Abstract
Purpose
Moyamoya disease is a chronic cerebrovascular disorder characterized by progressive stenosis of the circle of Willis with a compensatory collateral vessel network. Recent studies have identified the ring finger protein 213 gene (RNF213) as the unique susceptibility gene for moyamoya disease. The purpose of this study was to compare clinical features of moyamoya disease, especially angiographic findings, between patients with and without the RNF213 mutation.
Methods
Blood samples from 35 patients with moyamoya disease were obtained between May 2016 and May 2017. Information on age at the time of diagnosis, sex, and initial symptom were obtained via retrospective chart review. Angiographic records were evaluated.
Results
RNF213 variants were detected in the 28 of 35 patients (80%), including all pediatric patients (100%) and 18 of 25 adult patients (72%) in our cohort. Leptomeningeal collateral flow from posterior to anterior circulation was more frequent in the RNF213-negative group than in the RNF213-positive group (100% versus 38.9%; p = 0.020). Posterior cerebral arterial territorial involvement was more frequently observed in RNF213-positive patients than in RNF213-negative patients (50% versus 0%; p = 0.027).
Conclusions
RNF213 may play a significant role in the development of collateral anastomoses.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Moyamoya disease (MMD) is a chronic cerebrovascular disorder characterized by progressive stenosis of the circle of Willis with subsequent development of a compensatory collateral vessel network [27]. The prevalence of MMD is highest in East Asian countries, including Korea. A recent study reported that the annual incidence in 2011 was estimated to be 16.1 per 100,000 person-years in Korea [1, 13]. In 2011, two Japanese groups identified the ring finger protein 213 gene (RNF213) on chromosome 17q25.3 as the unique susceptibility gene for MMD. In these studies, most Japanese patients carried p.R4810K (c.14429G>A) [10, 18]. In previous studies, RNF213 p.R4810K variants were detected in approximately 80% of Korean MMD patients [14, 18, 19]. Although several risk factors for MMD have been identified, such as female sex, East Asian ethnicity, and family history of MMD, the etiology of MMD remains unclear. Furthermore, clinical features of MMD patients with RNF213 polymorphisms have not been fully investigated. A few studies have revealed that RNF213 polymorphisms are associated with high blood pressure, bilateral hemispheric involvement of MMD, early-onset MMD, and a poor prognosis [14, 16, 22]. Additionally, a recent animal study revealed a potential role of RNF213 in the development of pathological angiogenesis in ischemia [3, 9]. Therefore, we sought to compare clinical features, particularly angiographic findings, between MMD patients with and without an RNF213 mutation.
Methods
Patients
Between May 2016 and May 2017, RNF213 genetic variant testing was performed on patients who had been diagnosed with MMD and who had undergone follow-up for more than 1 year at our institute. We diagnosed 118 patients with MMD between January 2002 and May 2016 at our hospital on the basis of cerebral angiography and magnetic resonance angiography (MRA). Of 118 patients diagnosed with moyamoya disease, counseling for RNF213 genetic variant testing was performed and 35 patients provided written informed consent to be involved in this study. This study was approved by the Korea University Ansan Hospital’s Institutional Review Board (No. AS16032). Blood samples from 35 patients with MMD were obtained between May 2016 and May 2017. All patients were Koreans. A screening of RNF213 for c.14429G>A (p.Arg4810Lys/p.R4810K), which was proven to have a strong association with Korean MMD patients, was done [14, 19]. Information on age at time of diagnosis, patient sex, and initial symptoms at diagnosis were obtained via retrospective chart review. Angiographic records were evaluated to determine ICA (internal cerebral artery) staging, posterior cerebral artery (PCA) involvement, aneurysms, and collateral flow patterns, such as leptomeningeal and transdural collateral flow.
Diagnosis of MMD and ICA staging
All patients older than 18 years underwent cerebral angiography, including external carotid arteriography. All patients younger than 18 years underwent MRA. All patients were diagnosed based on published guidelines [4]. ICA staging of steno-occlusive lesions was classified according to the staging system as defined by Mugikura et al. [24]. The classification of ICA stage is described below: stage 1, mild to moderate stenosis around carotid bifurcation; stage 2, severe stenosis around carotid bifurcation of occlusion of either of proximal ACA or MCA (middle cerebral artery); stage 3, near occlusion of both proximal ACA and MCA, only a few of either ACA or MCA branches or both are faintly opacified in antegrade fashion; and stage 4, complete occlusion of both proximal ACA and MCA with absent or small amount of ICA moyamoya.
