Abstract
Purpose
To evaluate the relationship between the parameters of pulse waveform analysis in the optic nerve head using laser speckle flowgraphy (LSFG) and intima-media thickness (IMT) and severity of carotid arterial plaque.
Design
Prospective and cross-sectional study.
Methods
One hundred seventy-six subjects were studied. Mean IMT and plaque score obtained from high-resolution B-mode ultrasound were evaluated. If mean IMT was 1 mm or greater, abnormal carotid artery thickening was diagnosed; if the plaque score exceeded 10, severe atherosclerosis was diagnosed. Blowout time and blowout score, which are parameters of pulse waveform analysis using LSFG, were evaluated. Logistic regression analyses determined the independent factors for abnormal carotid artery thickening and severe atherosclerosis. The cutoff levels for abnormal carotid artery thickening and severe atherosclerosis were analyzed using a conventional receiver operating characteristic (ROC) curve.
Results
Logistic regression analysis showed that blowout time and body mass index contributed independently to abnormal carotid artery thickening, and blowout score contributed independently to severe atherosclerosis. The ROC curve showed that sensitivity and specificity of the blowout time and blowout score were equivalent to or greater than those of other factors. The cutoff level of the blowout time for abnormal carotid artery thickening was 46.4; the cutoff level of the blowout score for severe atherosclerosis was 71.8.
Conclusions
Measurement of blowout time and blowout score in the optic nerve head by LSFG can be useful for evaluating mean IMT and plaque score, which are parameters of carotid atherosclerosis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The development of atherosclerosis is significantly associated with aging, and aging is a major risk factor for cardiovascular disease and stroke [1, 2]. High-resolution B-mode ultrasound is a useful method for examining the walls of the peripheral arteries, and provides measurements of intima-media thickness (IMT) and plaque that may be indicative of early presymptomatic disease. IMT and carotid plaque formation have been reported to increase with age [3]. They have been proposed as quantitative indexes of atherosclerosis, and have been shown to be positively associated with coronary heart disease and stroke [4–10].
Laser speckle flowgraphy (LSFG), a method for determining ocular blood flow, is based on changes in the speckle pattern of laser light reflected from the ocular fundus [11, 12]. LSFG is dependent on the movement of erythrocytes in the retina and choroid [13], and the mean blur rate is a measure of the relative velocity of the erythrocytes. Changes in the mean blur rate have various pulse wave patterns that are synchronized with the cardiac cycle. In previous studies, we focused on the relationship between the pulse waveform in the optic nerve head and systemic arteriosclerosis, and reported that blowout time, a factor derived from pulse wave analysis, was significantly correlated with age and IMT in healthy subjects [14, 15]. Nagaoka et al. reported that retinal circulatory parameters measured using a laser Doppler velocimetry system were significantly associated with IMT in patients with coronary artery disease [16]. To the best of our knowledge, the relationship between the severity of carotid arterial plaque and ocular microcirculation remains unknown.
The goals of the current study were to reevaluate the relationship between the parameters of pulse waveform analysis in the optic nerve head using LSFG and the IMT in patients with various background factors and determine if there is a significant correlation with the severity of carotid arterial plaque.
Methods
The institutional review board of Toho University Sakura Medical Center approved the current study, which was cross-sectional in nature, and all patients provided informed consent for participation, according to the tenets of the Declaration of Helsinki. We studied 236 consecutive patients who visited the Vascular Function Section of the Cardiovascular Department of Toho University Sakura Medical Center between April 1, 2008, and April 1, 2011. All evaluations were performed between 15:00 and 17:00 hours before a meal on the same day.
Exclusion criteria
Patients were excluded from the study if they had a history of arrhythmias, atrial fibrillation, glaucoma, uveitis, optic neuropathy, vitreous or retinal disease, retinal or choroidal vascular disease, or previous intraocular surgery. A total of 176 patients met the study criteria.
