Abstract
Effects of feeding on pre-exercise VO2 and excess post-exercise oxygen consumption (EPOC) after exhaustive exercise were investigated in sedentary southern catfish, active herbivorous grass carp, omnivorous crucian carp, and sluggish omnivorous darkbarbel catfish to test whether feeding had different effects on EPOC and to compare EPOC in fishes with different ecological habits. For fasting fish, the pre-exercise and peak post-exercise VO2 were higher and recovery rates were faster in crucian carp and grass carp compared to those of darkbarbel catfish and southern catfish. EPOC magnitudes of grass carp and southern catfish were significantly larger than those of crucian carp and darkbarbel catfish. Feeding had no significant effect on peak post-exercise VO2, recovery rate, and EPOC magnitude in grass carp. Both the pre-exercise and peak post-exercise VO2 increased with meal size, while the EPOC magnitude and duration decreased significantly in the larger meal size groups of crucian carp and southern catfish. In darkbarbel catfish, both the pre-exercise and peak post-exercise VO2 increased with meal size, but the VO2 increment elicited by exercise was larger in feeding groups compared with the fasting group. These results suggest that (1) the characteristics of the post-exercise VO2 profile, such as peak post-exercise VO2 and recovery rate, were closely related to the activity of fishes, whereas the EPOC magnitude was not and (2) the effects of feeding on EPOC were more closely related to the postprandial increase in VO2.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Under natural conditions, animals often need to perform multiple tasks at the same time and undertake different activities simultaneously (Hicks and Bennett 2004). It may be important to exert priorities so that one activity can be emphasized at the expense of another. Unlike the functions of the brain and heart, digestion is not regarded as a priority physiological function and it is likely compromised when other physiological challenges place demands on an organism (Farrell et al. 2001). Farrell et al. suggested that experimentally stimulated enemy avoidance activity (i.e., struggling) would temporally delay or decrease the intensity of digestive processes in fishes. However, studies of competition between digestion and sustained aerobic swimming performance in fishes demonstrated that feeding compromises prolonged swimming performance (rather than swimming compromising feeding metabolism) (Alsop and Woods 1997; Thorarensen and Farrell 2006). Different fish species might have different physiological mechanisms involved in the competition between digestion and locomotion that depend on locomotion type and ecological relevance (Domenici and Blake 1997; Plaut 2001).
Locomotive and digestive capacities vary greatly in fishes with different ecological habits, thus researchers should consider the ecological habits of fishes when studying the relationship between digestion and locomotion. Furthermore, meal size might have a significant effect on the relationship between feeding and locomotion because it significantly affects postprandial metabolic rate in fishes (Fu et al. 2005). Previous research showed that the postprandial oxygen consumption rate (VO2) increased to 50–400% compared to that of fasting fish and that swimming speed might not be affected or might drop profoundly depending on the type of fish and the size of the consumed meal (Alsop and Woods 1997; Asaeda et al. 2001; Fu et al. 2005, 2006; Pang et al. 2009). These findings suggest that meal size is an important factor to consider in the investigation of physiological conflict between digestion and locomotion.
Like all other vertebrates, fish need time to recover after exhaustive activity. Gaesser and Brooks (1984) named the elevated VO2 following exhaustive exercise ‘excess post-exercise oxygen consumption’ (EPOC). The magnitude of EPOC is closely related to the anaerobic capacity of animals, whereas the maximum VO2 during the exercise recovery process (VO2peak) and the rate of decrease of VO2 during recovery might be limited by their aerobic capacity (Gaesser and Brooks 1984; Hancock and Gleeson 2002). The duration of EPOC also has important ecological relevance because the time required to restore the energy stores expended during exhaustive exercise will ultimately determine the frequency of maximal locomotion, which is important to survive (Milligan 1996; Lee et al. 2003a, b). Furthermore, the maximum VO2 immediately after exhaustive exercise could be used as a measure of VO2max for fishes (Cutts et al. 2002; Fu et al. 2008). Thus, the effect on EPOC of feeding with different meal sizes might provide valuable data about anaerobic and aerobic capacities of postprandial fishes.
