Abstract
The Himalayas are undergoing significant changes in temperature and moisture availability due to global climate change. Understanding how these changes affect tree growth is critical for conserving and managing this biodiverse region. To investigate the growth-limiting climatic factors, we collected tree-ring data from Abies pindrow old-growth mountain forests across 3190 to 3700 m a.s.l. in the inner valleys of the Garhwal Himalayas. This area faces significant temperature increases, reduced summer monsoon activity, and decreased winter precipitation. We found that over the past century, tree growth suffered from warm and dry springs and benefited from cool and moist springs, particularly at lower elevations (< 3400 m a.s.l.). Conversely, summer climate dynamics had minimal impact on radial growth. The sensitivity of Abies growth to spring climate can be attributed to the inner valleys’ continental climate, marked by spring water deficit. Additionally, higher elevation trees (> 3400 m a.s.l.) showed positive correlations with dormant period temperatures in prior October and January, with the positive effect of October warmth strengthening over time. A dynamic analysis of the impact of spring (March–May) temperature, precipitation and Standardized Precipitation-Evapotranspiration Index revealed a recent unification (the 1980–2010s) of the climate–growth relationship across elevations. Specifically, the low-elevation trees have become less sensitive to warm springs, while the high-elevation trees have only recently become sensitive to warm and dry springs. The varying growth responses along the elevation gradient highlight the necessity of site-dependent differentiation when assessing the impacts of global climate change on species performance and geographic range shifts.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The unprecedented changes in temperature and precipitation caused by global climate change significantly impact plant biomass growth and, thus, carbon sequestration (e.g., Babst et al. 2019; Gaire et al. 2023). Increasing temperatures and altered precipitation regimes affect physiological processes such as photosynthesis and water availability, affecting growth and primary production (Körner 2012). However, the impact of global climate change on plants varies significantly depending on their geographic location within species distributional ranges, as different constraints come into play, leading to varying responses to climate change (e.g., Doležal et al. 2010; Sohar et al. 2017). A multisite approach that compares plant growth responses in different habitats within a species’ range, including contrasting distributional boundaries, is needed to comprehensively understand how plants with wide ranges adapt to global climate change (e.g., Schurman et al. 2019; Altman et al. 2020). The general lack of information on the spatiotemporal variability of climate–growth interactions across species distributional ranges challenges our ability to predict plant vulnerability to climate change (e.g., Zheng et al. 2021; Malik et al. 2023).
Climate warming significantly influences the current changes in plant growth in the high mountains, where temperatures are rising much faster than in the lowlands (Shrestha et al. 2012; Dolezal et al. 2016; Sun et al. 2021). These changes manifest as spatiotemporal shifts in growth responses to climate across elevational gradients (Körner 2012; Gaire et al. 2020). In particular, climate warming in cool and moist high-elevation regions releases plants from low-temperature limitation by promoting increased plant growth, as seen in recent cases of greening in environments such as the Yukon mountains (Myers-Smith et al. 2020), the European Alps (Filippa et al. 2019) or the Himalayas (Anderson et al. 2020), and the Qinghai-Tibet Plateau (Wang et al. 2021). Conversely, increasing temperatures and altered precipitation patterns have more significant implications for reduced growth at warmer and drier lower elevations and latitudes (Altman et al. 2017; Dolezal et al. 2020). Understanding these elevation-dependent growth variations has implications for forest management and conservation strategies (Schickhoff et al. 2022).
Trees at different elevations display varying responses to climate change (Rai et al. 2020). As climate changes, the strength and direction of these responses might shift (Rai et al. 2023a), impacting growth trajectories differently at various elevations (Gaire et al. 2023). The negative impact of temperatures on growth diminishes with elevation (Thapa et al. 2017). Moreover, tree species may exhibit varying growth responses to climate change (e.g., Sohar et al. 2017; Zheng et al. 2021). Some species might thrive under altered conditions (Tiwari et al. 2017), while others might struggle due to ecological constraints and changing competitive dynamics (e.g., Thapa et al. 2017). Furthermore, the interplay between multiple climatic variables contributes to intricate climate–growth relationships across elevation gradients (Sohar et al. 2017). Due to the phenological shift in peak growth activities, certain factors, such as spring and autumn conditions, could gain greater prominence under shifting climatic conditions, in contrast to the significance of summer conditions (e.g., Sohar et al. 2017; Schurman et al. 2019; Rai et al. 2023a).
Finally, the relationship between elevation, climate, and tree growth might experience alterations over time (Rai et al. 2023b). Climate change can amplify or attenuate specific elevation-driven growth patterns, creating complex temporal trends (e.g., Altman et al. 2020; Gaire et al. 2023). Long-term studies offer valuable insights into the historical context of elevation-climate-growth relationships. Analyzing growth patterns over extended periods may help elucidate the impact of gradual climate shifts or abrupt changes triggered by extreme weather events. Dendroclimatology, the study of tree rings as climate archives, provides a powerful tool to examine historical climate-growth relationships and their modulation by elevation. Analyzing tree ring records across species’ elevational gradients allows researchers to infer past climatic conditions and assess how they influenced growth in time and space (e.g., Babst et al. 2019).
Despite increasing dendroclimatological research that is progressively enhancing our comprehension of elevation-climate-growth dynamics and their consequences for forest ecosystems amid the persisting climate change (Babst et al. 2019), the predominant portion of available knowledge stems from temperate and boreal mountain regions (e.g., Babst et al. 2013; Schurman et al. 2019). Tropical and subtropical mountain areas, where significant climate shifts are transpiring, are receiving increased attention (e.g., Liang et al. 2014; Sohar et al. 2017; Zheng et al. 2021). Trees inhabiting temperate and boreal mountain environments generally exhibit distinct short growing seasons influenced by singular or a few predominant climate drivers, such as summer temperatures (e.g., Doležal et al. 2010). Conversely, trees of subtropical and tropical mountains often face more intricate climate–growth associations due to their prolonged growing seasons (Altman et al. 2020; Gaire et al. 2023), which are often disrupted by excessively dry or excessively wet periods. A notable illustration can be observed in the Himalayas, where mountain trees confront dry conditions in the early growing season before the arrival of the monsoon (Rai et al. 2020), followed by water surpluses during the wet and chilly summer monsoon months, with both water deficit and surplus potentially reducing tree performance (Panthi et al. 2018).
