Abstract
Objective
To longitudinally evaluate effects of smoking cessation on quantitative CT in a lung cancer screening cohort of heavy smokers over 4 years.
Methods
After 4 years, low-dose chest CT was available for 314 long-term ex-smokers (ES), 404 continuous smokers (CS) and 39 recent quitters (RQ) who quitted smoking within 2 years after baseline CT. CT acquired at baseline and after 3 and 4 years was subjected to well-evaluated densitometry software, computing mean lung density (MLD) and 15th percentile of the lung density histogram (15TH).
Results
At baseline, active smokers showed significantly higher MLD and 15TH (-822±35 and -936±25 HU, respectively) compared to ES (-831±31 and -947±22 HU, p<0.01–0.001). After 3 years, CS again had significantly higher MLD and 15TH (-801±29 and -896±23 HU) than ES (-808±27 and -906±20 HU, p<0.01–0.001) but also RQ (-813±20 and -909±15 HU, p<0.05-0.001). Quantitative CT parameters did not change significantly after 4 years. Importantly, smoking status independently predicted MLD at baseline and year 3 (p<0.001) in multivariate analysis.
Conclusion
On quantitative CT, lung density is higher in active smokers than ex-smokers, and sustainably decreases after smoking cessation, reflecting smoking-induced inflammation. Interpretations of quantitative CT data within clinical trials should consider smoking status.
Key Points
• Lung density is higher in active smokers than ex-smokers.
• Lung density sustainably decreases after smoking cessation.
• Impact of smoking cessation on lung density is independent of potentially confounding factors.
• Smoke-induced pulmonary inflammation and particle deposition influence lung density on CT.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Chest multidetector computed tomography (CT) is the gold standard for imaging-based phenotyping of cigarette smoke-induced chronic obstructive pulmonary disease (COPD) [1,2,3]. Two main features, related to distinct clinical phenotypes, may be assessed: airway remodelling and emphysema [4,5,6,7,8,9]. Computational densitometry, or quantitative CT (QCT), of the lung parenchyma based on Hounsfield units (HU) is currently the method of choice for non-invasive objective quantification of smoke-induced parenchymal disorders, especially emphysema, which presents with decreased lung density values [1, 10, 11]. As such it has been implemented in various clinical trials including the COPDGene study [12, 13]. Its acceptance in clinical routine is rapidly broadening, leading to an implementation into the workflow of interventional emphysema therapy in many specialised centres [14]. There is, however, limited data available on the intra-individual time course of lung density on QCT. Moreover, the long-term effects of smoking cessation on QCT of the pulmonary parenchyma remain largely unknown. Some publications, amongst them a recent publication from the COPDGene cohort, have demonstrated that active smokers show a higher lung density and less signs of emphysema on QCT than former smokers [15,16,17,18]. Some reported that smoking cessation results in a decrease in lung density with a somewhat paradoxical increase of emphysema on QCT [19, 20]. Longitudinal data from larger populations is missing however. The present study was therefore conducted to longitudinally study effects of smoking cessation on CT-based metrics of lung density and emphysema in a large subcohort of heavy smokers from the first German Lung Cancer Screening Intervention Trial (LUSI) over a period of 4 years. Because this study included a standardised smoking cessation programme, patient groups could be designed to include past smokers, continuously active smokers and smokers who stopped smoking after study onset.
Materials and methods
Study population
Participants for the present study were identified within the Lung Cancer Screening Intervention Trial (LUSI) cohort, with inclusion criteria as published previously [21, 22]. Briefly, the recruitment procedure was population based in a study area mainly approximately 20–25 km around our centre, but partially up to 70 km. Recruitment with randomisation was carried out from November 2007 to April 2011. Because the CT scanner system needed to be replaced during the active conduct of the study, for the present investigation the following additional inclusion criteria were necessary: (1) Availability of CT data with 1.0-mm slice thickness and sharp convolution kernel reconstructions (Acquillion 16, Toshiba Medical Systems Corp., Otawara, Japan) at baseline. (2) Availability of CT data with 1.0-mm slice thickness and soft kernel reconstructions for years 3 and 4 (Definition Flash, Siemens Healthcare AG, Forchheim, Germany). (3) Spirometry performed at baseline.
Follow-up was conducted actively by annual questionnaire mailing and passively by repeated linkage to the local population registers and cancer registries [21]. With study inclusion, smoking-cessation counselling was offered to each participant as previously described [23].
