Abstract
The growth response of Himalayan pindrow fir (Abies pindrow) was investigated using tree rings at the treeline ecotone in Hirpora Wildlife Sanctuary in the northwestern Himalaya. The ring-width chronology was built based on 73 increment cores extracted from 40 healthy trees. The measurements were standardized to remove non-climatic growth trends, especially the age-related growth trends. The persistence, because of the effect of the previous year climate on current year growth, in standardized site chronology was removed by auto-regressive modelling. For dendroclimatic analysis, static and moving correlations were computed between site chronology and monthly temperature and precipitation data from Srinagar meteorological station. The climate data was divided into two parts pre-and post-1950 because of varying degrees of increase in the annual climate data during these two time periods. The growth rings formed post-1950 were bigger than those formed prior to 1950. Also, the growth-climate relationships varied between the time periods. The trees responded strongly to monthly temperature in the second part of the twentieth century, while the positive response to growing season precipitation weakened in the recent decades. The moving correlation analyses showed periods of strong and weak climatic responses consistent with the results from static correlations in the two studied time periods. This study will help in understanding the growth responses of Himalayan conifers to climate and hence would also aid in better predicting their future growth in response to predicted climate warming.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
20.1 Introduction
Forests not only play an important role in maintaining the mountain ecosystem functioning and also provide life-supporting ecosystem services that are valuable at local, regional and global scales. Worldwide, the ongoing climate change is leading to significant impacts on the forest ecosystems (Foster 2001; Field et al. 2007; Kirilenko and Sedjo 2007; Grimm et al. 2013). Climate change is expected to alter various eco-physiological processes resulting in shifting vegetation growth rates and distribution of species during the twenty-first century (Walther et al. 2002). Climate change is a great concern as it affects the human population, directly and indirectly, such as warmer winters, the spread of diseases, phenological shifts, seas level rise and frequent natural disasters (Kahn 2005; Webster 2005; Shepherd and Wingham 2007; IPCC 2014). High mountains around the World are among the most vulnerable ecosystems to climate change (Viviroli et al. 2011; Huggel et al. 2012; Rangwala and Miller 2012). The Himalaya is experiencing higher warming than average global warming (Bhutiyani et al. 2007; Shrestha et al. 2012) which can significantly impact the biodiversity, vegetation distribution and ecosystem structure in the region (Aryal et al. 2014; Rashid et al. 2015). The mean annual temperature in Himalaya is showing a more increasing trend than the global average (Pandey et al. 2018) up to 0.06–0.1 °C/y since the mid-1970s (Shrestha et al. 2012; Qin et al. 2013). More specifically, the mean annual temperature has increased by 1.6 °C during the last century in northwestern Himalaya with the maximum temperature showing a more rapid increase. Winter temperature has shown a more noticeable increase in the last century. The total annual precipitation has shown an overall decreasing trend in the northwestern Himalaya (Bhutiyani et al. 2007, 2009). The Himalaya plays an important role in climate regulation of the Indian subcontinent and supports the livelihood of millions of the people dependent on the ecosystem services provided by it (Gansser 1980; Singh and Singh 1987; Carrico et al. 2003; Anthwal et al. 2010; Viviroli et al. 2011; Ganjoo and Ota 2012). Despite being a global biodiversity hotspot, there are limited studies that have investigated the response of tree growth to changing climate in the Himalayan region (Schickhoff et al. 2015; Tiwari et al. 2017; Pandey et al. 2018). The plants growing in the Himalaya must quickly adapt to survive future climatic conditions. The variations in climate affect tree growth which significantly influence cambial phenology and wood formation (Camarero et al. 2010; Rossi et al. 2014).
Tree growth is largely influenced by endogenous factors like phytohormones and enzymes, and exogenous factors which include climatic variables, soil, slope and aspect. Climate is the most important factor for the regulation of growth particularly near the ecological boundaries of a species (Fritts 1972). Generally, the response of tree growth to climate is dependent on the habitat, species, age etc. Tree growth is sensitive to changes in regional environmental conditions (Korner 2007; McDowell et al. 2008). Given the predicted climate change, it is crucial to understand the tree growth and its response to climatic variability.
Himalayan conifers are known to have a huge dendroclimatic potential because of their age and climatic sensitivity (Ramesh et al. 1985, 1986a; Bhattacharyya et al. 1988). Several researchers have studied the role of climate in regulating the growth in various Himalayan conifers using annual growth rings (Borgaonkar et al. 1996, 2001; Yadav et al. 1997; Ahmed et al. 2010; Sohar et al. 2017) and wood micro-core samples for understanding the intra-annual dynamics of wood formation (Malik et al. 2020a, b). Trees in the treeline ecotone are strongly limited by temperature (Li et al. 2013, 2016, 2017), with an average growing season temperature around 6–7 °C (Körner 2003; Körner and Paulsen 2004). A detailed study on vascular cambium activity by Rossi et al., (2007a, b) at the alpine timberline showing that the cambium was active at a daily mean air temperature of 5.6–8.5 °C and a mean stem temperature of 7.2–9.0 °C. An increase in the growing season temperature would thus result in a longer time for cambial activity and hence more radial growth (Deslauriers et al. 2008a).
Tree rings have been commonly used as a proxy to investigate biological responses to regional/local climate as well as to study the temporal stability of these responses (Wang et al. 2005; Jump et al. 2007; Fan et al. 2009; Dang et al. 2013; He et al. 2013; Sohar et al. 2017). This approach provides a long time series at high spatiotemporal resolution and helps in better understanding of tree responses to ongoing climate change (Sohar et al. 2017). Many studies have shown that tree growth response to climate varies along the altitudinal range of a species (Kahle et al. 2002; Savva et al. 2006; Spiecker 2010; Dang et al. 2013; Kharal et al. 2015). Generally, trees respond positively to summer temperature in high altitudes to growing season precipitation in lower altitudinal limits. A better understanding of the climate-growth relationships in northwestern Himalaya will help in the assessment of the growth response of Himalayan conifers to changing climate and will help simulate the carbon sequestration potential of these forests in response to predicted climate change.
In the northwestern Himalayan region, tree rings have been widely used mostly for climate reconstruction (Ramesh et al. 1986a; Hughes 2001; Yadav 2009; Singh et al. 2009; Ram and Borgaonkar 2013, 2016; Yadav and Bhutiyani 2013; Shah et al. 2018). One of the most dominant conifer in the northwestern Himalayan coniferous forests is Abies pindrow (Royle ex D. Don) Royle. The climate sensitivity of A. pindrow has been utilized by dendroclimatologists to reconstruct regional temperature (Hughes 2001; Malik and Sukumar 2021) and precipitation (Borgaonkar et al. 1994a; Shah et al. 2018). Similarly, reconstruction of pre-monsoon relative humidity was carried out by Dhyani et al. (2021) using tree-ring data of Pinus roxburghii from Western Himalaya.
