyes of former preterm children observed that lower visual acuity was associated with a smaller FAZ and that preterm babies had a smaller FAZ compared to term controls. They reported that the VD at the SCP and DCP in the “central fovea,” i.e., central 1 mm of the grid, was increased in the preterm group compared to that in term controls. The variation, if any, between the VD of ROP treated and untreated eyes outside the central fovea was not analyzed. There is no agreement in literature on the foveal VD in preterms or its impact. Falavarjani et al. [13] reported “persistent foveal vasculature” in the central FAZ in three adults aged between 26 and 65 years who were born premature. Nonobe et al. [14] in ten patients who had undergone cryotherapy for ROP showed no difference in the VD in the central fovea (compared to controls), but significantly lower in the paracentral regions. However, their smaller number of cases did not allow subgroup analysis.
Our study aimed to compare the vascular characteristics in the central 1-mm and the paracentral 3-mm grids and investigate its correlation with the presence or absence of ROP. To study structural changes without its influence on visual acuity, our cases and controls were chosen to be comparable in visual acuity with both groups recording a CDVA of 20/30 or better. Our results point to an interesting observation that a smaller FAZ in preterms (with and without ROP), compared to terms, did not influence the visual acuity as the study cohort had a comparable and normal vision of 20/30 or better. Clinically they also had “normal” looking maculae. It is possible that the visual acuity is determined by multiple factors, only one of which may be the size of the FAZ. Our subgroup analysis of the GA showed a denser VD in the SCP and DCP in the central 1 mm zone in children who were born < 31 weeks of gestation but not in the 3 mm zone. Mean foveal VD was found to have a significant negative correlation with the GA in a study by Falavarjani et al. [15] In another study, the GA had a significant negative correlation with the deep foveal VD [16]. In both these studies, the GA of preterm children was between 24 and 31 weeks, and thus, analysis for preterm with GA more than 31 weeks is not available. Ganglion cell layer (GCL), inner nuclear layer, and inner plexiform layers (IPL) have been reported to be “thicker” in infants born between 30 and 32 weeks PMA compared to those between 33 and 36 weeks PMA on OCT [17]. The central 400 microns (but not beyond) have been reported to have thicker GCL and IPL in preterms compared to those in terms [18]. We postulate that, since the SCP is located in the GCL, whereas the DCP is in the INL, it could explain the difference between the SCP and DCP in children born < 31 weeks of GA that we found.
Conventional FFA in preterms has shown a small or absent FAZ [19]. It has variably been reported to be absent in 30% of preterm infants [13], 60% of the preterm infants who underwent laser therapy [15], and 10.4% of the preterms with ROP [20]. The ability to visualize deeper vascular layers on OCTA allows better visualization of the FAZ. We were able to delineate the FAZ in all our cases (type 1 and type 2 ROP). The mean size of the FAZ in our cohort was larger than that recorded in the Caucasian population [12, 15]. A possible reason is that larger and more mature infants are screened and treated in India compared to the West. We did not, however, find any difference in the size of FAZ between type 1 and type 2 ROP or preterms without ROP which is consistent with a Caucasian study [20].
Not much has been reported on the correlation between the SCP and DCP and the outcome of ROP. In 25 preterm children, Bowl et al. observed on OCTA that the foveal area was significantly reduced in both treated and spontaneously regressed ROP, but the foveal diameter was comparable [21]. They also observed a narrower superficial FAZ in eyes with treated and spontaneously regressed ROP, but the deep FAZ was comparable in all groups. While most studies have not analyzed FAZ at the DCP level [12, 15, 20, 22], we found that the FAZ at the level of the SCP and DCP was larger in type 1 ROP compared to that in type 2 ROP. While we are unsure at this time why this happens, we hypothesize that successful laser treatment (type 1 ROP cohort in our study) may, in fact, have a FAZ more normal akin to their term counterparts than those who had spontaneously regressed ROP (type 2) which causes remodeling. “Subclinical” SD-OCT findings [23] in the type 2 ROP foveae and its long-term effect on vision [24] suggest that even spontaneously regressed ROP can have central foveal changes that may influence structure and vision.
Studying the VD can help to non-invasively understand and monitor foveal development perinatally. Humans show foveal dip development between 30 and 32 weeks [17]. Henkind proposed from a work on the Rhesus monkey that the foveal center is fully vascularized and then remodels to form the capillary free zone of the FAZ [25]. In contrast, Engerman postulated that the capillaries arrest before reaching the foveal center in the Rhesus monkey [26]. Mintz-Hittner et al. suggested that the vascular meshwork at the FAZ undergoes remodeling by apoptosis and that preterm birth may alter these factors causing continued vascularization of the fovea [19]. Our GA subgroup analysis showed that children born at a lower GA (< 31) had a denser capillary plexus and a smaller FAZ than the older group born > 31 weeks or controls born > 37 weeks of age. Serial prospective OCTA imaging of GA cohorts will help to better understand the FAZ development in these children.
