Abstract
Background
The right ventricular ejection fraction (RVEF) is a surrogate marker of right ventricular function in pulmonary hypertension (PH), but its measurement is complicated and time consuming. The tricuspid annular plane systolic excursion (TAPSE) measures only the longitudinal component of RV contraction while the right ventricular fractional area change (RVFAC) takes into account both the longitudinal and the transversal components. The aim of our study was to evaluate the relationship between RVEF, RVFAC, and TAPSE according to hemodynamic severity in two groups of patients with PH: pulmonary arterial hypertension (PAH) and chronic thromboembolic pulmonary hypertension (CTEPH).
Methods and Results
Fifty-four patients with PAH (n = 15) and CTEPH (n = 39) underwent right heart catheterization and cardiac magnetic resonance (CMR). The ventricular volumes and areas, TAPSE, and eccentricity index were measured. The RVFAC was more strongly correlated with the RVEF (r = 0.81, p < 0.0001) than the TAPSE (r = 0.63, p < 0.0001). RVEF < 35% was better predicted by the RVFAC than the TAPSE (TAPSE: AUC = 0.77 and RVFAC: AUC = 0.91; p = 0.042). In the group with the worse hemodynamic status, the RVFAC correlated much better with the RVEF than the TAPSE. There were no significant differences in the CMR data analyzed between the groups of PAH and CETPH patients.
Conclusions
The RVFAC is a good index to estimate RVEF in PH patients; even better than the TAPSE in patients with more severe hemodynamic profile, possibly for including the transversal component of right ventricular function in its measurement. Furthermore, RVFAC performance was similar in the two PH groups (PAH and CTEPH).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pulmonary hypertension (PH) is an important cause of right ventricular (RV) overload, which ultimately leads to RV failure and death [1, 2]. In this sense, right ventricular function is critical for the prognosis in PH [3, 4] van de Veerdonk et al. [5] demonstrated that patients with PH and severe RV dysfunction (right ventricular ejection fraction < 35%) had worse prognosis despite the initial pulmonary vascular resistance (PVR); furthermore, a decreased right ventricular ejection fraction (RVEF) during follow-up was a stronger prognostic marker than an increased PVR, evidencing that beyond the hemodynamic impairment, the most important factor to determine the prognosis is the RV response.
Measuring the RVEF is the gold standard method to non-invasively determine RV function, but its measurement is laborious and time consuming due to the complex RV anatomy [4, 6, 7]. Taking into account the technical difficulties of non-invasively assessing RV function by measuring the RVEF, identifying other indices of RV function became necessary.
One of these indices is the tricuspid annular plane systolic excursion (TAPSE), which measures the longitudinal movement of the base of the heart toward the apex; the TAPSE is simple to obtain and highly reproductible [8]. Measurement of the TAPSE was strongly correlated with the RVEF [9] and was demonstrated to have prognostic utility in cardiopathies [10]. In PH, an initial TAPSE < 1.8 cm was associated with a worse prognosis [11]. Despite these results, only weak [12, 13] or moderate [14] correlations between the TAPSE and the RVEF were demonstrated in PH patients. The measurement of the longitudinal component seems to have limitations if there is more pronounced increases in RV afterload. Kind T et al. [13] analyzed the longitudinal and transversal components of RV contraction in patients with PH and demonstrated that the reduction in the transversal component correlated better to the RVEF than the reduction in the longitudinal axis. The transversal component better reflected the global contraction of the RV, possibly for including in its measurements the septal bulging into the LV.
In view of these findings, analysis of the right ventricular fractional area change (RVFAC) has been proposed. The RVFAC is a bidimensional measurement that includes both the longitudinal and transversal components of RV contraction (septal movement is included in the transversal component) with a strong correlation with the RVEF and good prognostic value in PH [15]. However, it is unclear if the RVFAC maintains its performance at different levels of disease severity, as well as in different forms of PH. The objective of this study was to evaluate the performance of the RVFAC, determined by cardiac magnetic resonance, in two groups of patients with PH (PAH and CTEPH) and according to different levels of hemodynamic impairment.