Generally, tree growth and climate relationships are assumed to be stable over time (Fritts et al. 1965). However, recent tree ring studies reported problems with this stable climate-growth relationship assumption. Some studies have found decrease or loss of temperature sensitivity (Jacoby and D’Arrigo 1995; Briffa et al. 1998; Smith et al. 1999; Jacoby et al. 2000; Solberg et al. 2002), whereas others have reported increased temperature sensitivity (Knapp et al. 2001; Wilmking 2005) and some studies have reported a shift from negative to positive temperature sensitivity or vice versa especially after the 1950s (Zhang et al. 2008; Wilmking and Myers-Smith 2008). These shifts in growth-climate relationships over time are probably because of some mechanisms, such as temperature-induced drought stress (Jacoby et al. 1996; Barber et al. 2000; Lloyd and Fastie 2002), delayed snowmelt and related change in seasonality (Vaganov et al. 1999), non-linear thresholds or time-dependent response to recent temperature warming (D’Arrigo et al. 2004; Wilmking et al. 2004; Rossi et al. 2007a), air pollution (Wilson and Elling 2004; Yonenobu and Eckstein 2006) and differential growth-climate relationships to mean, maximum and minimum temperatures (Wilson and Luckman 2002, 2003). There are other researchers who attribute these changed patterns in growth-climate relationships to methods of chronology development, biases in instrumental climate data and modelling (Cook and Peters 1997; Melvin et al. 2007; D’Arrigo et al. 2008).
In this study, we attempt to assess the response of radial growth of Abies pindrow (Royle ex D.Don) to monthly temperature and precipitation using standard dendroclimatological techniques. In addition, we investigated the temporal stability of growth-climate relationships using moving correlation analyses.
20.2 Materials and Methods
20.2.1 Study Site and Climate
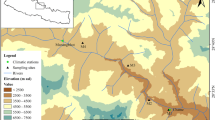
This study was carried out in Hirpora Wildlife Sanctuary (HWS) in district Shopian, Jammu and Kashmir, India (33o40′N, 74o42′E). The HWS lies in the Pir-Panjal range of northwestern Himalaya approximately 70 km south-west of Srinagar city covering an area of approximately 341.25 km2 (Ahmad et al. 2015) (Fig. 20.1). The Kashmir valley (Vale of Kashmir) is in the Northwest Himalaya lying between 33°20′–34°54′ N latitudes and 73°55′–75°35′ E longitudes covering an area of nearly 15, 948 km2 (Dar and Khuroo 2013) and is surrounded by the Karakoram range in the north, Pir-Panjal range in the south and west, Zanskar range in the east and Siwalik hills in the south (Pandey et al. 2016).
The vegetation of HWS is represented by moist temperate coniferous forest with sub-alpine and alpine grasslands at higher altitudes of the sanctuary. A. pindrow (silver fir/Himalayan pindrow fir) is the dominant conifer species, in association with Pinus wallichiana A. B. Jacks and Picea smithiana (Wall.) Boiss., growing on northern slopes from 2300 to 3200 m a.s.l. (Sharma and Baduni 2001). The forest type in HWS is a mixed coniferous forest with sub-alpine and alpine pastures at the higher altitudes (Ahmad et al. 2015).
The climate of the Kashmir valley is influenced by westerly disturbance (also known as ‘westerlies’) (Dimri et al. 2015). During winter, these westerlies originate from the Mediterranean and reach the Kashmir valley, bringing precipitation in the form of snow. Hirpora Wildlife Sanctuary has a sub-humid temperate climate. There is no weather station in the HWS, so climate data of the Srinagar meteorological station was used for this study (Fig. 20.2). The Srinagar station is the only station with more than 100 years of climate data in the Kashmir valley. The climate data from this station has earlier been used in many scientific studies (Ramesh et al. 1986b; Hughes 2001; Bhutiyani et al. 2007; Jeelani et al. 2012). Based on the monthly climatic data from Srinagar meteorological station, located at approximately 70 km from the study site, for the period 1901–2016, the mean annual temperature of the region is 13.4 °C, with July being the hottest month (mean maximum temperature = 24.2 °C) and January the coldest month (mean minimum temperature = 1.6 °C). Temperature goes to sub-zero in the winter months. The annual average precipitation is 680 mm, with March and April being the wettest months with an average rainfall of 102.8 and 90.8 mm respectively, while October and November are the driest months with total precipitation of 30 and 20.2 mm respectively. Most of the precipitation falls in the form of snow during the winter months.
The mean annual temperature has shown a significant increase in the last century as is evident from the Srinagar meteorological station data (Fig. 20.2). The magnitude of this increasing trend varies pre-and post-1950s with a greater increase post-1950s. There is no significant variation in the total annual precipitation in the two time periods. Because of the varying trends in the climate data of Srinagar, the growth-climate relationship analyses were done separately with these two time period climate data to understand the influence of recent climate warming on these relationships.
20.2.2 Data Collection
Near the highest distribution limits of the species in HWS, 40 healthy trees were selected for sampling. The diameter at breast height (DBH) of all the sampled trees was measured. A total of 73 increment cores were collected from the sampled trees. Usually, two cores per tree were collected parallel to the slope to avoid reaction wood. The samples were collected during 2014–2016. The cores were packed in straws and transported to the laboratory for further analysis. In the laboratory, the cores were glued to wooden holders and sequentially sanded using progressively finer grit sandpapers (80–3000 grit). The sanded cores were scanned in the high-resolution Espon Perfection V700 Photo scanner® at 2400 dpi to make ring boundaries visible. The scanned images were saved in JPEG format which is compatible with CooRecorder/CDendro software (Cybis Elektronik 2010). The ring-width measurements were done in CooRecorder/CDendro software package. Measurements were first visually cross-dated and then cross-checked with computer programme COFECHA (Holmes 1983) and dplR (Bunn 2010a, b) to ensure that each ring is assigned to its correct year of formation. COFECHA is a statistical tool in dendrochronology used for cross-dating purpose (Grissino-Mayer 2001; Speer 2010). The raw chronology was developed by taking the robust mean of all the series (Fig. 20.3). Various descriptive statistics such as mean ring-width, series-intercorrelation, the average mean sensitivity and first-order auto-correlation were computed for a general understanding of the site chronology. The ring width describes the radial growth in a site, mean sensitivity indicates the year-to-year variability between the consecutive tree-rings and the first-order auto-correlations describe the effect of the previous year growing conditions on the growth of the current year (Fritts 1972). The age of the trees was calculated by counting the number of annual growth rings. The mean ring-width of five innermost existing growth rings was used to estimate the number of years to pith in samples, wherein the pith was missing using CooRecorder software (Cybis Elektronik 2010).
20.2.3 Chronology Development
The raw ring-width series were standardized to convert them into dimensionless indices using dplR package (Bunn 2008) in the R version 4.0.5 statistical programme (R Core Team 2020). The raw ring-width series were detrended using cubic spline curves of 32 years to remove the non-climatic trends due to tree age, size and forest stand dynamics and the ratios were calculated (Cook et al. 1990). The main criteria for detrending were to minimize the non-climatic low-frequency trend and maximize the high-frequency trend. The detrended series were averaged by computing a bi-weight robust mean to make the standardized site chronology (Cook 1985). The standardized site chronology showed high first-order auto-correlation values because of the impact of the previous year growing season on the current year growth. This persistence in the standardized chronology was removed by autoregressive modelling and the resulting residual series were averaged by computing bi-weight robust mean to make residual site chronology, henceforth referred to as Site chronology (Fig. 20.4) (Cook 1985).