In this study, we selected four fishes with different locomotive and digestive performance (Ding 1994) (Table 1): southern catfish (Silurus meridionalis), which is a sedentary piscivorous fish; grass carp (Ctenopharyngodon idellus), an active herbivorous fish; red crucian carp (Carassius auratus), which is an active omnivorous fish; and darkbarbel catfish (Pelteobagrus fulvidraco), a sluggish omnivorous fish. We first conducted a pilot experiment to provide an index of feeding and locomotion capacity (Table 1). We then investigated the effect of feeding with different meal sizes (from fasting to maximum meal size) on postprandial VO2 increase and EPOC response. The aims of this study were (1) to provide and compare EPOC data for fish with different locomotive and digestive capacities and (2) to test whether feeding had different effects on EPOC in fishes with ecological habits.
Materials and methods
Experimental animals, diets, and acclimation
Juvenile catfish, crucian carp, grass carp, and darkbarbel catfish were obtained from local farmers and acclimated in a rearing system for at least 30 days before the experiment. The dechlorinated fresh water temperature was maintained at 25.0 ± 0.5°C and the oxygen content was kept above 7 mg L−1. During this period, the fish were fed to satiation once daily on a commercial diet (for crucian carp and grass carp) or fillets of freshly killed loach, Misgurnus anguillicaudatus (for southern catfish and darkbarbel catfish). Table 2 lists the composition of the formulated diet and loach cutlet. Analyses of chemical composition were conducted following standard methods (AOAC 1995).
Measurement of VO2
VO2 for individual fish was measured using a continuous-flow respirometer (Fu et al. 2005). The following formula was used to calculate the VO2 (mg h−1) of individual fish:
where ΔO2 is the difference (mgO2 L−1) in oxygen concentration between an experimental chamber and the control chamber (chamber without fish) and v is the water flow rate in the experimental chamber (L h−1). To account for variations in oxygen consumption rate due to size differences among the fish, VO2 was adjusted to a standard body mass of 1 kg using a mass exponent of 0.75 (Reidy et al. 2000; Fu et al. 2005). Standardized VO2 was calculated by the following formula:
where Xs is the standardized VO2, Xm is the measured VO2 per fish (mgO2 h−1 kg−1) calculated by Eq. (1), and m is the body mass of the fish (kg). Dissolved oxygen concentration was measured at the outlet of the chamber by an oxymeter (HQ20, Hach Company, Loveland, CO, USA). The flow rate of water through the respirometer chamber was measured by collecting the water outflow from each tube.
Experimental protocol
The EPOCs were measured at relative meal sizes of 0, 0.5, 1, 2, 4, 8, and 18% (maximum meal size group) body mass in southern catfish; 0, 0.5, 1, 2, 4, and 8% body mass in darkbarbel catfish; and 0, 0.5, 1, 2, and 4% body mass in red crucian carp and grass carp with either fish fillets or the formulated diet. The fish were placed in the respirometer chamber and allowed to acclimate for 1 week before the experiment. Then, after 48 h fasting, a prescribed quantity of fish fillet or formulated diet was offered. We fed 8–10 fish in each treatment group. The food was removed if the fish did not finish it within 30 min. Because some fish did not consume their food at the prescribed time, the sample number varied (Table 3). From published paper, we found the maximal postprandial VO2 achieved at 2 h in grass carp, crucian carp and darkbarbel catfish fed with any meal sizes from 0.5% meal size to maximum meal size, while maximal postprandial VO2 achieved at 4, 6, 10, and 10 h post-feeding fed diet of 2, 4, 8, and 18% meal sizes in southern catfish (Fu et al. 2005; Luo and Xie 2008; Pang et al. 2009). The pre-exercise VO2 was measured at 2 h post-feeding in grass carp, crucian carp, and darkbarbel catfish and at 4, 6, 10, and 10 h post-feeding with 2, 4, 8, and the 18% (maximum meal size) groups in southern catfish when each fish achieved its peak postprandial VO2 at the given meal size according to data obtained from previous studies. The fish were then removed from the chambers and individually exercised by manual chasing for 2–5 min until exhausted (Wood 1991; Milligan 1996; Kieffer et al. 2001; Wilkie et al. 2001). Following exercise, fish were returned to the chambers. The flow rate was ~0.6 L min−1 and 99% exchange of water was achieved within 1 min in the 0.1-L chamber (Steffensen 1989). Thus, the first post-exercise measurement was made 1 min after the end of exercise (when the fish was placed back into the chamber). The VO2 was then measured at 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, and 60 min post-exercise in grass carp, darkbarbel catfish, and southern catfish and at 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 15, 20, 25, and 30 min post-exercise in crucian carp (the duration was determined by the pilot experiment).