Smoking habits
Smoking habits were surveyed by a standardised questionnaire distributed to all subjects at inclusion and repetitively every 12 months after inclusion [23]. Amongst others, current and past smoking status and habits, cigarettes per day and total years smoked were assessed. Specifically, subjects who quitted smoking were to specify the duration of smoking cessation in categories of <1 month, 1–6 months, 7 months–1 year, 1–2 years, 3–5 years, 6–10 years, 11–15 years, 16–20 years and >20 years.
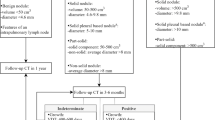
For the purpose of this study, two subgroups were designed at baseline: (1) ‘ex-smokers’ (ES) were defined as subjects who selected the category ‘quitted 1–2 years’ or more at baseline, and did not start active smoking again during the whole study period; (2) ‘all smokers’ were defined as subjects who actively smoked at baseline.
At the follow-up after 3 and 4 years we created three subgroups (Fig. 1): (1) The group of ES mentioned above who quitted 1–2 years or more at baseline. In addition, the above-mentioned group ‘all smokers’ was further subdivided into two groups as follows: (2) ‘continuous smokers’ (CS) were defined as subjects who continued to smoke from baseline during the whole study period (3) ‘recent quitters’ (RQ) were defined as subjects who were active smokers at baseline, selected category ‘quitted 1–2 years’ or more at year 3, and did not start smoking again during the remaining study period. In other words, ES had quitted smoking >12 months before baseline, and RQ quitted smoking after baseline but >12 months before the 3 years and >24 months before the 4 years’ follow-up CT. Of the 2,029 patients initially recruited for the LUSI trial [21], n=757 patients fulfilled the criteria for inclusion into the present study. Table 1 summarises patient characteristics.
Coronal CT images of a continuous smoker (CS; mean lung density (MLD) -801 HU), ex-smoker (ES; MLD -808 HU) and recent quitter (RQ; MLD -814 HU) with representative quantitative CT parameters for the corresponding group at time point T4. Hounsfield unit (HU) values <-950 are marked yellow, -950 to -900 HU orange, -900 to -850 HU red, -850 to -800 HU green, and ≥ -800 HU blue
Pulmonary function testing
Spirometry (Carefusion, Höchberg, Germany) was performed according to the guidelines of the European Respiratory Society and the standards of the American Thoracic Society (ATS) [24], and the European Coal and Steal Community (ECSC) predicted values served as the standard of reference [25]. The following lung function parameters (absolute and percent predicted values) were used for further analysis: forced expiratory volume in 1 s (FEV1), forced vital capacity (FVC) and FEV1/FVC (Tiffeneau index).
Patient Characteristics
According to spirometry at baseline the FEV1/FVC ratio, FEV1 as well as FEV1% predicted were not significantly different between the respective patient groups (Table 1). The criteria of the Global Initiative for Chronic Obstructive Lung Disease (GOLD) [2] were applied to all subjects upon inclusion into this trial to identify subjects with COPD. The prevalence of COPD was low in all groups, with 2.6–9.2 %, and did not show significant differences. The amount of pack years also did not show significant variability (Table 1). At baseline, the group of all smokers had a significantly higher duration of smoking compared to the ES group (38.5 ± 16.7 vs. 33.1 ± 9.7 years, p<0.001). The smoking duration of the CS and RQ groups did not differ significantly. ES and RQ showed younger age and higher percentage of male subjects than CS (Table 1).
Multidetector computed tomography
Non-enhanced thin-section low-dose CT was routinely performed in the supine position as previously described [5, 21]. Before scanning, all patients received an instructed training to achieve full end-inspiratory breath-hold. At baseline, all patients were scanned with a 16-slice Aquillion 16 system (Toshiba Medical Systems Corp., Otawara, Japan) with a dose-modulated protocol at 120 kV and 55 mAs (effective), 1.0 mm collimation and pitch 1.5. Reconstruction was performed using filtered back projection with a slice thickness of 1.0 mm and 0.8 mm increment in an FC51 convolution kernel. The FC51 convolution kernel is a standard lung kernel for Toshiba scanners. Using a high-pass filter algorithm it provides sharp image reconstructions with higher spatial frequencies (compared to soft tissue kernels) at the expense of greater noise. It works well in tissues with inherently high CT contrast and is frequently used in qualitative and quantitative chest imaging.