20.2.4 Dendroclimatic Analysis
The relationships between site chronology and monthly meteorological records were analysed by using Pearson’s correlation coefficients. The statistical significance of correlation coefficients was estimated by calculating bootstrapped confidence intervals. Monthly temperature and precipitation data from January to October, as well as mean of May–August temperature (mean-MJJA) and the sum of May–August precipitation (total-MJJA), was used for correlation analysis. The monthly correlation analysis was done for two time periods of 1901–1950 and 1951–2016 in addition to the full time period, henceforth referred to as pre-and post-1950s, to understand the impact of climate change on growth-climate relationships.
Further, the temporal stability of growth-climate relationships was investigated by running correlation analyses using 30-year moving intervals (climate data) with a 3-year offset to understand the variability of growth-climate relationships in smaller time periods of 30 years. The dynamic moving correlation analyses were run with the treeclim package (Zang and Biondi 2015) in R version 4.0.5 (R Core Team 2020).
20.3 Results
There is a stronger increasing trend in mean annual temperature after 1950s (R2 = 0.24; p < 0.001) than before (R2 = 0.096; p = 0.028), while there is no significant trend in total annual precipitation in both the analysed time periods (p > 0.05). This trend in mean annual temperature showed in growth-climate relationships in the two time periods of 1901–1950 and 1951–2016 respectively. The annual ring widths showed a significant variation between the two time periods (p = 0.01) (Fig. 20.5). The overall mean ring-width was 1.43 mm. The ring-width measured prior to 1950 were significantly smaller (p = 0.013) than the post-1950 time period with a mean ring-width of 1.25 and 1.33 mm respectively.
The average tree age was 229 years with a maximum age of 439 years (Table 20.1). There was a significant linear relationship between DBH and age of trees (p < 0.01). The age showed a linear increase with the increase in the diameter of individual trees (Fig. 20.6).
The chronology showed a good sensitivity to climatic conditions as depicted by a good mean sensitivity value (0.18). The first-order autocorrelation was high in the raw ring-width series (0.83) indicating the larger impact of previous year growth on current radial growth. The individual series showed lower auto-correlation after detrending and successive fitting of auto-regressive models (0.45). The expressed population signal (EPS) was more than 0.85 since 1765, which indicates that the chronology well represents the overall population, and can be reliably used for dendroclimatic studies. In addition, the strong series inter-correlation indicates the overall growth similarity between individual trees.
The response of radial growth to individual monthly climate changed during the two periods (Fig. 20.7). The positive response to January, February, March and April temperature in the pre-1950 changed to less positive or shifted to negative in the post-1950 time period, while May to October showing negative response in the pre-1950 period changed to less negative or responded positively in the later time period. The radial growth responded negatively to the mean of May–July temperature in the former time period, while the response was positive in the latter.
In pre-1950 period, the ring-width chronology showed strong negative response to October temperature (r = −0.37, p < 0.01), while the response was not significant to any other month temperature irrespective of the time period. The highest positive response was shown to January in 1901–2016 (r = 0.18, p = 0.05) and 1901–1950 (r = 0.24, p = 0.09) time periods, while July temperature showed maximum positive response during post-1950s time period (r = 0.23, p = 0.06). Likewise, the maximum negative response was shown to March temperature in 1901–2016 (r = −0.11, p = 0.22) and 1951–2016 (r = −0.18, p = 0.14) time periods, while the response was significantly negative to October temperature during 1901–1950 time period (r = −0.37, p < 0.01).
Similarly, the response to monthly precipitation varied in the two time periods as well. The overall response to growing season precipitation was weaker in the post-1950 time period. The response shifted from negative to either less negative or slightly positive, while there was a shift from positive to less positive or negative between the two studied time periods. Only August (r = 0.29, p < 0.05) and total-MJJA (r = 0.40, p < 0.01) precipitation showed significant positive response in the pre-1950s time period. None of the months in 1901–2016, as well as 1951–2016 time periods, responded significantly to radial growth at treeline site. The maximum positive response during 1901–2016 was shown to March precipitation (r = 0.15, p = 0.11), while August and total-MJJA precipitation responded positively during the pre-1950s time period. During 1951–2016 time period, the maximum positive response was shown to February precipitation (r = 0.24, p = 0.05). Similarly, the highest negative response was shown to September precipitation in all the studied time periods with correlation coefficients of −0.14, −0.21 and −0.11 in 1901–2016, pre-1950s and post-1950s time periods respectively.
The 30-year moving window analysis showed periods of low and high response to monthly climate data (Fig. 20.8). The general trend of responding to two time periods was seen in the moving correlation results as well. The strong negative response was shown to October month temperature during the 30-year window of 1901–1930 (r = −0.45, p < 0.01), while the strong positive response to shown to July temperature during 1976–2005 (r = 0.50, p < 0.01). The response to mean-MJJA temperature changed from being strongly negative (r = −0.34, p < 0.05) during 1922–1951 window to strongly positive (r = 0.50, p < 0.01) during the 30-year window of 1961–1990.
The moving correlation analysis with monthly precipitation data showed a strong negative response to October precipitation (r = −0.41, p < 0.05) during 1931–1960, while the response was strongly positive to July precipitation (r = 0.44, p < 0.05) during 1910–1939 time period. Likewise, the response to total growing season precipitation changed from strong negative (r = −0.27, p > 0.05) during 1985–2014 to strong positive (r = 0.49, p < 0.01) during the 1904–1933 time period.
20.4 Discussion
We present a dendroclimatology of A. pindrow that contributes to a better understanding of its response to recent climate change in the northwestern Himalaya. Due to its longevity and strong climate sensitivity, A. pindrow has been the subject of many dendroclimatological studies (Ramesh et al. 1985, 1986c; Borgaonkar et al. 1994b).
We found a linear relationship between tree age and diameter at breast-height (DBH). This relationship makes it easier to calculate the age of a forest stand at the treeline ecotone. Also, distinct differences were found between annual growth increments between two time periods. The relatively larger increase in average annual temperatures might have resulted in longer growing season duration and hence wider growth rings (Deslauriers et al. 2008b; Rossi et al. 2011; Malik et al. 2020a).
Radial growth in trees is affected by several biotic as well as abiotic factors (Briffa et al. 2004; Treydte et al. 2006; Yadav et al. 2011). Climate is one of the most important environmental factors affecting radial growth in trees. Temperate trees produce distinct growth rings which are easier to correlate with the regional climatic conditions. Trees annually record growth data in the form of the width of annual growth rings in their trunks, with wide rings during years of optimal climatic conditions and narrow rings during poor climatic conditions (Vaganov et al. 1999). Dendroclimatologists have developed time series of tree-ring widths from thousands of sites worldwide because of the well-established relationship between ring-width and climate (archived by the National Climate Data Center (NCDC) in the International Tree-Ring Data Bank (ITRDB; available online at http://www.ncdc.noaa.gov/paleo/treering.html). Several researchers have used statistical relationships between annual tree rings and climate to infer forest responses to future climate change.