Measurement of carotid atherosclerosis
High-resolution ultrasonographic imaging of the carotid artery in B-scan mode was performed using the EUB-8500 ultrasound system (Hitachi Co., Ltd., Tokyo, Japan), with probe frequency set to 7.5 MHz. Imaging was performed with patients in the supine position with their heads turned slightly away from the sonographer. The procedure involved scanning the near and far walls of the carotid artery every 1 cm proximal to the carotid bulb in the longitudinal view. Mean IMT was defined as the average of the maximum IMT 1 cm proximal and 1 cm distal to the carotid bulb [4, 17]. The mean IMT of the thickened side of the carotid artery was used for data analyses. If the mean IMT was ≥ 1 mm, abnormal carotid artery thickening was diagnosed. The mean IMT of the right carotid artery was used for data analyses.
The severity of the carotid arterial plaque was calculated using a plaque score [10, 17]. Briefly, carotid arterial plaques, defined as localized increases in IMT ≥ 1.1 mm, were detected by cross-sectional and longitudinal scanning of the bilateral common and internal carotid arteries. The plaque score was computed by adding the maximum thickness of each plaque in the bilateral carotid arteries. If the plaque score exceeded 10, severe atherosclerosis was diagnosed [9].
Determination of optic nerve head circulation by pulse waveform analysis of LSFG
The determination of the LSFG-NAVI (Softcare Co., Ltd., Fukutsu, Japan) from these images has been described in detail [12, 13, 18]. Briefly, the LSFG is obtained from a 21° section centered on the optic disc. This observation field comprises 750 (width) × 360 (height) pixels. LSFG uses the mean blur rate as an indicator of the relative velocity of the erythrocytes, which is determined by the formula: 2 × (normalized blur rate) 2.
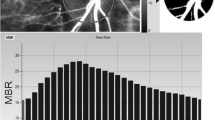
The normalized blur rate is calculated from the speckle pattern generated by the light reflected from the moving erythrocytes illuminated by an 830-nm wavelength diode laser beam [11, 13]. Initially, 118 mean blur rate images (118 frames) were recorded from the optic nerve head area over a period of 4 seconds. A grayscale map of the still images was then created by averaging the mean blur rate images (Fig. 1, top panel). On the analysis screen, the pulse wave of the changing mean blur rate corresponding to each cardiac cycle was obtained (Fig. 1, middle panel). Lastly, analysis of the screen normalized to one pulse was displayed (Fig. 1, bottom panel), and analysis of the pulse wave in the optic nerve head circulation was made on this screen. The maximum mean blur rate minus the minimum mean blur rate waves is labeled A (Fig. 1, bottom). The number of frames showing one-half the value of A is designated B, and the number of frames showing one cardiac cycle is labeled C. Finally, the average mean blur rate is labeled D.
Method for analyzing the pulse wave in the optic nerve head circulation using laser speckle flowgraphy. (Top) Grayscale map of the total measurement area. The circle designates the area of the optic disc measured. (Middle) Pulse waves show changes in the mean blur rate, which is tuned to the cardiac cycle for 4 s. Total number of frames is 118. (Bottom) Normalization of one pulse. A = maximum mean blur rate − minimum mean blur rate; B = number of frames spent at one-half the value of A; C = number of frames spent at one normalized pulse; D = average mean blur rate; T temporal, N nasal, S superior, I inferior
Analysis of the pulse wave in the optic nerve head circulation was determined by the following formulas: blowout time = 100 × (B)/(C) [14, 15] and blowout score = (2 – (A)/ (D))/2 × 100 [19].
We measured blowout time and blowout score of the optic nerve head area three times and used the average for statistical analyses. Only the data from the right eye were used for analysis. Blowout time and blowout score were calculated using LSFG software (LSFG Analyzer, version 3.1.45; Softcare Co., Ltd.). LSFG was performed after patients had rested for 10 min in a quiet room maintained at 24 °C. All subjects abstained from smoking, alcohol, and caffeine for at least 24 h before the measurements. All patients were measured in seated position, and pupils were dilated with 0.5 % tropicamide eye drops.