Variant detection of RNF213
In this study, we referred to NM_001256071.1 as the reference transcriptional sequence of RNF213. Genomic DNA was isolated from peripheral blood leukocytes. RNF213 on chromosome 17q25.3 was analyzed. Polymerase chain reaction and direct sequencing were performed for the selected mutation (c.14429G>A, p.R4810K in exon 60).
Statistical analysis
Differences in clinical features between patients with the RNF213 variant and those patients without the RNF213 variant were analyzed only in adult patients since all pediatric patients in our cohort carried the RNF213 variant. Statistical analysis was performed using a chi-squared test or Fisher’s exact probability test, as appropriate. Statistical analyses were performed using the Statistical Package for the Social Sciences version 18.0 for Windows (SPSS Inc.). Differences were considered significant at p values of less than or equal to 0.05.
Results
RNF213
RNF213 variants were detected in the 28 of 35 patients (80%). All genotypes of the minor allele frequency of the c.14429G>A variant were heterozygous. RNF213 variants were detected in all pediatric patients (100%) and in 18 of 25 adult patients (72%).
Pediatric group
A total of ten pediatric patients were included in our study. Unfortunately, all pediatric patients in our cohort carried the p.R4810K variant in RNF213, and therefore we could not analyze the difference between RNF213-negative and RNF213-positive pediatric patients. The mean age was 7.4 ± 3.2 years at the time of diagnosis. The male to female ratio was 1.5:1 (6 versus 4). Cerebral ischemia was observed in 90% of all patients including transient ischemic symptoms. One patient expressed having a headache. Leptomeningeal collateral flow from the posterior circulation to anterior circulation was observed in one patient. Significant transdural collateral flow from the middle meningeal artery (MMA) was observed in three patients (30%). Finally, 57% of cases were ICA stage 3 or 4. The characteristics of patients are shown in Table 1.
Adult group
A total of 25 adult patients were included in our study, of which 18 carried the RNF213 p.R4810K variant. The clinical and angiographic characteristics of the patients are shown in Table 1.
The mean age at diagnosis was 45 ± 10.4 years in the RNF213-positive group and 51.1 ± 7.7 years in the RNF213-negative group. No significant differences were observed in age (p = 0.443). Leptomeningeal collateral flow from posterior to anterior circulation was more frequently observed in the RNF213-negative group than in the RNF213-positive group (100% versus 39%; p = 0.020) (Fig. 1). Transdural collateral flow via the superficial temporal artery (STA) was more frequently observed in the RNF213-negative group than in the RNF213-positive group (57% versus 6%; p = 0.012), but the frequency of transdural collateral flow via the middle meningeal artery (MMA) was not significantly different between the two groups. PCA involvement was more frequently observed in the RNF213-positive than in the RNF213-negative group (50% versus 0%; p = 0.027) (Fig. 2). Otherwise, no significant differences were found in ICA stage, presence of aneurysm, sex, or symptoms during initial presentation.
Vertebral angiography of a 49-year-old RNF213(−) female patient. Note the leptomeningeal collateral flow to the anterior circulation from posterior circulation without moyamoya involvement of the proximal posterior cerebral artery (PCA). Anteroposterior (a) and lateral view (b) of vertebral angiography
Angiography of a 46-year-old RNF213(+) male patient. Note the prominent transdural collateral anastomoses via left external cerebral artery (a). Also note the moyamoya involvement of posterior circulation bilaterally and no significant leptomeningeal collateral flow to the anterior circulation from the posterior circulation was observed (b)
Discussion
The results of our study indicate that RNF213 is a major susceptibility gene for Korean MMD patients, which has been reported in previous studies [14, 19]. In this study, compared with patients without the variants in RNF213, fewer leptomeningeal collateral flow patterns from the PCA to anterior circulation and greater PCA involvement were observed in patients with RNF213 variants. Previous studies suggested that more PCA involvement and transdural collateral flow patterns might be found in more advanced stages of moyamoya disease [6, 12, 17, 28]. In our study, there was no statistically significant difference in the ICA stage between RNF213-positive and RNF213-negative patients, despite having differences in PCA involvement and collateral flow patterns in the two groups. We classified angiographic grade mainly according to their state of steno-occlusive changes of ICA, MCA, and ACA as defined by Mugikura et al. [24]. Suzuki’s staging system has been widely used to classify the severity of moyamoya disease [26, 27]. But the classification system was based mainly on serial angiographic changes in intensification and decrease in moyamoya vessels, and sometimes it cannot classify the angiographic findings accurately when ICA and PCA stages are far different [5, 24]. For example, in our study, there are some cases that complete occlusion of both proximal ACA and MCA without PCA involvement of moyamoya vessels. The staging system as defined by Mugikura et al. is more useful in the precise staging in such cases.
