Abstract
Nephrogenic systemic fibrosis (NSF) is a disease characterized by fibrosis of the systemic organs in patients with renal failure. Following the findings of recent epidemiological studies and the finding of gadolinium (Gd) in the skin tissue of NSF patients, it is now definitely known that the use of Gd contrast agents can trigger NSF. To date, however, the exact mechanism underlying the induction of fibrosis in various organs by Gd remains unexplained. This study was undertaken to evaluate the influence of Gd on the proliferation activity and collagen production of cultured fibroblasts. Normal human dermis-derived fibroblasts were incubated in the presence of gadodiamide (GA) in the concentration range of 5 × 10−7 to 5 × 10−3 M. The proliferation activity of the cells was assessed on the basis of the cell counts in the fibroblast growth curve and the DNA-synthetic activity of the cells (indicator; level of 3H-thymidine uptake by cells). The collagen production was evaluated by densitometric measurement of the quantity of collagen through electrophoresis and fluorography after incorporation of 3H-proline into the procollagens. Furthermore, the expression levels of the genes for type I and III collagen were measured by real-time reverse transcription polymerase chain reaction (RT-PCR) assay. The cell count tended to be higher when the fibroblasts were incubated in medium containing GA in the concentration range of 5 × 10−7 to 5 × 10−4M as compared to that in the GA-free control cultures; furthermore, the DNA-synthetic activity also rose in a concentration-dependent manner in the GA-treated group as compared to that in the control group. No significant changes in either the collagen production or the collagen gene expression levels were noted in cultures containing GA at concentrations between 5 × 10−7 and 5 × 10−3 M. These results suggest that the formation of sclerosing lesions in patients with NSF may be attributable to the effect of GA of enhancing the growth activity of fibroblasts.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nephrogeneic systemic flbrosis (NSF) was first reported in 2000 as a “scleromyxedema-like cutaneous disease,” through summarization of the characteristics of renal-dialysis patients presenting with skin sclerosis and pigmentation [4]. Later, cases with this condition were also found among patients with severe nephropathy not yet on hemodialysis. The name “NSF” was given to this entity, as the sclerosing lesions were noted not only in the skin, but also in various other organs [16]. Following the findings of recent epidemiological studies [24] and the detecion of gadolinium (Gd) in the skin tissue of NSF patients [14], it began to be considered that Gd-containing contrast agents (Gd-CA), such as gadodiamide (GA), may be involved as precipitating factors for NSF.
Symptoms of NSF largely occur in the skin (redness and hardness accompanied by pain and itching sensation of the extremities and trunk) and articular symptoms (contracture and articular immobility) [19]. The fibrosis-affected skin in NSF patients is histopathologically characterized by thickening of the collagen fiber bundles, presence of numerous fibroblast-like cells and positive results for Alcian blue staining (probably reflecting an increase of glycosaminoglycans such as hyaluronic acid) [7].
This study was designed to evaluate the influence of GA on the proliferation activity, collagen production activity and expression levels of the collagen genes in healthy human dermis-derived fibroblasts, with the goal of exploring the mechanism underlying the development of Gd-induced fibrosis.
Materials and methods
Materials
This study was approved by the Institutional Review Board of Dokkyo Medical University Hospital (ID: 1706), on the basis of the Ethical Guidelines for Clinical Research of the Ministry of Health, Labor and Welfare, Japan.
Fresh tissue specimens for fibroblast cultures were obtained from the abdominal skin of 15-year-old and 47-year-old healthy woman and 36-year-old man during reconstructive surgery. Gadodiamide (GA) (Omniscan®) as a pure liquid was a generous gift from DAIICHI SANKYO CO., LTD. (Tokyo, Japan). GA was dissolved in H20 and used at a final concentration of 5 × 10−7 to 5 × 10−3M.
Fibroblast cultures
Fibroblast cultures were established from the skin biopsy specimens by the outgrowth method described previously [8], and the cell strains obtained were designated N-15F, N-47F, and N-36 M, respectively. The cultures were maintained in Dulbecco’s modified Eaglemedium (DMEM) containing 10 % fetal bovine serum (FBS), in a CO2 incubator at 37 °C. The cells were used in each comparative study at population doubling levels (PDLs) of two to five.
Fibroblast growth assay [20]
We inoculated 24-well tissue culture plates with 2 × 104 fibroblasts per well, and the next day we started culturing the fibroblasts in DMEM containing 1 % FBS with 5 × 10−7 to 5 × 10−3 M gadodiamide (GA). We observed fibroblasts morphologically with a microscopy and counted the numbers of fibroblasts after 24, 48, and 72 h. A Burker-Turk hemocytometer was used to make the cell counts.