In this study, we explore the prominent climate factors associated with radial stem growth of West Himalayan firs (Abies pindrow (Royle ex D.Don) Royle) and consider their relative effects on growth dynamics over the past 110 years in contrasting populations across species’ elevational range in the central Himalayas. We chose Abies pindrow as the target species because it is widespread and one of the dendrochronologically potential trees in the Himalayas (Gautam et al. 2020). Abies pindrow grows in a wide elevational range (2000–3700 m a.s.l.), allowing retrospective assessment of climate impacts on growth dynamics of different populations exposed to contrasting environmental conditions. We collected stem increment data from several populations of Abies pindrow growing in montane forests in the Garhwal Himalaya encompassing elevations ranging from 3190 to 3700 m a.s.l. The Garhwal Himalaya has experienced an unprecedented increase in temperature over the past three decades, prominently during the winter season (Pant et al. 2018), accompanied by reduced monsoonal precipitation (Mishra 2014) and a rapid retreat of glaciers (Bhambri et al. 2011; Singh et al. 2017). The climate trends are particularly pronounced at higher elevations (Mishra 2014; Pant et al. 2018). In addition to those climate changes, the region experienced frequent extreme climate events leading to severe mudslides and flooding (Guri et al. 2015).
Given these circumstances, we focused on the following research questions: (a) Which climatic factors influence the growth of Abies pindrow at different elevations? (b) How sensitive is Abies pindrow to recent warming and diminished monsoon precipitation trends along the elevational gradient? Specifically, we expected: (a) a negative association between higher temperatures, water deficit and radial growth for trees at lower elevations, where trees are primarily limited by lower pre-monsoon spring precipitation and high evapotranspiration; (b) a positive association between higher temperatures and growth in trees from higher elevations, owing to their strong temperature limitation; (c) a negative association between high monsoon rainfall and the growth of high-elevation trees; owing to surplus water limitation; (d) a growth reduction of the low-elevation trees and a growth increase of the high-elevation trees due to the accelerated warming and diminished precipitation in the past three decades.
2 Materials and methods
2.1 Study area and data collection
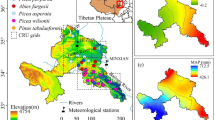
We collected cores from 278 West Himalayan fir (Abies pindrow (Royle ex D.Don) Royle) trees, hereafter referred to by their generic name, from nine different localities in the Garhwal Himalaya during two field expeditions from May 2014 to August 2015. These localities ranged in elevation from 3190 to 3700 m a.s.l. (Fig. 1; Table 1). We prioritized the largest and presumably oldest trees (Speer 2010) for constructing the large-scale tree-ring width chronologies necessary for subsequent dendroclimatic analysis. For the dendroclimatic investigation, we extracted one or two cores using an increment borer from each tree at breast height, parallel to the slope, to prevent any potential interference from reaction wood. We sampled predominantly live trees, with only a limited number of dead stems. Generally, the sampling procedure was designed to maintain a consistent elevation within each site (see average site elevations in Table 1).
2.2 Development of tree-ring chronologies
We followed standard dendrochronological procedures in the laboratory. Cross-sectional surfaces of cores were sliced using a core microtome (Gärtner and Nievergelt 2010) and coated with chalk to accentuate ring boundaries. Tree-ring widths (TRW) were measured to the nearest 0.01 mm employing the TimeTable measuring instrument and the PAST4 software (SCIEM). Visual assessments were conducted utilizing graphs in TSAP-Win (Rinntech) and supported by statistics in COFECHA (Holmes 1983) to ensure the quality of the time-series measurements. The matched radii were subsequently aggregated to form a tree-level TRW series. Following this, inter-tree comparisons involved cross-dating the TRW series from all individual trees against one another. A total of 235 trees (constituting 85% of the dataset) that correlated with the mean of all series within a site ≥ 0.3 were used for the subsequent growth chronologies.
Raw TRW measurements were transformed into dimensionless tree-ring indices (TRI) during the detrending process removing the non-climatic variability in the tree-ring records (Fritts 1976). Here, the negative exponential curve was used for detrending. This curve or a linear model with a negative or zero slope (to retain positive trends likely attributable to climate in the original data series) was fitted individually to each series. Indices were then derived from this curve by calculating ratios between observed TRW values and the corresponding curve values. Next, the detrended series underwent pre-whitening utilizing an autoregressive model to eliminate autocorrelation. The pre-whitened series were averaged across all series using a biweight robust mean estimation to eliminate the influence of outliers (Cook et al. 1990). In the same way, regional chronologies were constructed by combining data from multiple sites. The resultant residual chronologies were employed in subsequent dendroclimatic analyses. Detrending and chronology building was made using ARSTAN software (Cook 1985).
To assess the variability in Abies growth both within and between different sites, several descriptive statistics of tree-ring data were calculated using the COFECHA software. Standard deviation (SD), series intercorrelation (SI), average mean sensitivity (MS), and average first-order autocorrelation (AR) were computed based on raw TRW data for each site. To assess the reliability of the chronologies, the subsample signal strength (SSS) (Wigley et al. 1984) of the detrended series was calculated with a threshold of ≥ 0.85 in the “dplR” package (Bunn 2008) in the R environment (R Core Team 2021). To evaluate the degree of similarity in growth patterns among the residual chronologies over the common period with SSS ≥ 0.85, Baillie-Pilcher’s t-value (tBP) was calculated in TSAP-Win (Rinntech), with tBP ≥ 3.5 indicating good agreement among the series (Baillie and Pilcher 1973).