Methods
Study Population
This prospective study was conducted from January to December 2010 at the French referral center for severe pulmonary hypertension. The study complies with the Declaration of Helsinki. The data collection was part of a standardized diagnostic approach registry set up in agreement with the Commission Nationale de l’Informatique et des Libertés, the organization dedicated to information technology and civil rights in France (Approval Number 842,063). The population of the study consisted of patients evaluated for the first time at the Center. Patients over 18 years old presenting pre-capillary PH (mean pulmonary artery pressure ≥ 25 mmHg and pulmonary artery occlusion pressure ≤ 15 mmHg) at the time of right heart catheterization (RHC) were assessed for enrollment [2]. Patients who agreed to participate in the study had a CMR performed within 72 h of the RHC. Exclusion criteria included pregnancy, contraindications to having a CMR (known claustrophobia, pacemaker or implantable defibrillator, neurological metallic clip, ocular or cochlear metallic prosthesis, morbid obesity), PH owing to lung or left ventricular diseases, or PH with unclear multifactorial mechanisms.
Hemodynamic Evaluation
All right heart catheterizations were performed as described elsewhere [16]. The right atrial pressure, right ventricle pressures, mPAP pressure, PAoP, and heart rate were recorded for all patients. A sample of central venous blood was collected from 51 patients. Cardiac output was measured by the thermodilution technique, and a mean of three measurements was used as the final value. The PVR and stroke volume (SV) were calculated according to the following formulas:
Cardiac Magnetic Resonance Imaging
All CMRs were performed in a 1.5 T scanner (Magnetom Avanto, Siemens Medical Solutions, Germany) with electrocardiographic gating and inspiratory breath holds. Images were acquired in the short and long axes of the LV with balanced steady-state free precession pulse (SSFP) imaging. A stack of LV short-axis images was acquired to cover the whole LV from the base to the apex, with a 5 mm gap. Another stack of RV short-axis slices was acquired parallel to the tricuspid valve, without a gap, to cover the whole RV from the base to the apex, as well. Images in the four-chamber view were acquired perpendicular to the short axis of the LV. Cine images of the pulmonary artery were also acquired.
CMR Image Analysis
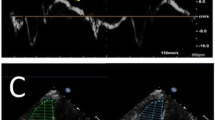
An experienced radiologist who was unaware of patients’ diagnoses and right heart catheterization results, analyzed all images. The RV end-diastolic area (RVEDA) and RV end-systolic area (RVESA) were measured by manually delineating the endocardial border of the RV in the four-chamber-view image, in end-diastole and end-systole, respectively (Fig. 1a, b). Papillary muscles were included in the volume. The RVFAC was calculated according to the following formula: RVFAC = (RVEDA − RVESA)/RVEDA.
RVFAC and TAPSE. Images in the four-chamber view. a, b illustrate the systole and diastole, respectively. RVFAC was calculated using the RV end-diastolic area (red shadow—a) and RV end-systolic area (yellow shadow—b), according to the following formula: RVFAC = (RVEDA − RVESA)/RVEDA. The TAPSE was estimated by the distance between the tricuspid annulus during diastole (point 1) and during systole (point 2)
The TAPSE was also measured in the four-chamber-view image by measuring the distance traveled by the tricuspid annulus (TA) from end-diastole to end-systole (Fig. 1c, d) [17]. The LV end-diastolic area (LVEDA) was also delineated in the four-chamber view image for the determination of the RVEDA/LVEDA.
The diameter between the anterior and posterior walls of the LV (DAW−PW) and the diameter between the septum and the LV free wall (DSEP−FW) were measured in the short-axis view at end-diastole and end-systole. The LV eccentricity index (EI) was calculated in end-diastole and end-systole as a ratio of DAW−PW/DSEP−FW.
For the measurement of the ejection fraction, the RV and LV end-diastolic and end-systolic areas were manually delineated in all slices acquired in the short-axis view for each ventricle, allowing for the assessment of end-diastolic and end-systolic volumes and the calculation of the RVEF by ARGUS Software (version VA 50C, Siemens Medical Solutions, Erlangen, Germany). In the pulmonary artery trunk cine images, the pulmonary artery trunk diameter was measured, and the pulmonary artery areas were manually delineated at maximal and minimal dilation (MaxPA and MinPA, respectively) to calculate the pulmonary artery pulsatility, where PA pulsatility = 100 x (MaxPA − MinPA)/MinPA.
Statistics
Values are presented as the mean ± standard deviation. Student’s t test was used for between-groups comparison of continuous variables; Chi-square or Fisher exact tests were used for categorical variables, as appropriate. Correlations were tested by linear regression, using the minimal squared mean. Receiver operating characteristic (ROC) curves were built to test the ability of the RVFAC and TAPSE to detect severe right ventricular dysfunction, defined by the presence of an RVEF < 35% [5, 13]. P values were considered significant when < 0.05. MedCalc® Software, version 12.2.1.0 (Mariakerke, Belgium) was used for the analysis.
