Abstract
Acute unilateral lesions of vestibular graviceptive pathways from the otolith organs and semicircular canals via vestibular nuclei and the thalamus to the parieto-insular vestibular cortex regularly cause deviations of perceived verticality in the frontal roll plane. These tilts are ipsilateral in peripheral and in ponto-medullary lesions and contralateral in ponto-mesencephalic lesions. Unilateral lesions of the vestibular thalamus or cortex cause smaller tilts of the perceived vertical, which may be either ipsilateral or contralateral. Using a neural network model, we previously explained why unilateral vestibular midbrain lesions rarely manifest with rotational vertigo. We here extend this approach, focussing on the direction-specific deviations of perceived verticality in the roll plane caused by acute unilateral vestibular lesions from the labyrinth to the cortex. Traditionally, the effect of unilateral peripheral lesions on perceived verticality has been attributed to a lesion-based bias of the otolith system. We here suggest, on the basis of a comparison of model simulations with patient data, that perceived visual tilt after peripheral lesions is caused by the effect of a torsional semicircular canal bias on the central gravity estimator. We further argue that the change of gravity coding from a peripheral/brainstem vectorial representation in otolith coordinates to a distributed population coding at thalamic and cortical levels can explain why unilateral thalamic and cortical lesions have a variable effect on perceived verticality. Finally, we propose how the population-coding network for gravity direction might implement the elements required for the well-known perceptual underestimation of the subjective visual vertical in tilted body positions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The peripheral and central vestibular systems are bilaterally organized in three major planes of action: the horizontal yaw plane, the sagittal pitch plane, and the frontal roll plane. Plane-specific disorders are determined by ocular motor, postural, and perceptual signs. Consequently, central vestibular disorders due to acute unilateral lesions manifest with signs and symptoms in one or several of these spatial planes. Vestibular tone imbalances in the roll plane are the most frequent. They are caused by a unilateral damage of graviceptive pathways, which mediate input from the otolith organs and vertical semicircular canals [1,2,3]. Typical roll plane signs are tilts of the perceived visual vertical, ocular torsion, skew deviation, and tilts of the head and body. This complete eye–head synkinesis is called the ocular tilt reaction (OTR). It was first observed during electrical stimulation of the rostral midbrain tegmentum in the monkey [4, 5] and was later described also in patients with unilateral meso-diencephalic lesions [6]. Various clinical studies have shown that tilts of perceived verticality are highly sensitive vestibular signs of unilateral peripheral [7,8,9,10] and central lesions of the vestibular circuitry in the brainstem [3, 11,12,13,14] as well as the vestibular thalamus [15,16,17] and the parieto-insular vestibular cortex [14, 18]. As regards the frequency of all components of the OTR in unilateral brainstem lesions, SVV tilts occurred in up to 80–90%, ocular torsion followed with 80%, skew deviation with 40%, and complete OTR with 20% [13, 16].
Human lesion studies have allowed clinically relevant interpretations on which the following topographic diagnostic rules are based: (1) complete OTR or its ocular motor components occur only with unilateral lesions from the labyrinth to the rostral midbrain tegmentum, including the INC and the rostral interstitial nucleus of the medial longitudinal fascicle (riMLF). Thus, reflexive control of eyes (vestibulo-ocular reflex and vestibulo-ocular tone imbalance), head, and body (vestibulo-spinal reflexes and tone imbalance, head tilt and body lateropulsion) is mediated at the brainstem/cerebellar level [19]. (2) Unilateral graviceptive tone imbalances caused by lesions of the centromedial or posterolateral vestibulo-thalamic subnuclei or the parieto-insular vestibular cortex (PIVC) manifest only with perceptual tilts and are determined by deviations of the perceived visual vertical. (3) All unilateral peripheral and caudal pontomedullary brainstem lesions (including the vestibular nuclei, VN) manifest with ocular motor and perceptual tilts that are ipsilateral. All unilateral lesions within the pontomesencephalic brainstem cause contralateral tilts due to a crossing of graviceptive pathways in the pons at a level above the vestibular nuclei. (4) Unilateral lesions at thalamic or cortical level manifest with either ipsilateral or contralateral tilts of the SVV [2, 15, 16]. The direction of the tilts is constant in the single patient (intraindividual consistency), but it varies interindividually, with about 50% ipsilateral and 50% contralateral. (5) The amount of the SVV tilts in degrees is less in thalamic and in cortical lesions (thalamus: mean 3°–5°; cortex: mean 4°–6° compared to brainstem lesions, mean 8°–14°) (Fig. 1; Table 1).