2.3 Climate data
Given the limitations of instrumental weather observations in high mountain regions, we utilized the CRU TS 4.01 0.5°×0.5°-gridded monthly temperature and precipitation datasets for the cell between four grid points at 30.0° to 31.0°N and 79.5° to 80.0°E covering the study sites in the period spanning 1901 to 2015 (Harris et al. 2014; climexp.knmi.nl; Fig. S1). Further, we used Standardized Precipitation-Evapotranspiration Index (SPEI) where CRU TS 4.03 precipitation and evapotranspiration data were accumulated over the previous 3 months for the same the cell in the period spanning 1902 to 2015 (Beguería et al. 2010, 2014; Vicente-Serrano et al. 2010; climexp.knmi.nl).
2.4 Dendroclimatic analysis
Pearson’s correlation coefficients (r) were employed to examine the associations between tree-ring residual chronologies and climate. Bootstrapped confidence intervals assessed the significance (p < 0.05) of the correlation coefficients. The analysis encompassed a 12-month timeframe of temperature, precipitation, SPEI variables, spanning from the prior October to the current September. Given the elimination of autocorrelation from the chronologies, the impact of weather from the prior growing season was not accounted for. Beyond the static relationships, the temporal consistency of dendroclimatic responses of temperature, precipitation and SPEI was determined by computing values within 50-year moving intervals, offset by 1 year. In addition, seasonal correlations were calculated for three-month periods. For dendroclimatic analysis the “treeclim” package (Zang and Biondi 2015) within the R environment (R Core Team 2021) was employed.
To analyze the dynamics of growth response to climate change, we broke our common (1901–2013/15) tree-ring chronologies down to four periods. The length of period was chosen as close to 30 years – the standard reference of climate – as possible, while still using similarly long periods (i.e., 27–29-year intervals: 1901–1928, 1929–1957, 1958–1986, 1987–2013/15, the latter interval depending on the length of chronology). Accordingly, the most significant monthly climate variables for tree growth were aggregated to respective seasonal characteristics and correlated with the Abies residual chronologies in each period. The climate–growth responses were plotted against elevation and assessed with linear regression models, comparing the regressions in the four periods.
3 Results
3.1 Tree-ring growth and chronologies
A total of 235 Abies trees (constituting 85% of the dataset) were used to construct nine site-specific and two regional residual chronologies (Table 1, Fig. S2). The remaining 15% of samples that did not reach the correlation threshold with the mean of all series within a site ≥ 0.3 were excluded from subsequent analyses. The amount of excluded samples was up to 16% per site, except sites A3 and A9, where 48% and 43% of samples were excluded, respectively. Mean TRW ranged from 0.79 mm to 1.25 mm across the various sites with generally narrower rings in the trees growing at their lower range limit (Table 1, Fig. S2).
Seven sites of nine showed negative growth trends through the last century (Fig. S3). There was no clear elevational difference between the growth trends, i.e. the significantly (p < 0.0001 for the linear fit) decreasing tree growths were evenly distributed along the elevational gradient. There was generally no clear shift towards faster decrease during the last decades, i.e. the decreases were rather uniform through the century.
The Abies trees have grown up to 550 years, thus facilitating the chronologies that extend over the centuries (Table 1; Fig. 2a). However, the extent of reliable chronologies was influenced by shared variability within the detrended series and replication of samples (as depicted by SSS in Fig. 2). Consequently, 1880–2013 emerged as the common period across the nine chronologies, with SSS ≥ 0.85. The residual chronologies were highly similar over the common period (Fig. 2b). There were weak negative spatial trends – the smaller the elevation difference or aerial distance, the more similar the sites were (R2 = 0.12, p < 0.05 and R2 = 0.13, p < 0.05 respectively; Fig. 2c). All nine chronologies qualified for dendroclimatic analyses spanning the period from 1902 to 2013/15 (Fig. 2a).
a Residual chronologies of nine Abies sites along the elevation gradient (black lines) and two regional chronologies (blue lines). Vertical red lines mark the year from which SSS ≥ 0.85. The darker violet area highlights the period covered with climate data and used in the dendroclimatic analysis. b tBP-values between the chronologies over the common period 1880–2013 (134 years) and the elevation differences between sites. c Effects of elevation difference (left) and aerial distance (right) on similarity between sites over the common period 1880–2013 (134 years)
Further data split according to similar elevation and climatic signal resulted in two aggregated regional chronologies (Table 1). Accordingly, sites A1 to A5 with 118 Abies trees growing at 3190–3390 m a.s.l. represented the lower elevations, while sites A6 to A9 with 117 trees growing at 3420–3700 m a.s.l. represented the higher elevations.
3.2 Temporal trends in climate
Based on interpolated CRU TS 4.01 (Harris et al. 2014; climexp.knmi.nl) monthly temperature and precipitation data for the cell between four grid points at 30.0° to 31.0°N and 79.5° to 80.0°E during 1901–2016, the most pronounced climatic trends in the study region are characterized by significant temperature increases (from September to April, p < 0.05), a notable reduction in summer monsoon activity (sum of precipitation in July and August, p < 0.05), and a decrease in winter precipitation (January, p < 0.05) (Fig. S4). Spring (March–April–May, MAM) temperature showed an approximate increase of 0.5 °C. In contrast, MAM precipitation showed no significant trend (Fig. S5). Since the spatial resolution of the data is too low, it was impossible to calculate spatial trends.
