Abstract
The amplitude of otoacoustic emissions (OAE) is known to decrease with increasing age, but it is still unclear whether this is due to aging alone or to age-related hearing loss. This study describes the exploration of a large database (5,142 patients from 0.4 to 89.8 years) collected in a routine clinical testing. Reliable pure tone audiograms, transitory evoked otoacoustic emissions (TEOAE) and distortion product otoacoustic emissions (DPOAE) recordings were available from 5,424 ears without conductive loss, acute sudden deafness or retrocochlear disorder. From this database, group 1 with behavioral thresholds of 10 dB HL or better at all frequencies from 1 to 4 kHz and group 2 with age-accordant thresholds after ISO 7029 were formed. In both groups, the OAE amplitude decreased with increasing age, but in group 1, the effect was significant only for DPOAE recorded at 3 and 4 kHz. In group 2, the loss of amplitude was steeper and highly significant for TEOAE as well as DPOAE at all frequencies, but most pronounced at high frequencies. These findings support the hypothesis that the reduction of OAE amplitude with increasing age is primarily caused by age-linked hearing loss and not by aging alone.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It is well known that the amplitude of transitory evoked otoacoustic emissions (TEOAE) and distortion product otoacoustic emissions (DPOAE) decreases with age. Nevertheless, the question whether this loss of OAE (otoacoustic emission) amplitude is caused by aging alone or by age-related hearing impairment remains unanswered. The literature reports dedicated to this topic are not consistent. Bonfils et al. [6] examined 151 ears of subjects of age 2–88 years and found that the incidence and amplitude of TEOAE decreased with increasing age, while the response threshold increased. Similarly, Collet et al. [9] reported that the presence of TEOAE decreased with increasing age, and their frequency peak was shifted to lower values. The first study related to the distortion products revealed that also the DPOAE magnitude decreased steadily with age, particularly at higher frequencies, and that the response threshold increased as a function of age [23]. This observation has been confirmed later by Castor et al. [7], who tested 60 subjects between “young” and 88 years and inferred that “the alterations found in TEOAE and DPOAE seem to be essentially related to age-linked hearing loss”. In another study on 115 ears of 142 subjects from 15 to 89 years, the DPOAE level was found to be weakly correlated with the age of the subject [18]. According to Bertoli and Probst [5], who analyzed the TEOAE of 201 subjects from 60 to 97 years, there was no apparent influence on TEOAE level due to aging alone. The benchmark data of these and further publications, which consistently report an influence of age on OAE parameters, are summarized in Table 1. The only statements with reference to the question whether aging alone or age-related hearing loss is responsible for the alteration of OAE are found in the studies of Stover and Norton [36], Prieve and Falter [29], He and Schmiedt [14] and Dorn et al. [11]. While the first and second assert that “age alone does not account for a significant portion of the variability in the data” and “age does not significantly reduce TEOAE level nor increase TEOAE threshold”, the latter two concede that “the differences in the pure tone thresholds … do not fully explain the differences in the … responses” and “there may be processes intrinsic to aging alone that act on DPOAE generation”.
In view of this background, it appeared opportune to perform a cross-sectional study on a large sample of individuals to separate the effect of the aging cochlear amplifier from that of decreasing hearing sensitivity. The availability of a large inventory of clinical OAE data motivated us to elaborate the analysis described in the present work. This data pool appeared to fulfill the essential condition, in that it included the elderly with normal threshold as well as younger subjects with elevated threshold. Hence, the material should be suitable for a fruitful enrichment and possibly clarification of the ongoing discussion on whether OAEs anticipate a loss of hearing sensitivity before it is clinically present.
