Abstract
Purpose
This study aimed at investigating the association of severity of obstructive sleep apnea with the markers of inflammation as fibrinogen to albumin ratio and C-reactive protein to albumin ratio.
Methods
We included 126 patients admitted to sleep disorders outpatient clinic. We constituted four groups by reference to the apnea–hypopnea index (mild-moderate-severe sleep apnea and control group). We investigated the difference of white blood cell, fibrinogen to albumin ratio, C-reactive protein to albumin ratio, neutrophil to lymphocyte ratio, platelet to lymphocyte ratio, and hemoglobin among the groups.
Results
Neutrophil to lymphocyte ratio, platelet to lymphocyte ratio, and hemoglobin did not significantly differ among four groups (p = 0.39, p = 0.06, and p = 0.31, respectively). A significant difference of fibrinogen to albumin ratio and C-reactive protein to albumin ratio was evident among four groups (p < 0.001). Fibrinogen to albumin ratio and C-reactive protein to albumin ratio did not significantly differ between the mild sleep apnea and control groups (p = 0.65, and p = 0.85, respectively), but were significantly greater in moderate sleep apnea group compared with the control group (p < 0.001 and p = 0.001, respectively). Also, fibrinogen to albumin ratio and C-reactive protein to albumin ratio were significantly greater in the severe sleep apnea group compared with the control group (p < 0.001). Fibrinogen to albumin ratio and C-reactive protein to albumin ratio did not significantly differ between the severe sleep apnea and moderate sleep apnea groups (p = 0.49, and p = 0.58, respectively).
Conclusion
Higher fibrinogen to albumin ratio and C-reactive protein to albumin ratio may be predictive of inflammation in patients with moderate-to-severe sleep apnea.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive sleep apnea syndrome (OSA), the most common sleep disorder, is characterized by recurrent episodes of apnea and hypopnea that bring about temporary oxygen desaturation. OSA influences approximately 4% of the population, also 35–45% of humans with obesity, and constitutes an important health hazard posing an additional risk for cardiovascular disorders like arrhythmias, arterial hypertension, coronary heart disease, and stroke [1]. Repetitive periods of hypoxia caused by transient respiratory interruption increases the oxidative stress and systemic inflammation that promotes the formation of tissue and cell damage in patients with OSA [2]. OSA-related systemic inflammation may contribute to the risk of development of various autoimmune diseases like systemic lupus erythematous, Sjogren syndrome, rheumatoid arthritis, and ankylosing spondylitis [3].
C-reactive protein (CRP), a well-known acute phase reactant, has been associated with a variety of diseases thus far, including acute coronary syndrome, ischemic stroke, and facial paralysis [4, 5]. In addition, a significant association between CRP levels and some types of cancer was reported [6]. Although levels of albumin, a negative acute phase protein, decrease in cases of acute inflammation, it decreases mainly in chronic inflammation and bad nutritional conditions. The evaluation of CRP to albumin ratio (CAR) may have a prognostic significance in short- and long-term inflammation. Another acute phase reactant, plasma fibrinogen, is one of the most important coagulation proteins increasing in inflammatory diseases [7]. Fibrinogen to albumin ratio (FAR) was reported as a predictive marker for the severity of various type of diseases [7,8,9].
Although several studies have evaluated the association of some inflammatory markers with OSA [10, 11], no previous study investigated the association of FAR and CAR with the severity of OSA, to the best of our knowledge.
The goal of this prospective, controlled, clinical study was to investigate the association of severity of OSA with the markers of chronic inflammation including FAR, CAR, Neutrophil to lymphocyte ratio (NLR), Platelet to lymphocyte ratio (PLR), and white blood cell (WBC). We included these blood parameters in our analysis because of their potential association with the systemic inflammatory base of OSA. Additionally, providing the cut-off values of significant inflammation markers was the secondary goal of this study.