Two alternative mechanisms for SVV tilt after unilateral vestibular lesion. a OTO model (see text). Lower row: body position of the patient, dark red cross indicates lesion side. Middle row: peripheral processing: unequal numbers of hair cells with left and right preferred directions on the intact utricle cause a utricular bias (dark red arrow), which is added to the remaining saccular and utricular discharge (light blue arrows) caused by gravity to yield the otolith vector (dark blue arrow). Top row: central processing: vectorial addition of normalized otolith vector (dark blue arrow) and idiotropic bias (light blue arrow) leads to the SVV estimate (red line). The resulting SVV is equal for left-ear-down and right-ear-down positions. b OTO model prediction for SVV tilt due to asymmetry of utricular hair cell proportion. The predicted SVV deviation at 90° is equal for ipsi- or contralateral lesions. c SCC model (see text). Lower row as in a. Middle row (peripheral): independent of body position, a torsional semicircular canal bias (dark orange arrow) rotates the otolith vector away from the affected ear. Top row: summation with idiotropic bias as in a. d Right: SCC model prediction for SVV tilt due to semicircular canal imbalance. At 90°, prediction for ipsi- and contralateral lesions differs by approximately the same amount as in the upright position. In c and d, the SVV deviation in the upright position has been set to about 12.5° corresponding to the data of [29]. In case of an asymmetry of utricular hair cells b the difference at 90° right-ear-down or left-ear-down positions (dashed vertical line) is close to zero. In contrast, the model with canal bias predicts a sustained difference for right-ear-down and left-ear-down positions
The last two findings are still unexplained. Why do unilateral lesions manifest at the thalamic or cortical level with perceptual tilts in either ipsi- or contralateral direction? Why are these deviations from true vertical quantitatively smaller than in brainstem and peripheral vestibular lesions? There is a partial crossing of the ascending graviceptive pathways in the rostral midbrain that provides the thalamus and the cortex with input from graviceptive sensors of both labyrinths [20]. This allows the right and left thalamocortical networks to operate separately (there is no direct interconnection between the two thalami) and mediate vestibular information from both labyrinths [21].
Another explanation for the difference between periphery/brainstem versus thalamus/cortex lesions could be based on different neuronal coding principles. In a recent study based on the clinical experience that caudal vestibular brainstem lesions cause rotational vertigo, whereas more rostral vestibular lesions cause non-rotational dizziness or only imbalance without vertigo. We mathematically simulated the different manifestations of vestibular disorders in the yaw plane using a neural network model of the head direction cell system [22]. This model was based on experimentally documented differences in neuronal coding for angular head velocity and head direction [23]. It explained and confirmed the clinical experience of vestibular vertigo syndromes in the horizontal semicircular canal plane. However, graviceptive vestibular function in the roll plane was not included in that model, although the majority of patients presented with roll plane signs.
We here hypothesize that the differential effects of unilateral graviceptive lesions in either the brainstem or thalamocortical vestibular circuitry can also be explained by a dysfunction of a graviceptive cell system that uses coding principles for spatial orientation in the roll (and pitch) planes similar to those exhibited by the head direction cell system for the horizontal yaw plane [23]. We therefore extended our model approach to the graviceptive frontal roll plane. Findings in a recent study on macaque monkey of the gravity-orientation tuning in anterior thalamic neurons [24] encouraged us to develop the following model.
Methods
Clinical study on published SVV tilts
A total of 15 published studies on acute unilateral peripheral or central vestibular lesions were reviewed as to the direction and amount of SVV tilts [2, 3, 7,8,9, 12,13,14, 16, 18, 25,26,27,28]. A statistical meta-analysis was not applicable since the methods used to determine the visual vertical differed across the studies, as did the lesion size and the grouping of the patients (complete or incomplete peripheral lesions; analyses of the entire brainstem versus lower and upper brainstem segments in central lesions). The data were obtained in upright body position. The mathematical model approach in the current study, however, also requires patient data on SVV measurements in tilted body positions, which are comparatively rare. We took into account two studies in which patients with peripheral vestibular lesions were tested in right-ear-down (RED) and left-ear-down (LED) positions [29, 30].
Model approach
In the following, only lateral tilt and perception of verticality in the roll plane are considered for clarity. Systems-level models for dynamic three-dimensional gravity or tilt perception have been published elsewhere [31,32,33,34,35,36,37 and other publications not cited here]. In contrast to these models, which focused on the mechanisms required to solve the problem of perception of verticality on a relatively abstract level, the current work posits a hypothesis as to how these mechanisms might be implemented at the neuronal level. Firing rate neurons (that is, simulated neurons that have firing rates as input and output) were used throughout, since these model neurons simplify the task of designing a network-level model implementing gravity perception with the aim of simulating lesions of this network.
Peripheral sensory input
The otolith organs, the sensory organs for gravity, decompose the gravity vector into one component aligned approximately with the interaural axis (utricle) and a second component aligned with the body-longitudinal axis (saccule). The primary otolith afferents are tuned to the angle of tilt in a sinusoidal manner. Since otolith hair cells on one otolith macula are polarized in both directions (e.g., left–right for the utricle and the roll plane), there are four populations of hair cells (two utricular and two saccular populations) for each side of the head. For example, the left utricle contains hair cells with preferred direction for leftward tilt, but also for rightward tilt. In addition to the sinusoidal tuning, each afferent fiber has a baseline firing rate. Taken together, the afferent fibers from each of the populations thus yield a population response with a baseline firing rate approximately proportional to the number of fibers [38].
In the following, we propose two mechanisms of how peripheral lesions might lead to roll tilt SVV deviations in the upright position and formulate them as computational models.
Otolith (OTO) model (Fig. 1a): the predominant explanation for SVV tilt after peripheral lesions is that the SVV bias is caused by the effect of the lesion on otolith afferents. To model this assumption, we assumed that hair cells on both sides of the striola (having opposite directions) are not equal in number (Ewald’s law for otolith organs) [39]. We refer to this model assumption as the “OTO model”. In a healthy system, the central fusion of utricular afferents from the left and right labyrinths and from both sides of the striola ensures an unbiased response. However, after a peripheral lesion, the unbalanced input from the remaining side causes an SVV deviation in the upright position (see Appendix for mathematical derivation).