Laboratory measurements and systemic parameters
The following values were measured: fasting blood sugar (FBS: mg/dl), glycosylated hemoglobin (HbA1c: %), total cholesterol (mg/dl), triglycerides (mg/dl), low-density lipoprotein cholesterol (LDL-C: mg/dl), high-density lipoprotein cholesterol (HDL-C: mg/dl), high-sensitivity C-reactive protein (mg/l), cystatin C (mg/l) obtained from fasting morning blood samples, and urinary albumin excretion (mg/l). HbA1c measurements were expressed based on the National Glycohemoglobin Standardization Program scale.
Body mass index (BMI: kg/m2), pulse pressure (mmHg), and mean arterial blood pressure (MABP: mmHg) were evaluated. MABP was determined by the formula: diastolic blood pressure + (systolic blood pressure - diastolic blood pressure)/3.
Ocular parameters
Ocular perfusion pressure (mmHg) was measured by applanation tonometry and defined as (2/3 MABP) – intraocular pressure.
Statistical analyses
Data are presented as means ± standard deviations of the continuous variables. Pearson correlation coefficients were used to determine the relationships between mean IMT, plaque score, blowout time, blowout score, and other parameters. Logistic regression analyses were used to determine independent factors for mean IMT ≥ 1 mm and plaque score > 10. Sensitivity and specificity with respect to the presence of mean IMT ≥ 1 mm and plaque score > 10 were analyzed using a conventional receiver operating characteristic (ROC) curve. A value of P < 0.05 was considered statistically significant. Statistical analyses were performed using the SPSS for Windows statistical software program, version 11.01.1 (SPSS Inc., Chicago, IL, USA).
Results
Table 1 shows patient characteristics (mean age, 61 ± 11 years) and the results for optic nerve head pulse waveform parameters, blowout time and blowout score, mean IMT, and plaque score. Mean IMT and plaque score were 0.89 ± 0.21 (mm) and 4.8 ± 4.7, respectively. Thirty-one percent of patients had mean IMT of ≥ 1 mm, and 13 % had a plaque score > 10. Blowout time and blowout score were 48.2 ± 3.9 and 75.2 ± 6.7, respectively.
The results of Pearson correlation analysis of factors contributing to values of mean IMT and plaque score are shown in Table 2. Mean IMT was significantly correlated with BMI (r = 0.15, P = 0.04), pulse pressure (r = 0.43, P < 0.0001), MABP (r = 0.15, P = 0.04), cystatin C (r = 0.23, P = 0.002), blowout time (r = −0.42, P < 0.0001), and blowout score (r = −0.34, P < 0.0001). Plaque score was significantly correlated with pulse pressure (r = 0.36, P < 0.0001), FBS (r = 0.17, P = 0.03), blowout time (r = −0.34, P < 0.0001), and blowout score (r = −0.39, P < 0.0001).
The results of logistic regression analyses of factors independently contributing to mean IMT of ≥ 1 mm are shown in Table 3. Explanatory variables were blowout time, BMI, pulse pressure, FBS, MABP, and cystatin C, which were significantly correlated with or tended to correlate with mean IMT. Blowout time (odds ratio [OR] 0.78, 95 % confidence interval [CI] 0.69–0.89; P = 0.0001) and BMI (OR 1.13, 95 % CI 1.03–1.25; P = 0.01) independently contributed to mean IMT of ≥ 1 mm.
The results of logistic regression analyses of factors independently contributing to a plaque scores > 10 are also shown in Table 3. The explanatory variables were the blowout score, HbA1c, FBS, pulse pressure, urinary albumin excretion, and cystatin C which were correlated significantly with or tended to be correlated with the plaque score. The blowout score independently contributed to a plaque score > 10 (OR 0.86, 95 % CI 0.79–0.94; P = 0.0009).
