Abstract
The effects of acclimation temperature (15, 20, 25 °C) on routine oxygen consumption and post-exercise maximal oxygen consumption rates (MO2) were measured in juvenile shortnose sturgeon (Acipenser brevirostrum LeSueur, 1818). The routine MO2 of shortnose sturgeon increased significantly from 126.75 mg O2 h−1 kg−1 at 15 °C to 253.13 mg O2 h−1 kg−1 at 25 °C. The temperature coefficient (Q 10) values of the routine metabolic rates ranged between 1.61 and 2.46, with the largest Q 10 values occurring between 15 and 20 °C. The average post-exercise MO2 of all temperature groups increased to a peak value immediately following the exercise, with levels increasing about 2-fold among all temperature groups. The Q 10 values for post-exercise MO2 ranged from 1.21 to 2.12, with the highest difference occurring between 15 and 20 °C. Post-exercise MO2 values of shortnose sturgeon in different temperature groups all decreased exponentially and statistically returned to pre-exercise (resting) levels by 30 min at 15 and 20 °C and by 60 min at 25 °C. The aerobic metabolic scope (post-exercise maximal MO2-routine MO2) increased to a maximum value ∼156 mg O2 h−1 kg−1 at intermediate experimental temperatures (i.e., 20 °C) and then decreased as the temperature increased to 25 °C. However, this trend was not significant. The results suggest that juvenile shortnose sturgeon show flexibility in their ability to adapt to various temperature environments and in their responses to exhaustive exercise.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The physiological response to exhaustive exercise in fish has been well studied for more than 50 years with an emphasis on salmonid species (for reviews, see Wood 1991; Milligan 1996; Kieffer 2000, 2010). It has been shown that various abiotic and biotic factors (e.g., temperature, body size, and training) influence the process of and the recovery from exhaustive exercise in fish (Kieffer 2000). It is generally agreed upon that temperature is among the most important abiotic factors affecting an ectothermic animal’s biology, ranging from modifying their distribution to their physiology (Brett 1971; Beitinger and Lutterschmidt 2011). Exhaustive exercise results in marked disturbances to acid-base, osmotic, and electrolyte balance, and increases in oxygen consumption rates in fish (Wood 1991, Kieffer 2000, for reviews), and that these processes and the recovery rate are affected by temperature (e.g., Kieffer et al. 1994; Galloway and Kieffer 2003; Zeng et al. 2010; Kieffer 2010).
Most work to date has focused on the physiological response to exhaustive exercise in teleost fish (e.g., Kieffer 2000; Kieffer and Cooke 2009), and less has focused on ancient fish, such as sturgeon and paddlefish (Barton et al. 2000; Kieffer et al. 2001; Beyea et al. 2005; Baker et al. 2005). The sturgeon (genus Acipenser) are freshwater chondrostean fish that have existed since the Jurassic period (approximately 200 million years ago). Sturgeon have retained many ancestral body characteristics and ways of living that distinguish them as relict fishes (Bemis et al. 1997). Almost all acipenserids are listed as threatened, vulnerable, and endangered throughout their ranges (Birstein and Bemis 1997). Thus, there has been an increased demand for information on all aspects of sturgeon biology, physiology, and evolutionary history (Billard and Lecointre 2001). It has been shown that the physiological response to exhaustive exercise and other forms of acute stressors (e.g., air emersion, hypoxia, confinement) is reduced in sturgeon compared with teleosts (Barton et al. 2000; Kieffer et al. 2001; Baker et al. 2005). In particular, blood and muscle lactate and plasma cortisol levels are not elevated greatly following exhaustive chasing/exercise (Barton et al. 2000; Kieffer et al. 2001; Beyea et al. 2005; Baker et al. 2005, Baker et al. 2008). Oxygen consumption rates increase rapidly and remain elevated for about 30 min following exercise in juvenile shortnose (Acipenser brevirostrum) and Atlantic (Acipenser oxyrinchus) (Kieffer et al. 2001) and Amur (Acipenser schrenckii) sturgeon (Cai et al. 2013). Of the available research on the effects of temperature on the exercise/stress response in sturgeon and paddlefish, studies have focused on critical swimming (Mayfield and Cech 2004; Deslauriers and Kieffer 2012; Aboagye and Allen 2014) and blood parameters following exercise/handling stress (Cataldi et al. 1998; Lankford et al. 2003). In general, a paucity of information exists on the effects of acclimation temperature on