Semicircular canal (SCC) model (Fig. 1c): an alternative explanation for SVV tilt following peripheral lesions is based on semicircular canal input. Gravity perception requires that the so-called tilt-translation ambiguity be solved [31,32,33,34,35,36,37, 40, 41]. The predominant view is that semicircular canal input is used by a central estimation mechanism to disentangle otolith input caused by tilt from that caused by translational acceleration. Under normal circumstances, canal input is transient and has no influence on static body positions. However, a peripheral unilateral lesion causes a continuous imbalance of semicircular canal afferent firing, since input from the lesioned side is missing. This imbalance is centrally interpreted as a head rotation, which manifests as vertigo and spontaneous nystagmus. However, it should also have an effect on the estimation of the direction of gravity. For this model, we assumed a basic gravity estimation mechanism proposed previously (2-D: [40]; 3-D: [31, 33, 34]). This dynamic model consists of a 3-D differential equation system whose static solution predicts an SVV deviation in the upright position that is approximately proportional to the semicircular canal bias (see Appendix). Since it is currently unknown how or where the brain implements gravity estimation at the neural level, we assumed for the SCC model a simple fusion of otolith and semicircular canal input (see Appendix) below the thalamus, for example, at the brainstem or cerebellar level (see for example [42, 43]).
Central processing
Otolith afferent fibers from both sides are fused (probably in the vestibular nuclei) via commissural connections, so that overall four populations of neurons can be assumed: those tuned for 2-D leftward, rightward, upward, and downward acceleration. While the components of gravitational acceleration in the periphery and supposedly in brainstem pathways are coded in sensory coordinates, central neurons show a tuning that is no longer restricted to the two directions of otolith organs. In central neurons [24], tuning is distributed over various directions similar to that of the head direction cell system [23, 44]. We thus implemented a tuning curve population model [45]. To implement the well-known bias of perception of the direction of gravity (Aubert effect, see for example [46]), we assumed a so-called rate change model, in which tuning curves are not homogeneous but depend on the direction of tuning ([45], Fig. 2 left), and from which the SVV direction (Fig. 2, right) is computed via population decoding. The tuning curve population model receives appropriately weighted input from utricular and saccular otolith afferents (in the case of the SCC model, after fusion with canal input). The perceptual bias implemented by the population of gravity-tuned neurons acts in a very similar way as Mittelstaedt’s idiotropic vector, which was applied to the normalized otolith organ vector [46]. The normalization, which causes the SVV to be approximately independent of gravity magnitude, is here implemented by transforming the vectorial representation of the gravity estimate (utricular and saccular components) to a population representation that codes the direction but not the magnitude of the vector.
Left: simulated neuronal tuning functions for graviceptive pathways above midbrain implementing rate change tuning. Right: subjective visual vertical derived from a tuning curve population model with neurons tuned to gravity direction as shown on the left. The typical Aubert effect (underestimation of tilt for larger angles) can be clearly seen (blue line)
Results
Clinical data on SVV tilts
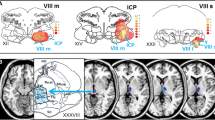
Unilateral peripheral lesions of the labyrinth or the eighth nerve caused ipsilateral SVV tilts in upright head and body position, which were maximal with complete vestibular loss due to neurectomy or labyrinthectomy [9, 10, 25] (Table 1; Fig. 3). Pontomedullary lesions of the vestibular nuclei also caused ipsilateral tilts of about the same amount. Above the crossing of graviceptive pathways in the lower pons (Table 1; Fig. 3), the tilts were contralateral up to the rostral midbrain tegmentum (INC) and showed somewhat less degrees of deviation from true vertical in the pons [3, 13, 14]. One exception from that rule has been described for anteromedian pontomesencephalic lesions close to and within the medial lemniscus which manifested with isolated ipsilateral SVV tilts of up to 6.6° [28]. Two studies have shown that lesions of the vestibular thalamic subnuclei caused either ipsilateral or contralateral SVV tilts had a lesser degree of up to 5° [15, 16]. The same is true for unilateral lesions of the parieto-insular vestibular cortex [1, 2]. For better understanding, the directions and ranges of the mean data have been schematically depicted in a simplified way (Fig. 3). This allowed us to demonstrate the special features of vestibular lesions in the thalamus and cortex, i.e., the smaller tilts to the ipsi- or contralateral side.
Schematic graviceptive pathways together with the amount (in deg) of SVV deviation for ipsi- and contralateral lesions depending on the level of acute unilateral vestibular damage. The range of the mean values was calculated from a total of 15 published studies (see Table 1). The four major messages are as follows. (1) In peripheral and pontomedullary brainstem lesions SVV tilts are ipsilateral. (2) In pontomesencephalic vestibular pathway lesions up to the INC, SVV tilts are contralateral. (3) In vestibular thalamic and cortical lesions, SVV tilts may be either ipsilateral or contralateral with an intraindividual consistency and an equal distribution interindividually. (4) The amount of SVV tilt is maximal in complete peripheral lesions, followed by tilt in brainstem lesions and less in lesions of the vestibular thalamus and cortex
Simulation results
Simulation of the intact network shows the typical Aubert effect on perception of verticality, that is, the underestimation of tilt observed for large tilt angles (Fig. 2, right). No particular effort was made to fit the response to experimental data so that the result shown in Fig. 2 should be considered an approximate response illustrating the principle. Note that the idiotropic vector [46], which can be interpreted in Bayesian terms as prior expectation for upright orientation [47,48,49], is implicitly realized by inhomogeneous tuning curves. Thus, there is no neuron or neuronal population implementing the idiotropic vector; instead, it is coded in the synaptic weights of the network.