Follow-up examinations after 3 and 4 years were performed with a 128-slice Definition Flash (Siemens Healthcare AG, Forchheim, Germany) with a dose-modulated protocol at 120 kV, 40 mAs (effective), a collimation of 0.6 mm and a pitch of 0.8. Reconstruction was performed using filtered back projection with a slice thickness of 1.0 mm and 0.7-mm increment in a soft B20f algorithm as recommended for densitometry [4, 7]. The B20f convolution kernel is a standard smooth soft tissue kernel developed for Siemens scanners, using low-pass filters to reduce higher frequency contributions and decrease image noise as well as spatial resolution.
Both systems were calibrated for water quarterly and after major maintenance, and daily for air. At baseline a CT dose index (CTDI) of 5.1 was used for the Toshiba scanner while the CTDI for the Siemens scanner at follow-up was 2.7. Assuming a scan length of 35 cm, this converts to effective doses of 3.0 mSv for the Toshiba scanner and 1.6 mSv for the Siemens scanner, approximately. All CT studies were visually inspected by a reader with more than 5 years of experience in chest radiology for adequate inspiration, absence of significant motion artefacts and inclusion of all parts of the chest, similar to the criteria of the COPDGene study [12].
Densitometry
QCT data from baseline as well as from 3 and 4 years after study onset were subjected to a well-evaluated in-house scientific software (YACTA version 2.5.4.3, programming by O.W.) for densitometry [26,27,28]. YACTA analysed each dataset fully automatically, as employed in previous studies [7, 26,27,28,29,30,31]. Neither user interaction nor manual correction of the segmentation were carried out. A lung voxel was assigned to emphysema if its density was equal to or lower than -950 HU. Prior to this, we applied a Gaussian filter [1, 4] and performed noise correction for voxels with -910 to -949 HU that needed at least four adjacent voxels with a density of < -950 HU to partially adjust for effects of image noise on QCT. The tracheal air was segmented and in order to minimise the influence of the tracheal wall on voxel densitory calculations the segmentation was eroded by 3 voxels. After 3 x 3 Gaussian filtering, the standard deviation for tracheal air HU was 36.9 HU for B20f reconstructions and 49.7 HU for FC51. The software further employs a dynamic threshold which adjusts the -950 HU threshold for emphysema in case the air density (outside the patient and inside the trachea) is different from -1,000 HU. Based on the CT data, the following variables were computed by the scientific software: the total lung volume in inspiration (LV), the total volume of lung areas with a density below -950 HU (emphysema volume, EV), and the fraction of emphysema volume in relation to the total lung volume in inspiration in percent (emphysema index, EI). We also calculated the mean lung density (MLD) in HU averaging CT-based density values from all voxels of the entire lung. Finally, the 15th percentile of lung density histogram (15TH) was calculated. This is defined as the density value in HU below which 15 % of the lung voxels are found.
Statistical analysis
Prior to statistical evaluation, computational results were reviewed by a reader with more than 7 years of expertise in chest radiology. All data were recorded in a dedicated database (Excel®, Microsoft Corp., Redmond, WA, USA) and analysed using SigmaPlot® (Systat Software GmbH, Erkrath, Germany) software. Parametric data are displayed as mean ± standard deviation, non-parametric data as median ± median average deviation. Since the extent of image noise decreased after baseline due to a CT scanner exchange, we thus did not focus on absolute QCT measures or intra-individual changes in QCT measures. Instead, we concentrated on relative intergroup differences in mean lung density and emphysema measures. Intra-individual changes were directly comparable only between the 3-year and 4-year follow-up. Intergroup comparison was performed using analysis of variance (ANOVA) for normal distributions and the Kruskal-Wallis test for skewed distributions. Student’s t-test with Bonferroni-Holm correction was used for post hoc analyses of parametric data. For non-parametric data, post hoc analysis was performed by the Mann-Whitney U test and Bonferroni correction. Categorical data were analysed using the chi-squared test or Fisher’s exact test where appropriate. For the evaluation of longitudinal changes, a paired t-test was applied on parametric data and Wilcoxon signed-rank test on non-parametric data.