Definition of parameters
-
1.
Pre-exercise VO2 (mgO2 h−1 kg−1): oxygen consumption before exercise.
-
2.
VO2peak (mgO2 h−1 kg−1): the observed maximum oxygen consumption during the exercise recovery process.
-
3.
EPOC magnitude (mgO2 kg−1): the excess oxygen consumption above the pre-exercise VO2 during the recovery process.
-
4.
Metabolic scope [VO2 increment (mgO2 h−1 kg−1)]: the difference between VO2peak and pre-exercise VO2.
-
5.
Duration (h): time from exercise to when post-exercise VO2 was not significantly different from pre-exercise VO2.
Data analysis
STATISTICA 4.5 (StatSoft Inc.) was used for data analysis. The data were subjected to ANOVA followed by post hoc multiple comparison (least significant difference). P values <0.05 were considered statistically significant and all data are presented as mean ± 1 SEM. Nonlinear estimation was used when necessary.
Results
Comparison of fasting EPOC curve and pre-exercise VO2 among fish species
For fasting fish, the peak post-exercise VO2 and pre-exercise VO2 of crucian carp and grass carp were significantly larger than those of darkbarbel catfish and southern catfish (P < 0.05). The EPOC magnitude of grass carp was significantly larger than that of southern catfish, while EPOC of southern catfish was significantly larger than those of crucian carp and darkbarbel catfish (P < 0.05). The duration of crucian carp was much shorter than that of the other fish species (Table 3).
Crucian carp
In this study, the maximum meal size was 5.11% body mass for crucian carp. The pre-exercise VO2 tended to increase with meal size. The pre-exercise VO2 was significantly different among different meal size groups, except for the 0.5 and 1% groups (P < 0.05). The value increased from about 50% in the 0.5 and 1% meal size groups to about 150% in the maximum meal size (5.11%) group.
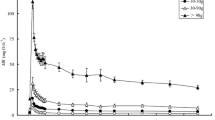
VO2 immediately increased after exhaustive exercise and returned to the pre-exercise level at 4–9 min. The relationship between post-exercise VO2 (Y, mgO2 h−1 kg−1) and time after exercise (X, min) was described by the following equation (Fig. 1):
where a is pre-exercise VO2, b is VO2 increment, and c is recovery rate. Table 4 lists the values of a, b, and c.
The post-exercise VO2 in fishes fed different sized meals (mean ± SE). The relationship between VO2 (Y, mgO2 h−1 kg−1) and time after exercise (X, min) was described by equation: Y = a + becX, where a is pre-exercise VO2, b is VO2 increment, and c is recovery rate. All VO2s were adjusted to a 1 kg body mass
Meal size significantly affected the EPOC profile in crucian carp. Feeding induced shorter duration (recovery time), smaller EPOC magnitude, and larger peak post-exercise VO2. The peak post-exercise VO2 of maximum meal size (5.11%) was significantly greater than those of the 1, 0.5%, and control groups. The EPOC magnitudes of the 2 and maximum meal size (5.11%) groups were significantly higher than those of the control and 1% groups. The peak post-exercise VO2 of the 1% meal size group was also significantly higher than that of the 0.5% meal size group. The VO2 during the recovery process decreased to a level significantly lower than that of the pre-exercise level in the maximum meal size (5.11%) group (P < 0.05).