ROC curves of the various factors were examined to determine the probability of patients having mean IMT of ≥ 1 mm based on blowout time × −1, pulse pressure, cystatin C, BMI, FBS, and MABP, and for determining the probability of patients having a plaque score > 10 based on a blowout score × −1, pulse pressure, cystatin C, urinary albumin excretion, FBS, and HbA1c (Fig. 2). The ROC curves show the fraction of true-positive results (sensitivity) and false-positive results (1-sensitivity) for various cutoff levels of each parameter. The cutoff level for the blowout time providing maximum sensitivity and specificity for a mean IMT ≥1 mm was 46.4. At the cutoff level, the sensitivity of the blowout time was 62 % and specificity was 74 %. The cutoff level of the blowout score providing maximum sensitivity and specificity for a plaque score > 10 was 71.8. At the cutoff level, the sensitivity of the blowout score was 78 % and specificity was 76 % (Table 4).
(Left) Receiver operating characteristic (ROC) curve for discriminating the probability of abnormal carotid artery thickening with a mean intima-media thickness ≥ 1 mm based on blowout time, pulse pressure, cystatin C, body mass index, fasting blood glucose, or mean arterial blood pressure. (Right) ROC curve for discriminating the probability of severe atherosclerosis with a plaque score > 10 based on blowout score, pulse pressure, cystatin C, urinary albumin excretion, fasting blood glucose, or glycated hemoglobin A1c (HbA1c). Curves show the fraction of true-positive results (sensitivity) and false-positive results (1-specificity) for various cutoff levels of each parameter
Discussion
Evaluation of the carotid artery using high-resolution B-mode ultrasound is effective in detecting carotid atherosclerosis and the presence of other atherosclerotic locations [20–22], and an increase in mean IMT is associated with primary cardiovascular risk factors [23–31]. The Atherosclerosis Risk in Communities (ARIC) study reported hazard rate ratios for stroke of 8.5 for women and 3.6 for men when comparing extreme intima-media thickness values (≥1 mm) to values less than 0.6 mm [7]. In addition, the incidence of stroke was 19 % in patients with severe atherosclerosis with plaque scores > 10 after an average 16 months of follow-up [9].
In our previous work, which focused on the relationship between pulse waveform analysis of the optic nerve head circulation using LSFG and risk factors for coronary artery disease and stroke, we found that blowout time may correlate with mean IMT, large-artery stiffness, chronic kidney disease, and age [14, 15, 32]. Thus we hypothesized that pulse waveform analysis of optic nerve head circulation would be effective for the detection of risk factors for coronary artery disease and stroke. The goals of the current study were to reevaluate the relationship between pulse waveform analysis of the optic nerve head and IMT in patients with various background factors by increasing the number of factors evaluated in a larger patient sample in order to determine whether there were significant correlations among blowout time, blowout score, and plaque score. Using the ROC curve, we also investigated the cutoff level for abnormal carotid artery thickening and severe atherosclerosis, which increase the risk for coronary artery disease and stroke.
Pearson correlation analysis showed that mean IMT was significantly correlated with BMI, pulse pressure, MABP, cystatin C, blowout time, and blowout score. The results of logistic regression analysis revealed that blowout time and BMI were independent risk factors for abnormal carotid artery thickening. Although obesity is recognized as a major risk factor for carotid artery thickening [33], blowout time was found to be the strongest contributor to abnormal carotid artery thickening (Tables 2 and 3). Pearson correlation analysis showed that plaque score was significantly correlated with pulse pressure, FBS, blowout time, and blowout score. Logistic regression analysis demonstrated that blowout score was the only independent factor contributing to severe atherosclerosis (Tables 2 and 3). Our results confirm that blowout time and blowout score, which are parameters of pulse waveform analysis of the optic nerve head, may reflect the status of carotid atherosclerosis and can detect abnormalities.