Results
Fifty-four consecutive patients who had a CMR and were diagnosed with PAH (n = 15) or CTEPH (n = 39) were included in the study. Clinical and hemodynamic data are presented in Table 1. The population had a mean age of 60 years old, with the majority being female and in New York Heart Association (NYHA) functional class III. The main diagnosis in the PAH group was idiopathic PAH (12 patients). The other diagnoses were PAH associated with: anorexigen use (1 patient) and veno-occlusive disease (2 patients). When we compared the group with PAH with the group with CTEPH, patients with CTEPH were older while presenting similar mPAP and PVR.
CMR Data
There was no significant difference between the mean heart rate measured during right heart catheterization and the mean heart rate measured during CMR (80.4 ± 13.2 and 79.5 ± 11.9, respectively, p = 0.599).
In the CMR data, no differences were found between the PAH and the CTEPH, (Table 2). Severe RV dysfunction (RVEF < 35%) was found in 37 patients. The RVEF was moderately correlated with the hemodynamic variables associated with RV afterload as mPAP (r = − 0.62; p < 0.0001) and PVR (r = − 0.60; p < 0.0001) and weakly correlated to right atrial pressure (r = − 0.27; p = 0.0542). The RVEF was also correlated with the hemodynamic variables associated with RV function: weakly with CO (r = 0.36; p = 0.006) and moderately with stroke volume (r = 0.51; p < 0.0001). The RVEF was more strongly correlated with the RV end-systolic volume (r = − 0.77; p < 0.0001) than to the end-diastolic volume (r = − 0.52; p < 0.0001). Although the end-diastolic and end-systolic areas are bidimensional measurements, they were strongly correlated with the end-diastolic and end-systolic volumes (RVEDA vs. RVEDV, r = 0.81; p < 0.0001 and RVESA vs. RVESV: r = 0.88; p < 0.0001).
Correlations Between the RVEF, RVFAC, and TAPSE
The RVEF was more strongly correlated with the RVFAC (r = 0.81, p < 0.0001) than to the TAPSE (r = 0.63, p < 0.0001) (Fig. 2).
We also analyzed the correlations between the RVEF, RVFAC, and TAPSE in each group of PH patients (PAH and CTEPH) separately. In the group with PAH, the correlation between the RVFAC and RVEF (r = 0.92, p < 0.0001) was better than the correlation between the TAPSE and RVEF (r = 0.73, p = 0.002). In the group with CTEPH, the results were similar, with the RVEF correlating better with the RVFAC than with the TAPSE (RVFAC r = 0.74, p < 0.0001; TAPSE r = 0.58, p = 0.001).
CMR Data According to Hemodynamic Severity
To study RV function in two groups with different hemodynamic severities we divided the population by the median of the pulmonary vascular resistance (Table 3).
The RVEF presented a stronger correlation to the RVFAC than to the TAPSE in both groups (PVR < 8.1 WU and PVR > 8.1 WU) (Fig. 3).
Correlations between the RVEF, TAPSE, and RVFAC according to the hemodynamic severity. a, c: group with PVR < 8.5 WU; b, d: group with PVR > 8.5 WU. PVR pulmonary vascular resistance, TAPSE tricuspid annular plane systolic excursion, RVFAC right ventricular fractional area change; RVEF right ventricular ejection fraction
The ability of the RVFAC and TAPSE to predict severe right ventricular dysfunction (RVEF < 35%) [5] was tested by the comparison of the ROC curves. The RVFAC demonstrated a better performance in predicting severe right ventricular dysfunction than the TAPSE [TAPSE: AUC 0.77 (0.633–0.872) and RVFAC: AUC 0.91 (0.797–0.969), p = 0.042] (Fig. 4). For detecting an RVEF < 35% and a TAPSE < 15 mm, the sensitivity was 84% and the specificity was 71%, whereas an RVFAC < 30% had a sensitivity of 90% and specificity of 83%.
Ventricular Interaction Measurements According to Hemodynamic Severity
To quantify septal bulging, ventricular interaction measurements were analyzed. The relationship between the RV end-diastolic area and LV end-diastolic area (RVEDA/LVEDA) was significantly increased in the group with worse hemodynamic severity. In a subgroup of 41 patients (n = 18 in the group with PVR < 8.1 WU and n = 23 in the group with PVR > 8.1 WU), measurements of the LV eccentricity index measured at end-diastole and end-systole were also significantly increased in the group with worse hemodynamic severity (Table 3).