Assay for DNA synthesis
The assay for DNA synthesis was performed as previously described [11]. Fibroblasts were cultured to confluence in 6-well tissue culture plates in DMEM containing 10 % FBS. The cells were then cultured for 48 h in DMEM containing 1 % FBS with various concentrations of GA. After the culture with GA, [6-3H] thymidine (spec. act. 15.0 Ci/mmol, Amersham, England) was added at a final concentration of 1 μCi/ml, and the cells were cultured for a further 3 h. Then, after being washed with PBS, they were processed with 5 % trichloroacetic acid and dissolved in 1 M NaOH, and the radioactivity was quantified using a liquid scintillation counter.
Isolation and measurement of newly synthesized collagen [10]
When the cell layers had become confluent, the cells were cultured for 48 h at 37 °C in DMEM containing 1 % FBS and supplemented with various concentrations of GA. They were then incubated with DMEM containing 1 % FBS and 5 μCi/ml 2, 3-[3H] proline (Amersham) in the presence of 50 μg/ml L-ascorbic acid 2-phosphate for 24 h. Labeled proteins secreted into the culture medium were precipitated by the addition of 5 % (final conc.) trichloroacetic acid. The precipitate was dissolved in 0.05 M acetic acid, and after being digested with 0.1 % pepsin was subjected to SDS-PAGE (5 % polyacrylamide gel containing 3.6 M urea). Radioactive bands were detected by fluorography, and the relative amounts of collagen were quantified by densitometry.
Quantitative real-time RT-PCR analysis [12]
Fibroblasts were cultured to confluence in DMEM containing 10 % FBS in 100 mm × 20 mm dishes, and then for 48 h in DMEM containing 1 % FBS and supplemented with GA at concentrations of 5 × 10−7 to 5 × 10−5 M. Total RNA was isolated using the guanidinium thio-cyanate-phenol–chloroform method. A 2 μg of total RNA was reverse transcribed in a 10 μl reaction using random primers and Transcriptor First Strand synthesis kit (Roche, USA). Real-time PCR assays were performed using MyiQ™ Single-Color Real-Time PCR Detection System (Bio-Rad iCycler). Amplification mixture (10 μl) contained 0.125 μg of cDNA, 0.25 μM of primer and 5 μl of iQ™ SYBR Green Supermix. Amplification was at 95 °C for 3 min, followed by a 40 cycles of 95 °C for 30 s, 60 °C for 1 min. All samples were analyzed in parallel for GAPDH expression as internal control. The fold change in the levels of genes of interest was determined by 2−ΔΔCT. To compare the different samples in an experiment, RNA expression in samples were compared to that of the control GAPDH in each experiment. The primers used were as previously described [3, 22], and as follows: COL1A1: forward 5′–CCAGAAGAACTGGTACATCAGCA–3′ and reverse 5′–CGCCATACTCGAACTGGGAAT–3′. COL1A2:forward 5′–GATGTTGAACTTGTTGCTGAGG-3′ and reverse 5′–TCTTTCCCCATTCATTTGTCTT–3′. COL3A1:forward 5′–CCATCTGCAGAGCAATGACT–3′ and reverse 5′–CCCTGTGGCATGCCAATGAA–3′. GAPDH :forward 5′–GGCCTCCAAGGAGTAAGACC–3′ and reverse 5′–CTGTGAGGAGGGGAGATTCA–3′.
Statistical analysis
One-Way Analysis of Variance (ANOVA) was used to determine the variance of data from six different groups using Prism (GraphPad, La Jolla, CA, USA). Subsequently, the effect of drug was determined by a student t test as a post hoc analysis. The P values <0.05 were defined as statistically significant.