3.3 Climate–growth relationships
The bootstrapped correlation coefficients (r) indicated that the radial growth of Abies trees was impacted negatively by higher spring temperature and positively by higher spring precipitation (Table 2, Fig. S6). This combined effect was particularly pronounced at sites A1, A2, A5, and A7 in elevations ranging from 3190 to 3470 m. The other sites showed either a spring temperature or spring precipitation signal. For instance, high spring temperature reduced tree growth at sites A3 (3350 m) and A9 (3700 m) while elevated spring precipitation enhanced growth at site A4 (3360 m). Seasonal correlations calculated for three-month periods (Fig. S7) revealed the strongest signal of temperature and precipitation from March–April–May (MAM) (Fig. S7).
Instead of spring precipitation, February snowfall positively affected trees at sites A3 and A6 (r = 0.23 and r = 0.21, respectively; Table 2, Fig. S6). However, preceding October precipitation negatively influenced trees at site A4 (r = − 0.25). January snow cover was the same at site A8 (r = − 0.21) and combined at site A7 (r = − 0.20). In addition, a significant positive influence of October temperature on the subsequent year’s growth was evident at five sites A3, A4, A6, A7, A8 (r = 0.27…0.37; Table 2, Fig. S6). This was sustained until January at site A8 (3620 m).
Generally, trees growing at lower elevations (combined from sites A1 to A5, 3190–3390 m) showed strong MAM precipitation, SPEI and temperature signals (r = 0.40, r = 0.40 and r = – 0.34, respectively; Figs. 3b and 4b). The trees suffered from July heat but only to a low extent (r = – 0.20). The moving correlation analysis showed that the negative effect of MAM temperature on growth was slightly more stable over the full observed period of 1902–2015 than the positive effect of MAM precipitation (Fig. 3b). However, trees growing at the higher elevations (sites A6 to A9, 3420–3700 m) reflected weaker MAM precipitation, SPEI and temperature signals (r = 0.21, r = 0.19, and r = – 0.18, respectively; Figs. 3a and 4a) and were more sporadic. In addition, the trees showed a significant positive correlation with temperature and negative correlations with precipitation and SPEI during the dormant months, particularly with prior October and January (r = 0.30, – 0.17 and – 0.25, and r = 0.29, – 0.21 and – 0.19, respectively). The positive temperature signal for prior October remained statistically significant almost throughout the entire observation period of 1902–2015, strengthening in time (Fig. 3a).
Climate response of Abies trees a in higher elevation sites A6–A9 based on 117 trees, b in lower elevation sites A1–A5 based on 118 trees. Pearson’s correlation coefficients between residual chronologies and monthly average temperature and precipitation sum from previous October to current September and from March to May (MAM) of 1902–2013/15. The left column shows static correlations (statistically significant relationships (p < 0.05) are indicated in black) and the right column shows dynamic correlations (using a 50-year interval with a 1-year offset; asterisks denote significant correlations (p < 0.05))
SPEI response of Abies trees a in higher elevation sites A6–A9 based on 117 trees, b in lower elevation sites A1–A5 based on 118 trees. Pearson’s correlation coefficients between residual chronologies and accumulated SPEI over the previous 3 months from previous October to current September of 1903–2013/15. The left column shows static correlations (statistically significant relationships (p < 0.05) are indicated in black) and the right column shows dynamic correlations (using a 50-year interval with a 1-year offset; asterisks denote significant correlations (p < 0.05))
3.4 Spatiotemporal trends in climate–growth relationships
As described above, the spring (from March to May, MAM) temperature and precipitation, as well as SPEI, were identified as the main factors controlling Abies growth. We further explored spatiotemporal variability in the growth responses, i.e. how their elevational pattern has changed in time (Fig. 5). The negative growth response to MAM temperature (Fig. 5a) and the positive response to MAM precipitation (Fig. 5b) and MAM SPEI (Fig. 5c) decreased with increasing elevation during the first three periods 1901–1928, 1929–1957, 1958–1986, without respective trends in the last (warmest) period 1987–2013/15 (Fig. 5). Specifically, the low-elevation trees (< 3400 m a.s.l.) have become less dependent on cool springs, while the high-elevation trees (> 3400 m a.s.l.) have developed dependence on cool and moist springs during the recent period.
4 Discussion
4.1 Which main climatic factors influence the growth of Abies pindrow?
We developed extended chronologies of tree-ring width spanning multiple centuries to examine radial growth patterns of Abies pindrow. These patterns were observed across an elevation gradient spanning 3190 to 3700 m within the inner valleys of the Garhwal Himalayan region in northwest India. Our study focused on understanding the growth dynamics of the Abies in response to climatic fluctuations over the past 110 years. Our findings indicate that similarly to other Himalayan forests in more continental climates (e.g., Kharal et al. 2017; Gaire et al. 2017; Sohar et al. 2017; Rai et al. 2020) that experience recurrent water deficits during the early growing season (Liang et al. 2014), the availability of moisture during the spring and early summer months (March to May) is the primary constraint on growth. Our research further points out spring warmth as a limiting factor.
This could result from an indirect impact caused by increased evaporative water loss or other unfavourable conditions due to higher spring temperatures and limited water availability for tree growth. These observations are consistent with larger trends in the interaction between elevation, climate, and growth (e.g., Schurman et al. 2019). Interestingly, in our Garhwal Abies trees, the summer climate had minimal influence on radial growth variations, except for a slight adverse effect of higher temperatures in June–July at lower elevations. Notably, until recent decades, the positive impact of cool, moist springs on Abies growth was primarily evident at lower elevation sites (3190–3390 m) in the Garhwal Himalayan region. However, our analysis uncovered that even the highest-elevation trees (3700 m) increasingly relied on cool and moist springs in the last few decades. This signifies a shift towards a different growth limitation pattern due to accelerated global warming.