For peripheral lesions, we simulated two different models. As described in “Methods”, the OTO model assumes that the SVV deviation in the upright position is caused by unequal numbers of hair cells on the utricular macula, an assumption found in the majority of the literature (for example, [29, 30]). The SCC model assumes that the SVV deviation is caused by a torsional bias of the vertical semicircular canals mediated by the central gravity estimator. Figure 4 shows simulation of both models in comparison to the data of [29], obtained after unilateral vestibular nerve section. Both models have been adjusted for the value reported in the upright position (12.4°): in the OTO model, the hair cell ratio was set to 0.436; in the SCC model the roll angular velocity bias was set to 2.8°/s, which, assuming a torsional VOR gain of 0.5, would approximately correspond to a hardly recognizable torsional nystagmus of 1.4°/s. Both models also predict some static ocular torsion due to utricular bias for the OTO model and the effect of sustained semicircular canal input for the SCC model [51].
SVV data from [32] obtained in patients with unilateral vestibular nerve section (blue, error bars denote SD) and model simulations of the SVV adjustments (red and yellow). The OTO model (yellow) assumes unequal distribution of hair cells with opposite tuning on the utricular macula (Ewald’s law for otolith organs); the SCC model (red) assumes that the afferent input of vertical semicircular canals is processed centrally by the gravity estimation mechanism. After a lesion, the semicircular canal bias causes a perceptual error of gravity direction that becomes visible as SVV tilt
While both models can simulate the SVV deviation in the upright position, the model predictions differ for tilted positions such as right-ear-down and left-ear-down positions (see Fig. 1b, d). As shown in Fig. 4, the SCC model clearly corresponds better to the patient data obtained in tilted head positions.
A unilateral lesion of the central population network was simulated by randomly removing half of the neurons in the network, that is, by setting their output firing rate to zero. To estimate the amount of the resulting SVV deviation, the lesion simulation was repeated 100 times (larger numbers yield similar results). With the present network, the average absolute SVV tilt was 5.0° for the upright position (Fig. 5); this is very close to the clinical findings at the thalamo-cortical level (see Fig. 3).
Average subjective visual vertical after simulated unilateral central lesion of the tuning curve population (red region shows standard deviation of the simulated lesion). To simulate the lesion, approximately 50% of the neurons have been removed randomly. For the upright position, the simulated mean SVV tilt is 0° ± 6.2° corresponding to an average absolute deviation of 5.0°
Simulated cosine tuning functions for population response of utricular afferents in the roll plane (blue: left utricle; yellow: right utricle). For each utricle, two functions corresponding to ipsi- and contralaterally polarized hair cells are shown. It is assumed that hair cell proportions are unequal so that both response gain and offset depend on hair cell polarization
Discussion
Modeling unilateral peripheral vestibular lesions
To explain the effect of unilateral peripheral lesions on SVV deviation, we considered two alternatives: for the OTO model unequal proportions of utricular hair cells with opposite tuning direction were assumed; for the SCC model we took into account that the SVV depends on tilt-translation disambiguation and that the central gravity estimator operates with semicircular canal input. Thus, SVV tilt following a peripheral lesion could be caused by an imbalance of utricular firing rate for ipsi- and contralesionally tuned hair cells, or by the well-known imbalance of semicircular canal input. Such an imbalance would act as rotational input to a central gravity estimator, which in turn leads to a deviation of the SVV.
While both models are able to generate an SVV deviation in the upright position, the model predictions for tilted body positions differ. The OTO model predicts that the difference of the SVV setting for right- and left-ear-down positions vanishes, since in both positions the SVV is determined only by utricular input independent of its amount (Fig. 1a, b). In contrast, the SCC model predicts that the SVV is rotated by an approximately constant amount to the ipsilateral side manifesting as a constant shift of the SVV independently of body position (Fig. 1c, d).
From our comparison of the predictions of the two models with data of the SVV adjustments in patients with unilateral vestibular lesions in left-ear-down and right-ear-down body positions [29], we propose that the SVV deviation in the upright position is indeed predominantly caused by the effect of semicircular canal bias on the gravity estimator and is not due to an utricular hair cell asymmetry. Corresponding to our assumption that SVV tilt is caused by semicircular canal imbalance, we previously have shown that static ocular torsion observed during prolonged galvanic vestibular stimulation can be fully explained by semicircular canal activation [50]. Damage to the gravity estimator has also recently been implicated in the generation of persistent horizontal positional nystagmus [51].
We also considered patient data from Müller et al. [30], who measured SVV in patients with unilateral peripheral vestibular damage in different body positions. However, due to the smaller SVV tilts found in their study, the differences between model predictions were not as conclusive as for the data from Böhmer and Rickenmann [29] (see Appendix Fig. 7). Müller and co-workers [30] also tested the hypothesis that SVV tilt due to complete vestibular nerve lesion should differ from that of a lesion of the superior branch, because the latter would spare the saccular nerve. The difference between both types of lesions in their data was negligible. An evident explanation compatible with our model is that SVV tilt with peripheral lesions is not caused by otolith organ dysfunction, but by the lesion of the vertical semicircular canal afferents, which are affected in both cases (even though only the superior canal is affected with superior nerve lesion).