Results
Influence of GA on cell growth activity
To determine whether GA affects the cell growth activity of fibroblasts or not, we first incubated the cells in the presence of GA and analyzed their growth curve. Two cell strains (N-47F, and N-36 M) were used for the experiments, and the results in both were similar. All experiments were performed in triplicate. The data from N-47F are shown in Fig. 1. The cell counts at 24 h after the start of incubation were about 1.3–2.3 times higher in the cells incubated in the presence of 5 × 10−3 M GA to 5 × 10−5 M GA and 5 × 10−7 M GA, and about 2.8 times higher in the cells cultured with 5 × 10−6 M GA, as compared to those in the GA-free control group. The cell counts at 48 h after the start of incubation were about 1.7–2.3 times higher in the GA 5 × 10−4 M, 5 × 10−5 M and 5 × 10−7 M groups, and about 3.4 times higher in the GA 5 × 10−6 M group, as compared to the count in the control group. The cell counts at 72 h were 1.1–1.3 times higher in the GA 5 × 10−4 M, 5 × 10−5 M and 5 × 10−7 M groups, and about 2.1 times higher in the GA 5 × 10−6 M group, as compared to the count in the control group. On the other hand, the cell count in the cultures containing GA at 5 × 10−3 M was about 56 % smaller than that in the control group. Floating and dying of cells were noted in the GA 5 × 10−3 M group at 48 and 72 h after the start of incubation. On observations by phase contrast images of fibroblasts, cell division (mitosis) images were more frequently recognized in the cultures containing GA at concentrations in the range of 5 × 10−7 to 5 × 10−4 M as compared to those in the control group (Fig. 2).
Effects of gadodiamide (GA) on the growth of cultured dermal fibroblasts. Two cell strains (N-47F and N-36 M) were used for the experiments, and the results in both were similar. All experiments were performed in triplicate. The data from N-36 M are shown. The cell growth curve was determined by inoculating 2 × 104 fibroblasts onto 24-well tissue culture plates and adding GA. The number of cells was counted 24 (day 1), 48 (day 2), and 72 (day 3) h after the start of incubation with GA. The results shown are mean ± SEM. * P < 0.05 vs untreated controls. ** P < 0.01 vs untreated controls
Influence of GA on the DNA synthesis activity
Effects of gadodiamide (GA) on DNA-synthetic activity by cultured dermal fibroblasts were analysed (Fig. 2). When fibroblasts were incubated in the presence of GA in the concentration range of 5 × 10−7 to 5 × 10−4M, fibroblasts showed a tendency towards concentration-dependent increase of the [6-3H] thymidine uptake. The [6-3H] thymidine uptake was significantly higher in the presence of GA at the concentration of 5 × 10−6M (about 1.5-fold higher) and 5 × 10−5M (about twofold higher) as compared to that in the control group. On the other hand, [6-3H] thymidine uptake was significantly lower as compared to that in the control group in the presence of GA at the concentration of 5 × 10−3M. Two cell strains (N-15F and N-36 M) were used for the experiments, and the results in both were similar. All experiments were performed in triplicate. The data from N-36 M are shown in Fig. 3.
Effects of gadodiamide (GA) on DNA-synthetic activity by cultured dermal fibroblasts. The cells were then cultured for 48 h in DMEM containing 1 % FBS with various concentrations of gadodiamide (GA). After the culture with GA, [6-3H] thymidine was added, and the cells were cultured for a further 3 h. Then, after being washed with PBS, they were processed with 5 % trichloroacetic acid and dissolved in 1 M NaOH, and the radioactivity was quantified using a liquid scintillation counter. The results shown are mean ± SEM. * P < 0.05 vs untreated controls. ** P < 0.01 vs untreated controls. Two cell strains (N-15F and N-36 M) were used for the experiments, and the results in both were similar. All experiments were performed in triplicate. The data from N-36 M are shown
Effects of GA on the production of collagens in cultured dermal fibroblasts
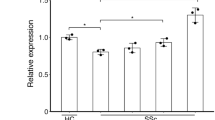
No significant changes in the production levels of α1(I) collagen, α2(I) collagen and α1(III) collagen in the culture medium were observed, as determined by SDS–polyacrylamide gel electrophoresis, fluorography and quantification with densitometry, when cultured fibroblasts were exposed to GA at concentrations in the range of 5 × 10−7 to 5 × 10−3M (Fig. 3a–d). Two cell strains (N-15F and N-36 M) were used for the experiments, and the results in both were similar. All experiments were performed in triplicate. The data from N-15F are shown in Fig. 4.