Overall, the radial growth in pindrow fir responded positively to monthly temperature in recent decades. The positive response of tree growth to the temperature at the treeline site is more of a general phenomenon (Wilson and Hopfmüller 2001; Takahashi et al. 2003). Trees at treeline did not respond to growing season temperature in the first half of the twentieth century and responded positively in the second half. This indicates that radial growth trees at the tree line ecotone might be benefitted from climate warming. The higher temperatures could increase the rate of photosynthesis and thus enhancing radial growth (White 1987; Zhang and Hebda 2004). A significantly lower number of tracheids are produced at the higher altitudinal limits compared to lower limits in the northwestern Himalaya (Malik et al. 2020a) which could be because of favourable temperatures for a relatively short duration of time. Increasing temperatures might potentially lead to a lengthening of growing season duration and hence results in more wood formation (Deslauriers et al. 2008a, b; Rossi et al. 2008, 2016; Moser et al. 2010; Li et al. 2016).
The impact of precipitation was weaker at the tree line site. The weaker impact of growing season precipitation on pindrow fir growth at its highest distributional limit is likely caused by good water availability due to snowmelt and rainfall during the growing season (Kim 2000; Körner 2007). The decreasing importance of precipitation at higher altitudes was reported in many other studies as well (Kahle et al. 2002; Jump et al. 2007). High temperatures during summer promote radial growth at higher altitudes. Generally, precipitation and soil moisture increase with increasing altitude, whereas air temperature decreases with increasing altitude, suggesting that radial growth in A. pindrow at upper limits was limited more by low temperature than by precipitation (Bradley and Fritts 1978). These results were in agreement with other studies showing that radial growth responds positively to growing season precipitation at lower altitudes, while radial growth responds positively to growing season temperature at higher altitudes (Wilson and Hopfmüller 2001; Takahashi et al. 2003; Dang et al. 2013).
Also, there are significant impacts of previous year climatic conditions on current year growth as is evident from higher first-order auto-correlations. As found by other researchers working with subalpine conifers, the previous season climatic conditions influence bud formation, increased foliage and production of photosynthetic assimilates, which in turn impacts the wood production in the subsequent growing season (Bradley and Fritts 1978; Savva et al. 2006).
The impact of climate on pindrow fir growth was not stable over time. The climatic conditions especially temperature varied between the two selected time periods, pre- and post-1950. The variation in climatic conditions is depicted in the climate-growth relationships as well. The response to monthly climate shows a variation between the two selected time periods. Before 1950, the radial growth responds negatively to growing season monthly temperature and positively to precipitation. These effects of temperature and precipitation on A. pindrow growth, however, changed in recent decades. Growth responded positively to monthly temperatures, while the response to monthly precipitation diminished in the latter part of the twentieth century. Because of temporal variability in climate, response to climatic conditions has varied during the twentieth century generally referred to as the ‘divergence effect’ (Briffa et al. 1998). The variation in growth-climate relationships in time is more of a general phenomenon (Carrer et al. 2010). The shift in growth-climate relationships is a reliable indicator of varying tree growth response to climate (Di Filippo et al. 2007a, b). The temporal instability of growth-climate relationships might be because of possible changes in climate and other environmental conditions (Carrer and Urbinati 2006; Di Filippo et al. 2007a, b; Leonelli et al. 2009; Dang et al. 2013). Many recent studies also showed the instability of growth-climate relationships mostly in the northern latitudes (Jacoby et al. 1996, 2000; Briffa et al. 1998; Smith et al. 1999; Knapp et al. 2001; Solberg et al. 2002; Wilmking et al. 2004; Wilmking 2005). The variations in the growth-climate relationships in pindrow fir might be attributed to regional climate and other local environmental changes, which disrupted the growth-climate relationships and subsequently resulted in changed climate sensitivity and growth trends (Carrer and Urbinati 2006; Leonelli et al. 2009; Dang et al. 2013; Sohar et al. 2017). The appearance or loss of significance of climate sensitivity of certain climatic variables might be because of threshold-controlled mechanisms, as were detected in other climate-growth relationship studies (D’Arrigo et al. 2004; Wilmking et al. 2004; Carrer and Urbinati 2006; Rossi et al. 2007b; Dang et al. 2013).
The results from moving correlation analyses are in good agreement with the results from static correlation analyses for two time periods, pre-and post-1950. The response to mean-MJJA temperature changed from being negative in the first half of the twentieth century to being positive in the latter part of the century as shown in both the static and moving correlation analyses. The shifting response indicates the positive impact of ongoing warming summer temperatures on the radial growth and hence overall carbon sequestration of the forest stands towards higher altitudinal limits. The response to total-MJJA precipitation shifted from being strongly positive in the first part of the century to not responding in the later part of the climate data. The weaker response to growing season precipitation towards the higher altitudinal limits of the species could be possibly the result of enough availability of soil moisture, due to delayed snowmelt as well as good rainfall during this period.
The investigation of growth-climate relationships in different time periods gives a better indication of varying responses of radial growth to climate. Such studies are important for a better understanding of the growth responses of Himalayan tree species to climate change. Better clarity regarding growth responses to local climate will have management implications for afforestation programmes keeping in view the future carbon sequestration potential of Himalayan forests.
20.5 Conclusion
This study provides insights into the response of radial growth in A. pindrow to monthly temperature and precipitation at its highest distribution limit in the northwestern Himalaya. The climate data used in this study showed an overall increase in annual temperature and more or less stable annual precipitation. The growth responded positively to the growing season monthly temperature in the recent decades. The positive response of monthly precipitation on radial growth decreased in recent decades. Specifically, the study provides evidence of varying response of pindrow fir growth to recent climate change. The radial growth is strongly assisted by recent warming, while the positive impact of precipitation diminished in recent decades. In general, pindrow fir in the northwestern Himalaya is responsive to growing season temperature, and therefore the increase in temperature in combination with enriched soil moisture will possibly lead to growth enhancement in this species near its higher distribution limits. We highlight the importance of studying the temporal stability of growth-climate relationships to provide evidence about the impacts of climate change on forest ecosystems.