The significant impact of spring’s climate on the growth of Abies trees, rather than the typical influence of the summer months, is a result of the distinctly continental climate of the deep inner valleys of the Garhwal Range. These areas are beyond the reach of the summer monsoon, which generally brings cool and moist conditions to the lower regions of the outer Himalayas. Consequently, while conifer forests in the cool and humid outer Himalayas primarily thrive due to the positive influence of summer temperatures (Panthi et al. 2018; Gaire et al. 2023), the growth dynamics of our Garhwal fir populations show a stark contrast. In contrast to the outer Himalayan ranges, species like Abies pindrow, Picea smithiana, Juniperus spp., and Cedrus deodara, growing in the continental interior Himalayan valleys, heavily rely on rain or snow and cooler weather during otherwise dry springs, while their growth is impeded by high spring temperatures (e.g., Sohar et al. 2017; Rai et al. 2020). This reliance on water availability is due to towering mountain barriers like Nanda Devi, Kalanka, and Dunagiri in the Garhwal Range, which block the summer monsoons. Consequently, coniferous forests on the lee side of inner Himalayan valleys receive less than 1000 mm of precipitation compared to over 3000 mm in the southern foothills (Viviroli and Weingartner 2004).
Furthermore, the positive correlations between Abies growth and dormant period temperatures (prior October, January) at the higher sites (especially at 3620 m) correspond with previous studies (e.g., Fan et al. 2009; Sohar et al. 2017; Panthi et al. 2018). Several potential mechanisms, such as warmer soil, reduced root damage, and earlier growth initiation following milder winters, can explain this relationship. Conversely, although cambial activity remains inactive, warmer winters may protect the needles from frost damage (Panthi et al. 2018). Furthermore, our findings indicate a strengthening positive temperature trend from the preceding October over time. Consequently, the fir trees at higher elevations in the Garhwal Himalayan region could benefit from climate warming, similar to observations in high- to mid-elevation conifers in the central Hengduan Mountains (Fan et al. 2009). Furthermore, our findings regarding the inverse correlation between Abies growth and winter precipitation (October and January) at high elevations have been previously observed in Abies spectabilis thriving around 3400 m in Nepal (Kharal et al. 2017). We hypothesize that a thicker snowpack leading to a postponed onset of growth could account for this pattern.
The interplay between elevated temperatures and precipitation, and lower evaporation and their impact on tree growth can be subject to various influencing factors. These factors encompass variations in macroclimatic conditions associated with changes in elevation and microclimate across diverse locations, which are modulated by geographical features such as slopes, aspects, and local topographical attributes (Körner 2012). For example, trees in the Dharansi sites A6 (3420 m) and A8 (3620 m) exhibited no adverse influence of high spring temperature or diminished spring precipitation but instead showed a strong positive relationship with temperature and a negative correlation with SPEI during the preceding autumn and winter months (from October to January), particularly at site A8 (3620 m). This discrepancy contrasts the observations of most of our Abies populations and likely results from the geographical location of the Dharansi sites on the north-facing slopes of the Nanda Devi Sanctuary. These slopes generally maintain cooler conditions compared to the elevations facing south or west. This variation in temperature due to the aspect of the slopes can have repercussions on moisture availability. Cooler air on the north-facing slopes could lead to condensation and increased humidity, facilitating soil moisture retention and ultimately reducing water stress and maintaining radial growth.
4.2 Does tree growth respond to climate change differently at different elevations?
Our Abies trees showed decreasing growth trends across all elevations. Elevation has been shown to significantly modulate past and current climate-growth responses in the Himalayas (Rai et al. 2020) and beyond (e.g., Doležal et al. 2010; Panthi et al. 2018; Altman et al. 2020). We also found that climatic signals for Abies pindrow growth are population-specific across the species elevational range. The contrasting effect of temperature and moisture on tree growth at higher elevations compared to populations at lower elevations is consistent with previous observations made on trees, shrubs, and herbs across broad climatic gradients (Altman et al. 2017; Babst et al. 2019; Weijers et al. 2012; Dolezal et al. 2021). In particular, the diminishing positive effect of moist and cool springs on tree growth toward the high elevations was consistent with previous studies from the Himalayas (e.g., Rai et al. 2023a). This spatial contrast assumes a stronger limiting factor at one end of the species’ elevation range but also context-dependency in the effect of higher temperature as a function of soil moisture, consistent with recent experimental evidence from other cold ecosystems (Ronk et al. 2020) and global observational studies (Myers-Smith et al. 2020; Zhang et al. 2020). This context dependence was evident in two aspects: firstly, the pronounced adverse impact of warmer temperatures during March–May on growth in the low-elevation, attributed to a pronounced water deficit before the arrival of the summer monsoon season; and secondly, the substantial positive influence of October and January temperatures on growth during the subsequent year. This positive effect is attributed to ample soil moisture following the summer monsoon and the continued assimilation activity of evergreen trees.
As a whole, global climate change, marked by unparalleled temperature increases over the last thirty years (Pant et al. 2018), has led to significant elevational changes in climate factors driving tree growth. Before the period of accelerated climate changes, throughout most of the previous century, our lower-mountain trees (< 3400 m a.s.l.) benefited from moist and cool springs without a similar response from the high-mountain trees (> 3400 m a.s.l.). The higher-mountain tree-growth response shifted closer to the lower-elevation pattern only during the last three decades – the period of accelerated climate warming. This suggests that, similarly to the lower-elevation trees, spring precipitation is becoming an increasingly important water resource while cold-related limitations on trees at higher elevations are easing and the start of tree growth is drawing into the precarious spring. Higher evapotranspiration is possibly a connecting factor in the response pattern to temperature and precipitation (Grossiord et al. 2020). Thus, our findings provide a novel contribution to understanding the growth patterns and climatic responses of West Himalayan fir across the full elevational gradient (Schickhoff et al. 2022). The rising spring temperatures and moisture stress potentially have a negative impact on tree growth, reducing growth rates and contributing to forest decline across the species’ elevation range.