ROC analysis of abnormal carotid artery thickening revealed that the area under the ROC of the blowout time was equivalent to or greater than other contributing factors associated with mean IMT. A cutoff level of 46.4 for blowout time yielded maximum sensitivity and specificity for abnormal carotid artery thickening. Similarly, ROC analysis of severe atherosclerosis showed that the area under the ROC of the blowout score was equivalent to or greater than other factors contributing to the plaque score. The cutoff level of the blowout score was 71.8 (Fig. 2, Table 4). These results suggest that blowout time and blowout score obtained from LSFG could be additional tools for discriminating patients with a high risk of coronary artery disease and stroke.
LSFG is a simpler method than high-resolution B-mode ultrasound for measuring carotid artery atherosclerosis, as it is non-invasive, with a measurement time of only 4 s [11, 12, 18]. In addition, previous research found that the coefficient of variation of measurement in the optic nerve head area using LSFG was less than 3 % [34], and thus measurement of optic nerve head circulation using LSFG has shown favorable reproducibility. These observations suggest that measurement of ocular circulation with LSFG enables early detection of risk factors for coronary artery disease and stroke.
The current study had certain limitations. First, the entire optic nerve head area was measured, as it had the highest correlation with carotid atherosclerotic parameters in preliminary investigations (data not shown). However, as it has been suggested that blowout time and blowout score of the optic nerve head reflect arterial blood flow and venous blood flow, a more in-depth study is needed to separately evaluate arterial and venous blood flow. Second, atorvastatin calcium (Lipitor; Pfizer Inc., New York, NY, USA), the mainstay treatment for hyperlipidemia, decreased the IMT compared with baseline [35]. Because the design of the current study was cross-sectional, we did not investigate whether blowout time and blowout score changed simultaneously with the changes in mean IMT and plaque score. A prospective study should evaluate whether blowout time and blowout score can detect changes in mean IMT and plaque score. Finally, the current study included a small number of patients. A multi-center study is needed for evaluation of a larger number of patients of various backgrounds.
In conclusion, our results show that measurement of blowout time and blowout score in the optic nerve head by LSFG can be useful for evaluating risk factors for coronary artery disease and stroke, including mean IMT and plaque score, which are parameters of carotid atherosclerosis.
Change history
26 October 2021
A Correction to this paper has been published: https://doi.org/10.1007/s00417-021-05458-9
References
Pinto E (2007) Blood pressure and ageing. Postgrad Med J 83:109–114
Kubo M, Kiyohara Y, Kato I, Tanizaki Y, Arima H, Tanaka K, Nakamura H, Okubo K, Iida M (2003) Trends in the incidence, mortality, and survival rate of cardiovascular disease in a Japanese community: the Hisayama study. Stroke 34:2349–2354
Sun Y, Lin CH, Lu CJ, Yip PK, Chen RC (2002) Carotid atherosclerosis, intima media thickness and risk factors-an analysis of 1781 asymptomatic subjects in Taiwan. Atherosclerosis 164:89–94
O’Leary DH, Polak JF, Kronmal RA, Manolio TA, Burke GL, Wolfson SK Jr (1999) Carotid artery intima and media thickness as a risk factor for myocardial infarction and stroke in older adults. N Engl J Med 340:14–22