Patients and methods
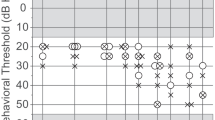
The data were selected for evaluation out of 10,284 ears examined in the time period from November 1992 to May 2005 in the Audiology Unit of the Univ.-HNO-Klinik Heidelberg, in accordance with the approval by the institutional review board of the university. In spite of the long period during which data were acquired, all patients were tested with the same or equivalent methods and equipments. The age of all patients in the unselected database ranged from 0.4 to 89.8 years. Patients with conductive hearing loss, retrocochlear disorders and those during the acute phase after sudden hearing loss were excluded from evaluation. Among the remaining cases, patients were considered only if a reliable pure tone audiogram (PTA) was available (this restriction includes the limitation to subjects older than 10 years) and OAE were recorded under standard conditions (see next paragraph for details). After this selection, the database contained 5,424 ears for further analysis. Out of these, one group containing only normally hearing ears (group 1) and a second group containing ears with a threshold within the normal limits according to age (group 2) were constituted. The criterion for inclusion into group 1 was a pure tone threshold lower than or equal to 10 dB HL at all frequencies between 1 and 4 kHz. It was fulfilled by 499 ears. The criterion for inclusion into group 2 was a pure tone threshold within the 90 percentile around the median, according to the ISO 7029 standard for otologically normal persons [17] in the frequency range 1–4 kHz. This criterion was fulfilled by 1,005 ears. Patients belonging to group 1 exhibited normal hearing thresholds (limit = 10 dB HL) independent of age and frequency, whereas the thresholds of group 2 patients were normal at frequencies below 1 kHz and elevated at higher frequencies, the elevation depending on gender and age and amounting to more than 40 dB HL at 4 kHz (Fig. 1).
Pure tone audiometry was conducted in a sound-treated booth using an Auritec AT 335 audiometer with Beyer DT-48 supra-aural headphones calibrated according to ISO standards. OAEs were recorded from both ears using the ILO92 system (software V5.60) with the patient sitting comfortably in a sound-treated booth and separated spatially from the PC and examiner. Nonlinear TEOAE mode with standard parameters (20 ms repetition rate, 260 averages) was applied with stimulus levels adjusted in the individual ear canal to 80 dB SPL pe with ±5 dB tolerance. Data with a residual noise level exceeding the limit of 5 dB SPL were excluded from the analysis. DPOAE were elicited by two primary tones with frequencies f 1 and f 2 = 1.2 × f 1. A DP-gram was recorded for f 2 = 1, 1.5, 2, 3 and 4 kHz at L 1 = L 2 = 70 dB SPL with ±3 dB tolerance. The high levels of stimuli are not especially suitable for the extraction of frequency-specific responses, but most common in clinical routine testing because they are known to elicit stable responses in the majority of ears with less than 50 dB hearing loss [15]. Responses were regarded present if the true amplitude of pure OAE (corrected for residual noise assuming a linear superposition of the corresponding variances) exceeded the estimated background noise by at least one standard deviation. All data that did not fulfill this condition were excluded from further analysis. This ensures that only reliable OAE amplitudes are considered, although it produces a bias with respect to the OAE capability of detecting hearing loss.
After selection of data, true (corrected) OAE amplitudes were classified in groups according to age and hearing loss and analyzed in terms of scatter plots, linear regression and correlation, frequency distributions, mean values, standard deviation and standard errors. The significance of correlations was tested by an analysis of variance according to Student’s F distribution.
Results
Whenever OAE amplitudes of different individuals are compared and analyzed numerically, the most impressive feature is their large variability. This holds true even in ears with normal hearing threshold (hearing loss between 0 and 10 dB HL, group 1) and within restricted age limits. As can be seen in Fig. 2, the OAE amplitudes observed in group 1 are extremely variable but in the average they decrease with increasing age. This is illustrated by the linear regression, which shows a negative slope for click-evoked TEOAE as well as for DPOAE measured at five different stimulus frequencies. The absolute value of the slope ranges from 0.019 to 0.089 dB/year (Table 2). This decay of amplitude corresponds to a loss of 6 dB (bisection of amplitude) in 67–324 years (“half-life period”).
The relation between age and OAE amplitude can be described quantitatively by the correlation coefficients, which are derived from the linear regression. Within the normal hearing group 1, all coefficients are small, the correlation between age and OAE amplitude being significant for DPOAE at 3 kHz and highly significant for DPOAE at 4 kHz (Table 2). This means that elderly subjects exhibit significantly reduced OAE amplitudes at high frequencies, though their hearing thresholds lie at or below 10 dB HL for all frequencies in the range of 1–4 kHz.
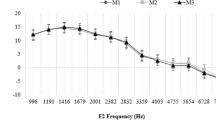
Mean values and standard deviations of OAE levels have been calculated for each decade of age. In Fig. 3, these mean values and corresponding standard errors are shown for group 1.