5 Conclusions
We conducted a dendrochronological analysis of the growth patterns and the pivotal climatic influences shaping the growth of Abies pindrow across its high and low-elevation habitats in the Himalayas. Our investigation utilized tree-ring width data spanning multiple centuries to explore growth variations along a wide elevation gradient. This study provided valuable insights into the intricate relationship between climate factors and the growth patterns of Abies pindrow specifically within the inner continental valleys of the Garhwal Himalaya in northwest India. Spring moisture and coolness are major climate factors that have boosted growth over the past century, particularly in the lower elevations. In addition, our results underscore the impact of spring climate warming, causing higher-elevation Abies pindrow growth patterns to draw closer to those of lower-elevation trees. Moreover, we show that high-elevation treeline trees benefit from milder and drier winters. This study significantly augments our understanding of the evolving dynamics in tree growth responses driven by global climate shifts. Over the past three decades, rising temperatures have fundamentally reshaped growth dynamics. Consequently, constraints related to spring moisture and cool temperatures have intensified across the entire elevation range of Abies pindrow. As the influence of global climate change persists, these intricate interactions assume greater significance in predicting the growth trajectories of species like Abies pindrow. The variable growth responses along elevation gradients highlight the necessity of site-dependent differentiation when assessing the impacts of global climate change on species performance and geographic range shifts. This knowledge is a cornerstone for formulating effective management and conservation strategies for Himalayan forest ecosystems amidst the continuing environmental change.
Data availability
The datasets generated from lab measurements can be provided in the future upon reasonable request to JA. CRU TS 4.01 monthly temperature and precipitation, and CRU TS 4.03 SPEI time-series were retrieved from https://climexp.knmi.nl.
References
Altman J, Fibich P, Santruckova H, Dolezal J, Stepanek P, Kopacek J, Hunova I, Oulehle F, Tumajer J, Cienciala E (2017) Environmental factors exert strong control over the climate-growth relationships of Picea abies in Central Europe. Sci Total Environ 609:506–516. https://doi.org/10.1016/j.scitotenv.2017.07.134
Altman J, Treydte K, Pejcha V, Cerny T, Petrik P, Srutek M, Song J-S, Trouet V, Dolezal J (2020) Tree growth response to recent warming of two endemic species in Northeast Asia. Clim Change 162:1345–1364. https://doi.org/10.1007/s10584-020-02718-1
Anderson K, Fawcett D, Cugulliere A, Benford S, Jones D, Leng R (2020) Vegetation expansion in the subnival Hindu Kush Himalaya. Glob Change Biol 26:1608–1625. https://doi.org/10.1111/gcb.14919
Babst F, Poulter B, Trouet V, Tan K, Neuwirth B, Wilson R, Carrer M, Grabner M, Tegel W, Levanic T (2013) Site-and species‐specific responses of forest growth to climate across the European continent. Glob Ecol Biogeogr 22:706–717. https://doi.org/10.1111/geb.12023
Babst F, Bouriaud O, Poulter B, Trouet V, Girardin MP, Frank DC (2019) Twentieth century redistribution in climatic drivers of global tree growth. Sci Adv 5:eaat4313. https://doi.org/10.1126/sciadv.aat4313
Baillie MGL, Pilcher JR (1973) A simple crossdating program for tree-ring research. Tree-Ring Bull 33:7–14
Beguería S, Vicente-Serrano SM, Angulo-Martínez M (2010) A Multiscalar Global Drought dataset: the SPEIbase: a New Gridded product for the analysis of Drought variability and impacts. Bull Amer Meteor Soc 91:1351–1356. https://doi.org/10.1175/2010BAMS2988.1
Beguería S, Vicente-Serrano SM, Reig F, Latorre B (2014) Standardized precipitation evapotranspiration index (SPEI) revisited: parameter fitting, evapotranspiration models, tools, datasets and drought monitoring. Int J Climatol 34:3001–3023. https://doi.org/10.1002/joc.3887
Bhambri R, Bolch T, Chaujar RK, Kulshreshtha SC (2011) Glacier changes in the Garhwal Himalaya, India, from 1968 to 2006 based on remote sensing. J Glaciol 57:543–556. https://doi.org/10.3189/002214311796905604
Bunn AG (2008) A dendrochronology program library in R (dplR). Dendrochronologia 26:115–124. https://doi.org/10.1016/j.dendro.2008.01.002
Cook ER (1985) A time series analysis approach to tree ring standardization. PhD Dissertation, University of Arizona, Tucson, AZ
Cook ER, Shiyatov S, Mazepa V (1990) Estimation of the mean chronology. In: Cook ER, Kairiukstis LA (eds) Methods of dendrochronology: applications in the environmental sciences. Kluwer Academic, Dordrecht, pp 123–132
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. www.R-project.org
Dolezal J, Dvorsky M, Kopecky M, Liancourt P, Hiiesalu I, Macek M, Altman J, Chlumska Z, Rehakova K, Capkova K, Borovec J, Mudrak O, Wild J, Schweingruber F (2016) Vegetation dynamics at the upper elevational limit of vascular plants in Himalaya. Sci Rep 6:24881. https://doi.org/10.1038/srep24881