the post-exercise metabolic rate changes in sturgeon, yet some recent work exists for routine metabolic rates at various temperatures (Mayfield and Cech 2004; Kieffer et al. 2014). As such, the purpose of this study was to investigate the effects of acclimation temperature (15, 20, and 25 °C) on the routine (routine metabolic rate (RMR)) and post-exercise maximal (maximum metabolic rate (MMR)) metabolic rates and aerobic scope in juvenile shortnose sturgeon. We chose to measure oxygen consumption rates because these are non-lethal measurements and multiple calculations (e.g., routine metabolic rate, maximal metabolic rate, metabolic scope) can be made. Assessing the maximum aerobic metabolic rate and aerobic scope of fish helps in understanding the capacity for performing oxygen consuming physiological capacities (Killen et al. 2016). Previous research has shown that maximum metabolic rate can be influenced by abiotic factors, such as temperature (Norin and Clark 2016). In addition, the metabolic recovery can be easily assessed using relatively low sample sizes. It is important to understand whether temperature affects recovery processes as it has been suggested that a fast recovery process could facilitate the speed at which subsequent exercise bouts could take place (Milligan 1996, for a review). Moreover, the production of sturgeon in intensive aquaculture settings has increased worldwide because of the high market value of caviar (Feshalami et al. 2016). Assessing the potential effects of aquaculture practices, such as exercise/handling stress, on the physiology of sturgeon can help to understand the metabolic costs associated with these events.
Materials and methods
Young-of-the-year shortnose sturgeon were obtained from Acadian Sturgeon and Caviar, Inc. (New Brunswick, Canada; http://www.acadian-sturgeon.com). Groups of fish were assigned to one of three acclimation temperatures: 15, 20, or 25 °C. The tanks were supplied with a flow-through of fresh, aerated, de-chlorinated, municipal tap water at 10 °C. To achieve the required temperatures, the incoming water was heated at a rate of 1 °C/day until the required temperature was attained. Once the desired temperature was achieved, the fish were held at that temperature (i.e., 15, 20, or 25 °C) for 4 weeks prior to the beginning of the experiment (see in the following). Fish were fed twice daily to satiation (Corey Aquafeed: 1.5 mm optimum, 52% protein), but fish were fasted for 24 h prior to the experiments. A 12-h light: 12-h dark photoperiod was maintained throughout the study.
The evening before trials began, an individual sturgeon (∼100 g) was removed from its holding tank (at one of the acclimation temperatures), weighed in water, and placed into a custom-made clear respirometer in fresh water (Loligo® Systems, Denmark; http://www.loligosystems.com; see in the following) held at the acclimation temperature (N = 8 fish at each acclimation temperature). The following morning, baseline measurements of MO2 (mg O2 kg−1 h−1) were taken over 2 h (the mean of this time period was used as the routine metabolic rate) before the exercise. Following this, each fish was removed from the respirometer and exercised to exhaustion via 5-min manual chasing (Kieffer et al. 2001; Baker et al. 2005). Chasing was performed at the same temperature as the experimental exposure. Following exercise, fish were quickly (within a minute) placed back in the respirometer at the same temperature, and MO2 was measured for a further 8 h.
Respirometer setup and oxygen consumption measurement
Custom-made 3.5L respirometers (Loligo® Systems, Denmark; http://www.loligosystems.com) were submerged in a water table with a constant supply of fresh, aerated water at the appropriate acclimation temperature (either 15, 20, or 25 °C). Using a series of pumps, the respirometers were set up to allow for either (i) flow-through from the water table (during flushing) or (ii) recirculation of water within the respirometer (during measurement). An oxygen electrode (Loligo® Systems, Denmark) was located within the recirculation pump circuit tubing to allow for direct measurements of system oxygen levels. All respirometers were set up with visual barriers according to Penny and Kieffer (2014). All measurements of oxygen consumption were calculated using an automated intermittent respirometry program (RESP_EDU, Loligo® Systems, Denmark). Flush/recirculation periods were adjusted for each fish to optimize R 2 values (>0.9) while insuring oxygen concentrations were maintained at or above 75% saturation (400–500 s).