Effects of gadodiamide (GA) on production of collagens by cultured dermal fibroblasts. The cells were incubated for 48 h at 37 °C in DMEM supplemented with 5 × 10−7 to 5 × 10−3 M concentrations of GA and then cultured for 24 h in DMEM containing 5 μCi/ml 2, 3-[3H] proline. Labeled proteins secreted into the culture medium were digested with pepsin, and they were subjected to SDS-PAGE. Radioactive bands were detected by fluorography (a), and the relative amounts of collagen were quantified by densitometry and corrected for cell number. The results of densitometry scanning of the α1(I) collagen (b), α2(I) collagen (c) and α1(III) collagen (d) from cultures treated with various concentrations of GA are expressed as percentages of the values in untreated control cultures. Two cell strains (N-15F and N-36 M) were used for the experiments, and the results in both were similar. All experiments were performed in triplicate. The data from N-15F are shown. The results shown are mean ± SEM
Effects of GA on the gene expression levels of the genes encoding α1(I) collagen, α2(I) collagen and α1(III) collagen in cultured dermal fibroblasts
No significant changes in the gene expression levels of α1(I) collagen (Fig. 4a), α2(I) collagen (Fig. 4b) and α1(III) collagen (Fig. 4c) were observed, as determined by real-time reverse transcriptase-polymerase chain reaction(PCR) analysis of the corresponding genes, when cultured fibroblasts were exposed to GA at concentrations in the range of 5 × 10−7 to 5 × 10−3M. Two cell strains (N-15F and N-47F) were used for the experiments, and the results in both were similar. All experiments were performed in triplicate. The data from N-47F are shown in Fig. 5.
Effects of GA on the gene expression levels of the genes encoding α1(I) collagen, α2(I) collagen and α1(III) collagen in cultured dermal fibroblasts Real-time reverse transcriptase- polymerase chain reaction (PCR) analysis of the genes encoding α1(I) collagen (a), α2(I) collagen (b) and α1(III) collagen (c) and isolation of total RNA were performed from cultures treated with various concentrations of GA. The mRNA levels were expressed as a percentage relative to the levels in untreated control cultures. Two cell strains (N-15F and N-47F) were used for the experiments, and the results in both were similar. All experiments were performed in triplicate. The data from N-47F are shown. The results shown are mean ± SEM
Discussion
According to the report of Broome DR published in 2008 [2], As of February 1, 2008 there have been 190 biopsy-proven cases of NSF published in the peer-reviewed literature with the following associations: 157 gadodiamide (Omniscan, GE Healthcare), 8 gadopentetate (Magnevist, Bayer Healthcare), 3 gadoversetamide (OptiMARK, Covidien), and 18 unspecified Gd-CA, and 4 confounded cases with more than one Gd-CA. Five cases of NSF were unassociated with Gd-CA.
Symptoms of NSF include skin symptoms (redness and hardness accompanied by pain and itching of the extremities and trunk) and articular symptoms (contracture and joint immobility). Fatal cases of NSF have also been reported [19]. The histopathological features of the fibrotic skin lesions in NSF include thickened collagen bundles with several clefts, numerous plump fibroblasts-like cells, and strongly positive Alcian blue staining, suggesting elevated deposition of sulphated glycosaminoglycans and hyaluronic acid [7].
To elucidate the mechanism underlying the induction of fibrosis in the body organs by Gd, Edward et al. [7] first compared the levels of synthesis of hyaluronic acid and collagen between the fibroblasts obtained from NSF patients and fibroblasts derived from healthy individuals, reporting that hyaluronic acid synthesis was 2.8 times higher and collagen synthesis was 3.3 times higher in the cells from the NSF group as compared to those from the healthy group. They additionally reported that as compared to serum from healthy individuals, the serum from NSF patients enhanced hyaluronic acid synthesis by up to sevenfold and collagen synthesis by up to 2.4-fold in the fibroblasts from the healthy group. Furthermore, they reported that the addition of Gd to the culture medium of healthy individual-derived fibroblasts resulted in elevation of the cell growth activity and up to a 2.3-fold increase of hyaluronic acid synthesis, while no influence was observed on the collagen synthesis. Do et al. [6] reported that the addition of Gd-CA to the culture medium of human foreskin fibroblasts resulted in dose-dependent of fibronectin generation and transforming growth factor-β(TGFβ)production. In this study, we added Gd to the culture medium of healthy individual-derived fibroblasts and demonstrated elevation of the cell growth activity, similar to the finding reported by Edward et al. [7].
TGFβ, a cytokine derived from various cell types, or including T cells, fibroblasts, and endothelial cells, is known to play a central role in fibrosis [23]. Jiménez et al. 15] reported increased expression of TGFβ1 in affected skin of patients with NSF. TGFβ1 polymorphisms in codon 10 (Leu10Pro) and codon 25 (Arg25Pro) have been implicated in the development and progression of several other fibrotic processes, including pulmonary, renal, and hepatic fibrosis [1, 5, 9, 13]. Le et al. [18] reported fibrosis-associated single-nucleotide polymorphisms in TGFβ1 and CAV1 are not associated with the development of NSF. Koreishi et al. [17] reported that in early lesions of NSF skin, a progressive thickening of the dermis is accompanied by the accumulation of stromal cells with long dendric processes. Quatresooz et al. [21] analyzed immunohistochemical aspects of the fibrogenic pathway in NSF, they found that the cell proliferation highlighted by the MIB/Ki67 immunoreactivity was unusually high in the interstitial stromal cells. Wagner et al. [25] suggested that fibrosis of NSF is from bone marrow-derived fibroblasts by the results of their experiments with animal models.