Multivariate linear regression analysis was performed with baseline data and data from year three in order to identify potential predictors of MLD. In both models lung volume, age, gender, FEV1%, current smoking status, BMI and pack years were chosen as explanatory variables. Previously, the explanatory variables were tested for collinearity using Pearson’s product-moment correlation coefficient for normal distributions and Spearman's rank order correlation coefficient for skewed distributions. A p-value of <0.05, corrected with the Bonferroni-Holm method in case of multiple comparisons, was considered statistically significant [32].
Results
QCT results at baseline
At the time of the first assessment, ES had significantly higher QCT signs of emphysema in terms of EV, EI, MLD and 15TH than all active smokers (p<0.01–0.001) (Table 2).
QCT 3 years after study inclusion
Three years after study onset, two groups evolved out of the active smokers. CS again showed lower QCT parameters for emphysema (p<0.01–0.001). Furthermore, RQ who just ceased to smoke now showed a significantly reduced MLD and 15TH (p<0.05–0.001) compared to CS, which were even lower compared to the group of ES (n.s.) (Table 2). Of note, baseline values of CS and RQ were not significantly different (data not shown). A longitudinal intragroup comparison cannot be made between baseline and follow-up due to an exchange of the CT scanner and use of a sharper reconstruction kernel after baseline.
Longitudinal changes in QCT
We did not detect a significant change of QCT parameters from year 3 to year 4, irrespective of smoking habits (Table 3). Four years after study onset, the abovementioned relationships between ES, CS and RQ for emphysema in QCT remained largely unchanged (Table 2).
At baseline, multivariate linear regression analysis revealed the following seven variables to be independent predictors of MLD, in descending order of predictive value as expressed by the standardised regression coefficient: LV, sex (male), smoking status (current smoker), BMI, FEV1%, age and pack years (p<0.001) (Table 4). The regression model showed a high coefficient of determination with R2=0.572. After 3 years, the above-mentioned variables would again significantly predict MLD (p<0.001), with a similar predictive value compared to baseline (Table 4). Interestingly, the predictive value of the smoking status tended to increase from baseline to the 3-year follow-up (standardised regression coefficient = 0.310 vs. 0.349). For the sake of completeness, it should be mentioned that FEV1/FVC showed no meaningful association with MLD. The remaining variables listed in Tables 1 and 2 were excluded from the regression model due to excessive collinearity with other independent variables.
Discussion
It has been hypothesised that active smoking perpetuates inflammatory processes in the lung parenchyma and airways and thus can partially mask signs of emphysema on QCT by increasing lung tissue density. Further, a deposition of tar and increased mucus production may play a role [19, 20]. The influence of subclinical smoking-related interstitial lung diseases (ILD) such as respiratory-bronchiolitis interstitial lung disease (RB-ILD) or desquamative interstitial pneumonia (DIP) has also not been systematically examined. Recently, Zach et al. could demonstrate in a cross-sectional study with as many as 6,762 subjects from the COPDGene cohort that active smokers have lower signs of emphysema and air-trapping on QCT [18], and thus confirmed previous reports from studies smaller in size. Specifically, they suggested adding 3.5 % to the LAA%950 (equal to the emphysema index EI in our study) for active smoking status to gain a more precise estimation of emphysema in these patients [18]. They also reported a significant and strong influence on MLD and 15TH, without providing quantitative data. Time since quit did not show a significant influence in regression analyses [18]. The 15th percentile density PD15 (15TH in our study) was also addressed by Ashraf et al., who have shown in a longitudinal study with 1-year follow-up that cessation may significantly decrease PD15 only during the first year after quitting [20]. This is complementary to the data from our study, which can be interpreted as follows: (1) Ex-smokers (>12 months before study onset) had a lower MLD and 15TH than active smokers at baseline, as well as after 3 and 4 years. (2) Those who ceased to smoke during the study >12 months before the follow-up examination again had a lower MLD and 15TH than continuous smokers. (3) The aforementioned recent quitters continued to have a lower MLD and 15TH >24 months after cessation, and had a MLD and 15TH close to the long-term ex-smokers.