Materials and methods
Patients, groups* and study design
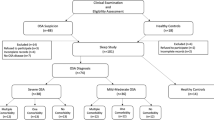
This prospective, controlled, clinical study was conducted in line with the dictates of the World Medical Association Declaration of Helsinki and approved by the local ethic committee (IRB Number: 90139838-000-E.44913). We included 126 patients from two different institutions who were admitted to sleep disorders outpatient clinic. We took a detailed medical history of all patients, performed a comprehensive otolaryngologic and rhinologic examination, a laboratory analysis and a whole-night PSG (type 1) (Alice® 5 Diagnostic Sleep System, Respironics, Murrysville, PA, USA, linked to the Alice® Sleepware™ software). Excluded from the study were the patients with any systemic, autoimmune, inflammatory and/or nutritional disease that might affect the level of blood parameters, pregnancy, acute trauma, and a history of malignancy. We recruited the data of the patients who were admitted to outpatient clinic of sleep disorders with any compliant related to sleep disorders including snoring, witnessed apnea, daytime sleepiness, insomnia and periodic leg movements, etc., and those patients that had undergone PSG for the anxiety of having OSA (without any symptom of OSA). Among these patients, the patients of who we were sure that they did not have OSA [those with apnea–hypopnea index (AHI) under 5] were included in the control group. Thus, we ensured that our control group did not contain any patient with OSA. The study groups consisted of the patients with the diagnosis of OSA (those with AHI over 5). For the comparison of blood test parameters, we constituted four groups by reference to the AHIs of the patients: The control group (AHI ≤ 5), the mild OSA group (AHI = 6–15), the moderate OSA group (AHI = 16–30) and the severe OSA group (AHI > 30).
Laboratory evaluation
We used an automated blood cell counter for complete blood count (Mindray BC-6000, Shenzhen, China). We analyzed Serum albumin levels using Abbott C8000i (Abbott Park, IL, USA) automatic photometry commercial kits. We measured Serum CRP levels using nephelometric method (AU5800 System; Beckman Coulter Inc, Brea, CA, USA) and fibrinogen levels by Clauss method using a BCS Analyzer (Multifibren U; Siemens Healthcare, Erlangen, Germany). We noted the blood test parameters including hemoglobin (Hgb), fibrinogen, CRP, neutrophil count, lymphocyte count, platelet count, WBC and albumin. Then, we calculated FAR, CAR, NLR and PLR values of the groups.
First, we compared the FAR, CAR, NLR, PLR, Hbg and WBC values of the groups. Then, we investigated the most significant indicator parameter associated with the severity of OSA.
Statistical analysis
Results are presented as mean ± standard deviation for normally distributed data and median (min–max) for abnormally distributed data. To investigate the distribution pattern of the data, we used Kolmogorov–Smirnov normality test. We used One-way analysis of variance (ANOVA) to compare the mean WBC values of the groups and Kruskal–Wallis test to compare the median FAR, CAR, NLR, PLR and Hbg values of the groups. For advanced comparisons of FAR and CAR, we used Mann–Whitney U test, as post-hoc test. To detect the most significantly associated parameter with the severity of OSA, and to determine the cut-off values, we used receiving operator characteristics (ROC) curve analysis test. For statistical analysis of all data, we used SPSS 16.0 software for Windows (SPSS Inc., Chicago, IL, USA). We considered a p value less than 0.05 as statistically significant. Additionally, for post-hoc comparison tests, we used Bonferroni correction of four groups (hexed combination) and a p value less than 0.008 (0.05/6) was considered statistically significant.
Results
One hundred twenty-six patients were eligible for the study. The mild OSA group consisted of 39 patients (20 males and 19 females, mean age: 48 ± 12), the moderate OSA group consisted of 33 patients (17 males and 16 females, mean age: 49 ± 10), the severe OSA group consisted of 26 patients (13 males and 13 females, mean age: 50 ± 10) and the control group consisted of 28 patients (13 males and 15 females, mean age: 48 ± 11). The groups were age and gender- matched (p = 0.9).