Data handling and analysis
RMR was defined as the post-absorptive MO2 value for a given fish. MMR was defined as the MO2 value immediately following exercise. From these two values, the absolute (MMR – RMR) aerobic and factorial (MMR/RMR) metabolic scopes were determined for each fish. To determine the effects of acclimation temperature on RMR, and the absolute metabolic scopes, a one-way ANOVA was used. Linear regressions were also carried out to determine the relationship between temperature and the endpoint (e.g., RMR, MMR). Temperature quotients (Q 10) were calculated using the formula provided by Prosser (1991): Q 10 = (k 2/k 1)10/(t2 − t1), where k 1 and k 2 are the rates of reaction (rate constants) at temperatures t 1 and t 2, respectively. All values are presented as mean ± SEM (N = 8 fish per group). When the ANOVA revealed significance (P < 0.05), Tukey’s tests were used to test for where these differences between pairs of temperatures occurred. Alpha in all cases was 5% (P < 0.05).
In addition to the previously mentioned, a repeated measures ANOVA followed by a Dunnett’s multiple comparison test was used to compare post-exercise (MMR) values with their respective routine (RMR) values at each acclimation temperature. To test whether differences in the rate of recovery of oxygen consumption (MO2) between fish at the various acclimation temperatures, a two-way repeated measure analysis of variance was used (factors: acclimation temperature and time). A Tukey post hoc test was used if significant differences were found. Values are presented as means ± standard error of the means (SEM). In all cases, P < 0.05 was the accepted level of significance.
Results
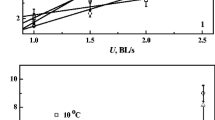
The routine and post-exercise maximal MO2 of A. brevirostrum was affected by acclimation temperature (Fig. 1). The routine MO2 of A. brevirostrum increased significantly from 126.75 mg O2 h−1 kg−1 at 15 °C to 253.13 mg O2 h−1 kg−1 at 25 °C (Fig. 1a; one-way ANOVA, df 2,23, F = 8.9, P < 0.002; linear regression: MO2 = (12.6 × temperature) − 59.8; R 2 = 0.46, P < 0.001). The Q 10 values of the resting MO2 ranged between 1.61 and 2.46, with the largest Q 10 values occurring between 15 and 20 °C (Table 1). Following exercise, the average post-exercise maximal MO2 of all temperature groups increased to a peak value immediately following the exercise (Fig. 1; one-way ANOVA, df 2,23, F = 15.8, P < 0.001; linear regression: MO2 = (14.8 × temperature) + 35.2; R 2 = 0.554, P < 0.001), with levels increasing about 2-fold among all temperature groups. The Q 10 values for post-exercise maximal MO2 ranged from 1.21 to 2.12, with the highest difference occurring between 15 and 20 °C (Table 1). The results of a two-way repeated measures ANOVA indicate that acclimation temperature (P < 0.001) and time following exercise (P < 0.001) had an overall effect on metabolic rate of shortnose sturgeon. Post-exercise maximal MO2 values of A. brevirostrum in different temperature groups all decreased exponentially and returned to pre-exercise (resting) levels by 30 min at 15 and 20 °C and by 60 min at 25 °C (P < 0.05) (Fig. 2). The aerobic metabolic scope (post-exercise MO2- routine MO2) increased to a maximum value ∼156 mg O2 h−1 kg−1 at intermediate experimental temperatures (i.e., 20 °C) and then decreased as the temperature increased to 25 °C (Fig. 1). However, this trend was not significant (P > 0.05). Factorial metabolic scope was consistent (2.02) at 15 and 20 °C but decreased slightly (to 1.61) at 25 °C (Fig. 1).
The relationships between temperature and post-exercise maximal MO2 (MMR; indicated by open symbols with a dashed line) and routine MO2 (RMR; indicated by closed symbols with a solid line) in juvenile shortnose sturgeon (A. brevirostrum). The larger number between the two lines represents the aerobic metabolic scope (MMR-RMR), and the smaller number represents the factorial metabolic scope (MMR/SMR). Different lowercase letters indicate significant differences (P < 0.05) between temperature groups for RMR. Different uppercase letters indicate significant differences (P < 0.05) between temperature groups for MMR. The data represent means ± SEM at specific temperatures
Discussion
While there have been several studies that have examined the effects of temperature on the routine metabolic rates of sturgeon (see Fig. 3), few studies have examined the effects of temperature on post-exercise responses. It has been suggested that the mean global surface temperature has increased by 0.7 °C over the past 100 years (IPCC 2007, in Lassalle et al. 2010). These temperatures are expected to increase which could have significant effects on the distribution and survival of many species (Lassalle et al. 2010). Thus, the findings from the present study are important from both biological and applied perspectives (e.g., aquaculture) and can provide the foundation and some of the parameters to guide future experiments examining the thermal tolerance of sturgeon species.