In our study population, most risk factors potentially influencing lung density did not show significant variability between the patient groups. However, ex-smokers and recent quitters showed slightly younger age and higher percentage of male subjects than continuously active smokers (Table 1). While the amount of pack years did not differ significantly between the patient groups, active smokers at baseline showed significantly longer smoking duration compared to ex-smokers, who in turn had a higher number of cigarettes smoked per day. To further explore separate effects of smoking cessation and potential confounders such as age and sex on the observed decrease in lung density, multivariate analysis was applied. In this context, the smoking status was shown to be a significant predictor of MLD and 15TH, independent from other potentially contributing factors (Table 4). Since a causality between the relatively long smoking duration and the elevated MLD in the group of baseline smokers seems inconceivable, and smoking duration is confounded by inclusion criteria (smoking cessation before inclusion in case of ex-smokers), the smoking duration was not included in the multivariate model,
Of note, our study population had low signs of emphysema on QCT with an EI of 5.1 % and 8.4 % at baseline (Table 2), and practically no emphysema (EI=0–0.3 %) after scanner and protocol change at follow-up. In part this may be explained by the low-dose CT acquisition protocol, which will significantly influence CT densitometry [33, 34]. The presence of emphysema observed at baseline but not at follow-up may be largely based on increased image noise due to the relatively sharp convolution kernel (Toshiba FC51) applied at baseline compared to the soft and low-noise kernel used during follow-up (Siemens B20f). In this context, Gierada et al. showed that the emphysema index tends to increase with sharper kernels [35]. For the COPDGene study, for example, a standard-dose CT protocol was carried out [12]. Normal reference values for emphysema and emphysema progression have not been established [36], but values around 5 % for EI compare well to GOLD stage 0 smokers in this age group and may be considered as ‘normal’ when using -950 HU as a threshold [7]. Values for MLD and 15TH also were close to results previously obtained by our group in GOLD stage 0 smokers, and much higher than in GOLD stage III or IV [7]. PFT results of our population support this notion of normal smokers (Table 1).
In the present population of relatively normal smokers, we did not detect a significant annual change of QCT parameters for emphysema in any group, regardless of smoking status. Of note, we could not detect a significant annual change in the emphysema index, mean lung density or 15th percentile of lung density histogram from year 3 to year 4 in our present study, despite including a reasonable number of patients. For repeated low-dose scans in a 3-month interval, Gietema et al. found a mean difference for the emphysema score (equals EI in our study) of -0.1 % with a limit of agreement from -1.3 to 1.1 % at a threshold of -950 HU [37]. Hence, the short-term interscan intra-individual variability is low. Soejima et al. reported an annual change between 0.7 % and 2.3 % for relatively low attenuation areas (threshold of -912 HU) during a 5-year observational period for 47 current or former smokers, without significant impact of the smoking status. For MLD, they reported a mean annual change between and -3.5 and +0.5 [38]. In a more recent study, Mohamed Hoesein et al. reported a mean annual increase of emphysema of 1.07 % (confidence interval 1.06–1.09) in 3,670 former and current smokers at -950 HU [17]. Interestingly, the reported long-term data showed a variability that is within the limits of reported short-term variability [39]. Thus it is not surprising that we did not find significant results in our population. We speculate that the signal of annual increase in QCT parameters was too low to be detected by our CT protocol in a population of this size. Potentially, emphysema progression may accelerate with higher emphysema severity, and thus, on the other hand, may be very slow in our relatively normal population. Longitudinal data from the large COPDGene cohort are currently being anticipated, and may clarify the process of emphysema progression.
Some limitations of our study need to be addressed. Because scanner systems were exchanged after study onset, a direct longitudinal comparison of QCT between baseline and years 3 to 4 is not possible since CT attenuation measurements usually show significant variability between different scanner types [40]. The quantitative CT software YACTA required the use of thin section image data, which were provided by the imaging centre with the soft B20f kernel for the Siemens scanner, and the sharper FC51 kernel for the Toshiba scanner as defined by the protocol of the screening study. Unfortunately, the soft reconstructions (FC13) of the Toshiba scanner had a high slice thickness of 3 mm. In this context, the quantitative analysis of images with different CT reconstruction kernels can produce different results, and sometimes a higher emphysema threshold (-960 instead of -950 HU) is recommended for sharp kernels. However, the quantitative software YACTA employs a Gaussian filter to smoothen sharp kernel images partially adjusting for effects of the reconstruction kernel on QCT. Further, it employs a dynamic threshold that adjusts the -950 HU threshold for emphysema in case the air density (outside the patient and inside the trachea) is different from -1,000 HU. As the B20f kernel seems more comparable to the FC51 because of slice thickness, these kernels were subsequently used for analysis. Since this still poses a limitation, we focused on inter-group differences at each time point of QCT, rather than on intra-individual longitudinal changes of lung density. Thus, potential variations in densitometry due to different scanner types or convolution kernels do not affect the findings of this study, nor does the chosen emphysema threshold. Further, we assessed routine screening CT, which was not controlled by a spirometer, and thus levels of inspirations may vary [41, 42]. On the other hand, we refrained from computational correction of individual lung volume between scans because this would not be correct for a true annual change in lung volume due to hyperinflation. Furthermore, we did not detect a significant change in lung volume from year 3 to year 4. This allows the interpretation of relative differences between the groups in our study.