It appears that, in addition to the advanced stage of moyamoya disease, RNF213 may have a significant role in vascular pathology, such as PCA involvement and angiogenesis in the development of collateral vessels. Tomohito et al. [5, 7] reported that pediatric patients with posterior circulation involvement had significantly fewer advanced ICA lesions than did adult patients. Furthermore, they found that posterior circulation involvement at the initial onset was significantly correlated with poorer outcomes. Moreover, recent animal studies have shown enhanced post-ischemic angiogenesis in mice lacking RNF213 [9]. The study showed that angiogenesis under chronic ischemic conditions was significantly better in RNF213 knockout mice. For these reasons, we speculated that greater PCA involvement of moyamoya vessels and more aggressive neoangeogenesis, such as transdural collateral anastomoses rather than leptomeningeal collateral flow, would be found in patients with RNF213 variants. Yet, we observed that transdural collateral anastomoses via MMA occurred at similar frequencies between the two groups in our study. Our cohort of patients was small, so more studies will be needed in the future.
Previous studies have reported the clinical significance of posterior circulation involvement in MMD. The prevalence of posterior circulation involvement in MMD was reported to be 27–38% [5, 7, 14]. Additionally, some studies have demonstrated that clinical presentation, particularly cerebral ischemia, is significantly correlated with the severity of posterior circulation involvement in MMD [21, 27].
It appears that not only the severity of steno-occlusive lesions in MMD but also collateral status may influence clinical outcomes. Collateral status, such as leptomeningeal collateral flow from the PCA to anterior circulation and transdural collateral flow, could influence clinical outcomes [5, 15, 23]. Moreover, Armide et al. [25] revealed that the presence of preoperative transdural collaterals significantly correlated with better postoperative angiographic results.
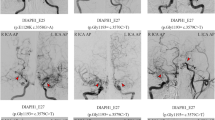
There are several surgical revascularization methods available to treat MMD, including direct revascularization of the STA and middle cerebral artery (MCA) anastomoses [11] and indirect revascularization via encephaloduroarteriosynangiosis (EDAS) [20]. Recent studies have emphasized that combining direct and indirect bypass surgeries produce good outcomes in pediatric and adult patients [2, 8]. We believe that different outcomes after indirect bypass or direct bypass surgery are expected in accordance with the presence of the RNF213 mutation since different developmental collateral flow patterns were observed in this study. Although statistical analysis was not performed on surgical outcomes in our study due to the lack of postoperative angiographic evaluations, two patients with RNF213 polymorphisms revealed good angiographic outcomes after indirect bypass surgery (Figs. 3 and 4). We believe that indirect bypass surgery may be helpful not only in pediatric patients but also in adult patients with the RNF213 polymorphism. Combined direct and indirect bypass surgeries may produce better outcomes than direct bypass surgery alone in those cases. Future studies will be needed to clarify this point.
Angiography of a 36-year-old RNF213(+) female patient who had a history of external ventricular drainage surgery via right Kocher’s point for intraventricular hemorrhage 10 years ago. Note well-developed transdural anastomoses via burr hole site from the external carotid artery to the anterior cerebral artery (ACA) and middle cerebral artery (MCA) territories. Anteroposterior (a) and lateral views (b) of external carotid angiography
There were several limitations in our study. First, the number of patients was small, and the study was retrospective, not randomized, and incorporated data from only one institute. Furthermore, only angiographic findings at initial diagnosis were analyzed, whereas data on perfusion were not analyzed in this study due to a lack of documented clinical results. Second, every pediatric patients involved in this study carried the RNF213 polymorphism, so we could not analyze the differences. More number of cases will be needed to analyze the differences between RNF213-postive and RNF213-negative groups in pediatric population. Third, all patients who had undergone surgical revascularization already had operations before checking RNF213 polymorphism. This means that we did not know whether the patients had RNF213 polymorphism or not at the time of surgery. Only EDASs were performed on all patients who had undergone surgical revascularization in this study. Among them, we thought that RNF213-positive cases had better postoperative revascularization than RNF213-negative cases. But there is no sufficient postoperative angiographic evaluation so we just introduced two cases of RNF213-positive cases with good postoperative revascularization. However, we currently take the postoperative angiographic evaluation on the patients and hope to compare the surgical outcomes between RNF213-positive and RNF213-negative cases in the near future. Nevertheless, we provide key findings that will assist in future efforts to identify the best surgical treatment for MMD patients according to their RNF213 mutation. Thus, a prospective study is needed.