Dolezal J, Kurnotova M, Stastna P, Klimesova J (2020) Alpine plant growth and reproduction dynamics in a warmer world. New Phytol 228:1295–1305. https://doi.org/10.1111/nph.16790
Dolezal J, Jandova V, Macek M, Mudrak O, Altman J, Schweingruber FH, Liancourt P (2021) Climate warming drives himalayan alpine plant growth and recruitment dynamics. J Ecol 109:179–190. https://doi.org/10.1111/1365-2745.13459
Doležal J, Ishii H, Kyncl T, Takahashi K, Vetrova VP, Homma K, Sumida A, Hara T (2010) Climatic factors affecting radial growth of Betula Ermanii and Betula Platypylla in Kamchatka. Can J Res 40:273–285. https://doi.org/10.1139/X09-179
Fan Z-X, Bräuning A, Cao K-F, Zhu S-D (2009) Growth–climate responses of high-elevation conifers in the central Hengduan Mountains, southwestern China. Ecol Manage 258:306–313. https://doi.org/10.1016/j.foreco.2009.04.017
Filippa G, Cremonese E, Galvagno M, Isabellon M, Bayle A, Choler P, Carlson BZ, Gabellani S, di Morra U, Migliavacca M (2019) Climatic drivers of Greening trends in the Alps. Remote Sens 11:2527. https://doi.org/10.3390/rs11212527
Fritts HC (1976) Tree rings and Climate. Academic, London, p 567
Gaire NP, Koirala M, Bhuju DR, Carrer M (2017) Site-and species-specific treeline responses to climatic variability in eastern Nepal Himalaya. Dendrochronologia 41:44–56. https://doi.org/10.1016/j.dendro.2016.03.001
Gaire NP, Fan Z-X, Bräuning A, Panthi S, Rana P, Shrestha A, Bhuju DR (2020) Abies spectabilis shows stable growth relations to temperature, but changing response to moisture conditions along an elevation gradient in the central Himalaya. Dendrochronologia 60:125675. https://doi.org/10.1016/j.dendro.2020.125675
Gaire NP, Zaw Z, Bräuning A, Grießinger J, Sharma B, Rana P, Bhandari S, Basnet S, Fan Z-X (2023) The impact of warming climate on himalayan silver fir growth along an elevation gradient in the Mt. Everest region. Agric Meteorol 339:109575. https://doi.org/10.1016/j.agrformet.2023.109575
Gärtner H, Nievergelt D (2010) The core-microtome: a new tool for surface preparation on cores and time series analysis of varying cell parameters. Dendrochronologia 28:85–92. https://doi.org/10.1016/j.dendro.2009.09.002
Gautam D, Basnet S, Karki P, Thapa B, Ojha P, Poudel U, Gautam S, Adhikari D, Sharma A, Miya MS, Khatri A (2020) A review on dendrochronological potentiality of the major tree species of Nepal. J for Res 9:227. https://doi.org/10.35248/2168-9776.20.9.227
Grossiord C, Buckley TN, Cernusak LA, Novick KA, Poulter B, Siegwolf RT, Sperry JS, McDowell NG (2020) Plant responses to rising vapor pressure deficit. New Phytol 226:1550–1566. https://doi.org/10.1111/nph.16485
Guri PK, Ray PK, Patel RC (2015) Spatial prediction of landslide susceptibility in parts of Garhwal Himalaya, India, using the weight of evidence modelling. Environ Monit Assess 187:324. https://doi.org/10.1007/s10661-015-4535-1
Harris I, Jones PD, Osborn TJ, Lister DH (2014) Updated high-resolution grids of monthly climatic observations – the CRU TS3.10 dataset. Int J Climatol 34:623–642. https://doi.org/10.1002/joc.3711
Holmes RL (1983) Computer-assisted quality control in tree-ring dating and measurement. Tree-Ring Bull 43:69–78
Kharal DK, Thapa UK, George SS, Meilby H, Rayamajhi S, Bhuju DR (2017) Tree-climate relations along an elevational transect in Manang Valley, central Nepal. Dendrochronologia 41:57–64. https://doi.org/10.1016/j.dendro.2016.04.004
Körner C (2012) Alpine treelines: functional ecology of the global high elevation tree limits. Springer, Basel, p 220
Liang E, Dawadi B, Pederson N, Eckstein D (2014) Is the growth of birch at the upper timberline in the Himalayas limited by moisture or by temperature? Ecology 95:2453–2465. https://doi.org/10.1890/13-1904.1
Malik RA, Sukumar R (2023) Response of Radial Growth in Abies pindrow (Royle ex D.Don) Royle to Climate at Treeline Ecotone in the northwestern Himalaya. In: Singh SP, Reshi ZA, Joshi R (eds) Ecology of Himalayan Treeline Ecotone. Springer, Singapore, pp 483–504. https://doi.org/10.1007/978-981-19-4476-5_20
Mishra A (2014) Changing climate of Uttarakhand, India. J Geol Geosci 3:163. https://doi.org/10.4172/2329-6755.1000163
Myers-Smith IH, Kerby JT, Phoenix GK et al (2020) Complexity revealed in the greening of the Arctic. Nat Clim Chang 10:106–117. https://doi.org/10.1038/s41558-019-0688-1
Pant GB, Kumar PP, Revadekar JV, Singh N (2018) Climate change in the Himalayas. Springer, Cham, p 145
Panthi S, Bräuning A, Zhou Z-K, Fan Z-X (2018) Growth response of Abies georgei to climate increases with elevation in the central Hengduan Mountains, southwestern China. Dendrochronologia 47:1–9. https://doi.org/10.1016/j.dendro.2017.11.001
Rai S, Dawadi B, Wang Y, Lu X, Ru H, Sigdel SR (2020) Growth response of Abies spectabilis to climate along an elevation gradient of the Manang valley in the central Himalayas. J Res 31:2245–2254. https://doi.org/10.1007/s11676-019-01011-x
Rai S, Altman J, Kopecký M, Sohar K, Fibich P, Pejcha V, Doležal J (2023a) Contrasting impacts of climate warming on Himalayan Hemlock growth: Seasonal and elevational variations. Dendrochronologia 82:126144. https://doi.org/10.1016/j.dendro.2023.126144