The analysis of ears with age-appropriate hearing loss (group 2) yields results, which exhibit some similarities to those of the normal hearing group (Figs. 4, 5). Since the reference values in ISO 7029 are gender specific, the results for male and female subjects are presented separately (Fig. 4a–f, g–l). The common feature of all scatter plots is the large variability of the amplitude data and the negative slope of the linear regression. Systematic differences can be seen in the absolute values, which are slightly larger in females than in males for TEOAE and for DPOAE measured at high frequencies.
In contrast to Fig. 2 (group 1), the inclination of the lines fitted to the data is not independent of emission type and stimulus frequency. This holds true especially in male subjects, where a steep gradient is found for DPOAE measured at high stimulus frequencies (Fig. 4g–l). The absolute value of the slopes ranges from 0.073 to 0.185 dB/year for female ears (Table 3a) and from 0.055 to 0.196 dB/year for male subjects (Table 3b). This decay of amplitude corresponds to half-life periods (50% amplitude reduction) between 32 (31) and 82 (109) years for female (male) subjects. All correlation coefficients describing the relation between age and DPOAE amplitude are larger than in group 1. As can be seen from the test parameter t 0, all correlations are highly significant and larger than in group 1.
Regarding the mean values and standard errors, the age-dependent amplitude loss of OAE in group 2 (persons with age-accordant threshold, Fig. 5) is very similar to that observed in group 1 (persons with normal threshold, Fig. 2). As in the scatter plots, significant differences between the groups occur only at high frequencies (f 2 = 3 and 4 kHz) and mainly for male subjects. This is not surprising since according to ISO 7029, substantial hearing loss is encountered only at high frequencies, especially in males (see Fig. 1). The amplitude of DPOAE evoked by tone stimuli of low frequency (f 2 = 1 and 1.5 kHz), as well as the amplitude of TEOAE, shows the same behavior in normal hearing ears and in ears with age-accordant hearing loss. This can be explained by the fact that low-frequency threshold is only marginally affected by aging. Click-evoked TEOAE are fed by low as well as high-frequency stimulus components and therefore less sensitive to high-frequency hearing loss.
It is the central purpose of the present investigation to explore the effect of hearing loss and age on OAE amplitude and to separate both effects. The results presented so far originated from the analysis of defined groups constituted from the pool of available data, which suggest that the largest portion of amplitude loss can be explained by the threshold shift caused by aging and not by aging alone. The analysis of all data without any selection shows a substantial influence of age on both hearing loss and OAE amplitude (Fig. 6). All correlations r are highly significant as has been evidenced by considering the test quantity \( t_{0} = r\sqrt {{\frac{n - 2}{{1 - r^{2} }}}} \) (n = number of data pairs), which follows the F distribution of Student. The similarity of the graphs showing the age dependence of hearing loss (Fig. 6a) and of OAE amplitude (Fig. 6b) support the finding that the loss of OAE amplitude with increasing age is mediated by the shift in age-related threshold.
In the interpretation of data, there are some special aspects of the data acquisition methods that deserve particular attention. For clinical purposes, TEOAE are conventionally recorded in the nonlinear mode, which eliminates all signal components that grow linearly with increasing stimulus. The conclusions drawn from the TEOAE data are therefore somewhat limited in that they are blind to the effects that are determined by the linear operation of the cochlea. Some effects of aging may therefore be invisible, particularly since cochlear nonlinearities are supposed to be only marginally affected by age [14]. Another restriction results from the fact that nonlinear TEOAE recording operates in the range of saturated responses. Therefore, the nonlinear mode may be less suitable than the linear mode to reflect subtle changes in cochlear performance [16]. Similarly, the inclusion of spontaneous OAE (SOAE) and synchronized SOAE (SSOAE) could have augmented the amount of information since their incidence and amplitude are known to depend on age [29]. With respect to the exploration of DPOAE, a resolution of two frequencies per octave was selected. With this default setting, the observation of DPOAE fine structure, the age dependence of which has been described elsewhere [14], is not possible. All these restrictions are necessary to combine the collection of experimental data with the demands of practicability in a clinical environment.
Discussion
The objective assessment of auditory function is one of the most important challenges in current audiology. In the context of early intervention and prevention, the reliable detection of hearing loss has special importance in newborns and in the elderly. Otoacoustic emissions (OAE) are particularly suitable for the detection of inner ear dysfunctions and are increasingly applied in clinical and practical audiology. Unfortunately, the results obtained from OAE measurements are of limited use because of their large variability. Age could be one of the factors accounting for this variability. Its influence on OAE amplitude is subject matter of the present work. The question of interest is whether age is an independent factor influencing the properties of OAE or whether the well-known age dependence of OAE amplitude is a consequence of age-linked hearing loss.