Conclusions
RNF213 is the major susceptibility gene in MMD patients. Our study indicates that collateral flow patterns are different according to the presence of the RNF213 mutation. Leptomeningeal collateral flow patterns from posterior circulation to anterior circulation were observed more frequently in the RNF213-negative group. Meanwhile, PCA involvement is more common in the RNF213-positive group and may have a significant role in the development of collateral anastomoses. We suggest that different outcomes after indirect bypass or direct bypass surgery may be observed in accordance with RNF213 mutations because different collateral flow patterns were observed in our study. Future research is needed to further elucidate the relationship between RNF213 variants and collateral flow patterns.
References
Ahn IM, Park DH, Hann HJ, Kim KH, Kim HJ, Ahn HS (2014) Incidence, prevalence, and survival of moyamoya disease in Korea: a nationwide, population-based study. Stroke 45:1090–1095. https://doi.org/10.1161/STROKEAHA.113.004273
Amin-Hanjani S, Singh A, Rifai H, Thulborn KR, Alaraj A, Aletich V, Charbel FT (2013) Combined direct and indirect bypass for moyamoya: quantitative assessment of direct bypass flow over time. Neurosurgery 73:962–968
Fujimura M, Sonobe S, Nishijima Y, Niizuma K, Sakata H, Kure S, Tominaga T (2014) Genetics and biomarkers of moyamoya disease: significance of RNF213 as a susceptibility gene. J Stroke 16:65–72. https://doi.org/10.5853/jos.2014.16.2.65
Fukui M (1997) Guidelines for the diagnosis and treatment of spontaneous occlusion of the circle of Willis (‘moyamoya’ disease). Clin Neurol Neurosurg 99:S238–S240
Hishikawa T, Tokunaga K, Sugiu K, Date I (2013) Assessment of the difference in posterior circulation involvement between pediatric and adult patients with moyamoya disease. J Neurosurg 119:961–965. https://doi.org/10.3171/2013.6.JNS122099
Hishikawa T, Tokunaga K, Sugiu K, Date I (2013) Clinical and radiographic features of moyamoya disease in patients with both cerebral ischaemia and haemorrhage. Br J Neurosurg 27:198–201
Hishikawa T, Tokunaga K, Sugiu K, Date I (2014) Long-term outcomes in adult patients with ischemic-type moyamoya disease involving posterior circulation. Acta Neurochir 156:1745–1751. https://doi.org/10.1007/s00701-014-2136-3
Imai H, Miyawaki S, Ono H, Nakatomi H, Yoshimoto Y, Saito N (2015) The importance of encephalo-myo-synangiosis in surgical revascularization strategies for moyamoya disease in children and adults. World Neurosurg 83:691–699. https://doi.org/10.1016/j.wneu.2015.01.016
Ito A, Fujimura M, Niizuma K, Kanoke A, Sakata H, Morita-Fujimura Y, Kikuchi A, Kure S, Tominaga T (2015) Enhanced post-ischemic angiogenesis in mice lacking RNF213; a susceptibility gene for moyamoya disease. Brain Res 1594:310–320. https://doi.org/10.1016/j.brainres.2014.11.014
Kamada F, Aoki Y, Narisawa A, Abe Y, Komatsuzaki S, Kikuchi A, Kanno J, Niihori T, Ono M, Ishii N (2011) A genome-wide association study identifies RNF213 as the first moyamoya disease gene. J Hum Genet 56:34–40
Karasawa J, Kikuchi H, Furuse S, Kawamura J, Sakaki T (1978) Treatment of moyamoya disease with STA-MCA anastomosis. J Neurosurg 49:679–688
Kim J-M, Lee S-H, Roh J-K (2009) Changing ischaemic lesion patterns in adult moyamoya disease. J Neurol Neurosurg Psychiatry 80:36–40
Kim T, Lee H, Bang JS, Kwon O-K, Hwang G, Oh CW (2015) Epidemiology of moyamoya disease in Korea: based on national health insurance service data. J Korean Neurosurg Soc 57:390–395