The relationship between routine oxygen consumption and temperature variation that exists for various sturgeon and paddlefish species (e.g., green sturgeon, Mayfield and Cech 2004; shortnose sturgeon, Kieffer et al. 2014; Atlantic sturgeon, Secor and Gunderson 1998; paddlefish, Patterson et al. 2013)
Routine metabolic rates
The overall mean RMR of the shortnose sturgeon increased with increases in acclimation temperature (Figs. 1 and 3). This pattern is similar (although the absolute values lower) to our previous study using ∼10 g shortnose sturgeon (Kieffer et al. 2014). Despite these parallels, studies for other species of sturgeon and paddlefish report rates of routine metabolism that were quite dissimilar to the present study (Fig. 3). For example, the mass-specific metabolic rate of 22-g green sturgeon (Acipenser medirostris) was lower than shortnose sturgeon at cooler temperatures (Kieffer et al. 2014). In addition, the relationship between metabolic rate and temperature appears to vary between species of sturgeon. Mayfield and Cech (2004) showed that metabolic rates increased exponentially with increases in temperature in both 22- and 851-g green sturgeon; a similar trend is noted for small (280–500 g) and larger (11–12 kg) paddlefish (Polyodon spathula), but not medium (2.4–3.8 kg) paddlefish (Patterson et al. 2013). Evidence for white sturgeon (Acipenser transmontanus, Crocker and Cech 1997) and paddlefish (Patterson et al. 2013) shows that an interaction exists between fish size and temperature on the metabolic rate of these species. Relative to some of the other species of sturgeon, however, the pattern for shortnose sturgeon (Kieffer et al. 2014) showed that the routine metabolic rate increased to a large extent at the lower temperatures tested (Kieffer et al. 2014) and then showing a plateau. These findings are supported by the larger Q 10 values noted in the present study between 15 and 20 and 15 and 25 °C (Table 1). The relationships between temperature and metabolic rate in sturgeon might reflect the importance of fish size/age, or it may be related to differences in thermal sensitivities (e.g., critical thermal maxima) of the fish species or the range of temperatures used. Thus, future studies are needed to further our understanding of temperature and resting metabolic rate relationships in sturgeon.
Post-exercise maximum metabolic rates and recovery patterns
Similar to that for routine metabolism, there was an increase in post-exercise metabolic rates with increases in temperature (Fig. 1) and these rates were about two times higher than routine values across temperatures (see Table 2). Compared to routine values, post-exercise metabolic rates were less affected (i.e., lower Q 10 values) by temperature. These changes of the routine and post-exercise metabolic rate initially led to an increase in aerobic metabolic scope (between 15 and 20 °C) and then a plateau trend (between 20 and 25 °C). These patterns for absolute metabolic scope have been shown in other species of fish, including freshwater and marine species (Tirsgaard et al. 2015; Eliason and Farrell 2016). In contrast, the findings for factorial metabolic scope (maximum metabolic rate/routine rate) suggest a slight decrease in factorial scope at the higher temperatures, a pattern also noted for Pacific salmon (Oncorhynchus spp.; reviewed in Eliason and Farrell 2016). This may suggest that the ability to do metabolic work at higher temperatures in shortnose sturgeon might be compromised, which has recently been shown by Deslauriers and Kieffer (2012) who showed that critical swimming speed (UCrit) in shortnose sturgeon began to plateau at 15 through 25 °C. From a practical perspective, it is known that maximum metabolic rate determination may be affected by the experimental protocol utilized (see Tirsgaard et al. 2015, Norin and Clark 2016; Killen et al. 2016; for background), such as a chase protocol (as used here) and critical swimming tests (UCrit), specific dynamic action (SDA) test, or critical thermal maximum test (CTmax) to achieve the maximum metabolic rate of the test species. From our experience, the absolute metabolic and factorial scopes following exhaustive exercise in the current study were about 75% of the values noted during a critical thermal maximum test using similar-sized shortnose sturgeon (Zhang and Kieffer, unpublished). However, the burst, chasing stress protocol used within the current study represents a scenario related to exercise responses in fishes and is considered a relevant stressor within the fisheries and aquaculture sectors (e.g., catch-and-release fisheries, commercial fishing throwback, netting stress, transportation and handling stresses; Wood 1991, Kieffer and Cooke 2009).