Rai S, Altman J, Kopecký M, Pejcha V, Svoboda M, Doležal J (2023b) Global warming alters himalayan hemlock’s climate sensitivity and growth dynamics. Clim Dyn 16:1–15. https://doi.org/10.1007/s00382-023-07026-9
Ronk A, Liancourt P, Boldgiv B, Petraitis PS, Casper BB (2020) Greater effect of warming on community composition with increased precipitation and in moister landscape location. J Veg Sci 31:3–13. https://doi.org/10.1111/jvs.12813
Schickhoff U, Bobrowski M, Mal S, Schwab N, Singh RB (2022) The World’s mountains in the Anthropocene. In: Schickhoff U, Singh R, Mal S (eds) Mountain landscapes in Transition. Effects of land use and climate change. Sustainable Development Goals Series. Springer, Cham, pp 1–44. https://doi.org/10.1007/978-3-030-70238-0_1
Schurman JS, Babst F, Björklund J, Rydval M, Bače R, Čada V, Janda P, Mikolas M, Saulnier M, Trotsiuk V, Svoboda M (2019) The climatic drivers of primary Picea forest growth along the Carpathian arc are changing under rising temperatures. Glob Change Biol 25:3136–3150. https://doi.org/10.1111/gcb.14721
Shrestha UB, Gautam S, Bawa KS (2012) Widespread climate change in the Himalayas and associated changes in local ecosystems. PLoS ONE 7:e36741. https://doi.org/10.1371/journal.pone.0036741
Singh DS, Tangri AK, Kumar D, Dubey CA, Bali R (2017) Pattern of retreat and related morphological zones of Gangotri Glacier, Garhwal Himalaya, India. Quatern Int 444:172–181. https://doi.org/10.1016/j.quaint.2016.07.025
Sohar K, Altman J, Lehečková E, Doležal J (2017) Growth–climate relationships of himalayan conifers along elevational and latitudinal gradients. Int J Climatol 37:2593–2605. https://doi.org/10.1002/joc.4867
Speer JH (2010) Fundamentals of tree-ring research. University of Arizona, Tucson, p 333
Sun M, Li J, Cao R, Tian K, Zhang W, Yin D, Zhang Y (2021) Climate-growth relations of Abies georgei along an Altitudinal gradient in Haba Snow Mountain, Southwestern China. Forests 12:1569. https://doi.org/10.3390/f12111569
Thapa UK, St. George S, Kharal DK, Gaire NP (2017) Tree growth across the Nepal Himalaya during the last four centuries. Prog Phys Geogr 41:478–495. https://doi.org/10.1177/0309133317714247
Tiwari A, Fan Z-X, Jump AS, Li S-F, Zhou Z-K (2017) Gradual expansion of moisture sensitive Abies spectabilis forest in the Trans-Himalayan zone of central Nepal associated with climate change. Dendrochronologia 41:34–43. https://doi.org/10.1016/j.dendro.2016.01.006
Vicente-Serrano SM, Beguería S, López-Moreno JI (2010) A Multiscalar Drought Index sensitive to global warming: the standardized precipitation Evapotranspiration Index. J Clim 23:1696–1718. https://doi.org/10.1175/2009JCLI2909.1
Viviroli D, Weingartner R (2004) The hydrological significance of mountains: from regional to global scale. Hydrol Earth Syst Sci 8:1017–1030. https://doi.org/10.5194/hess-8-1017-2004
Wang Z, Cui G, Liu X, Zheng K, Lu Z, Li H, Wang G, An Z (2021) Greening of the Qinghai–Tibet Plateau and its response to Climate variations along Elevation gradients. Remote Sens 13:3712. https://doi.org/10.3390/rs13183712
Weijers S, Greve Alsos I, Bronken Eidesen P, Broekman R, Loonen MJ, Rozema J (2012) No divergence in Cassiope tetragona: persistence of growth response along a latitudinal temperature gradient and under multi-year experimental warming. Ann Bot 110:653–665. https://doi.org/10.1093/aob/mcs123
Wigley TML, Briffa KR, Jones PD (1984) On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J Appl Meteorol Climatol 23:201–213
Zang C, Biondi F (2015) Treeclim: an R package for the numerical calibration of proxy-climate relationships. Ecography 38:431–436. https://doi.org/10.1111/ecog.01335
Zhang Y, Parazoo NC, Williams AP, Zhou S, Gentine P (2020) Large and projected strengthening moisture limitation on end-of-season photosynthesis. PNAS 117:9216–9222. https://doi.org/10.1073/pnas.191443611
Zheng L, Shi P, Song M, Zhou T, Zong N, Zhang X (2021) Climate sensitivity of high altitude tree growth across the Hindu Kush Himalaya. Ecol Manage 486:118963. https://doi.org/10.1016/j.foreco.2021.118963
Acknowledgements
We thank Eva Navratova for her help with tree-ring width measurement.
Funding
This work was funded by the Czech Science Foundation (projects no. 21–26883S and 24–11954S) and by the Czech Academy of Sciences (Grant Number: RVO 67985939). Kristina Sohar was supported by the Mobilitas Pluss returning researcher’s project MOBTP35 financed by the European Regional Development Fund.
Author information
Authors and Affiliations
Contributions
KS, JA and JD designed the research. JA, MK, VP and JD collected the field data. KS performed data analyses with help of JA and JD. KS and JD wrote the original draft with significant contribution from JA. All authors contributed to the discussion and writing of the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sohar, K., Altman, J., Kopecký, M. et al. Elevational variations in climate warming impacts on West Himalayan fir growth. Clim Dyn (2024). https://doi.org/10.1007/s00382-024-07409-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00382-024-07409-6