Grass carp
For grass carp, the maximum meal size was 3.89% body mass. Feeding only elicited a 50% increase in the pre-exercise VO2. The pre-exercise VO2s of the feeding groups were significantly higher than that of the fasting group (P < 0.05), but there was no significant difference among all meal size groups and control group.
VO2 immediately increased after exhaustive exercise and returned to the pre-exercise level at 4 to more than 60 min depending on meal size used. Exercise elicited a 300 mgO2 h−1 kg−1 peak post-exercise VO2 in grass carp, which was >5-fold higher than that of pre-exercise VO2. There was no significant difference in peak post-exercise VO2, VO2 increment, and EPOC magnitude among all meal size groups and control group.
Darkbarbel catfish
The maximum meal size was 8.03% body mass in darkbarbel catfish. Maximal feeding elicited about a 70% increase in the pre-exercise VO2. The pre-exercise VO2 of the maximum meal size (8.03%) group was significantly greater than that of all other groups except the 4% meal size group, while the pre-exercise VO2s of the 4 and 1% groups were significantly higher than that of the control group (P < 0.05).
For darkbarbel catfish, the peak post-exercise VO2 for all feeding groups was significantly higher than that of the control group, but there was no difference among feeding groups. VO2 increments of the 1, 2, and 4% groups were significantly higher than that of the control group. There was no significant difference in EPOC magnitude among all groups.
Southern catfish
In this study, the maximum meal size for the southern catfish was 18.57% body mass. The pre-exercise VO2 tended to increase with meal size, as it did in crucian carp. VO2 differed significantly between fasted and fed fish, except for the 2% meal size group, which did not differ from those of the 4% and control groups. The pre-exercise VO2 increased from about 15% in the 2% meal size group to about 230% in the maximum meal size group. VO2 immediately increased after exhaustive exercise and returned to the pre-exercise level at 15 to more than 60 min depending on meal size used.
Meal size significantly affected EPOC in southern catfish. The EPOC magnitudes of the 8 and maximum meal size (18.57%) groups were significantly lower than those of control, 2, and 4% meal size groups (P < 0.05). The peak post-exercise VO2 tended to increase with increased meal size. The peak post-exercise VO2 of maximum meal size (18.57%) group was significantly higher than those of all other groups. The peak post-exercise VO2s of the 4 and 8% groups were significantly higher than that of the control group. The duration decreased from 50 min in the control group to 15 and 20 min in the 8% and maximum meal size (18.57%).
Discussion
The pre-exercise VO2 of fasting and feeding fish, the peak post-exercise VO2 elicited by exercise, the increment of VO2, the recovery rate, and the total VO2 (EPOC magnitude) during the recovery process varied greatly among the four fish species studied. The factorial increase after exhaustive exercise was about 3–5 in southern catfish, darkbarbel catfish, crucian carp, and grass carp, which is within the range reported in previous studies (Scarabello et al. 1992; Lee et al. 2003a; Peng et al. 2007). The EPOC magnitudes found in this study were similar to those of rainbow trout (Scarabello et al. 1992) and coho salmon (Lee et al. 2003a). The characteristics of the post-exercise VO2 profile, such as peak post-exercise VO2, VO2 recovery rate, and pre-exercise VO2 of fasting fishes (but not magnitude), were closely related to the activity of fishes, whereas the effects of feeding on EPOC were more closely related to increase in VO2 in fish fed with a certain meal size.
Comparison of the fasting EPOC curve and pre-exercise VO2
Exercise elicited a much larger VO2 increment in active crucian carp and grass carp than it did in the sit-and-wait southern catfish, which had the lowest VO2 increment. Hence, the VO2 increment of active fishes was larger to support locomotion activities. The recovery rates of post-exercise VO2 were also faster in active fish, which suggests that active fish may have higher cardio-respiratory capacity and can take up more oxygen from the environment and distribute it to tissues (Milligan 1996). However, the cost of such a high-capacity cardio-respiratory system is a high expenditure of maintenance energy, as suggested by the higher pre-exercise VO2 in fasting crucian carp and grass carp. Interestingly, peak post-exercise VO2 of fasting crucian carp was similar to that of fasting grass carp, whereas its pre-exercise VO2 was about 25% higher (significantly different) than that of fasting grass carp. This difference might be due to the higher costs required for the crucian carp’s higher locomotive capacity, as suggested by its faster critical swimming speed (U crit, which is faster in crucian carp than in grass carp), and its faster EPOC recovery rate (high turnover of metabolites). However, the cost of having a larger digestive capacity (suggested by the higher postprandial VO2 in this study and the shorter digestion time in a previous study; Pang et al. 2009) is an alternate explanation.