The mean WBC values and the median FAR, CAR, NLR, PLR and Hgb values of the groups are presented in Table 1. One-way ANOVA analysis revealed that the mean WBC values did not significantly differ among four groups (p = 0.24). According to the Kruskal–Wallis test, median NLR, PLR and Hgb values did not significantly differ among four groups (p = 0.39, p = 0.06, and p = 0.31, respectively). However, a statistically significant difference of median FAR and CAR was evident among four groups (p < 0.001).
Advanced comparisons revealed that both median FAR and CAR values did not significantly differ between the mild OSA group and the control group (p = 0.65, and p = 0.85, respectively). On the other hand, both median FAR and CAR values were significantly greater in the moderate OSA group compared with the control group (p < 0.001, and p = 0.001, respectively). Additionally, both median FAR and CAR values were significantly greater in the severe OSA group compared with the control group (p < 0.001). However, median FAR and CAR values did not significantly differ between the severe OSA and moderate OSA groups (p = 0.49, and p = 0.58, respectively). Thus, we found that moderate to severe OSA was significantly associated with elevated FAR and CAR levels.
Figure 1 represents the graph of ROC analysis in which FAR and CAR were included as an indicator of moderate to severe OSA. The area under curve of FAR was greater (0.856) than of CAR (0.766). The cut-off value of FAR for moderate to severe OSA was found as 70.76, with a sensitivity of 79% and specificity of 72%. In addition, the cut-off value of CAR for moderate to severe OSA was found as 0.81, with a sensitivity of 74% and specificity of 72%.
Discussion
Increased levels of inflammatory parameters as FAR and CAR might have a clinical implication as markers of the severity of OSA. In this study, the FAR and CAR levels of the patients with moderate-to-severe OSA were significantly greater compared with the control patients. On the other hand, FAR and CAR levels were not associated with mild OSA. These results suggest the FAR and CAR as the predictors of moderate-to-severe OSA.
OSA is caused by repetitive upper respiratory collapse that occurs during sleep and might have a significant association with the increase in systemic inflammatory markers. Hypoxia, caused by obstruction in the upper respiratory tract, might be the main cause of this increase in inflammatory process and markers. In addition, a positive correlation was found between OSA and cardiovascular/cerebrovascular diseases [12]. In patients with OSA, oxygen desaturation caused by apnea periods, along with increased negative intrathoracic pressure and sympathetic system activation, triggers a series of humoral, neural, metabolic and thrombotic events that may cause atherosclerosis, and many of these conditions are associated with inflammation [13]. Previous studies investigated the association of several inflammatory markers like WBC, NLR and PLR with OSA. Wu et al. [14] published a meta-analysis about the hematological parameters used to determine the severity of OSA. Koseoglu et al. [15] reported decreased PLR levels in patients with OSA, but NLR levels of OSA patients were not different from the NLR levels of the controls. In our study, no statically significant difference of WBC, NLR and PLR was evident between OSA groups and control group. Our results suggest that WBC, NLR and PLR might be insufficient for prediction of the severity of OSA.
CRP, an acute phase reactant, is produced by hepatocytes in the liver in response to cytokines during inflammatory events and significantly increases due to inflammation and infection. In the prior literature, a positive correlation was reported between inflammation/infection severity and CRP levels [16]. Albumin, a negative acute phase protein, helps interpret the prognosis of related events in inflammation and infection processes and is also closely associated with the oxidative stress. In our study, we found significantly greater CAR values in moderate to severe OSA patients compared with the control group. Increased incidence of cardiovascular and autoimmune diseases related to chronic inflammation in patients with OSA might be associated with higher CAR values. Based on our results, we can assume that greater CAR values in moderate-to-severe OSA patients might be predictive of inflammation and the complications that may develop.