Data from [30] (blue, complete vestibular nerve lesion, error bars denote SD) compared to SCC model prediction (red) for SVV tilt (left, due to semicircular canal bias) and OTO model prediction (right, due to utricular haircell asymmetry). Model parameters have been adjusted to approximately match the data. Due to the small bias in the upright position, the difference between model predictions is within the variability of the data, even though it is in the same direction as in Fig. 4 (asymmetric errors for the OTO model, right panel)
To further clarify the relative contribution of the otolith or canal biases to pathological SVV deviations after unilateral lesions, SVV data from patients not only in the upright position but also in tilted positions are required. Such data acquired in patients with brainstem or midbrain lesions might also help to elucidate where otolith–canal fusion for gravity estimation takes place—already in the cerebellum [42, 43] or higher at midbrain levels, thus comparable to the generation of the head direction cell signal [22, 23].
Modeling central vestibular lesions at thalamic level
For the central lesions, we proposed a tuning function network that codes gravity direction in a distributed fashion similar to the coding of heading direction in the head direction cell network [22, 23]. The recent finding of gravity-tuned neurons in the thalamus [24] prompted us to propose that neuronal coding of gravity in the thalamus, and possibly in regions below and above, follows similar principles as known for the head direction cell system [23]. The effect of a unilateral lesion of such a network depends on its implementation and read-out mechanisms. Here, we assumed that thalamic gravity-encoding neurons converge onto a cortical population-tuning network with similar coding properties; this is followed by standard population decoding to derive the gravity direction encoded in the network activity. In addition, we proposed that the perceptual bias in tilted positions, i.e., the Aubert effect, which has been explained as idiotropic vector or, in a Bayesian framework, as prior expectation for the upright position, is implemented at the neural level as inhomogeneous tuning functions via synaptic weights. Interestingly, the transformation from vectorial coding (utricular/horizontal and saccular/vertical components) to population coding also implements what is [50] called normalization, that is, representation of the angle with respect to gravity independently of the amplitude of the otolith vector. To achieve normalization in a vectorial coding scheme would require the implementation of non-linear mathematical operations, specifically division, which is difficult to realize neuronally. However, the transformation from a vector code to a population code proposed here implements the normalization as emergent property and without needing mathematical division.
Unilateral central lesion of this model network showed that in the upright position, a random bias with an average deviation of 5° emerges. The direction of the bias depends on the distribution of neurons (or neuronal assemblies) on both sides of the thalamus. A perfectly equal distribution would not cause any bias after a unilateral lesion. We thus hypothesize that the SVV tilt observed after unilateral thalamic or cortical lesions, which is independent of the lesion side, is due to the population coding of the direction of gravity and the relative distribution of neuronal assemblies in the population. The experimentally observed distribution of lateral deviations is unlikely to be related to hemispheric vestibular dominance, since this would imply a dependence of tilt magnitude on the side of the lesion, which was not found.
Instead of representing gravity direction using a feedforward network, we could as well implement the gravity-tuned population model by an attractor network with recurrent connectivity as supposed to exist for head direction cells [22, 23, 44]. The results of such an attractor network concerning the effect of unilateral lesions is, according to our simulations, similar, but the implementation as feedforward network used here is simpler and therefore preferable to illustrate our main hypotheses.
However, an attractor solution would have several additional advantages:
-
1.
Since an attractor network implements a short-term memory, during short periods of weightlessness, such as while jumping down from a rock, the perception of the direction of gravity would be sustained despite zero input from the otolith organs. In a pure feedforward network, it would rapidly decay.
-
2.
During longer periods of weightlessness, the subjective perception of verticality does not vanish but aligns with the body midline [52, 53]. This is exactly what happens in an attractor network with inhomogeneous tuning curves due to drift of the bump of activity toward the region with maximal tuning. Again, in a feedforward network, the perception of verticality would simply vanish during zero g.
-
3.
As mentioned above, all systems-level models for gravity estimation and tilt-translation disambiguation ([31,32,33,34,35,36,37] and others) posit that an internal estimate of gravity is updated not only by otolith input, but also by angular velocity signals derived from semicircular canal input. While we assumed here that gravity estimation is performed below the level of population coding, for example by cerebellar circuits [43], an attractor network could solve this updating using a similar integration mechanism as applied in the head direction cell network to update head direction while turning [22].
Conclusions
The presented model explains the clinical data summarized in Fig. 3: (1) ipsilateral tilt of perceived verticality in unilateral peripheral lesions, which are compatible with SVV tilts measured in upright or head-tilted positions; (2) ipsi- or contralateral tilts in vestibular lesions of the thalamus or cortex; (3) SVV tilts in degree are about 50% smaller in thalamus and cortex lesions compared to a peripheral deficit. The pathophysiology of the latter two clinical findings has been hitherto unexplained. Our model approach to coding of the direction of gravity in central areas is well in line with animal data that suggest a 3-D ‘brain compass’ comprising head direction and gravity direction cells [44]. Furthermore, comparison of our model simulations with patient data in the right-ear-down and left-ear-down positions suggests that static deviations of the SVV after unilateral peripheral lesions may not be caused by otolith organ imbalance as suggested previously, but may be a consequence of a vertical semicircular canal imbalance acting upon the central gravity estimator.