Discussion
Our study is the first study to demonstrate that in patients with more severe hemodynamic impairment, RVFAC is a better marker of right ventricular dysfunction. RVFAC performed better than TAPSE, possibly for including the transversal component of right ventricular function in its measurement. Furthermore, RVFAC better performance was similar in the two PH groups (PAH and CTEPH).
The TAPSE and RVFAC are classical indices of right ventricular function used in echocardiography, both related to survival in PH [11, 15, 18] The more regular a chamber is, more intrinsically correlated are its areas and volumes; also true for longitudinal measurements. This is the case for RV of patients with pulmonary hypertension, since the RV progressively looses its characteristic C-shape. Our study demonstrated that RV area evaluation remains strongly correlated with RVEF at later phases of the disease, whilst the longitudinal measurement progressively looses its value, even considering that the shape of RV is more “regular.” This finding suggests a change in the dynamics of the RV contraction in more severe cases.
TAPSE has been demonstrated to have a strong correlation with the RVEF, supporting that longitudinal shortening is the main component of RV function [19]. In patients with increased RV afterload, there is significant RV dilation; studies have demonstrated that, in this specific group of patients, the transversal component of RV function increases in importance; therefore, the RVFAC would be a better index of the RVEF [15, 20]. Our study confirms the findings of Mauritz et al. [15] and Kind et al. [13] by demonstrating a stronger correlation of the RVEF with the RVFAC than with the TAPSE in patients with PH, even more pronounced in later phases of the disease.
When the study groups were divided according to hemodynamic severity, the RVFAC proved to be an even better marker of the RVEF than the TAPSE in the most severe group. The strong correlation between the RVFAC and RVEF was documented regardless of the hemodynamic severity (as reflected by the pulmonary vascular resistance level), whereas the correlation between the TAPSE and RVEF was weaker in the most severe group. This finding emphasizes that in patients with pronounced increases in RV afterload, isolated analysis of the longitudinal component of the RV contraction might not be representative of RV function; thus, it is necessary to also evaluate the transversal component. The area correlation seems to remain strong, even at later phases of the disease, whilst the longitudinal measurement progressively looses its value. A low TAPSE remains as a marker of worse prognosis, however it looses sensitivity in reflecting the progressive loss of RV function, while RVFAC remains closely correlated.
Septal bulging into the LV was demonstrated in patients with PH, and this bulging is associated with reduced LV diameter measurements [21,22,23]. Marcus et al. [24] also demonstrated that septal bulging was correlated with significantly decreased LV diastolic volumes and to LV diastolic dysfunction. Accordingly, our study demonstrated that in patients with worse hemodynamics, the RVFAC, which also includes the transversal component of RV contraction, performed better than the TAPSE, which measures only the longitudinal component. We performed the measurements of ventricular interaction indices (eccentricity index and RVEDA/LVEDA) in a subgroup of patients. The worse the hemodynamic profile, the greater was the RVEDA/LVEDA ratio, reflecting RV dilation with LV compression. We observed no difference in the RV end-diastolic areas between both groups of different hemodynamic severity but did observe a significant reduction in the LV end-diastolic area in the group with worse hemodynamic severity. The LV eccentricity index was also significantly increased in the group with worse hemodynamic severity, reflecting more septal bulging with increased LV compression in this group.
When we separately compared the correlations of the RVFAC and the TAPSE with the RVEF in the two groups (PAH and CTEPH), we observed that the correlations were similar to those found in the total study population, suggesting that the type of PH does not affect these correlations. In the preliminary analysis of our data, we demonstrated that in 23 patients with PAH and 23 patients with CTEPH (paired by sex and age), there were no significant differences in the CMR data [25]. The findings of the present study reinforce the similarity of the CMR findings in PAH and CTEPH patients.
To better understand the determinants of the RVEF, the correlations of the RVEF with different hemodynamic variables were assessed. The RVEF was weakly correlated with the hemodynamic variables associated with RV overload (mPAP, PVR, and right atrial pressure) and RV function (CO and stroke volume). Furthermore, the RVEF was better correlated with the RV end-systolic volume than to the RV end-diastolic volume. Studies demonstrating increased levels of brain natriuretic peptide (BNP) in patients with PH also support the presence of increased cardiac wall stress associated with RV overload [26]. These data support the concept that RVEF is a reflection of both: the RV overload and the myocardial response to it.