In conclusion, we could show in a lung cancer screening population with relatively normal smokers that lung density is higher in active smokers than ex-smokers, and sustainably decreases after smoking cessation. Importantly, the impact of smoking cessation on lung density was demonstrated to be independent from potentially confounding factors. The results of our study add four findings to previous data: (1) We could reproduce that active smokers have less signs of emphysema on QCT; (2) we could confirm that smoking cessation results in an increase of signs of emphysema on QCT; (3) we could demonstrate that the difference between active smokers and ex-smokers is maintained over a period of 1 year for a group of recent quitters; and (4) over a period of at least 4 years for long-term non-smokers. These results support the notion that active smoking induces inflammation, leads to particle deposition in the lungs and may trigger subclinical interstitial lung disease, all influencing QCT. Thus, it may be advisable to consider the effects of smoking status when interpreting QCT data from clinical trials, for example when correlating lung density with clinical biomarkers or genotypes in large observational cohort studies, as the sole consideration of cumulative pack-years may not be sufficient.
Abbreviations
- 15TH:
-
15th percentile of lung density histogram
- ATS:
-
American Thoracic Society
- BMI:
-
Body mass index
- COPACETIC COPD Pathology:
-
Addressing Critical Gaps, Early Treatment and Diagnosis and Innovative Concepts.
- COPD:
-
Chronic obstructive pulmonary disease
- CS:
-
Continuous smokers
- CT:
-
Multidetector computed tomography
- DFG:
-
German Research Council
- ECSC:
-
European Coal and Steal Community
- EI:
-
Emphysema index
- ES:
-
Ex-smokers
- EV:
-
Emphysema volume
- FEV1%:
-
Forced expiratory volume in 1 s in percent predicted
- FEV1/FVC:
-
Tiffenau index
- FEV1:
-
Forced expiratory volume in 1 s
- FVC:
-
Forced vital capacity
- GOLD:
-
Global Initiative for Chronic Obstructive Lung Disease
- HU:
-
Hounsfield units
- LAA%950 :
-
Percentage of low attenuation areas at a threshold of -950 HU
- LUSI:
-
Lung Cancer Screening Intervention Trial
- LV:
-
Lung volume
- MLD:
-
Mean lung density
- PD15:
-
15th percentile of lung density histogram
- PFT:
-
Pulmonary function testing
- QCT:
-
Quantitative CT
- RB-ILD:
-
Respiratory bronchiolitis interstitial lung disease
- RQ:
-
Recent quitters
- YACTA:
-
Yet Another CT Analyzer
References
Coxson HO, Mayo J, Lam S, Santyr G, Parraga G, Sin DD (2009) New and current clinical imaging techniques to study chronic obstructive pulmonary disease. Am J Respir Crit Care Med 180:588–597
Rabe KF, Hurd S, Anzueto A et al (2007) Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med 176:532–555
Lynch DA, Austin JH, Hogg JC et al (2015) CT-definable subtypes of chronic obstructive pulmonary disease: a statement of the Fleischner Society. Radiology. doi:https://doi.org/10.1148/radiol.2015141579:141579
Coxson HO, Rogers RM (2005) Quantitative computed tomography of chronic obstructive pulmonary disease. Acad Radiol 12:1457–1463
Kauczor HU, Wielputz MO, Owsijewitsch M, Ley-Zaporozhan J (2011) Computed tomographic imaging of the airways in COPD and asthma. Journal of thoracic imaging 26:290–300
Ley-Zaporozhan J, van Beek EJ (2010) Imaging phenotypes of chronic obstructive pulmonary disease. Journal of magnetic resonance imaging : JMRI 32:1340–1352
Heussel CP, Herth FJ, Kappes J et al (2009) Fully automatic quantitative assessment of emphysema in computed tomography: comparison with pulmonary function testing and normal values. Eur Radiol 19:2391–2402
Hoffman EA, Simon BA, McLennan G (2006) State of the Art. A structural and functional assessment of the lung via multidetector-row computed tomography: phenotyping chronic obstructive pulmonary disease. Proc Am Thorac Soc 3:519–532