The data presented in this cross-sectional analysis confirm the observation that OAE amplitude decreases with increasing age and strongly support the hypothesis that this amplitude loss is primarily an effect of age-related hearing impairment. The effect only of aging was separated from that of hearing loss due to aging by selecting patients with normal or near normal hearing (maximum hearing loss 10 dB HL within the frequency range of 1–4 kHz) out of the largest sample of clinical data, which has ever been described in this context. Our data show a moderate loss of TEOAE and DPOAE amplitude in the group of normal hearing persons and a much more pronounced age dependence in the group of persons with age-accordant (worse) hearing threshold within the limits defined by ISO 7029.
In the literature synopsis given in “Introduction”, the inconsistency of statements concerning the interrelation between OAE and aging becomes obvious. Various authors [1, 2, 10, 13, 20, 22, 24, 27, 28, 30, 34] articulate their conviction that OAE parameters may generally be able to indicate a beginning hearing loss caused by age, noise or ototoxic agents before it is measurable with pure tone threshold, while others [4, 8, 21] accentuate that this might well be possible, but may not yet be regarded as proven. Considering this survey, it must be kept in mind that the changes in cochlear functionality caused by aging are different from those resulting from noise exposure or ototoxicity. Nevertheless, the general capability of OAE to anticipate age-related cochlear damage remains controversial.
Our data show a consistent and significant amplitude loss of TEOAE and DPOAE with increasing age in subjects exhibiting a cochlear hearing loss typical for their age according to ISO 7029 (Table 3), but no significant loss of OAE amplitude (exception: DPOAE recorded at f 2 = 3 and 4 kHz) in the group of normal hearing subjects (Table 2). In practical audiology, the age dependence of OAEs in ears with threshold according to ISO 7029 is associated with an amplitude loss of 0.6 dB per decade of age (for TEOAE and DPOAE recorded at low frequencies) to 2.0 dB per decade (for DPOAE recorded at high frequencies).
The weak age dependence of OAE amplitude observed in normal hearing ears is significant only at high frequencies, where it might be attributed to a possible hearing loss at frequencies beyond the frequency range considered here (1–4 kHz). If the criterion for inclusion in the normal hearing group is extended to higher frequencies, the number of data falls below the limit of a meaningful statistical evaluation. On the other hand, if normal threshold is claimed only for one frequency (e.g., threshold at 1 kHz for DPOAE measured with f 2 = 1 kHz), a strong negative correlation between age and OAE amplitude is observed, which in reality is caused by threshold elevations at adjacent frequencies. In fact, the amplitude of DPOAE correlates dominantly with the hearing loss at the stimulus frequency, but the influence of neighboring frequency regions is considerable (Fig. 7). The correlation between TEOAE amplitude and pure tone hearing loss is most pronounced at high audiometric frequencies (Fig. 7). This means that small TEOAE amplitudes indicate a hearing loss at high frequencies, even higher than those represented in the TEOAE spectrum.
In view of these results, the statement “… intrinsic aging of the cochlear amplifier affects sensitivity and OAEs differentially” [36] appears in a new light. Alterations of the properties of OAEs without affection of the sensitivity of hearing as it is mirrored in pure tone threshold cannot be ruled out definitely, but they may be present, if ever, only to a very small extent. Hypothetically, a decrement of OAE amplitude in normal hearing ears could be ascribed to the growth of the external ear canal. This effect can be ruled out in our results since the data analysis has been confined to subjects older than 10 years. Alternatively, a loss of cochlear sensitivity may well be present in ears, which exhibit a normal threshold in the range of frequencies between 1 and 4 kHz. Correlations between OAE magnitude and the “ultra-high-frequency hearing threshold” (8–16 kHz) have been described in humans [3, 33] and earlier in guinea pigs [2]. According to these observations, the OAE could reflect a cochlear damage located in cochlear regions, which are tuned to frequencies higher than the OAE frequencies. This coincides with the observation that the hearing threshold in the extended high-frequency range is a sensitive early indicator of noise-induced hearing loss [28], although other reports are contradictory [32]. Apart from the impact of noise, there is evidence for a tight relation between the amplitude of TEOAE and DPOAE and a better hearing sensitivity in the extended high-frequency region [33]. On the other hand, even for frequencies within the standard audiometric range, the loss of hearing acuity induced by aging has been reported to be more pronounced than the alteration of some OAE parameters, suggesting that presbycusis is related rather to a dysfunction of the stria vascularis than to a functional loss of hair cells [12]. Irrespective of these implications, which remain to be investigated, the findings presented in this work do not support the hypothesis that otoacoustic emissions may be suitable for early detection of cochlear dysfunctions, which have no counterpart in routine pure tone audiometry. The largest share of age-related decay of OAE amplitude can be attributed to the age-linked elevation of pure tone threshold, especially at high frequencies.