To date, the report by Edward et al. [7] is the only published report of evaluation of the influence of Gd on collagen synthesis by cultured healthy individual-derived fibroblasts. They showed no influence of the level on collagen synthesis. Their report was based on quantification of C-terminal propeptide of type I collagen (CICP) as an indicator of collagen synthesis. This study was designed to measure the collagen synthesis level more directly after adding Gd to a culture medium of healthy individual-derived fibroblasts, by incorporating radioactive proline into the procollagens and measuring the quantity of collagen synthesized. Furthermore, the collagen gene expression levels were also analyzed by real-time reverse transcription polymerase chain reaction (RT-PCR) assay in this study.
Since collagen amounts in affected tissue of patients with NSF are enhanced histologically, but no alterations in collagen synthesis and collagen gene expression were detected in this study. Therefore, we should also consider the presence of alterations of collagen turnover such as inactivation of collagenase (MMP-1). The gene expression level of MMP-1 in the fibroblasts cultured with 5 × 10−7 to 5 × 10−4 M GA measured by real-time RT-PCR assay was not decreased (data not shown).
In our experiment conducted thus, the cell proliferation was reduced and the DNA-synthetic potential was markedly lower in the fibroblasts incubated with 5 × 10−3M GA as compared to the observations in the GA-free control fibroblasts. This result suggests that 5 × 10−3 M is the toxic dose of GA.
We conclude that, addition of GA to the culture medium resulted in elevation of the fibroblast growth activity, while the collagen expression level did not differ significantly between the GA-added group and the control group. These results suggest that the formation of sclerosing lesions in patients with NSF may be attributable to the effect of GA of enhancing the growth activity of fibroblasts.
References
Brazova L, Sismova K, Vavrova V, Bartosova J, Macek M Jr, Lauschman H, Sediva A (2006) Polymorphisms of TGF-beta1 in cystic fibrosis patients. Clin Immunol 121:350–357
Broome DR (2008) Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: a summary of the medical literature reporting. Eur J Radiol 66:230–234
Bujor MA, Pannu J, Bu S, Smith AE, Muise-Helmericks CR, Trojanowska M (2008) Akt blockade downregulates collagen and upregulates MMP1 in human dermal fibroblasts. J Invest Dermatol 128:1906–1914
Cowper SE, Robin HS, Steinberg SM, Su LD, Gupta S, LeBoit PE (2000) Scleromyxoedema-like cutaneous diseases in renal-dialysis patients. Lancet 356:1000–1001
Dixon JB, Bhathal PS, Jonsson JR, Dixon AF, Powell EE, O’Brien PE (2003) Pro-fibrotic polymorphisms predictive of advanced liver fibrosis in the severely obese. J Hepatol 39:967–971
Do C, Barnes JL, Tan C, Wagner B (2014) Type of mri contrast, tissue gadolinium, and fibrosis. Am J Physiol Renal Physiol 307:F844–F855
Edward M, Quinn JA, Mukherjee S, Jensen MB, Jardine AG, Mark PB, Burden AD (2008) Gadodiamide contrast agent ‘activates’ fibroblasts: a possible cause of nephrogenic systemic fibrosis. J Pathol 214:584–593
Fleischmajer R, Perlish JS, Krieg T, Timpl R (1981) Variability in collagen and fibronectin synthesis by scleroderma fibroblasts in primary culture. J Invest Dermatol 76:400–403
Gewaltig J, Mangasser-Stephan K, Gartung C, Biesterfeld S, Gressner AM (2002) Association of polymorphisms of the transforming growth factor-beta1 gene with the rate of progression of HCV-induced liver fibrosis. Clin Chim Acta 316:83–94
Hata R, Kurata S, Shinkai H (1988) Existence of malfunctioning pro alpha2 (I) collagen genes in a patient with a pro alpha 2 (I)–chain–defective variant of Ehlers-Danlos syndrome. Eur J Biochem 174:231–237