References
Ahmad F, ul Hassan Z, Farooq S, Shah JA (2015) Assessment of land use/land cover change in Hirpora wildlife sanctuary, Kashmir. Asian J Earth Sci 8:64–73. https://doi.org/10.3923/ajes.2015.64.73
Ahmed M, Khan N, Wahab M (2010) Climate response function analysis of Abies Pindrow (Royle) Spach. Preliminary results. Pak J Bot 42:165–171
Anthwal A, Gupta N, Sharma A et al (2010) Conserving biodiversity through traditional beliefs in sacred groves in Uttarakhand Himalaya, India. Resour Conserv Recycl 54:962–971. https://doi.org/10.1016/j.resconrec.2010.02.003
Aryal A, Brunton D, Raubenheimer D (2014) Impact of climate change on human-wildlife-ecosystem interactions in the trans-Himalaya region of Nepal. Theor Appl Climatol 115:517–529. https://doi.org/10.1007/s00704-013-0902-4
Barber VA, Juday GP, Finney BP (2000) Reduced growth of Alaskan white spruce in the twentieth century from temperature-induced drought stress. Nature 405:668–673. https://doi.org/10.1038/35015049
Bhattacharyya A, Lamarche V, Telewski F (1988) Dendrochronological reconnaissance of the conifers of Northwest India. Tree Ring Bull 48:21–30
Bhutiyani MR, Kale VS, Pawar NJ (2007) Long-term trends in maximum, minimum and mean annual air temperatures across the northwestern Himalaya during the twentieth century. Clim Chang 85:159–177. https://doi.org/10.1007/s10584-006-9196-1
Bhutiyani MR, Kale VS, Pawar NJ (2009) Climate change and the precipitation variations in the northwestern Himalaya: 1866–2006. Int J Climatol. https://doi.org/10.1002/joc.1920
Borgaonkar HP, Pant GB, Rupa Kumar K (1994a) Dendroclimatic reconstruction of summer precipitation at Srinagar, Kashmir, India, since the late-eighteenth century. The Holocene 4:299–306
Borgaonkar HP, Pant GB, Rupa Kumar K (1994b) Dendroclimatic reconstruction of summer precipitation at Srinagar, Kashmir, India, since the late-eighteenth century. The Holocene 4:299–306. https://doi.org/10.1177/095968369400400309
Borgaonkar HP, Pant GB, Rupa Kumar K (1996) Ring-width variations in Cedrus deodara and its climatic response over the western Himalaya. Int J Climatol 16:1409–1422
Borgaonkar HP, Kumar KR, Pant GB et al (2001) Climatic implications of tree-ring density variations in Himalayan conifers. Palaeobotanist 50:27–34
Bradley RS, Fritts HC (1978) Tree rings and climate. Arct Alp Res 10:144. https://doi.org/10.2307/1550667
Briffa KR, Schweingruber FH, Jones PD et al (1998) Reduced sensitivity of recent tree-growth to temperature at high northern latitudes. Nature 391:678–682. https://doi.org/10.1038/35596
Briffa KR, Osborn TJ, Schweingruber FH (2004) Large-scale temperature inferences from tree rings: a review. Glob Planet Chang 40:11–26. https://doi.org/10.1016/S0921-8181(03)00095-X
Bunn AG (2008) A dendrochronology program library in R (dplR). Dendrochronologia 26:115–124. https://doi.org/10.1016/j.dendro.2008.01.002
Bunn AG (2010a) Statistical and visual crossdating in R using the dplR library. Dendrochronologia 28:251–258. https://doi.org/10.1016/j.dendro.2009.12.001
Bunn AG (2010b) Statistical and visual crossdating in R using the dplR library. Dendrochronologia 28:251–258
Camarero JJ, Olano JM, Parras A (2010) Plastic bimodal xylogenesis in conifers from continental Mediterranean climates. New Phytol 185:471–480. https://doi.org/10.1111/j.1469-8137.2009.03073.x
Carrer M, Urbinati C (2006) Long-term change in the sensitivity of tree-ring growth to climate forcing in Larix decidua. New Phytol 170:861–872. https://doi.org/10.1111/j.1469-8137.2006.01703.x
Carrer M, Nola P, Motta R, et al (2010) Contrasting tree-ring growth to climate responses of Abies alba toward the southern limit of its distribution area published by : Wiley on behalf of Nordic society Oikos stable URL: https://www.jstor.org/stable/20779075 linked references are available on. 119:1515–1525. https://doi.org/10.1111/j.l
Carrico CM, Bergin MH, Shrestha AB et al (2003) The importance of carbon and mineral dust to seasonal aerosol properties in the Nepal Himalaya. Atmos Environ 37:2811–2824. https://doi.org/10.1016/S1352-2310(03)00197-3
Cook ER (1985) A time series analysis approach to tree ring standardization
Cook ER, Peters K (1997) Calculating unbiased tree-ring indices for the study of climatic and environmental change. The Holocene 7:361–370. https://doi.org/10.1177/095968369700700314
Cook ER, Briffa K, Shiyatov S, Mazepa V (1990) Tree-ring standardization and growth-trend estimation
Cybis Elektronik (2010) CDendro and CooRecorder., http://www.cybis.se/forfun/dendro/index.htm
D’Arrigo RD, Kaufmann RK, Davi N et al (2004) Thresholds for warming-induced growth decline at elevational tree line in the Yukon Territory, Canada, Global Biogeochem Cycles. 18. https://doi.org/10.1029/2004GB002249
D’Arrigo R, Wilson R, Liepert B, Cherubini P (2008) On the ‘divergence problem’ in northern forests: a review of the tree-ring evidence and possible causes. Glob Planet Chang 60:289–305. https://doi.org/10.1016/j.gloplacha.2007.03.004
Dang H, Zhang Y, Zhang K et al (2013) Climate-growth relationships of subalpine fir (Abies fargesii) across the altitudinal range in the Shennongjia Mountains, Central China. Clim Chang 117:903–917. https://doi.org/10.1007/s10584-012-0611-5
Dar GH, Khuroo AA (2013) Floristic diversity in the Kashmir Himalaya: Progress, problems and prospects. Sains Malays 42:1377–1386
Deslauriers A, Rossi S, Anfodillo T, Saracino A (2008a) Cambial phenology, wood formation and temperature thresholds in two contrasting years at high altitude in southern Italy. Tree Physiol 28:863–871. https://doi.org/10.1093/treephys/28.6.863
Deslauriers A, Rossi S, Anfodillo T, Saracino A (2008b) Cambial phenology, wood formation and temperature thresholds in two contrasting years at high altitude in southern Italy. Tree Physiol 28:863–871
Dhyani R, Shekhar M, Joshi R et al (2021) Reconstruction of pre-monsoon relative humidity since 1800 C.E. based on tree-ring data of Pinus roxburghii Sarg. (chir–pine) from Pithoragarh, Western Himalaya. Quat Int. https://doi.org/10.1016/j.quaint.2021.04.026
Di Filippo A, Biondi F, Čufar K et al (2007a) Bioclimatology of beech (Fagus sylvatica L.) in the eastern Alps: spatial and altitudinal climatic signals identified through a tree-ring network. J Biogeogr 34:1873–1892. https://doi.org/10.1111/j.1365-2699.2007.01747.x