The exploratory, cross-sectional nature of this study has some limitations. We included children who had a visual acuity better than 20/30. Whereas this normal range of vision helps us study the influence of demographic factors and retinal layers with OCTA, it does not help correlate structure with visual acuity. Future research on correlation between vision and OCTA must consider systemic parameters like hypoglycemia, which can influence visual acuity. Other than visual acuity, one of the exciting future directions includes evaluating the association between functional outcomes such as contrast sensitivity threshold and anatomical and vascular differences at the fovea in preterm infants [27].
The study highlights the influence of ROP and GA on foveal vessel density, and the FAZ in preterm infants with and without ROP. As we improve our understanding of foveal maturation in preterm infants [20, 21, 24], the availability of hand-held OCTA in the future holds exciting prospects of helping us understand the effect of angiogenesis in prematurity, disease, and treatment of ROP.
Data availability
All data has been included in the manuscript.
References
Spaide RF, Klancnik JM Jr, Cooney MJ (2015) Retinal vascular layers imaged by fluorescein angiography and optical coherence tomography angiography. JAMA Ophthalmol 133(1):45–50. https://doi.org/10.1001/jamaophthalmol.2014.3616
Campbell JP, Zhang M, Hwang TS, Bailey ST, Wilson DJ, Jia Y, Huang D (2017) Detailed vascular anatomy of the human retina by projection-resolved optical coherence tomography angiography. Sci Rep;7(42201. DOI https://doi.org/10.1038/srep42201.
Dimitrova G, Chihara E, Takahashi H, Amano H, Okazaki K (2017) Quantitative retinal optical coherence tomography angiography in patients with diabetes without diabetic retinopathy. Invest Ophthalmol Vis Sci 58(1):190–196. https://doi.org/10.1167/iovs.16-20531
Sambhav K, Grover S, Chalam KV (2017) The application of optical coherence tomography angiography in retinal diseases. Surv Ophthalmol 62(6):838–866. https://doi.org/10.1016/j.survophthal.2017.05.006
Moussa M, Leila M, Bessa AS, Lolah M, Abou Shousha M, El Hennawi HM, Hafez TA (2019) Grading of macular perfusion in retinal vein occlusion using en-face swept-source optical coherence tomography angiography: a retrospective observational case series. BMC Ophthalmol;19(1):127. DOI https://doi.org/10.1186/s12886-019-1134-x.
Stanga PE, Papayannis A, Tsamis E, Chwiejczak K, Stringa F, Jalil A, Cole T, Biswas S (2016) Swept-source optical coherence tomography angiography of paediatric macular diseases. Dev Ophthalmol;56(166-173. DOI https://doi.org/10.1159/000442809.
Vinekar A, Chidambara L, Jayadev C, Sivakumar M, Webers CA, Shetty B (2016) Monitoring neovascularization in aggressive posterior retinopathy of prematurity using optical coherence tomography angiography. J aapos 20(3):271–274. https://doi.org/10.1016/j.jaapos.2016.01.013
Hsu ST, Chen X, Ngo HT, House RJ, Kelly MP, Enyedi LB, Materin MA, El-Dairi MA, Freedman SF, Toth CA, Vajzovic L (2019) Imaging infant retinal vasculature with OCT angiography. Ophthalmol Retina 3(1):95–96. https://doi.org/10.1016/j.oret.2018.06.017
Vinekar A, Jayadev C, Mangalesh S, Shetty B, Vidyasagar D (2015) Role of tele-medicine in retinopathy of prematurity screening in rural outreach centers in India - a report of 20,214 imaging sessions in the KIDROP program. Semin Fetal Neonatal Med 20(5):335–345. https://doi.org/10.1016/j.siny.2015.05.002
Quinn GE, Vinekar A (2019) The role of retinal photography and telemedicine in ROP screening. Semin Perinatol 43(6):367–374. https://doi.org/10.1053/j.semperi.2019.05.010
Vinekar A, Gilbert C, Dogra M, Kurian M, Shainesh G, Shetty B, Bauer N (2014) The KIDROP model of combining strategies for providing retinopathy of prematurity screening in underserved areas in India using wide-field imaging, tele-medicine, non-physician graders and smart phone reporting. Indian J Ophthalmol 62(1):41–49. https://doi.org/10.4103/0301-4738.126178
Balasubramanian S, Borrelli E, Lonngi M, Velez F, Sarraf D, Sadda SR, Tsui I (2019) Visual function and optical coherence tomography angiography features in children born preterm. Retina 39(11):2233–2239. https://doi.org/10.1097/iae.0000000000002301
Falavarjani KG, Sarraf D, Tsui I (2018) Optical coherence tomography angiography of the macula in adults with a history of preterm birth. Ophthalmic Surg Lasers Imaging Retina 49(2):122–125. https://doi.org/10.3928/23258160-20180129-06
Nonobe N, Kaneko H, Ito Y, Takayama K, Kataoka K, Tsunekawa T, Matsuura T, Suzumura A, Shimizu H, Terasaki H (2019) Optical coherence tomography angiography of the foveal avascular zone in children with a history of treatment-requiring retinopathy of prematurity. Retina 39(1):111–117. https://doi.org/10.1097/iae.0000000000001937
Falavarjani KG, Iafe NA, Velez FG, Schwartz SD, Sadda SR, Sarraf D, Tsui I (2017) Optical coherence tomography angiography of the fovea in children born preterm. Retina 37(12):2289–2294. https://doi.org/10.1097/iae.0000000000001471
Chen YC, Chen YT, Chen SN (2019) Foveal microvascular anomalies on optical coherence tomography angiography and the correlation with foveal thickness and visual acuity in retinopathy of prematurity. Graefes Arch Clin Exp Ophthalmol 257(1):23–30. https://doi.org/10.1007/s00417-018-4162-y
Vajzovic L, Hendrickson AE, O’Connell RV, Clark LA, Tran-Viet D, Possin D, Chiu SJ, Farsiu S, Toth CA (2012) Maturation of the human fovea: correlation of spectral-domain optical coherence tomography findings with histology. Am J Ophthalmol;154(5):779-789.e772. DOI https://doi.org/10.1016/j.ajo.2012.05.004.