The fact that the RVEF also reflects this myocardial involvement in its measurements reinforces the concept that the RVEF has prognostic importance beyond its association with the hemodynamic profile [5]. The recognition of the importance of RV function in PH might be relevant for the treatment of PH because new drugs aiming to treat RV might be used [27, 28]. It is important to emphasize that even though surrogate markers of RV function maybe important in clinical practice because they are less time consuming, thus facilitating their use in the follow-up assessment of PH patients, measurements of RV volumes and RVEF remain important to better understand many of the aspects associated with disease progression, particularly the RV remodeling process.
Our study has limitations that must be acknowledged. The study was conducted in a single center, with a potential selection bias. Right heart catheterization was not performed at the same time as CMR for technical reasons. Despite the 72 h delay, the heart rates measured during both exams were similar, reflecting essentially the same basal conditions. The CMRs were reviewed by a single expert radiologist, therefore reproducibility could not be evaluated; nevertheless, high interobserver agreement in RV function measurements through CMR has been previously demonstrated [6, 13, 29]. Furthermore, the inclusion of patients with PAH and CTEPH limits the extrapolation of our findings to other forms of PH.
In conclusion, our study demonstrated that the RVFAC, assessed by CMR, is a good index of RV function in PAH and CTEPH patients, performing better than the TAPSE in predicting severe RV dysfunction, independent of the hemodynamic severity.
References
Woods J, Monteiro P, Rhodes A (2007) Right ventricular dysfunction. Curr Opin Crit Care 13:532–540
McLaughlin VV, McGoon MD (2006) Pulmonary arterial hypertension. Circulation 114:1417–1431
van Wolfereng SA, Marcus JT, Boonstra A, Marques KMJ, Bronwaer JGF, Spreeuwenberg MD et al (2007) Prognostic value of right ventricular mass, volume, and function in idiopathic pulmonary arterial hypertension. Eur Heart J 28:1250–1257
Vonk-Noordegraaf A, Souza R (2012) Cardiac magnetic resonance imaging: what can it add to our knowledge of the right ventricle in pulmonary arterial hypertension? Am J Cardiol 110:25S–31S
van de Veerdonk M, Kind T, Marcus T, Mauritz GJ. Heymans MW, Bogaard HJ et al (2011) Progressive right ventricular dysfunction in patients with pulmonary arterial hypertension responding to therapy. J Am Coll Cardiol 58:2511–2519
Nijveldt R, Germans T, McCann G, Beek AM, van Rossum AC (2008) Semi-quantitative assessment of right ventricular function in comparison to a 3D volumetric approach: a cardiovascular magnetic resonance study. Eur Radiol 18:2399–2405
Kosiborod M, Wackers FJT (2003) Assessment of right ventricular morphology and function. Semin Respir Crit Care Med 24:245–261
Germing A, Gotzmann M, Rausse R, Brodherr T, Holt S, Lindstaedt M, Dietrich J et al (2010) Normal values for longitudinal function of the right ventricle in healthy women > 70 years of age. Eur J Echocardiogr 11:725–728
Ueti OM, Camargo EE, Ueti AA, Lima-Filho EC, Nogueira EA (2002) Assessment of right ventricular function with Doppler echocardiographic indices derived from tricuspid annular motion: comparison with radionuclide angiography. Heart 88:244–248
Lamia B, Teboul J, Monnet X, Richard C, Chemla D (2007) Relationship between the tricuspid annular plane systolic excursion and right and left ventricular function in critically ill patients. Intensive Care Med 33:2143–2149
Forfia PR, Fisher MR, Mathai SC, Hausten-Harris T, Hemnes AR, Borlaug AB, Chamera E et al (2006) Tricuspid annular displacement predicts survival in pulmonary hypertension. Am J Respir Crit Care Med 174:1034–1041
Anavekar NS, Skali H, Bourgoun M, Ghali JK, Kober L, Maggioni AP, McMurray JJ et al (2008) Usefulness of right ventricular fractional area change to predict death, heart failure, and stroke following myocardial infarction (from de VALIANT ECHO Study). Am J Cardiol 101:607–612