Di Filippo A, Biondi F, Čufar K et al (2007b) Bioclimatology of beech (Fagus sylvatica L.) in the eastern Alps: spatial and altitudinal climatic signals identified through a tree-ring network. J Biogeogr 34:1873–1892. https://doi.org/10.1111/J.1365-2699.2007.01747.X
Dimri AP, Niyogi D, Barros AP et al (2015) Western disturbances: a review. Rev Geophys 53:225–246. https://doi.org/10.1002/2014RG000460
Fan Z-X, Bräuning A, Cao K-F, Zhu S-D (2009) Growth–climate responses of high-elevation conifers in the central Hengduan Mountains, southwestern China. For Ecol Manag 258:306–313. https://doi.org/10.1016/j.foreco.2009.04.017
Field CB, Lobell DB, Peters HA, Chiariello NR (2007) Feedbacks of terrestrial ecosystems to climate change. Annu Rev Environ Resour 32:1–29
Foster P (2001) The potential negative impacts of global climate change on tropical montane cloud forests. Earth-Sci Rev 55:73–106
Fritts HC (1972) Tree rings and climate. Sci Am 226:92–100. https://doi.org/10.1038/scientificamerican0572-92
Fritts HC, Smith DG, Cardis JW, Budelsky CA (1965) Tree-ring characteristics along a vegetation gradient in northern Arizona. Ecology 46:393–401. https://doi.org/10.2307/1934872
Ganjoo RK, Ota SB (2012) Mountain environment and early human adaptation in NW Himalaya, India: a case study of Siwalik Hill range and Leh valley. Quat Int 269:31–37. https://doi.org/10.1016/j.quaint.2011.11.033
Gansser A (1980) The significance of the Himalayan suture zone. Tectonophysics 62:37–52. https://doi.org/10.1016/0040-1951(80)90134-1
Grimm NB, Chapin FS, Bierwagen B et al (2013) The impacts of climate change on ecosystem structure and function. Front Ecol Environ 11:474–482
Grissino-Mayer HD (2001) Evaluating crossdating accuracy: a manual and tutorial for the computer program COFECHA. Tree-Ring Res 57:205–221. https://doi.org/S
He M, Yang B, Bräuning A (2013) Tree growth–climate relationships of Juniperus tibetica along an altitudinal gradient on the southern Tibetan plateau. Trees 27:429–439. https://doi.org/10.1007/s00468-012-0813-5
Holmes RL (1983) Computer-assisted quality control in tree-ring dating and measurement. Tree-Ring Bull. https://doi.org/10.1016/j.ecoleng.2008.01.004
Huggel C, Clague JJ, Korup O (2012) Is climate change responsible for changing landslide activity in high mountains? Earth Surf Process Landforms 37:77–91. https://doi.org/10.1002/esp.2223
Hughes MK (2001) An improved reconstruction of summer temperature at Srinagar, Kashmir since 1660 AD, based on tree-ring width and maximum latewood density of Abies pindrow [Royle] Spach. Palaeobotanist 50:13–19
IPCC (2014) Climate change 2013: the physical science basis: working group I contribution to the IPCC fifth assessment report. Annex 1 1–85. https://doi.org/10.1017/CBO9781107415324.Summary
Jacoby GC, D’Arrigo RD (1995) Tree ring width and density evidence of climatic and potential forest change in Alaska. Global Biogeochem Cycles 9:227–234. https://doi.org/10.1029/95GB00321
Jacoby GC, D’Arrigo RD, Davaajamts T (1996) Mongolian tree rings and 20th-century warming. Science (80-) 273:771–773. https://doi.org/10.1126/science.273.5276.771
Jacoby GC, Lovelius NV, Shumilov OI et al (2000) Long-term temperature trends and tree growth in the Taymir region of northern Siberia. Quat Res 53:312–318. https://doi.org/10.1006/qres.2000.2130
Jeelani G, Feddema JJ, van der Veen CJ, Stearns L (2012) Role of snow and glacier melt in controlling river hydrology in Liddar watershed (western Himalaya) under current and future climate. Water Resour Res 48. https://doi.org/10.1029/2011WR011590
Jump A, Hunt J, Penuelas J (2007) Climate relationships of growth and establishment across the altitudinal range of Fagus sylvatica in the Montseny Mountains, northeaast Spain. Ecoscience 14:507–518
Kahle H, Neumann U, Mäkinen H et al (2002) Radial growth variation of Norway spruce (Picea abies (L.) karst.) across latitudinal and altitudinal gradients in central and northern Europe. For Ecol Manag 171:243–259. https://doi.org/10.1016/S0378-1127(01)00786-1
Kahn ME (2005) The death toll from natural disasters: the role of income, geography, and institutions. Rev Econ Stat 87:271–284. https://doi.org/10.1162/0034653053970339
Kharal DK, Meilby H, Rayamajhi S et al (2015) Tree ring variability and climate response of Abies spectabilis along an elevation gradient in mustang, Nepal. Banko Janakari 24:3. https://doi.org/10.3126/banko.v24i1.13473
Kim E-SY-S (2000) Possibility of climate change and simulation of soil moisture content on Mt. Hallasan National Park, Chejudo Island, Korea. Korean J Ecol 23:117–123
Kirilenko AP, Sedjo RA (2007) Climate change impacts on forestry. Proc Natl Acad Sci 104:19697–19702
Knapp PA, Soulé PT, Grissino-Mayer HD (2001) Detecting potential regional effects of increased atmospheric CO 2 on growth rates of western juniper. Glob Chang Biol 7:903–917. https://doi.org/10.1046/j.1365-2486.2001.00452.x
Körner C (2003) Plant ecology at high elevations. In: Alpine Plant Life. Springer, Berlin, pp 1–7
Korner C (2007) Significance of temperature in plant life. In: Plant growth and climate change. Blackwell Publishing Ltd, Oxford, pp 48–69
Körner C (2007) The use of ‘altitude’ in ecological research. Trends Ecol Evol 22:569–574. https://doi.org/10.1016/J.TREE.2007.09.006
Körner C, Paulsen J (2004) A world-wide study of high altitude treeline temperatures. J Biogeogr 31:713–732. https://doi.org/10.1111/j.1365-2699.2003.01043.x
Leonelli G, Pelfini M, Battipaglia G, Cherubini P (2009) Site-aspect influence on climate sensitivity over time of a high-altitude Pinus cembra tree-ring network. Clim Chang 96:185–201. https://doi.org/10.1007/s10584-009-9574-6
Li X, Liang E, Gricar J et al (2013) Age dependence of xylogenesis and its climatic sensitivity in Smith fir on the south-eastern Tibetan plateau. Tree Physiol 33:48–56
Li X, Rossi S, Liang E, Julio Camarero J (2016) Temperature thresholds for the onset of xylogenesis in alpine shrubs on the Tibetan plateau. Trees 30:2091–2099. https://doi.org/10.1007/s00468-016-1436-z
Li X, Liang E, Gričar J et al (2017) Critical minimum temperature limits xylogenesis and maintains treelines on the southeastern Tibetan plateau. Sci Bull 62:804–812
Lloyd AH, Fastie CL (2002) Spatial and temporal variability in the growth and climate response of treeline trees in Alaska. Clim Chang 52:481–509. https://doi.org/10.1023/a:1014278819094