Yanni SE, Wang J, Chan M, Carroll J, Farsiu S, Leffler JN, Spencer R, Birch EE (2012) Foveal avascular zone and foveal pit formation after preterm birth. Br J Ophthalmol 96(7):961–966. https://doi.org/10.1136/bjophthalmol-2012-301612
Mintz-Hittner HA, Knight-Nanan DM, Satriano DR, Kretzer FL (1999) A small foveal avascular zone may be an historic mark of prematurity. Ophthalmology 106(7):1409–1413. https://doi.org/10.1016/s0161-6420(99)00732-0
Takagi M, Maruko I, Yamaguchi A, Kakehashi M, Hasegawa T, Iida T (2019) Foveal abnormalities determined by optical coherence tomography angiography in children with history of retinopathy of prematurity. Eye (Lond) 33(12):1890–1896. https://doi.org/10.1038/s41433-019-0500-5
Bowl W, Bowl M, Schweinfurth S, Holve K, Knobloch R, Stieger K, Andrassi-Darida M, Lorenz B (2018) OCT angiography in young children with a history of retinopathy of prematurity. Ophthalmol Retina 2(9):972–978. https://doi.org/10.1016/j.oret.2018.02.004
Miki A, Yamada Y, Nakamura M (2019) The size of the foveal avascular zone is associated with foveal thickness and structure in premature children. J Ophthalmol;2019(8340729. DOI https://doi.org/10.1155/2019/8340729.
Vinekar A, Avadhani K, Sivakumar M, Mahendradas P, Kurian M, Braganza S, Shetty R, Shetty BK (2011) Understanding clinically undetected macular changes in early retinopathy of prematurity on spectral domain optical coherence tomography. Invest Ophthalmol Vis Sci 52(8):5183–5188. https://doi.org/10.1167/iovs.10-7155
Vinekar A, Mangalesh S, Jayadev C, Bauer N, Munusamy S, Kemmanu V, Kurian M, Mahendradas P, Avadhani K, Shetty B (2015) Macular edema in Asian Indian premature infants with retinopathy of prematurity: impact on visual acuity and refractive status after 1-year. Indian J Ophthalmol 63(5):432–437. https://doi.org/10.4103/0301-4738.159879
Henkind P, Bellhorn RW, Murphy ME, Roa N (1975) Development of macular vessels in monkey and cat. Br J Ophthalmol 59(12):703–709. https://doi.org/10.1136/bjo.59.12.703
Engerman RL (1976) Development of the macular circulation. Invest Ophthalmol 15(10):835–840
Thomas S, Thomas R, Vinekar A, Mangalesh S, Mochi TB, Munusamy S, Sarbajna P (2019) Shetty B; Evaluating contrast sensitivity in Asian Indian preterm infants with and without retinopathy of prematurity. Invest. Ophthalmol. Vis. Sci. 60(9):4753
Code availability
Not applicable.
Author information
Authors and Affiliations
Contributions
All authors contributed equally.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the Narayana Nethralaya Ethics Committee.
Consent to participate
All participants gave a written informed consent.
Consent for publication
All participants gave a written informed consent for publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vinekar, A., Sinha, S., Mangalesh, S. et al. Optical coherence tomography angiography in preterm-born children with retinopathy of prematurity. Graefes Arch Clin Exp Ophthalmol 259, 2131–2137 (2021). https://doi.org/10.1007/s00417-021-05090-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-021-05090-7