Kim EH, Yum MS, Ra YS, Park JB, Ahn JS, Kim GH, Goo HW, Ko TS, Yoo HW (2016) Importance of RNF213 polymorphism on clinical features and long-term outcome in moyamoya disease. J Neurosurg 124:1221–1227. https://doi.org/10.3171/2015.4.JNS142900
Kim DY, Son JP, Yeon JY, Kim GM, Kim JS, Hong SC, Bang OY (2017) Infarct pattern and collateral status in adult moyamoya disease: a multimodal magnetic resonance imaging study. Stroke 48:111–116. https://doi.org/10.1161/STROKEAHA.116.014529
Koizumi A, Kobayashi H, Liu W, Fujii Y, Senevirathna ST, Nanayakkara S, Okuda H, Hitomi T, Harada KH, Takenaka K, Watanabe T, Shimbo S (2013) P.R4810K, a polymorphism of RNF213, the susceptibility gene for moyamoya disease, is associated with blood pressure. Environ Health Prev Med 18:121–129. https://doi.org/10.1007/s12199-012-0299-1
Kuroda S, Ishikawa T, Houkin K, Iwasaki Y (2002) Clinical significance of posterior cerebral artery stenosis/occlusion in moyamoya disease. No shinkei geka Neurol Surg 30:1295–1300
Liu W, Morito D, Takashima S, Mineharu Y, Kobayashi H, Hitomi T, Hashikata H, Matsuura N, Yamazaki S, Toyoda A (2011) Identification of RNF213 as a susceptibility gene for moyamoya disease and its possible role in vascular development. PLoS One 6:e22542
Liu W, Hitomi T, Kobayashi H, Harada KH, Koizumi A (2012) Distribution of moyamoya disease susceptibility polymorphism p. R4810K in RNF213 in East and Southeast Asian populations. Neurol Med Chir 52:299–303
Matsushima T, Fukui M, Kitamura K, Hasuo K, Kuwabara Y, Kurokawa T (1990) Encephalo-duro-arterio-synangiosis in children with moyamoya disease. Acta Neurochir 104:96–102
Miyamoto S, Kikuchi H, Karasawa J, Nagata I, Ikota T, Takeuchi S (1984) Study of the posterior circulation in moyamoya disease: clinical and neuroradiological evaluation. J Neurosurg 61:1032–1037
Miyawaki S, Imai H, Shimizu M, Yagi S, Ono H, Mukasa A, Nakatomi H, Shimizu T, Saito N (2013) Genetic variant RNF213 c.14576G>A in various phenotypes of intracranial major artery stenosis/occlusion. Stroke 44:2894–2897. https://doi.org/10.1161/STROKEAHA.113.002477
Mugikura S, Takahashi S, Higano S, Shirane R, Kurihara N, Furuta S, Ezura M, Takahashi A (1999) The relationship between cerebral infarction and angiographic characteristics in childhood moyamoya disease. Am J Neuroradiol 20:336–343
Mugikura S, Takahashi S, Higano S, Shirane R, Sakurai Y, Yamada S (2002) Predominant involvement of ipsilateral anterior and posterior circulations in moyamoya disease. Stroke 33:1497–1500
Storey A, Michael Scott R, Robertson R, Smith E (2017) Preoperative transdural collateral vessels in moyamoya as radiographic biomarkers of disease. J Neurosurg Pediatr 19:289–295
Suzuki J, Kodama N (1983) Moyamoya disease--a review. Stroke 14:104–109
Suzuki J, Takaku A (1969) Cerebrovascular moyamoya disease: disease showing abnormal net-like vessels in base of brain. Arch Neurol 20:288–299
Yamada I, Himeno Y, Suzuki S, Matsushima Y (1995) Posterior circulation in moyamoya disease: angiographic study. Radiology 197:239–246
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Kim, WH., Kim, SD., Nam, MH. et al. Posterior circulation involvement and collateral flow pattern in moyamoya disease with the RNF213 polymorphism. Childs Nerv Syst 35, 309–314 (2019). https://doi.org/10.1007/s00381-018-3985-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-018-3985-5