Conclusion
The observations described in the present paper corroborate the hypothesis that the reduction of OAE amplitude with increasing age is primarily caused by age-related hearing loss and not by aging alone. Only a small amount of amplitude loss at high frequencies was observed in ears with normal thresholds (maximum hearing loss 10 dB in the frequency range from 1 to 4 kHz). Even this OAE amplitude loss may be related rather to hearing loss at higher frequencies than to pure effects of aging alone.
References
Attias J, Furst M, Furman V, Reshef I, Horowitz G, Bresloff I (1995) Noise-induced otoacoustic emission loss with or without hearing loss. Ear Hear 16(6):612–618
Avan P, Bonfils P, Loth D, Elbez M, Erminy M (1995) Transient-evoked otoacoustic emissions and high-frequency acoustic trauma in the guinea pig. J Acoust Soc Am 97(5):3012–3020
Avan P, Elbez M, Bonfils P (1997) Click-evoked otoacoustic emissions and the influence of high-frequency hearing losses in humans. J Acoust Soc Am 101(5):2771–2777
Balatsouras DG, Tsimpiris N, Korres S, Karapantzos I, Papadimitrou N, Danielidis V (2005) The effect of impulse noise on distortion otoacoustic emissions. Int J Audiol 44:540–549
Bertoli S, Probst R (1997) The role of transient-evoked otoacoustic emissions testing in the evaluation of elderly persons. Ear Hear 18(4):286–293
Bonfils P, Bertrand Y, Uziel A (1988) Evoked otoacoustic emissions: normative data and presbycusis. Audiology 27:27–35
Castor X, Veuillet E, Morgon A, Collet L (1994) Influence of aging on active cochlear micromechanical properties and on the medial olivocochlear system in humans. Hear Res 77(1–2):1–8
Chan VSW, Wong ECM, McPherson B (2004) Occupational hearing loss: screening with distortion-product otoacoustic emissions. Int J Audiol 43:323–329
Collet L, Moulin A, Gartner M, Morgon A (1990) Age-related changes in evoked otoacoustic emissions. Ann Otol Rhinol Laryngol 99:993–997
Dhooge I, Dhooge C, Geukens S, De Clerk B, De Vel E, Vinck BM (2006) Distortion product otoacoustic emissions: an objective technique for the screening of hearing loss in children treated with platin derivates. Int J Audiol 45:337–343
Dorn PA, Piskorski P, Keefe DH, Neely ST, Gorga MP (1998) On the existence of an age/threshold/frequency interaction in distortion product otoacoustic emissions. J Acoust Soc Am 104(2):964–971
Gates GA, Mills D, Nam B, D’Agostino R, Rubel EW (2002) Effects of age on the distortion product otoacoustic emission growth functions. Hear Res 163:53–60
Hall AJ, Lutman ME (1999) Methods for early identification of noise-induced hearing losses. Audiology 38:277–280
He NJ, Schmiedt RA (1996) Effect of aging on the fine structure of the 2f1–f2 acoustic distortion product. J Acoust Soc Am 99(2):1002–1015
Hoth S (1996) Der Einfluß von Innenohrhörstörungen auf verzögerte otoakustische Emissionen (TEOAE) und Distorsionsprodukte (DPOAE) (The influence of inner ear hearing loss on transitory evoked and distortion product otoacoustic emissions). Laryngol Rhinol Otol 75:709–718
Hoth S, Polzer M, Neumann K, Plinkert P (2007) TEOAE amplitude growth, detectability, and response threshold in linear and nonlinear mode and in different time windows. Int J Audiol 46:407–418
ISO 7029 (2001) Acoustics: statistical distribution of hearing thresholds as a function of age. German version EN 7029:2000. Beuth, Berlin
Kimberley BP, Hernadi I, Lee AM, Brown DK (1994) Predicting pure tone thresholds in normal and hearing-impaired ears with distortion product emission and age. Ear Hear 15:199–209
Kon K, Inagaki M, Kaga M (2000) Developmental changes of distortion product and transient evoked emissions in different age groups. Brain Dev 22(1):41–46