Shortnose sturgeon exhibited a physiological stress response to chasing stress as indicated by increases in whole-animal oxygen consumption rates. These responses, however, are considerably lower compared with those previously documented for teleost fishes (Scarabello et al. 1992; Wakefield et al. 2004; Kieffer 2000). Of interest is the fast post-exercise recovery process in shortnose sturgeon. The post-exercise oxygen consumption rates in sturgeon acclimated to 15 and 20 °C return to routine levels within 30 min and by 60 min at 25 °C. Kieffer et al. (2001) noted a similar return of oxygen consumption rate (i.e., 30 min) following exhaustive exercise in shortnose and Atlantic sturgeon at 12 °C. In the same way, the oxygen consumption rate rapidly decreased after exercise by approximately 50 min in juvenile (32 g) Amur sturgeon (A. schrenckii) at 20 °C (Cai et al. 2013). Patterns of metabolic recovery following exhaustive exercise have been shown to be influenced by acclimation temperature in Nile tilapia (Oreochromis niloticus McKenzie et al. 1996) and juvenile southern catfish (Pterygoplichthys anisitsi; Zeng et al. 2010), also with the slowest recovery occurring at the warmest temperature. Pang et al. (2015) also showed a similar and significant effect of temperature on the post-exercise recovery process in juvenile qingbo (Spinibarbus sinensis) following a constant acceleration test. It has been suggested that a fast recovery process could facilitate the speed at which subsequent exercise bouts could take place (Milligan 1996, for a review). Whether this applies to sturgeon is still not entirely clear as little work has been done on multiple exercise bouts in sturgeon (Cai et al. 2014) and at various temperatures. Future work should address whether temperature-related changes in exercise capacity vary with multiple exercise bouts.
In conclusion, the findings from the present study indicate that the routine metabolic rates of shortnose sturgeon, although are impacted by temperature, show a different response compared to documented cases for some other sturgeon species. In addition, while exercise increases metabolic rates in shortnose sturgeon, the magnitude of the response is muted compared with other studied species, such as salmonids (Eliason and Farrell 2016). Lastly, the post-exercise recovery process is relatively fast and affected by temperature in sturgeon. Future research should examine the effects of temperature on the metabolic costs associated with aerobic, sustained swimming in sturgeon, as this is not well known. From a practical perspective, the findings from the present study suggest that recovery from other relevant stressful events (e.g., husbandry practices, hatchery practices, angling, stocking densities) might be relevant and important to understand. As various sturgeon species are being grown in an aquaculture setting, it may be important to understand the impacts of temperature on the stress response (e.g., transport stress, culling of various size classes of fish). However, a more detailed examination of the importance of temperature on sturgeon biology and physiology (e.g., changes in metabolites, production of heat shock proteins, ion changes, cardiac function, and circulatory physiology) is warranted.
References
Aboagye DL, Allen PJ (2014) Metabolic and locomotor responses of juvenile paddlefish Polyodon spathula to hypoxia and temperature. Comp Bioch Physiol 169A:51–59
Baker DW, Wood AM, Litvak MK, Kieffer JD (2005) Haematology of juvenile Acipenser oxyrinchus and Acipenser brevirostrum at rest and following forced activity. J Fish Biol 66:208–221
Baker DW, Peake SJ, Kieffer JD (2008) The effect of capture, handling, and tagging on hematological variables in wild adult lake sturgeon. North Am J Fish Manag 28:296–300
Barton BA, Bollig H, Hauskins BL, Jansen CR (2000) Juvenile pallid (Scaphirhynchus albus) and hybrid pallid × shovelnose (S. albus X platorynchus) sturgeons exhibit low physiological responses to acute handling and severe confinement. Comp Biochem Physiol 126A:125–134