Coxson HO, Dirksen A, Edwards LD et al (2013) The presence and progression of emphysema in COPD as determined by CT scanning and biomarker expression: a prospective analysis from the ECLIPSE study. The Lancet Respiratory medicine 1:129–136
Gevenois PA, De Vuyst P, de Maertelaer V et al (1996) Comparison of computed density and microscopic morphometry in pulmonary emphysema. Am J Respir Crit Care Med 154:187–192
Coxson HO, Rogers RM, Whittall KP et al (1999) A quantification of the lung surface area in emphysema using computed tomography. Am J Respir Crit Care Med 159:851–856
Regan EA, Hokanson JE, Murphy JR et al (2010) Genetic epidemiology of COPD (COPDGene) study design. COPD 7:32–43
Han MK, Kazerooni EA, Lynch DA et al (2011) Chronic obstructive pulmonary disease exacerbations in the COPDGene study: associated radiologic phenotypes. Radiology 261:274–282
Sciurba FC, Ernst A, Herth FJ et al (2010) A randomized study of endobronchial valves for advanced emphysema. N Engl J Med 363:1233–1244
Grydeland TB, Dirksen A, Coxson HO et al (2009) Quantitative computed tomography: emphysema and airway wall thickness by sex, age and smoking. The European respiratory journal 34:858–865
Camiciottoli G, Cavigli E, Grassi L et al (2009) Prevalence and correlates of pulmonary emphysema in smokers and former smokers. A densitometric study of participants in the ITALUNG trial. Eur Radiol 19:58–66
Mohamed Hoesein FA, Zanen P, de Jong PA et al (2013) Rate of progression of CT-quantified emphysema in male current and ex-smokers: a follow-up study. Respiratory research 14:55
Zach JA, Williams A, Jou SS et al (2016) Current Smoking Status Is Associated With Lower Quantitative CT Measures of Emphysema and Gas Trapping. Journal of thoracic imaging 31:29–36
Shaker SB, Stavngaard T, Laursen LC, Stoel BC, Dirksen A (2011) Rapid fall in lung density following smoking cessation in COPD. COPD 8:2–7
Ashraf H, Lo P, Shaker SB et al (2011) Short-term effect of changes in smoking behaviour on emphysema quantification by CT. Thorax 66:55–60
Becker N, Motsch E, Gross ML et al (2012) Randomized study on early detection of lung cancer with MSCT in Germany: study design and results of the first screening round. J Cancer Res Clin Oncol 138:1475–1486
Becker N, Motsch E, Gross ML et al (2015) Randomized Study on Early Detection of Lung Cancer with MSCT in Germany: Results of the First 3 Years of Follow-up After Randomization. Journal of thoracic oncology : official publication of the International Association for the Study of Lung Cancer 10:890–896
Bade M, Bahr V, Brandt U et al (2016) Effect of smoking cessation counseling within a randomised study on early detection of lung cancer in Germany. J Cancer Res Clin Oncol 142:959–968
Miller MR, Hankinson J, Brusasco V et al (2005) Standardisation of spirometry. The European respiratory journal 26:319–338
Quanjer PH, Tammeling GJ, Cotes JE, Pedersen OF, Peslin R, Yernault JC (1993) Lung volumes and forced ventilatory flows. Report Working Party Standardization of Lung Function Tests, European Community for Steel and Coal. Official Statement of the European Respiratory Society. Eur Respir J Suppl 16:5–40
Weinheimer O, Achenbach T, Heussel CP, Düber C (2011) Automatic Lung Segmentation in MDCT Images Fourth International Workshop on Pulmonary Image Analysis 2011, pp 241-255
Wielputz MO, Weinheimer O, Eichinger M et al (2013) Pulmonary emphysema in cystic fibrosis detected by densitometry on chest multidetector computed tomography. PLoS One 8:e73142
Wielputz MO, Bardarova D, Weinheimer O et al (2014) Variation of densitometry on computed tomography in COPD--influence of different software tools. PloS one 9:e112898
Heussel CP, Kappes J, Hantusch R et al (2009) Contrast enhanced CT-scans are not comparable to non-enhanced scans in emphysema quantification. Eur J Radiol 74:473–478
Wielputz MO, Eichinger M, Weinheimer O et al (2013) Automatic airway analysis on multidetector computed tomography in cystic fibrosis: correlation with pulmonary function testing. Journal of thoracic imaging 28:104–113