Hatamochi A, Arakawa M, Takeda K, Ueki H (1994) Activation of fibroblast proliferation by Werner’s syndrome fibroblast-conditioned medium. J Dermatol Sci 7:210–216
Hayashi S, Ikeda M, Kitamura Y, Hamasaki Y, Hatamochi A (2012) UVA irradiation following treatment with topical 8-methoxypsoralen improves bleomycin-induced scleroderma in a mouse model, by reducing the collagen content and collagen gene expression levels in the skin. J Dermatol Sci 67:20–25
Helmig S, Belwe A, Schneider J (2009) Association of transforming growth factor beta1 gene polymorphisms and asbestos-induced fibrosis and tumors. J Investig Med 57:655–661
High WA, Ayers RA, Chandler J, Zito G, Cowper SE (2006) Gadolinium is detectable within the tissue of patients with nephrogenic systemic fibrosis. J Am Acad Dermatol 56:21–26
Jiménez SA, Artlett CM, Sandorfi N, Derk C, Latinis K, Sawaya H, Haddad R, Shanahan JC (2004) Dialysis-associated systemic fibrosis (nephrogenic fibrosing dermopathy): study of inflammatory cells and transforming growth factor beta1 expression in affected skin. Arthritis Rheum 50:2660–2666
Kay J (2008) Nephrogenic systemic fibrosis: a gadolinium-associated fibrosing disorder in patients with renal dysfunction. Ann Rheum Dis 67(suppl 3):66–69
Koreishi AF, Nazarian RM, Saenz AJ, Klepeis VE, McDonald AG, Farris AB, Colvin RB, Duncan LM, Mandal RV, Kay J (2009) Nephrogenic systemic fibrosis: a pathologic study of autopsy cases. Arch Pathol Lab Med 133:1943–1948
Le LP, Garibyan L, Lara D, Finberg KE, Lafrate AJ, Duncan LM, Kay J, Nazarian RM (2013) Fibrosis-associated single-nucleotide polymorphisms in TGFB1 and CAV1 are not associated with the development of nephrogenic systemic fibrosis. Am J Dermatopathol 35:351–356
Morcos SK, Thomsen HS (2008) Nephrogenic systemic fibrosis: more questions and some answers. Nephron Clin Pract 110:24–31
Namikawa H, Sunazuka T, Kitamura Y, Suzuki T, Hamasaki Y, Yamazaki S, Omura S, Hatamochi A (2010) Effect of erythromycin A and its new derivative EM201 on type I collagen production by cultured dermal fibroblasts. Arch Dermatol Res 302:341–348
Quatresooz P, Paquet P, Hermanns-Lê T, Piérard GE (2010) Immunohistochemical aspects of the fibrogenic pathway in nephrogenic systemic fibrosis. Appl Immunohistochem Mol Morphol 18:448–452
Reyes-Gordillo K, Shah R, Arellanes-Robledo J, Hemandez-Nazara Z, Rincon-Sanchez RA, Inagaki Y, Rojkind M, Raj Lakshman M (2014) Mechanisms of action of acetaldehyde in the Up-regulation of the human α2(I) collagen gene in hepatic stellate cells: key roles of Ski, SMAD3, SMAD4, and SMAD7. Am J Pathol 184:1458–1467
Shah M, Revis D, Herrick S, Baillie R, Thorgeirson S, Ferguson M, Roberts A (1999) Role of elevated plasma transforming growth factor-beta1 levels in wound healing. Am J Pathol 154:1115–1124
Todd DJ, Kagan A, Chibnik LB, Kay J (2007) Cutaneous changes of nephrogenic systemic fibrosis: predictor of early mortality and association with gadolinium exposure. Arthritis Rheum 56:3433–3441
Wagner B, Tan C, Barnes JL, Ahuja S, Davis TL, Gorin Y, Jimenez F (2012) Nephrogenic systemic fibrosis: evidence for oxidative stress and bone marrow-derived fibrocytes in skin, liver, and heart lesions using a 5/6 nephrectomy rodent model. Am J Pathol 181:1941–1952
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors state no conflicts of interest. Authors also observed the protocol approved by the Institutional Review Board of Dokkyo Medical University Hospital (ID: 1706).
Rights and permissions
About this article
Cite this article
Ozawa, Y., Hayashi, S., Hamasaki, Y. et al. Effects of Gadodiamide on cell proliferation and collagen production in cultured human dermal fibroblasts. Arch Dermatol Res 308, 695–701 (2016). https://doi.org/10.1007/s00403-016-1689-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-016-1689-9