EPOC includes many components, such as the oxygen required to restore tissue and cellular stores of oxygen and high-energy phosphates (Bahr 1992), biochemical imbalances in metabolites such as lactate and glycogen (Rose et al. 1988), and ionic and osmotic balance (Stainsby and Barclay 1970). For a long time, post-exercise excess metabolism was used as an index of anaerobic capacity. Our results showed that the anaerobic capacities of crucian carp and darkbarbel catfish were lower than those of grass carp and southern catfish. The active crucian carp may not need high anaerobic capacity because it has higher aerobic capacity. In their research on transgenic and ocean-ranched coho salmon (Oncorhynchus kisutch), Lee et al. (2003a) found that the more active ocean-ranched salmon had a higher VO2peak, fast recovery rate, and lower EPOC magnitude compared to transgenic fish. We found that the sluggish darkbarbel catfish was not very sensitive to chasing, did not show typical fast-start movement when being chased, and was not totally exhausted after 5 min of chasing. Therefore, it is not surprising that darkbarbel catfish had a lower EPOC magnitude. As for the sit-and-wait piscivorous southern catfish with lower aerobic capacity, higher anaerobic capacity might be crucial for its ambush hunting activity. Because the southern catfish is at the top of the food chain in its habitat and has a habit of hiding motionless most of the time, a lower recovery rate from exercise is not a physiological problem. Herbivorous grass carp must graze for food all day and have a semi-migratory habit, so having a higher capacity for both aerobic and anaerobic might be beneficial for its physiological activities, such as hunting, migration, and predator avoidance.
EPOC magnitude consists of various components, some of which are not positively related to anaerobic capacity. Several researchers found that post-exercise VO2 was affected by fluctuations in levels of circulating catecholamines (Gladden et al. 1982; Gaesser and Brooks 1984) and by substrate cycling within both the gluconeogenic–glycolytic and triglyceride–fatty acid cycles after exercise (Bahr 1992). Scarabello et al. (1992) called these responses ‘non-specific increases in VO2,’ meaning they are not directly associated with the processing of metabolites but instead are due to the psychological aspects of exercise stress. Of the fish species investigated in this study, the grass carp was the most sensitive fish to chasing. It showed typical fast-start movement and quickly became completely exhausted after 1–2 min of being chased. The large EPOC magnitude of this species might also include some non-specific stress response. This result means that some fish expend more energy for recovery when exposed to similar anaerobic stress.
In general, active fish usually have higher pre-exercise VO2, peak post-exercise VO2, and recovery rate of post-exercise metabolism compared to sluggish or sit-and-wait fishes. However, the magnitude might not be related to activity due to the complicated component involved in recovery process of exhaustive exercise.
The effect of feeding on postprandial VO2 and EPOC among fish species
Before this study, data about the effect of feeding on EPOC was only available in one fish species (Fu et al. 2007). It suggested that feeding had no effect on EPOC in the southern catfish. Only one meal size was used in the previous study.