Abbreviations
- AVS:
-
Acute vestibular syndrome
- OTR:
-
Ocular tilt reaction
- SVV:
-
Subjective visual vertical
References
Brandt T, Dieterich M (1994) Vestibular syndromes in the roll plane: topographic diagnosis from brainstem to cortex. Ann Neurol 36:337–347
Baier B, Suchan J, Karnath HO, Dieterich M (2012) Neural correlates of disturbed perception of verticality. Neurology 78:728–735
Yang TH, Oh SY, Kwak K, Lee JM, Shin BS, Jeong SK (2014) Topology of brainstem lesions associated with subjective visual vertical tilt. Neurology 82:1968–1975
Westheimer G, Blair SM (1975) The ocular tilt reaction—a brainstem ocular motor routine. Investig Ophthalmol 14:833–839
Westheimer G, Blair SM (1975) Synkinesis of head and eye movements evoked by brainstem stimulation in the alert monkey. Exp Brain Res 24:89–95
Halmagyi GM, Brandt T, Dieterich M, Curthoys IS, Starck RJ, Hoyt WF (1990) Tonic contraversive ocular tilt reaction due to meso-diencephalic lesions. Neurology 40:1503–1509
Friedmann G (1970) The judgement of the visual vertical and horizontal with peripheral and central vestibular lesions. Brain 93:313–328
Halmagyi GM, Gresty MA, Gibson WPR (1979) Ocular tilt reaction with peripheral vestibular lesions. Ann Neurol 6:80–83
Böhmer A (1997) Zur Beurteilung der Otolithenfunktion mit der subjektiven visuellen Vertikalen. HNO 45:533–537
Anastasopoulos D, Haslwanter T, Bronstein A, Fetter M, Dichgans J (1997) Dissociation between the perception of body verticality and the visual vertical in acute peripheral vestibular disorder in humans. Neurosci Lett 233:151–153
Dieterich M, Brandt T (1992) Wallenberg’s syndrome: lateropulsion, cyclorotation and subjective visual vertical in 36 patients. Ann Neurol 31:399–408
Dieterich M, Brandt T (1993) Ocular torsion and tilt of subjective visual vertical are sensitive brainstem signs. Ann Neurol 33:292–299
Brandt T, Dieterich M (1993) Skew deviation with ocular torsion, a vestibular brainstem sign of topographic diagnostic value. Ann Neurol 33:528–534
Baier B, Thömke F, Wilting J, Heinze C, Geber C, Dieterich M (2012) A pathway in the brainstem for roll-tilt of the subjective visual vertical: evidence from a lesion-behavior mapping study. J Neurosci 32:14854–14858
Dieterich M, Brandt T (1993) Thalamic infarctions: differential effects on vestibular function in the roll plane (35 patients). Neurology 43:1732–1740
Baier B, Conrad J, Stephan T, Kirsch V, Vogt T, Wilting J, Müller-Forell W, Dieterich M (2016) Vestibular thalamus: two distinct graviceptive pathways. Neurology 86:134–140
Elwischger K, Rommer P, Prayer D, Mueller C, Auff E, Wiest G (2012) Thalamic astasia from isolated centromedian thalamic infarction. Neurology 78:146–147
Brandt T, Dieterich M, Danek A (1994) Vestibular cortex lesions affect the perception of verticality. Ann Neurol 35:403–412
Dieterich M, Brandt T (2015) The bilateral central vestibular system: its pathways, functions, and disorders. Ann N Y Acad Sci 84:1–5
Kirsch V, Keeser D, Hergenroeder T, Erat O, Ertl-Wagner B, Brandt T, Dieterich M (2016) Structural and functional connectivity mapping of the vestibular circuitry from human brainstem to cortex. Brain Struct Funct 221:1291–1308
Brandt T, Dieterich M (2018) Functional and structural benefits of separately operating right and left thalamo-cortical networks. J Neurol. https://doi.org/10.1007/s00415-018-8824-9
Dieterich M, Glasauer S, Brandt T (2018) Why acute unilateral vestibular midbrain lesions rarely manifest with rotational vertigo: a clinical and modelling approach to head direction cell function. J Neurol 265:1184–1198
Clark BJ, Taube JS (2012) Vestibular and attractor network basis of the head direction cell signal in subcortical circuits. Front Neural Circuits 6:7
Laurens J, Kim B, Dickman JD, Angelaki DE (2016) Gravity orientation tuning in macaque anterior thalamus. Nat Neurosci 19:1566–1568
Vibert D, Häusler R, Safran AB (1999) Subjective visual vertical in peripheral unilateral vestibular diseases. J Vestib Res 9:145–152
Böhmer A, Mast F, Jarchow T (1996) Can a unilateral loss of otolith function be clinically detected by assessment of the subjective visual vertical? Brain Res Bull 40:423–429
Bisdorff AR, Wolsley CJ, Anastasopoulos D, Bronstein AM, Gresty MA (1996) The perception of body verticality (subjective postural vertical) in peripheral and central vestibular disorders. Brain 119:1523–1534
Zwergal A, Büttner-Ennever J, Brandt T, Strupp M (2008) An ipsilateral vestibulothalamic tract adjacent to the medial lemniscus in humans. Brain 131:2928–2935
Böhmer A, Rickenmann J (1995) The subjective visual vertical as a clinical parameter of vestibular function in peripheral vestibular diseases. J Vestib Res 5:33–45
Müller JA, Bockisch CJ, Tarnutzer AA (2016) Spatial orientation in patients with chronic unilateral vestibular hypofunction is ipsilesionally distorted. Clin Neurophysiol 127:3243–3251
Glasauer S (1992) Interaction of semicircular canals and otoliths in the processing structure of the subjective zenith. Ann N Y Acad Sci 656:847–849