Malik R, Sukumar R (2021) June–July temperature reconstruction of Kashmir Valley from tree rings of Himalayan Pindrow Fir. Atmosphere (Basel) 12:410. https://doi.org/10.3390/atmos12030410
Malik R, Rossi S, Sukumar R (2020a) Cambial phenology in Abies pindrow (Pinaceae) along an altitudinal gradient in northwestern Himalaya. IAWA J 41:186–201. https://doi.org/10.1163/22941932-bja10007
Malik R, Rossi S, Sukumar R (2020b) Variations in the timing of different phenological stages of cambial activity in Abies pindrow (Royle) along an elevation gradient in the north-western Himalaya. Dendrochronologia 59:125660. https://doi.org/10.1016/j.dendro.2019.125660
McDowell N, Pockman WT, Allen CD et al (2008) Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought? New Phytol 178:719–739
Melvin TM, Briffa KR, Nicolussi K, Grabner M (2007) Time-varying-response smoothing. Dendrochronologia 25:65–69. https://doi.org/10.1016/j.dendro.2007.01.004
Moser L, Fonti P, Buntgen U et al (2010) Timing and duration of European larch growing season along altitudinal gradients in the Swiss Alps. Tree Physiol 30:225–233. https://doi.org/10.1093/treephys/tpp108
Pandey U, Shah SK, Mehrotra N (2016) Tree-ring studies from Kashmir Valley: present status and future perspectives tree-ring studies from Kashmir Valley: present status. Geophytology 46:207–220
Pandey S, Carrer M, Castagneri D, Petit G (2018) Xylem anatomical responses to climate variability in Himalayan birch trees at one of the world’s highest forest limit. Perspect Plant Ecol Evol Syst 33:34–41
Qin C, Yang B, Melvin TM et al (2013) Radial growth of qilian juniper on the northeast tibetan plateau and potential climate associations. PLoS One. https://doi.org/10.1371/journal.pone.0079362
R Core Team (2020) R: a language and environment for statistical computing. R Found. Stat. Comput, Vienna, Austria. https//www.R-project.org/
Ram S, Borgaonkar HP (2013) Growth response of conifer trees from high-altitude region of western himalaya. Curr Sci 105(2):225–231
Ram S, Borgaonkar HP (2016) Reconstruction of heat index based on tree-ring width records of western Himalaya in India. Dendrochronologia 40:64–71. https://doi.org/10.1016/j.dendro.2016.06.003
Ramesh R, Bhattacharya SK, Gopalan K (1985) Dendroclimatological implications of isotope coherence in trees from Kashmir Valley, India. Nature 317:802–804. https://doi.org/10.1038/317802a0
Ramesh R, Bhattacharya SK, Gopalan K (1986a) Climatic correlations in the stable isotope records of silver fir (Abies pindrow) trees from Kashmir, India. Earth Planet Sci Lett 79:66–74
Ramesh R, Bhattacharya SK, Gopalan K (1986b) Climatic correlations in the stable isotope records of silver fir (Abies pindrow) trees from Kashmir, India. Earth Planet Sci Lett 79:66–74. https://doi.org/10.1016/0012-821X(86)90041-5
Ramesh R, Bhattacharya SKK, Gopalan K (1986c) Climatic correlations in the stable isotope records of silver fir (Abies pindrow) trees from Kashmir, India. Earth Planet Sci Lett 79:66–74. https://doi.org/10.1016/0012-821X(86)90041-5
Rangwala I, Miller JR (2012) Climate change in mountains: a review of elevation-dependent warming and its possible causes. Clim Chang 114:527–547. https://doi.org/10.1007/s10584-012-0419-3
Rashid I, Romshoo SA, Chaturvedi RK et al (2015) Projected climate change impacts on vegetation distribution over Kashmir Himalayas. Clim Chang 132:601–613. https://doi.org/10.1007/s10584-015-1456-5
Rossi S, Deslauriers A, Anfodillo T, Carraro V (2007a) Evidence of threshold temperatures for xylogenesis in conifers at high altitudes. Oecologia 152:1–12. https://doi.org/10.1007/s00442-006-0625-7
Rossi S, Deslauriers A, Anfodillo T, Carraro V (2007b) Evidence of threshold temperatures for xylogenesis in conifers at high altitudes. Oecologia 152:1–12
Rossi S, Deslauriers A, Griçar J et al (2008) Critical temperatures for xylogenesis in conifers of cold climates. Glob Ecol Biogeogr 17:696–707. https://doi.org/10.1111/j.1466-8238.2008.00417.x
Rossi S, Morin H, Deslauriers A, Plourde P-Y (2011) Predicting xylem phenology in black spruce under climate warming. Glob Chang Biol 17:614–625
Rossi S, Girard MJ, Morin H (2014) Lengthening of the duration of xylogenesis engenders disproportionate increases in xylem production. Glob Chang Biol 20:2261–2271
Rossi S, Anfodillo T, Čufar K et al (2016) Pattern of xylem phenology in conifers of cold ecosystems at the northern hemisphere. Glob Chang Biol 22:3804–3813. https://doi.org/10.1111/gcb.13317
Savva Y, Oleksyn J, Reich PB et al (2006) Interannual growth response of Norway spruce to climate along an altitudinal gradient in the Tatra Mountains, Poland. Trees 20:735–746. https://doi.org/10.1007/s00468-006-0088-9
Schickhoff U, Bobrowski M, Böhner J et al (2015) Do Himalayan treelines respond to recent climate change? An evaluation of sensitivity indicators. Earth Syst Dynam 6:245–265
Shah SK, Pandey U, Mehrotra N (2018) Precipitation reconstruction for the Lidder Valley, Kashmir Himalaya using tree-rings of Cedrus deodara. Int J Climatol 38:e758–e773. https://doi.org/10.1002/joc.5405
Sharma CM, Baduni NP (2001) Effect of aspect on the structure of some natural stands of Abies pindrow in Himalayan moist temperate forest. Environmentalist 20:309–317
Shepherd A, Wingham D (2007) Recent Sea-Level Contributions of the Antarctic and Greenland Ice Sheets. Science (80-) 315:1529–1532. https://doi.org/10.1126/science.1136776
Shrestha UB, Gautam S, Bawa KS (2012) Widespread climate change in the Himalayas and associated changes in local ecosystems. PLoS One 7:e36741. https://doi.org/10.1371/journal.pone.0036741
Singh JS, Singh SP (1987) Forest vegetation of the Himalaya. Bot Rev 53:80–192. https://doi.org/10.1007/BF02858183
Singh J, Yadav RR, Wilmking M (2009) A 694-year tree-ring based rainfall reconstruction from Himachal Pradesh, India. Clim Dyn 33:1149–1158. https://doi.org/10.1007/s00382-009-0528-5
Smith KT, Cufar K, Levanic T (1999) Temporal stability and dendroclimatology in silver fir and red spruce. In: Phyton - Annales Rei Botanicae, pp 117–122
Sohar K, Altman J, Lehečková E, Doležal J (2017) Growth-climate relationships of Himalayan conifers along elevational and latitudinal gradients. Int J Climatol 37:2593–2605. https://doi.org/10.1002/joc.4867