Many previous studies reported that patients with OSA had an increased risk of cardiovascular and cerebrovascular events [17]. Fibrinogen, another acute phase reactant, is the main plasma coagulation factor synthesized by hepatocytes. On the other hand, there is a reverse relationship between serum levels of albumin and inflammatory conditions. Additionally, OSA may cause a decrease in serum albumin levels due to an increased inflammatory process. An association of hypoalbuminemia with endothelial dysfunction, another possible cause of cardiovascular events in OSA patients, was found; and decreased albumin levels was reported to cause an increase in blood viscosity, and thence endothelial dysfunction [18]. Thus, increased FAR was identified as a potential risk factor for cardiovascular diseases [8]. We determined that the FAR values were significantly greater in moderate to severe OSA group compared to the controls (p < 0.001). Thus, we can hypothesize that greater FAR values in patients with moderate to severe OSA might be an indicator of cardiovascular complications. Moreover, our comprehensive literature review revealed that no previous study investigated the association of FAR with OSA.
In the ROC curve analysis to test the capacity of FAR and CAR in demonstrating the severity of OSA, the area under curve of FAR was greater than of CAR. The cut-off value of FAR for moderate to severe OSA was found as 70.76, with a sensitivity of 79% and specificity of 72%. In addition, the cut-off value of CAR for moderate to severe OSA was found as 0.81, with a sensitivity of 74% and specificity of 72%. These results suggest that both FAR and CAR are associated with moderate to severe OSA; however, FAR has a higher predictive value for severity of OSA. Nonetheless, physicians may use both parameters for prediction of OSA severity, since the sleep- disordered patients with FAR value over 70.76 and CAR value over 0.81 carry the risk of having moderate to severe OSA. However, no inflammatory marker (including FAR and CAR) can be replaced to polysomnography, because whole night polysomnography is the gold standard method for diagnosing and classifying (based on the severity) the patients with OSA. FAR and CAR cannot be an alternative of polysomnography but might be used by sleep physicians to determine the priority of OSA patients for polysomnography. A priority for polysomnography (as an earlier appointment) might be given to the patients with increased FAR and CAR because of the increased risk of having moderate-to-severe OSA.
Our study provided the predictive blood parameters of moderate to severe OSA, however, the major limitation was the relatively small sample size, leading the lack of generalization. Thus, our results need a confirmation by further prospective-multicenter investigations utilizing larger study populations.
Conclusion
In conclusion, higher FAR and CAR values may be predictive for inflammation in patients with moderate-to-severe OSA. Both FAR and CAR might be considered as the novel potential parameters to predict the severity of OSA. The sleep-disordered patients with FAR value over 70.76 and CAR value over 0.81 have the risk of having moderate-to-severe OSA.
References
Tan S, Liu X, Xu Y, Luo L, Zhou S, Gao Y (2017) Serum high-density lipoprotein correlates with serum apolipoprotein M and A5 in obstructive sleep apnea hypopnea syndrome. Sleep Breath 21(1):37–44
DeMartino T, Ghoul RE, Wang L, Bena J, Hazen SL, Tracy R, Patel SR, Auckley D, Mehra R (2016) Oxidative stress and inflammation differentially elevated in objective versus habitual subjective reduced sleep duration in obstructive sleep apnea. Sleep 39(7):1361–1369
Kang J-H, Lin H-C (2012) Obstructive sleep apnea and the risk of autoimmune diseases: a longitudinal population-based study. Sleep Med 13(6):583–588
Zhang X, Wang A, Zhang J, Singh M, Liu D, Zuo Y, Wu L, Song M, Wang W, Feigin VL, Wang Y, Zheng D (2019) Association of plasma C-reactive protein with ischemic stroke: a Mendelian randomization study. Eur J Neurol. https://doi.org/10.1111/ene.14113