Merfeld DM, Young LR, Oman CM, Shelhamer MJ (1993) A multidimensional model of the effect of gravity on the spatial orientation of the monkey. J Vestib Res 3:141–161
Glasauer S, Merfeld DM (1997) Modeling three dimensional vestibular responses during complex motion stimulation. In: Fetter M, Haslwanter T, Misslisch H, Tweed D (eds) Three-dimensional kinematic principles of eye-, head, and limb movements in health and disease. Harwood, Amsterdam, pp 387–398
Bos JE, Bles W (2002) Theoretical considerations on canal–otolith interaction and an observer model. Biol Cybern 86:191–207
Glasauer S, Brandt T (2007) Noncommutative updating of perceived self-orientation in three dimensions. J Neurophysiol 97:2958–2964
MacNeilage PR, Ganesan N, Angelaki DE (2008) Computational approaches to spatial orientation: from transfer functions to dynamic Bayesian inference. J Neurophysiol 100:2981–2996
Laurens J, Angelaki DE (2017) A unified internal model theory to resolve the paradox of active versus passive self-motion sensation. Elife 6:e28074
Cousins S, Kaski D, Cutfield N, Seemungal B, Golding JF, Gresty M, Glasauer S, Bronstein AM (2013) Vestibular perception following acute unilateral vestibular lesions. PLoS One 8:e61862
Dai MJ, Curthoys IS, Halmagyi GM (1989) Linear acceleration perception in the roll plane before and after unilateral vestibular neurectomy. Exp Brain Res 77:315–328
Mayne R (1974) A systems concept of the vestibular organs. In: Kornhuber HH (ed) Handbook of sensory physiology, vol VI. Vestibular system Part 2: psychophysics, applied aspects and general interpretations. Springer, Berlin, pp 493–580
Green AM, Angelaki DE (2010) Multisensory integration: resolving sensory ambiguities to build novel representations. Curr Opin Neurobiol 20:353–60
Wilden A, Glasauer S, Kleine JF, Büttner U (2002) Modelling transfer characteristics of vestibular neurons in the fastigial nucleus of the behaving monkey on the basis of canal-otolith interaction. Neuroreport 13:799–804
Laurens J, Meng H, Angelaki DE (2013) Neural representation of orientation relative to gravity in the macaque cerebellum. Neuron 80:1508–1518
Laurens J, Angelaki DE (2018) The brain compass: a perspective on how self-motion updates the head direction cell attractor. Neuron 97:275–289
Schwartz O, Hsu A, Dayan P (2007) Space and time in visual context. Nat Rev Neurosci 8:522–535
Mittelstaedt H (1983) A new solution to the problem of the subjective vertical. Naturwissenschaften 70:272–281
Eggert T (1998) Der Einfluss orientierter Texturen auf die subjektive visuelle Vertikale und seine systemtheoretische Analyse. Dissertation, Technical University of Munich
Vingerhoets RA, De Vrijer M, Van Gisbergen JA, Medendorp WP (2009) Fusion of visual and vestibular tilt cues in the perception of visual vertical. J Neurophysiol 101:1321–1333
Alberts BB, de Brouwer AJ, Selen LP, Medendorp WP (2016) A Bayesian account of visual-vestibular interactions in the rod-and-frame task. eNeuro. https://doi.org/10.1523/ENEURO.0093-16.2016
Schneider E, Glasauer S, Dieterich M (2002) Comparison of human ocular torsion patterns during natural and galvanic vestibular stimulation. J Neurophysiol 87:2064–2073
Choi JY, Glasauer S, Kim JH, Zee D, Kim JS (2018) Characteristics and mechanism of apogeotropic central positional nystagmus. Brain 141:762–775
Glasauer S, Mittelstaedt H (1998) Perception of spatial orientation in microgravity. Brain Res Brain Res Rev 28:185–93
Harris LR, Jenkin M, Jenkin H, Zacher JE, Dyde RT (2017) The effect of long-term exposure to microgravity on the perception of upright. NPJ Microgravity 3:3
Cnyrim CD, Rettinger N, Mansmann U, Brandt T, Strupp M (2007) Central compensation of deviated subjective visual vertical in Wallenberg's syndrome. J Neurol Neurosurg Psychiatry 78(5):527–528
Cnyrim CD, Newman-Toker D, Karch C, Brandt T, Strupp M (2008) Bedside differentiation of vestibular neuritis from central “vestibular pseudoneuritis”. J Neurol Neurosurg Psychiatry 79(4):458–460
Acknowledgements
This work was supported by funds from the German Federal Ministry of Education and Research (BMBF Grant code 01 EO 0901 to TB, MD, SG), the Hertie Foundation (TB), the Deutsche Forschungsgemeinschaft (DFG GL 342/3-1 to SG), and the German Foundation for Neurology (MD). We thank Judy Benson for copy-editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
There are no conflicts of interest.
Ethical approval
An approval by an ethics committee was not applicable.
Additional information
This manuscript is part of a supplement sponsored by the German Federal Ministry of Education and Research within the funding initiative for integrated research and treatment centers.
Appendix
Appendix
OTO model
If the static deviation of the SVV found after unilateral lesions results from unequal numbers of hair cells on both sides of the utricular striola, we have to consider four populations of utricular afferents (two for each utricle) and two populations of saccular afferents (one tuned for upright posture and one for upside-down posture). Each population has an offset and a gain corresponding to the proportion of contributing hair cells (see Fig. 6).