Solberg BO, Hofgaard A, Hytteborn H (2002) Shifts in radial growth responses of coastal Picea abies induced by climatic change during the 20th century, Central Norway. Ecoscience 9:79–88
Speer JHJH (2010) Fundamentals of tree-ring research. University of Arizona Press, Tucson, p 333. https://doi.org/10.1002/gea.20357
Spiecker H (2010) Growth responses of Abies alba Mill and Picea abies (L.) Karst at different altitudes in south-western Germany, vol. 8, pp 21–25
Takahashi K, Azuma H, Yasue K (2003) Effects of climate on the radial growth of tree species in the upper and lower distribution limits of an altitudinal ecotone on mount Norikura, Central Japan. Ecol Res 18:549–558. https://doi.org/10.1046/j.1440-1703.2003.00577.x
Tiwari A, Fan Z-X, Jump AS, Zhou Z-K (2017) Warming induced growth decline of Himalayan birch at its lower range edge in a semi-arid region of trans-Himalaya, Central Nepal. Plant Ecol 218:621–633. https://doi.org/10.1007/s11258-017-0716-z
Treydte KS, Schleser GH, Helle G et al (2006) The twentieth century was the wettest period in northern Pakistan over the past millennium. Nature 440:1179–1182. https://doi.org/10.1038/nature04743
Vaganov EA, Hughes MK, Kirdyanov AV et al (1999) Influence of snowfall and melt timing on tree growth in subarctic Eurasia. Nature 400:149–151. https://doi.org/10.1038/22087
Viviroli D, Archer DR, Buytaert W et al (2011) Climate change and mountain water resources: overview and recommendations for research, management and policy. Hydrol Earth Syst Sci 15:471–504. https://doi.org/10.5194/hess-15-471-2011
Walther G-R, Post E, Convey P et al (2002) Ecological responses to recent climate change. Nature 416:389–395
Wang T, Ren H, Ma K (2005) Climatic signals in tree ring of Picea schrenkiana along an altitudinal gradient in the central Tianshan Mountains, northwestern China. Trees 19:736–742. https://doi.org/10.1007/s00468-005-0003-9
Webster PJ (2005) Changes in Tropical Cyclone Number, Duration, and Intensity in a Warming Environment. Science (80-) 309:1844–1846. https://doi.org/10.1126/science.1116448
White TL (1987) Drought tolerance of southwestern Oregon Douglas-fir. For Sci 33:283–293. https://doi.org/10.1093/forestscience/33.2.283
Wilmking M (2005) Increased temperature sensitivity and divergent growth trends in circumpolar boreal forests. Geophys Res Lett 32:L15715. https://doi.org/10.1029/2005GL023331
Wilmking M, Myers-Smith I (2008) Changing climate sensitivity of black spruce (Picea mariana mill.) in a peatland–forest landscape in interior Alaska. Dendrochronologia 25:167–175. https://doi.org/10.1016/j.dendro.2007.04.003
Wilmking M, Juday GP, Barber VA, Zald HSJ (2004) Recent climate warming forces contrasting growth responses of white spruce at treeline in Alaska through temperature thresholds. Glob Chang Biol 10:1724–1736. https://doi.org/10.1111/j.1365-2486.2004.00826.x
Wilson R, Elling W (2004) Temporal instability in tree-growth/climate response in the lower Bavarian Forest region: implications for dendroclimatic reconstruction. Trees - Struct Funct 18:19–28. https://doi.org/10.1007/s00468-003-0273-z
Wilson RJS, Hopfmüller M (2001) Dendrochronological investigations of Norway spruce along an elevational transect in the Bavarian Forest, Germany. Dendrochronologia 19:67–79
Wilson RJS, Luckman BH (2002) Tree-ring reconstruction of maximum and minimum temperatures and the diurnal temperature range in British Columbia, Canada. Dendrochronologia 20:257–268. https://doi.org/10.1078/1125-7865-00023
Wilson RJS, Luckman BH (2003) Dendroclimatic reconstruction of maximum summer temperatures from upper treeline sites in interior British Columbia, Canada. The Holocene 13:851–861. https://doi.org/10.1191/0959683603hl663rp
Yadav RR (2009) Tree ring imprints of long-term changes in climate in western Himalaya, India. J Biosci 34:699–707. https://doi.org/10.1007/s12038-009-0058-7
Yadav RR, Bhutiyani MR (2013) Tree-ring-based snowfall record for cold arid western Himalaya, India since a.D. 1460. J Geophys Res Atmos 118:7516–7522. https://doi.org/10.1002/jgrd.50583
Yadav RR, Park W-K, Bhattacharyya A (1997) Dendroclimatic reconstruction of April–May temperature fluctuations in the Western Himalaya of India since A.D. 1698. Quat Res 48:187–191. https://doi.org/10.1006/qres.1997.1919
Yadav RR, Braeuning A, Singh J (2011) Tree ring inferred summer temperature variations over the last millennium in western Himalaya, India. Clim Dyn 36:1545–1554. https://doi.org/10.1007/s00382-009-0719-0
Yonenobu H, Eckstein D (2006) Reconstruction of early spring temperature for central Japan from the tree-ring widths of Hinoki cypress and its verification by other proxy records. Geophys Res Lett 33. https://doi.org/10.1029/2006GL026170
Zang C, Biondi F (2015) Treeclim: an R package for the numerical calibration of proxy-climate relationships. Ecography (Cop) 38:431–436. https://doi.org/10.1111/ecog.01335
Zhang Q-B, Hebda RJ (2004) Variation in radial growth patterns of Pseudotsuga menziesii on the central coast of British Columbia, Canada. Can J For Res 34:1946–1954. https://doi.org/10.1139/x04-078
Zhang Y, Wilmking M, Gou X (2008) Changing relationships between tree growth and climate in Northwest China. In: Forest ecology. Springer, Dordrecht, pp 39–50
Acknowledgements
We thank Divecha Centre for Climate Change, IISc Bangalore for providing funding support for this project. R. Malik received the Merit Scholarship from Fonds de Recherche du Quebec – Nature et Technologies (FRQNT), Quebec, Canada. R. Malik also received Junior Research Fellowship from University Grants Commission (UGC), New Delhi. RS was a JC Bose Fellow during the tenure of this study. Also, thanks to Jammu and Kashmir Wildlife Department for giving permission to carry out this work in Hirpora Wildlife Sanctuary, J & K.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Malik, R.A., Sukumar, R. (2023). Response of Radial Growth in Abies pindrow (Royle ex D.Don) Royle to Climate at Treeline Ecotone in the northwestern Himalaya. In: Singh, S.P., Reshi, Z.A., Joshi, R. (eds) Ecology of Himalayan Treeline Ecotone. Springer, Singapore. https://doi.org/10.1007/978-981-19-4476-5_20
Download citation
DOI: https://doi.org/10.1007/978-981-19-4476-5_20
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-4475-8
Online ISBN: 978-981-19-4476-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)