Konopka W, Pawlaczyk-Lusczynska M, Sliwinska-Kowalska M, Grzanka A, Zalewski P (2005) Effects of impulse noise on transiently evoked otoacoustic emission in soldiers. Int J Audiol 44:3–7
Lapsley Miller JA, Marshall L, Heller LM (2004) A longitudinal study of changes in evoked otoacoustic emissions and pure-tone thresholds as measured in a hearing conservation program. Int J Audiol 43:307–322
Lapsley Miller JA, Marshall L, Heller LM, Hughes LM (2006) Low-level otoacoustic emissions may predict susceptibility to noise-induced hearing loss. J Acoust Soc Am 120(1):280–296
Lonsbury-Martin BL, Cutler WM, Martin GK (1991) Evidence for the influence of aging on distortion-product otoacoustic emissions in humans. J Acoust Soc Am 89(4):1749–1759
Lucertini M, Moleti A, Sisto R (2002) On the detection of early cochlear damage by otoacoustic emission analysis. J Acoust Soc Am 111(2):972–978
Morant-Ventura A, Marco Algarra J, Sequi Canet J, Caballero Mallea A, Mir Planas B (1999) Modifications of evoked otoacoustic emissions: study of age groups. Acta Otorrinolaringol Esp 50(5):355–358
Oeken J, Lenk A, Bootz F (2000) Influence of age and presbyacusis on DPOAE. Acta Otolaryngol 120:396–403
Plinkert PK, Hemmert W, Wagner W, Just K, Zenner HP (1999) Monitoring noise susceptibility: sensitivity of otoacoustic emissions and subjective audiometry. Br J Audiol 33:367–382
Plinkert PK, Hemmert W, Zenner HP (1995) Methodenvergleich zur Früherkennung einer Lärmvulnerabilität des Innenohres (A comparison of different audiometric methods for early detection of a vulnerable cochlea). HNO 43:89–97
Prieve BA, Falter SR (1995) COAEs and SSOAEs in adults with increased age. Ear Hear 16:521–528
Probst R, Harris FP (1997) Otoacoustic emissions. In: Alford BR, Jerger J, Jenkins HA (eds) Electrophysiologic evaluation in otolaryngology. Adv Otolaryngol 53. Karger, Basel, pp 182–204
Satoh Y, Kanzaki J, O-Uchi T, Yoshihara S (1998) Age-related changes in transiently evoked otoacoustic emissions and distortion product otoacoustic emissions in normal hearing ears. Auris Nasus Larynx 25(2):121–130
Schmuziger N, Patscheke J, Probst R (2007) An assessment of threshold shifts in nonprofessional pop/rock musicians using conventional and extended high-frequency audiometry. Ear Hear 28(5):643–648
Schmuziger N, Probst R, Smurzynski J (2005) Otoacoustic emissions and extended high-frequency hearing sensitivity in young adults. Int J Audiol 44:24–30
Stavroulaki P, Apostolopoulos N, Segas J, Tsakanikos M, Adamopoulos G (2001) Evoked otoacoustic emissions—an approach for monitoring cisplatin-induced ototoxicity in children. Int J Ped Otorhinolaryngol 59:47–57
Stenklev NC, Laukli E (2003) Transient evoked otoacoustic emissions in the elderly. Int J Audiol 42:132–139
Stover L, Norton SJ (1993) The effects of aging on otoacoustic emissions. J Acoust Soc Am 94(5):2670–2681
Acknowledgment
The authors gratefully acknowledge the valuable comments and recommendations of Prof. Dr. Rudolf Probst who left his initial role as an anonymous reviewer and contributed substantially to the final version of this paper.
Conflict of interest statement
The authors declare that they do not have any financial relationship to organizations that might have sponsored the research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hoth, S., Gudmundsdottir, K. & Plinkert, P. Age dependence of otoacoustic emissions: the loss of amplitude is primarily caused by age-related hearing loss and not by aging alone. Eur Arch Otorhinolaryngol 267, 679–690 (2010). https://doi.org/10.1007/s00405-009-1106-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-009-1106-5