In a static body position, the gravitational acceleration is the only input to the otolith organs; thus, the utricular and saccular components can be written as \(\left[ {\begin{array}{*{20}{c}} {{f_y}} \\ {{f_z}} \end{array}} \right]=\left[ {\begin{array}{*{20}{c}} {\sin (\alpha )} \\ {\cos (\alpha )} \end{array}} \right]\) with α being the angle of tilt. The utricular firing rates of one utricle are thus \({u_i}=c \cdot \left( {{f_y}+{u_0}} \right)\) for the ipsilateral population and \({u_c}=\left( {1 - c} \right) \cdot \left( { - {f_y}+{u_0}} \right)\) with c < 1 being the proportion of hair cells tuned to the ipsilateral direction and u0 being a resting discharge (assumed to be large enough so that the steady-state firing rates always remain larger than zero). For an equal number of hair cells on both sides, c would be 0.5. The saccular populations (up and down) are \({s_{u/d}}= \pm {f_z}+{s_0}.\)
The overall otolith organ output used to estimate the direction of gravity can thus be computed as \({g_y}=u={u_{{\text{ri}}}}+{u_{{\text{lc}}}} - {u_{{\text{rc}}}} - {u_{{\text{li}}}}\) and \({g_z}=s={s_u} - {s_d}\). In the healthy case, the factor c and the resting discharges cancel out.
In case of a unilateral lesion, \(u={u_{{\text{ri}}}} - {u_{{\text{rc}}}}={f_y}+(2c - 1) \cdot {u_0}\) and \(s={f_z}\). For the upright position the expected SVV deviation can be calculated by setting fy = 0 and fz = 1 as
With a resting discharge corresponding to 2 g and a hair cell proportion of 0.55, this would yield an SVV deviation of 11.5°. The influence of the idiotropic bias can be ignored in the upright position.
In the right-ear-down and left-ear-down positions, fy = ± 1 and fz = 0. Thus, \(u={u_{{\text{ri}}}} - {u_{{\text{rc}}}}= \pm 1+\left( {2c - 1} \right) \cdot {u_0}\). Even though the utricular discharge in the left- and right-ear-down positions is not equal in amplitude, the estimated head orientation \(\beta ={\tan ^{ - 1}}\left( {u/s} \right)\) is ± 90° and thus results in the same SVV in both positions after applying the idiotropic bias.
SCC model
To explain a static SVV tilt on the basis of a unilateral semicircular canal lesion, we have to consider how canal and otolith input is fused centrally for gravity estimation.
The basic differential equation governing the dynamic fusion of canal and otolith signals [31, 33, 34] is given by:
with g being the estimated gravity vector, ω the angular velocity vector (measured by the semicircular canals), and f the gravito-inertial acceleration (measured by the otolith organs). In our 2-D case, only fy and fz corresponding to utricular and saccular afferent discharge are used. If participants are in a static body position, f is equal to the true gravity vector. The time constant τ is estimated by [34] to be about 5 s, and by [37] as 1.3 s.
To estimate the static deviation caused by a unilateral lesion, we consider the steady state of the equation by setting the left side to zero. This results in
For simplicity, we only consider tilt in the roll plane caused by a rotation around the naso-occipital axis (the x-axis). Then the equation above simplifies to:
which can be solved for the estimated gravity vector g as
The SVV deviation in upright can then be calculated by setting fy = 0 and fz = 1. For small ωx, the SVV deviation is \(\beta \approx {\omega _x} \cdot \tau\). Thus, with τ = 5 s a torsional semicircular canal bias of ωx = 2°/s corresponds approximately to an SVV tilt in the upright position of 10°. The estimated gravity components are used for central processing.
In right-ear-down and left-ear-down positions, fy = ± 1 and fz = 0. Therefore, the estimated angle is \(\beta \approx 90^\circ \pm {\omega _x} \cdot \tau\), which means that in both positions the error corresponds to a rotation of the SVV in the same direction and amount as in upright.
Note that for a unilateral lesion, not only a torsional semicircular canal bias has to be assumed, but also that otolith input from one side is completely missing (see OTO model above). For the SVV model, this results in otolith input being reduced to half of its normal amplitude. However, due to the implicit normalization in the central processing (see below), this reduction of amplitude only affects the variability of the response.
Central processing
The two graviceptive inputs gy and gz (corresponding to interaural and longitudinal gravity components) derived by the OTO or SCC models described above are fed to a dynamic population of gravity-tuned neurons with firing rates fj and tuning direction φj by applying synaptic weights sin(φj) and cos(φj):
with b(t) being a time-dependent bias term derived from an inhibitory neuron dynamically adjusting the overall firing rate, τ the time constant, and [.]+ denoting the linear-threshold activation function given by g(x) = [x]+ with g(x) = 0 for x ≤ 0 and g(x) = x otherwise, to provide only positive firing rates. The second, otherwise identical layer of neurons applies synaptic weights wjk implementing the idiotropic bias. The Gaussian synaptic weight between neuron j of the first layer and neuron k of the second layer is given by \({w_{jk}}={c_k} \cdot {e^{ - {{\left( {j - k} \right)}^2}/{\sigma ^2}}}\) with ck being a scaling factor depending on the tuning direction and σ the tuning width. The resulting Gaussian tuning functions are schematically depicted in Fig. 2 (left). The SVV was read out from the network layer by standard population decoding. For simulation of lesions, we randomly set the firing rates of approximately 50% of the neurons in layer 1 to zero (Fig. 7).
Rights and permissions
About this article
Cite this article
Glasauer, S., Dieterich, M. & Brandt, T. Neuronal network-based mathematical modeling of perceived verticality in acute unilateral vestibular lesions: from nerve to thalamus and cortex. J Neurol 265 (Suppl 1), 101–112 (2018). https://doi.org/10.1007/s00415-018-8909-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-018-8909-5