Beitinger TJ, Lutterschmidt WI (2011) Measures of thermal tolerance. In: Farrell AP, Stevens ED, Cech JJ, Richards JG (eds) Encyclopedia of fish physiology—from genome to environment. Academic Press, Waltham
Bemis WE, Findeis EK, Grande L (1997) An overview of Acipenseriformes. Env Biol Fish 48:25–71
Beyea MM, Benfey TJ, Kieffer JD (2005) Hematology and stress physiology of juvenile diploid and triploid shortnose sturgeon (Acipenser brevirostrum). Fish Physiol Biochem 31:303–313
Billard R, Lecointre G (2001) Biology and conservation of sturgeon and paddlefish. Rev Fish Biol Fisher 10:355–392
Birstein VJ, Bemis WE (1997) How many species are there within the genus Acipenser? Envir Biol Fish 48:157–163
Brett JR (1971) Energetic responses of salmon temperature. A study of thermal relations in the physiology and freshwater ecology of sockeye salmon (Oncorhynchus nerka). Am Zool 11:99–113
Cai L, Taupier R, Johnson D, Tu Z, Liu G, Huang Y (2013) Swimming capability and swimming behaviour of juvenile Acipenser schrenckii. J Exp Zool 319A:149–155
Cai L, Johnson D, Mandal P, Gan M, Yuan X, Tu Z, Huang Y (2014) Integrating water flow, locomotor performance and respiration of Chinese sturgeon during multiple fatigue-recovery cycles. PLoS One 9:e94345
Cai L, Johnson D, Mandal P, Gan M, Yuan X, Tu Z, Huang Y (2015) Effect of exhaustive exercise on the swimming capability and metabolism of juvenile Siberian sturgeon. Trans Am Fish Soc 144:532–538
Cataldi E, Di Marco P, Mandich A, Cataudella S (1998) Serum parameters of Adriatic sturgeon Acipenser naccarii (Pisces: Acipenseriformes): effects of temperature and stress. Comp Biochem Physiol 121A:351–354
Crocker CE, Cech JJ (1997) Effects of environmental hypoxia on oxygen consumption rate and swimming activity in juvenile white sturgeon, Acipenser transmontanus, in relation to temperature and life intervals. Environ Biol Fish 50:383–389
Deslauriers D, Kieffer JD (2012) The effects of temperature on swimming performance of juvenile shortnose sturgeon (Acipenser brevirostrum). J Appl Ichth 28:176–181
Eliason EJ, Farrell AP (2016) Oxygen uptake in Pacific salmon Oncorhynchus spp.: when ecology and physiology meet. J Fish Biol 88:359–388
Feshalami MY, Amiri F, Nikpey M, Mortazavizadeh MT (2016) Influence of stocking density on growth and physiological responses of Beluga, Huso huso (Brandt, 1869), and Ship sturgeon, Acipenser nudiventris (Lovetsky, 1828), juveniles in a flow-through system. J World Aquacult Soc Doi. doi:10.1111/jwas.12376
Galloway BJ, Kieffer JD (2003) The effects of an acute temperature change on the metabolic recovery from exhaustive exercise in juvenile Atlantic salmon (Salmo salar). Phys Biochem Zool 76:652–662
Kieffer JD (2000) Limits to exhaustive exercise in fish. Comp Biochem Physiol 126A:161–179
Kieffer JD (2010) Perspective-exercise in fish: 50+ years and going strong. Comp Biochem Physiol 156A:163–168
Kieffer JD, Cooke SJ (2009) Physiology and organismal performance of centrarchids. In: Cooke SJ, Philipp DP (eds) Centrarchid fishes: diversity, biology and conservation. Oxford Press, Wiley, pp 207–263
Kieffer JD, Currie S, Tufts BL (1994) Effects of environmental temperature on the metabolic and acid–base responses of rainbow trout to exhaustive exercise. J Exp Biol 194:299–317
Kieffer JD, Wakefield AM, Litvak MK (2001) Juvenile sturgeon exhibit reduced physiological responses to exercise. J Exp Biol 204:4281–4289
Kieffer JD, Penny FM, Papadopoulos V (2014) Temperature has a reduced effect on routine metabolic rates of juvenile shortnose sturgeon (Acipenser brevirostrum). Fish Physiol Biochem 40:551–559
Killen SS, Norin T, Halsey LG (2016) Do method and species lifestyle affect measures of maximum metabolic rate in fishes? J Fish Biol Doi. doi:10.1111/jfb.13195
Lankford SE, Adams TE, Cech JJ (2003) Time of day and water temperature modify the physiological stress response in green sturgeon, Acipenser medirostris. Comp Biochem Physiol 135A:294–302
Lassalle G, Crouzet P, Gessner J, Rochard E (2010) Global warming impacts and conservation responses for the critically endangered European Atlantic sturgeon. Biol Conserv 30:2441–2452