In the present study, with the increase of meal size, pre-exercise VO2 and EPOC responded differently among different fish species. EPOC magnitude, recovery rate, and peak post-exercise VO2 were not significantly different among all meal size groups in the grass carp, possibly because of its low digestive capacity (as suggested by the lower postprandial VO2 response). It suggested that locomotion could use all of its cardio-respiratory capacity because the peak post-exercise VO2 elicited solely by exhaustive exercise of fasting grass carp was not significantly different from that of fish fed the maximum size meal. For crucian carp and southern catfish, peak post-exercise VO2s elicited by exercise were significantly greater than those of fasting fish, which means that neither recovery metabolism after exhaustive exercise nor feeding metabolism could use up all of the cardio-respiratory capacity and both processes could co-occur to some extent. The EPOC magnitude and duration decreased significantly in the larger meal size groups in both crucian carp and southern catfish, which clearly suggests that the postprandial EPOC process was profoundly curtailed in fishes with higher digestive capacity. However, because the anaerobic capacity in both crucian carp and southern catfish did not significantly decrease, the oxygen required to restore tissue and cellular stores of oxygen and metabolites might not account for all of the difference observed in EPOC between feeding and fasting fish. It is possible that the ‘non-specific increase in VO2’ decreased, because fed fish are usually less sensitive to chasing. Furthermore, because struggling would temporally delay or decrease the intensity of digestive processes (Farrell et al. 2001), during the recovery process following exhaustive exercise, digestive metabolism might be depressed. This might be why the post-exercise VO2 from 5 to 30 min after exercise ended was lower than the pre-exercise level. For southern catfish, a similar situation was detected when we compared the VO2 profile of the 18% meal size group to those of the small meal size and control groups.
Although the effect of feeding on the postprandial VO2 response in darkbarbel catfish was similar to that of grass carp, the increment of VO2peak in the feeding group was significantly larger than that of the fasting group. EPOC magnitude was also a little larger. This suggests that feeding enhances the aerobic and/or anaerobic metabolic capacity in this species. This interesting phenomenon needs to be studied further. However, from this study we conclude that when investigating the effect of feeding on physiological processes, meal size is a very important parameter to consider because different meal sizes might have different effects on physiological processes.
References
Alsop DH, Wood CM (1997) The interactive effects of feeding and exercise on oxygen consumption, swimming performance and protein usage in juvenile rainbow trout (Oncorhynchus mykiss). J Exp Biol 200:2337–2346
Asaeda T, Priyadarshana T, Manatunge J (2001) Effects of satiation on feeding and swimming behavior of planktivores. Hydrobiologia 443:47–157
Bahr R (1992) Excess post-exercise oxygen consumption magnitude, mechanisms, and practical implications. Acta Physiol Scand Suppl 605:1–70
Cutts CJ, Metcalfe NB, Taylor AC (2002) Juvenile Atlantic salmon (Salmo salar) with relatively high standard metabolic rates have small metabolic scopes. Funct Ecol 16:73–78
Ding RH (1994) The fishes of Sichuan, China. Sichuan Science and Technology Press, Sichuan
Domenici P, Blake RW (1997) The kinematics and performance of fish fast-start swimming. J Exp Biol 200:1165–1178
Farrell AP, Thorarensen H, Axelsson M, Crocker CE, Gamperl AK, Cech JJ Jr (2001) Gut blood flow in fish during exercise and severe hypercapnia. Comp Biochem Physiol A 128:549–561
Fu SJ, Xie XJ, Cao ZD (2005) Effect of meal size on postprandial metabolic response in southern catfish (Silurus meridionalis). Comp Biochem Physiol A 140:445–451
Fu SJ, Cao ZD, Peng JL (2006) Effect of meal size on postprandial metabolic response in Chinese catfish (Silurus asotus). J Comp Physiol B 176:489–495