Hillen T, Nieczaj R, Munzberg H, Schaub R, Borchelt M, Steinhagen-Thiessen E (2000) Carotid atherosclerosis, vascular risk profile and mortality in a population-based sample of functionally healthy elderly subjects: the Berlin ageing study. J Intern Med 247:679–688
Touboul PJ, Elbaz A, Koller C, Lucas C, Adraï V, Chédru F, Amarenco P (2000) Common carotid artery intima-media thickness and brain infarction. GENIC case–control study. Circulation 102:313–318
Chambless LE, Folsom AR, Clegg LX, Sharrett AR, Shahar E, Nieto FJ, Rosamond WD, Evans G (2000) Carotid wall thickness is predictive of incident clinical stroke. The Atherosclerosis Risk in Communities (ARIC) study. Am J Epidemiol 151:478–487
Sakaguchi M, Kitagawa K, Nagai Y, Yamagami H, Kondo K, Matsushita K, Oku N, Hougaku H, Ohtsuki T, Masuyama T, Matsumoto M, Hori M (2003) Equivalence of plaque score and intima-media thickness of carotid ultrasonography for predicting severe coronary artery lesion. Ultrasound Med Biol 29:367–371
Handa N, Matsumoto M, Maeda H, Hougaku H, Kamada T (1995) Ischemic stroke events and carotid atherosclerosis. Results of the Osaka Follow-up Study for Ultrasonographic Assessment of Carotid Atherosclerosis (the OSACA Study). Stroke 26:1781–1786
Hougaku H, Matsumoto M, Handa N, Maeda H, Itoh T, Tsukamoto Y, Kamada T (1994) Asymptomatic carotid lesions and silent cerebral infarction. Stroke 25:566–570
Tamaki Y, Araie M, Kawamoto E, Eguchi S, Fujii H (1995) Non-contact, two-dimensional measurement of tissue circulation in choroid and optic nerve head using laser speckle phenomenon. Exp Eye Res 60:373–383
Isono H, Kishi S, Kimura Y, Hagiwara N, Konishi N, Fujii H (2003) Observation of choroidal circulation using index of erythrocytic velocity. Arch Ophthalmol 121:225–231
Fujii H (1994) Visualisation of retinal blood flow by laser speckle flow-graphy. Med Biol Eng Comput 32:302–304
Shiba T, Takahashi M, Hori Y, Maeno T (2012) Pulse-wave analysis of optic nerve head circulation is significantly correlated with brachial–ankle pulse-wave velocity, carotid intima–media thickness, and age. Graefes Arch Clin Exp Ophthalmol 250:1275–1281
Shiba T, Takahashi M, Hori Y, Maeno T, Shirai K (2012) Optic nerve head circulation determined by pulse wave analysis is significantly correlated with cardio ankle vascular index, left ventricular diastolic function, and age. J Atheroscler Thromb 19:999–1005
Nagaoka T, Ishii Y, Takeuchi T, Takahashi A, Sato E, Yoshida A (2005) Relationship between the parameters of retinal circulation measured by laser Doppler velocimetry and a marker of early systemic atherosclerosis. Invest Ophthalmol Vis Sci 46:720–725
Takiuchi S, Kamide K, Miwa Y, Tomiyama M, Yoshii M, Matayoshi T, Horio T, Kawano Y (2004) Diagnostic value of carotid intima-media thickness and plaque score for predicting target organ damage in patients with essential hypertension. J Hum Hypertens 18:17–23
Sugiyama T, Araie M, Riva CE, Schmetterer L, Orgul S (2010) Use of laser speckle flowgraphy in ocular blood flow research. Acta Ophthalmol 88:723–729
Tsuda S, Kunikata H, Shimura M, Aizawa N, Omodaka K, Shiga Y, Yasuda M, Yokoyama Y, Nakazawa T (2014) Pulse-waveform analysis of normal population using laser speckle flowgraphy. Curr Eye Res 39:1207–1215
O’Leary DH, Polak JF, Kronmal RA, Kittner SJ, Bond MG, Wolfson SK Jr, Bommer W, Price TR, Gardin JM, Savage PJ (1992) Distribution and correlates of sonographically detected carotid artery disease in the Cardiovascular Health Study. Stroke 23:1752–1760
Bots ML, Witteman JC, Grobbee DE (1993) Carotid intima-media thickness in elderly women with and without atherosclerosis of the abdominal aorta. Atherosclerosis 102:99–105