Mayfield RB, Cech JJ (2004) Temperature effects on green sturgeon bioenergetics. Trans Am Fish Soc 133:961–970
McKenzie DJ, Serrini G, Piraccini G, Bronzi P, Bolis CL (1996) Effects of diet on responses to exhaustive exercise in Nile tilapia (Oreochromis nilotica) acclimated to three different temperatures. Comp Biochem Physiol 114A:43–50
McKenzie DJ, Cataldi E, Romano P, Owen SF, Taylor EW, Bronzi P (2001) Effects of acclimation to brackish water on the growth, respiratory metabolism, and swimming performance of young-of-the-year Adriatic sturgeon (Acipenser naccarii). Can J Fish Aquat Sci 58:1104–1112
Milligan CL (1996) Metabolic recovery from exhaustive exercise in rainbow trout. Comp Biochem Physiol 113A:51–60
Norin T, Clark TD (2016) Measurements and relevance of maximum metabolic rate in fishes. J Fish Biol 88:122–151
Pang X, Yuan X-Y, Cao Z-D, Zhang Y-G, Fu S-J (2015) The effect of temperature on repeat performance in juvenile qingbo (Spinibarbus sinensis). Fish Physiol Biochem 41:19–29
Patterson JT, Mims SD, Wright RA (2013) Effects of body mass and temperature on routine metabolism of American paddlefish Polodon spathula. J Fish Biol 82:1269–1280
Penny FM, Kieffer JD (2014) Oxygen consumption and haematology of juvenile shortnose sturgeon, Acipenser brevirostrum, during an acute 24 hour saltwater challenge. J Fish Biol 84:1117–1135
Prosser CL (1991) Environmental and metabolic animal physiology. Wiley, New York, pp 109–166
Scarabello M, Heigenhauser GJF, Wood CM (1992) Gas exchange, metabolic status and excess post-exercise oxygen consumption after repetitive bouts of exhaustive exercise in juvenile rainbow trout. J Exp Biol 167:155–169
Secor DH, Gunderson TE (1998) Effects of hypoxia and temperature on survival, growth, and respiration of juvenile Atlantic sturgeon, Acipenser oxyrinchus. Fish Bull 96:603–613
Svendsen JC, Genz J, Anderson WG, Stol JA, Watkinson DA, Enders EC (2014) Evidence of circadian rhythm, oxygen regulation capacity, metabolic repeatability and positive correlations between forced and spontaneous maximal metabolic rates in lake sturgeon Acipenser fulvescens. PLoS One 9:e94693
Tirsgaard B, Behrens JW, Steffensen JF (2015) The effect of temperature and body size on metabolic scope of activity in juvenile Atlantic cod Gadus morhua L. Comp Biochem Physiol 179A:89–94
Wakefield AM, Cunjak RA, Kieffer JD (2004) Metabolic recovery in Atlantic salmon fry and parr. J Fish Biol 65:920–932
Wood CM (1991) Acid-base and ion balance, metabolism, and their interactions, after exhaustive exercise in fish. J Exp Biol 160:285–308
Zeng L-Q, Zhang Y-G, Cao Z-D, Fu S-J (2010) Effect of temperature on excess post-exercise oxygen consumption in juvenile southern catfish (Silurus meridionalis Chen) following exhaustive exercise. Fish Physiol Biochem 36:1243–1252
Acknowledgements
Financial support was provided by a discovery grant from the Natural Sciences and Engineering Research Council of Canada (NSERC) and New Brunswick Innovation Fund (NBIF) to J. Kieffer. The authors also thank the MADSAM fish group for their continued support. The experimental protocol was approved by the University of New Brunswick Animal Care Committee, meeting Canadian Council of Animal Care guidelines.
Author information
Authors and Affiliations
Corresponding author
Additional information
What is already known
Acclimation temperature has significant impacts on the routine oxygen consumption rate in teleost fish. It also affects the post-exercise maximal oxygen consumption rate and exercise/stress response.
What this study adds
The current study shows that acclimation temperature significantly increases the routine oxygen consumption rate and post-exercise oxygen consumption rate in shortnose sturgeon.
Rights and permissions
About this article
Cite this article
Zhang, Y., Kieffer, J.D. The effect of temperature on the resting and post-exercise metabolic rates and aerobic metabolic scope in shortnose sturgeon Acipenser brevirostrum . Fish Physiol Biochem 43, 1245–1252 (2017). https://doi.org/10.1007/s10695-017-0368-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-017-0368-x