Cayir S, Hizli O, Kayabasi S (2019) Is C-reactive protein to albumin ratio an indicator of poor prognosis in Bell's palsy? Eur Arch Otorhinolaryngol. https://doi.org/10.1007/s00405-019-05691-3
Liu Z, Jin K, Guo M, Long J, Liu L, Liu C, Xu J, Ni Q, Luo G, Yu X (2017) Prognostic value of the CRP/Alb ratio, a novel inflammation-based score in pancreatic cancer. Ann Surg Oncol 24(2):561–568. https://doi.org/10.1245/s10434-016-5579-3
He Y, Tang J, Wu B, Yang B, Ou Q, Lin J (2019) Correlation between albumin to fibrinogen ratio, C-reactive protein to albumin ratio and Th17 cells in patients with rheumatoid arthritis. Clin Chim Acta. https://doi.org/10.1016/j.cca.2019.10.009
He D, Jiao Y, Yu T, Song J, Wen Z, Wu J, Duan W, Sun N, Sun Z, Sun Z (2019) Prognostic value of fibrinogen-to-albumin ratio in predicting 1-year clinical progression in patients with non-ST elevation acute coronary syndrome undergoing percutaneous coronary intervention. Exp Ther Med 18(4):2972–2978. https://doi.org/10.3892/etm.2019.7890
Wang YY, Liu ZZ, Xu D, Liu M, Wang K, Xing BC (2019) Fibrinogen-albumin ratio index (FARI): a more promising inflammation-based prognostic marker for patients undergoing hepatectomy for colorectal liver metastases. Ann Surg Oncol 26(11):3682–3692. https://doi.org/10.1245/s10434-019-07586-3
Bozkus F, Dikmen N, Samur A, Bilal N, Atilla N, Arpag H (2018) Does the neutrophil-to-lymphocyte ratio have any importance between subjects with obstructive sleep apnea syndrome with obesity and without obesity? Tuberk Toraks 66(1):8–15. https://doi.org/10.5578/tt.66535
Korkmaz M, Korkmaz H, Kucuker F, Ayyildiz SN, Cankaya S (2015) Evaluation of the association of sleep apnea-related systemic inflammation with CRP, ESR, and neutrophil-to-lymphocyte ratio. Med Sci Monit 21:477–481. https://doi.org/10.12659/msm.893175
Pafili K, Steiropoulos P, Papanas N (2015) The relationship between obstructive sleep apnoea and coronary heart disease. Curr Opin Cardiol 30(4):439–446
Shamsuzzaman AS, Gersh BJ, Somers VK (2003) Obstructive sleep apnea: implications for cardiac and vascular disease. JAMA 290(14):1906–1914
Wu M, Zhou L, Zhu D, Lai T, Chen Z, Shen H (2018) Hematological indices as simple, inexpensive and practical severity markers of obstructive sleep apnea syndrome: a meta-analysis. J Thorac Dis 10(12):6509–6521. https://doi.org/10.21037/jtd.2018.10.105
Koseoglu S, Ozcan KM, Ikinciogullari A, Cetin MA, Yildirim E, Dere H (2015) Relationship between neutrophil to lymphocyte ratio, platelet to lymphocyte ratio and obstructive sleep apnea syndrome. Adv Clin Exp Med 24(4):623–627. https://doi.org/10.17219/acem/47735
Sproston NR, Ashworth JJ (2018) Role of C-reactive protein at sites of inflammation and infection. Front Immunol 9:754. https://doi.org/10.3389/fimmu.2018.00754
Baratta F, Pastori D, Fabiani M, Fabiani V, Ceci F, Lillo R, Lolli V, Brunori M, Pannitteri G, Cravotto E, De Vito C, Angelico F, Del Ben M (2018) Severity of OSAS, CPAP and cardiovascular events: a follow-up study. Eur J Clin Investig 48(5):e12908. https://doi.org/10.1111/eci.12908
Zhang W-J, Frei B (2002) Albumin selectively inhibits TNFα-induced expression of vascular cell adhesion molecule-1 in human aortic endothelial cells. Cardiovasc Res 55(4):820–829
Funding
The authors declared that this study has received no financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of local ethical committee of Giresun University (IRB Number: 90139838-000-E.44913).
Informed consent
Informed consent was obtained from the participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on sleep apnea syndrome. Guest Editors: Manuele Casale, Rinaldi Vittorio.
Rights and permissions
About this article
Cite this article
Hizli, O., Cayir, S., Coluk, Y. et al. The novel indicators of moderate to severe sleep apnea: fibrinogen to albumin ratio vs. CRP to albumin ratio. Eur Arch Otorhinolaryngol 278, 851–855 (2021). https://doi.org/10.1007/s00405-019-05770-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-019-05770-5