Gariepy J, Simon A, Massonneau M, Linhart A, Levenson J (1995) Wall thickening of carotid and femoral arteries in male subjects with isolated hypercholesterolemia. Atherosclerosis 113:141–151
Heiss G, Sharrett AR, Barnes R, Chambless LE, Szklo M, Alzola C (1991) Carotid atherosclerosis measured by B-mode ultrasound in populations: associations with cardiovascular risk factors in the ARIC study. Am J Epidemiol 134:250–256
Salonen R, Salonen JT (1991) Determinants of carotid intima-media thickness: a population-based ultrasonography study in Eastern Finnish men. J Intern Med 229:225–231
Bots ML, Breslau PJ, Briët E, de Bruyn AM, van Vliet HH, van den Ouweland FA, de Jong PT, Hofman A, Grobbee DE (1992) Cardiovascular determinants of carotid artery disease: the Rotterdam elderly study. Hypertension 19:717–720
Willeit J, Kiechl S (1993) Prevalence and risk factors of asymptomatic extracranial carotid artery atherosclerosis: a population-based study. Arterioscler Thromb 13:661–668
Bonithon-Kopp C, Touboul PJ, Berr C, Leroux C, Mainard F, Courbon D, Ducimetière P (1996) Relation of intima-media thickness to atherosclerotic plaques in the carotid arteries: the EVA study. Arterioscler Thromb Vasc Biol 16:310–316
Lassila HC, Tyrrell KS, Matthews KA, Wolfson SK, Kuller LH (1997) Prevalence and determinants of carotid atherosclerosis in healthy postmenopausal women. Stroke 28:513–517
Wendelhag I, Wiklund O, Wikstrand J (1992) Arterial wall thickness in familial hypercholesterolemia: ultrasound measurement of intima-media thickness in the common carotid artery. Arterioscler Thromb 12:70–77
Zureik M, Touboul PJ, Bonithon-Kopp C, Courbon D, Berr C, Leroux C, Ducimetière P (1999) Cross-sectional and longitudinal associations between brachial pulse pressure and common carotid intima-media thickness in a general population: the EVA study. Stroke 30:550–555
Gnasso A, Irace C, Mattioli PL, Pujia A (1996) Carotid intima-media thickness and coronary heart disease risk factors. Atherosclerosis 119:7–15
Shiba T, Takahashi M, Maeno T (2014) Pulse-wave analysis of optic nerve head circulation is significantly correlated with kidney function in patients with and without chronic kidney disease. J Ophthalmol 2014:291687
Yamasaki Y, Kawamori R, Matsushima H, Nishizawa H, Kodama M, Kajimoto Y, Morishima T, Kamada T (1994) Atherosclerosis in carotid artery of young IDDM patients monitored by ultrasound high-resolution B-mode imaging. Diabetes 43:634–639
Aizawa N, Yokoyama Y, Chiba N, Omodaka K, Yasuda M, Otomo T, Nakamura M, Fuse N, Nakazawa T (2011) Reproducibility of retinal circulation measurements obtained using laser speckle flowgraphy-NAVI in patients with glaucoma. Clin Ophthalmol 5:1171–1176
Smilde TJ, van Wissen S, Wollersheim H, Trip MD, Kastelein JJ, Stalenhoef AF (2001) Effect of aggressive versus conventional lipid lowering on atherosclerosis progression in familial hypercholesterolaemia (ASAP): a prospective, randomized, double-blind trial. Lancet 357:577–581
Acknowledgments
The authors did not receive funds, sponsorship, or other support for this study. There are no conflicts of interest to report. No statistical consultation or assistance was used.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rina, M., Shiba, T., Takahashi, M. et al. Pulse waveform analysis of optic nerve head circulation for predicting carotid atherosclerotic changes. Graefes Arch Clin Exp Ophthalmol 253, 2285–2291 (2015). https://doi.org/10.1007/s00417-015-3123-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-015-3123-y