Lim HJ, Weinheimer O, Wielputz MO et al (2016) Fully Automated Pulmonary Lobar Segmentation: Influence of Different Prototype Software Programs onto Quantitative Evaluation of Chronic Obstructive Lung Disease. PLoS One 11:e0151498
Holm S (1979) A simple sequentially rejective multiple test procedure. Scandinavian Journal of Statistics 6:65–70
Zaporozhan J, Ley S, Weinheimer O et al (2006) Multi-detector CT of the chest: influence of dose onto quantitative evaluation of severe emphysema: a simulation study. Journal of computer assisted tomography 30:460–468
Yuan R, Mayo JR, Hogg JC et al (2007) The effects of radiation dose and CT manufacturer on measurements of lung densitometry. Chest 132:617–623
Gierada DS, Bierhals AJ, Choong CK et al (2010) Effects of CT section thickness and reconstruction kernel on emphysema quantification relationship to the magnitude of the CT emphysema index. Acad Radiol 17:146–156
Smith BM, Barr RG (2013) Establishing normal reference values in quantitative computed tomography of emphysema. Journal of thoracic imaging 28:280–283
Gietema HA, Schilham AM, van Ginneken B, van Klaveren RJ, Lammers JW, Prokop M (2007) Monitoring of smoking-induced emphysema with CT in a lung cancer screening setting: detection of real increase in extent of emphysema. Radiology 244:890–897
Soejima K, Yamaguchi K, Kohda E et al (2000) Longitudinal follow-up study of smoking-induced lung density changes by high-resolution computed tomography. Am J Respir Crit Care Med 161:1264–1273
Kauczor HU, Heussel CP, Herth FJ (2013) Longitudinal quantitative low-dose CT in COPD: ready for use? The Lancet Respiratory medicine 1:95–96
Sieren JP, Newell JD, Judy PF et al (2012) Reference standard and statistical model for intersite and temporal comparisons of CT attenuation in a multicenter quantitative lung study. Medical physics 39:5757–5767
Stoel BC, Putter H, Bakker ME et al (2008) Volume correction in computed tomography densitometry for follow-up studies on pulmonary emphysema. Proc Am Thorac Soc 5:919–924
Madani A, Van Muylem A, Gevenois PA (2010) Pulmonary emphysema: effect of lung volume on objective quantification at thin-section CT. Radiology 257:260–268
Acknowledgements
We thank all participants for their willingness to contribute to this study. The expert technical assistance of Julia Schliebus and Martina Jochim are gratefully appreciated. This work contains parts of the doctoral thesis of Mila Trauth, Heidelberg, Germany.
Funding
This study has received funding by the Dietmar Hopp Foundation (2007–2010) and the German Research Association (DFG) (2007–2013). The COPACETIC study was funded by EU FP7 grant 201379.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Mark O. Wielpütz.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
Ethical approval
Institutional Review Board approval was obtained (approved by the Ethics Committee of the Medical Faculty of the University of Heidelberg (073/2001) and the federal radiation protection authority (22462/2 2006-045)).
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Study subjects or cohorts overlap
The study subjects have been previously reported in J Thorac Oncol. 2015 Jun;10(6):890-6, Am J Respir Crit Care Med. 2015 Mar 1;191(5):547-56, J Cancer Res Clin Oncol. 2012 Sep;138(9):1475-86, and Eur J Radiol. 2014 Mar;83(3):600-5, investigating data or aspects that were significantly different from the present study.
Methodology
• prospective
• randomised controlled trial
• observational
• performed at one institution
Rights and permissions
About this article
Cite this article
Jobst, B.J., Weinheimer, O., Trauth, M. et al. Effect of smoking cessation on quantitative computed tomography in smokers at risk in a lung cancer screening population. Eur Radiol 28, 807–815 (2018). https://doi.org/10.1007/s00330-017-5030-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-5030-6