Fu SJ, Cao ZD, Peng JL (2007) Effect of feeding and fasting on excess post-exercise oxygen consumption in juvenile southern catfish (Silurus meridionalis Chen). Comp Biochem Physiol A 146:435–439
Fu SJ, Cao ZD, Xie XJ (2008) Feeding metabolism and locomotion metabolism in fishes. Chin J Zool 43:150–159
Gaesser GA, Brooks GA (1984) Metabolic bases of excess post-exercise oxygen consumption: a review. Med Sci Sports Exerc 16:29–43
Gladden LB, Stainsby WN, MacIntosh BR (1982) Norepinephrine increases canine skeletal muscle VO2 during recovery. Med Sci Sports Exerc 16:29–43
Hancock TV, Gleeson TT (2002) Metabolic recovery in the desert iguana (Dipsosaurus dorsalis) following activities of varied intensity and duration. Funct Ecol 16:40–48
Hicks JWH, Bennett AF (2004) Eat and run: prioritization of oxygen delivery during elevated metabolic states. Res Physiol Neurol 144:215–224
Kieffer JD, Wakefield AM, Litvak MK (2001) Juvenile sturgeon exhibit reduced physiological responses to exercise. J Exp Biol 204:4281–4289
Lee CG, Devlin RH, Farrell AP (2003a) Swimming performance, oxygen consumption and excess post-exercise oxygen consumption in adult transgenic and ocean-ranched coho salmon. J Fish Biol 62:753–766
Lee CG, Farrell AP, Lotto A, Hinch SG, Healey MC (2003b) Excess post-exercise oxygen consumption in adult sockeye (Oncorhynchus nerka) and coho (O. kisutch) salmon following critical speed swimming. J Exp Biol 206:3253–3260
Luo YP, Xie XJ (2008) Effects of temperature on the specific dynamic action of the southern catfish, Silurus meridionalis. Comp Biochem Physiol A 149:150–156
Milligan CL (1996) Metabolic recovery from exhaustive exercise in rainbow trout. Comp Biochem Physiol A 113:51–60
Pang X, Cao ZD, Fu SJ (2009) Effect of meal size on postprandial metabolic response in crucian carp. J Chongqing Normal Univ (Nat Sci) 26:34–36
Peng JL, Zeng LQ, Cao ZD, Fu SJ (2007) Effects of chase and air exposure on oxygen consumption and respiration frequency in Silurus meridionalis. Freshwater Fisheries 37:7–10
Plaut I (2001) Critical swimming speed: its ecological relevance. Comp Biochem Physiol A 131:41–50
Reidy SP, Kerr SR, Nelson JA (2000) Aerobic and anaerobic swimming performance of individual Atlantic cod. J Exp Biol 203:347–357
Rose RJ, Hodgson DR, Kelso TB, McCutcheon LJ, Reid TA, Bayley WM, Golnick PD (1988) Maximum O2, uptake O2, EPOC, and deficit and muscle metabolites in thoroughbred horses. J Appl Physiol 64:781–788
Scarabello M, Heigenhauser GJ, Wood CM (1992) Gas exchange, metabolite status and excess post-exercise oxygen consumption after repetitive bouts of exhaustive exercise in juvenile rainbow trout. J Exp Biol 167:155–169
Stainsby WN, Barclay JK (1970) Exercise metabolism: O2 deficit, steady level O2 uptake and O2 uptake for recovery. Med Sci Sports 2:177–181
Steffensen JF (1989) Some errors in respirometry of aquatic breathers: how to avoid and correct for them. Fish Physiol Biochem 6:49–59
Thorarensen H, Farrell AP (2006) Postprandial intestinal blood flow, metabolic rate, and exercise in Chinook salmon (Oncorhynchus tshawytscha). Physiol Biochem Zool 79:688–694
Wilkie MP, Bradshaw PG, Joanis V, Claude JF, Swindell SL (2001) Rapid metabolic recovery following vigorous exercise in burrow-dwelling larval sea lampreys (Petromyzon marinus). Physiol Biochem Zool 74:261–272
Wood CM (1991) Acid–base and ion balance, metabolism and their interactions, after exhaustive exercise in fish. J Exp Biol 160:285–308
Acknowledgments
This study was funded by the National Science Foundation of China (NSFC 30700087), the Chongqing Science and Technology Commission (CSTC 2007BB1226), and the Research Project of Chongqing Education Committee (KJ080823). We declare that the experiments comply with the current laws of the country in which the experiments were performed.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Heldmaier.
Rights and permissions
About this article
Cite this article
Fu, SJ., Zeng, LQ., Li, XM. et al. Effect of meal size on excess post-exercise oxygen consumption in fishes with different locomotive and digestive performance. J Comp Physiol B 179, 509–517 (2009). https://doi.org/10.1007/s00360-008-0337-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-008-0337-x