Kind T, Mauritz GJ, Marcus JT, van de Veerdonk M, Westhof N, Vonk-Noordegraaf A (2010) Right ventricular ejection fraction is better reflected by transverse rather than longitudinal wall motion in pulmonary hypertension. J Cardiovasc Magn Reson 12:35
Leary PJ, Kurtz CE, Hough CL, Waiss M-P, Ralph DD, Sheehan FH (2012) Three-dimensional analysis of right ventricular shape and function in pulmonary hypertension. Pulm Circ 2:34–40
Mauritz G, Kind T, Marcus JT, Bogaard H, van de Veerdonk M, Postmus PE, Boonstra A et al (2012) Progressive changes in right ventricular geometric shortening and long-term survival in pulmonary arterial hypertension. Chest 141:935–943
Provencher S, Herve P, Sitbon O, Humbert M, Simonneau G, Chemla D (2008) Changes in exercise haemodynamics during treatment in pulmonary arterial hypertension. Eur Respir J 32:393–398
Ahmad H, Mor-Avi V, Lang RM, Nesser HJ, Weinert L, Tsang W et al (2012) Assessment of right ventricular function using echocardiographic speckled tracking of the tricuspid annular motion: coparison with cardiac magnetic resonance. Echocardiography 29:19–24
Jurcut R, Giusca S, La Gerche A, Vasile S, Ginghina C, Voigt JU (2010) The echocardiographic assessment of the right ventricle: what to do in 2010? Eur J Ecocardiogr 11:81–96
Badano LP, Ginghina C, Easaw J, Muraru D, Grillo MT, Lancellotti P et al (2010) Right ventricle in pulmonary arterial hypertension. Haemodynamics, structural changes, imaging, and proposal of a study protocol aimed to assess remodeling and treatment effects. Eur J Echocardiogr 11:27–37
Brown SB, Raina A, Katz D, Szerlip M, Wiegers S, Forfia P (2011) Longitudinal shortening accounts for the majority of right ventricular contraction and improves after pulmonary vasodilator therapy in normal subjects and in pulmonary arterial hypertension. Chest 140:27–33
Dellegrottaglie S, Sanz J, Poon M, Viles-Gonzalez JF, Sulica R, Goyenechea M, Macaluso F et al (2007) Pulmonary hypertension: accuracy of detection with left ventricular septal-to-free wall curvature ratio measured at cardiac MRI. Radiology 243:63–69
Roeleveld RJ, Marcus JT, Faes TJ, Gan TJ, Boonstra A, Postmus PE et al (2005) Interventricular septal configuration at MR imaging and pulmonary arterial pressure in pulmonary hypertension. Radiology 234:710–717
Alluni JP, Degano B, Arnaud C, Tétu L, Blot-Soulétie N, Didier A et al (2010) Cardiac MRI in pulmonary artery hypertension: correlations between morphological and function parameters and invasive measurements. Eur Radiol 20:1149–1159
Marcus JT, Vonk-Noordegraaf A, Roeleveld RJ, Pieter E, Postmus PE, Heethaar RM et al (2001) Impaired left ventricular filling due to right ventricular pressure overload in primary pulmonary hypertension: noninvasive monitoring using MRI. Chest 119:1761–1765
Hoette S, Creuze N, Musset D, Jaïs X, Savale L, Natali D et al (2011) Comparing cardiac magnetic resonance imaging in group 1 and group 4 pulmonary hypertension. European Respiratory Society Annual Meeting in Amsterdam, NR 2309
Souza R, Bogossian HB, Humbert M, Jardim C, Rabelo R, Amato MB et al (2005) N-terminal-pro-brain natriuretic peptide as a haemodynamic marker in idiopathic pulmonary arterial hypertension. Eur Respir J 25:509–513
Naeije R, Ghio S (2015) More on the right ventricle in pulmonary hypertension. Eur Respir J 45:33–35
Humbert M, Sitbon O, Simonneau G (2004) Treatment of pulmonary arterial hypertension. N Engl J Med 351:1425–1436
Gnanappa GK, Rashid I, Celermajer D, Ayer J, Puranik R (2017) Reproducibility of cardiac magnetic resonance imaging (CMRI)-derived right ventricular parameters in repaired tetralogy of fallot (ToF). Heart Lung Circ. https://doi.org/10.1016/j.hlc.2017.04.017
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hoette, S., Creuzé, N., Günther, S. et al. RV Fractional Area Change and TAPSE as Predictors of Severe Right Ventricular Dysfunction in Pulmonary Hypertension: A CMR Study. Lung 196, 157–164 (2018). https://doi.org/10.1007/s00408-018-0089-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-018-0089-7