Abstract
The aim of this study was to investigate the presence of subclinical atherosclerosis among psoriatic arthritis (PsA) patients without any cardiovascular disease (CVD) or traditional cardiovascular risk factors through measurement of endothelial function and carotid intima-media thickness (IMT) and correlated with disease-related risk factors. Twenty patients with PsA according to classification criteria for psoriatic arthritis and 20 age- and sex-matched controls were included. Patients with risk factors for cardiovascular disease were excluded. Carotid IMT was measured using two-dimensional carotid ultrasonography (USG). Endothelial function was determined by measuring flow-mediated endothelial-dependent vasodilatation (FMD %) and nitrate-induced dilatation (NID %) using brachial artery USG. Additionally, serum asymmetric dimethylarginine (ADMA) level was obtained using ELISA methodology. In this cross-sectional study, FMD % was significantly more decreased among PsA patients versus control group [mean 11 % (median (range) %10.5 (8–15 %)] and mean 13.2 % [median (range) 12, 8 % (8.1–17.6 %), respectively; p = 0.01]. There was no significant difference in NID %, ADMA level and mean IMT or maximum IMT results. FMD % did not show a significant correlation with clinical and laboratory data of PsA patients. This study showed that endothelial dysfunction may be present in PsA patients with no CVD and traditional cardiovascular risk factors. The study findings lend support to the previous reports that suggested a potential relationship between PsA and atherosclerotic disorders.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Psoriatic arthritis (PsA) is a type of inflammatory arthritis which develops in people who have psoriasis [1]. Atherosclerotic vascular diseases (AVD) are a leading cause of morbidity and mortality in PsA patients [2, 3]. However, the exact mechanism of increased early atherosclerosis as observed in PsA patients is still unclear. Chronic inflammation involved in the early stage of atherosclerosis has been implicated in the development of AVD via a synergistic effect with vascular disease risk factors in rheumatic diseases and primarily rheumatoid arthritis (RA) [4]. Inflammation may accelerate atherosclerosis both directly and indirectly by causing endothelial dysfunction through insulin resistance, dyslipidemia and lipid oxidation [4]. Additionally, PsA patients were shown to have a greater prevalence of obesity, hypertension, dyslipidemia and insulin resistance compared with the general population [5, 6]. It was also reported that PsA patients have a more severe course of subclinical atherosclerosis due to increased burden of inflammation compared to patients who have psoriasis alone [6].
Asymmetric dimethylarginine (ADMA) is an endogeneous inhibitor of nitric oxide synthase, and thus, a risk factor for endothelial dysfunction associated with increased atherosclerosis [7, 8]. ADMA was shown to predict cardiovascular risk and to be elevated in the setting of atherosclerotic vascular diseases [9]. Increased ADMA level was found in RA patients and patients with active PsA in the early stage of the disease. Also, ADMA level was reported to be a marker for endothelial dysfunction and subclinical atherosclerosis [10].
Endothelial dysfunction is a hallmark for several disorders associated with atherosclerosis and considered to be an early finding of atherogenesis [11, 12]. Endothelium modulates vascular tone by secreting a number of vasoactive hormones including nitric oxide as a response to mechanical stress [11]. Endothelial dysfunction is defined as the impaired ability of the arteries to dilate as a result of increased breakdown or decreased secretion of nitric oxide in response to physical or chemical stimuli [11, 13]. Endothelial function may be assessed noninvasively using sensitive brachial artery USG through measurement of postocclusion flow-mediated endothelial-dependent vasodilatation (FMD) of the brachial artery [12].
Carotid intima-media thickness (IMT) that is another marker of AVD may be used to predict a future vascular event [14]. For an absolute carotid IMT difference of 0.1 mm, the future risk of MI increases by 10–15 %, and the stroke risk increases by 13–18 %. Impaired endothelial dysfunction [15] and increased carotid IMT [16] were reported in PsA patients without any traditional cardiovascular risk factors.
In the current study, we aimed to investigate the presence of subclinical atherosclerosis in a cohort of psoriatic arthritis patients without clinical CVD or traditional cardiovascular risk factors who met the criteria for minimal disease activity (MDA) [17].
Materials and methods
Study population
Twenty patients who admitted to the Rheumatology outpatient clinic of Kocaeli University Hospital between November 2011 and April 2012 and met classification criteria for PsA as defined by CASPAR study group [18] were enrolled cross sectionally. Patients were receiving treatment on a regular basis for at least 1 year and met the MDA criteria at the time of presentation [17]. Patients with documented hypertension (systolic blood pressure >140 mmHg and/or diastolic blood pressure >90 mmHg), preexisting dyslipidemia (fasting plasma total cholesterol and/or triglyceride >240 and/or >160 mg/dl), diabetes mellitus (history of diabetes, on a diabetic diet, receiving oral hypoglycemic medication or insulin, fasting blood glucose >126 mg/dl), chronic renal disease (creatinine >1.3 mg/dl), established obesity (body mass index >30 kg/m2) or a history of cardiovascular and/or cerebrovascular disease were excluded. Current non-smokers or patients who have ceased smoking at least 5 years prior to the study date were allowed to participate. Twenty randomly selected subjects matched for age, sex and body mass index were included as controls. Control group did not have known history of hypertension, diabetes mellitus, dyslipidemia, renal failure, obesity, smoking or cardiovascular and/or cerebrovascular disease. Approval for the study was obtained from the Ethics Committee of Kocaeli University Medical Faculty. The study was conducted in accordance with the principles set forth in the World Medical Association’s Declaration of Helsinki. Written informed consent was obtained from all patients and healthy controls.
Clinical assessment of PsA
Patient and physician global assessments were rated on a 100-mm visual analog scale. During joint examination, a tender joint count using 66 joints and a swollen joint count using 66 joints (66/66-joint counts) were assessed and recorded in the protocol form along with the number of painful entheseal sites. Joint activity was assessed using the Disease Activity Score in 28 joints (DAS 28) in patients with peripheral joint involvement [19]. Psoriasis Area and Severity Index (PASI) was used to evaluate disease activity of skin psoriasis and recorded in the protocol form [20]. Results of the Health Assessment Questionnaire (HAQ) which were previously completed by the patients were also recorded in the form [21].
Carotid IMT and plaques
Carotid IMT was measured from both the right and the left common carotid arteries. The measurements were taken from the far wall 10 mm proximal to the carotid bifurcation with 2D examination by a semi-automated, quick, safe and reproducible computer program (M’ATH©). The presence of plaques in the common carotid artery, bifurcation and internal carotid artery was evaluated in 2D views. Plaque was defined as a focal structure encroaching into the arterial lumen of at least 0.5 mm or 50 % of the surrounding IMT or demonstrates a thickness of greater than or equal to 1.5 mm from the media adventitia interface to the intima lumen interface. A carotid IMT ≥0.9 mm and/or the presence of atheromatous plaques was defined as subclinical atherosclerosis [22]. Reproducibility of IMT and detection of plaque have been well documented [23–25].
Brachial artery reactivity
Measurements of endothelial function were taken in a temperature-controlled vascular research laboratory according to Celermajer method [26]. Patients were examined in the morning after having abstained from alcohol, caffeine and food for nearly 8 h and having a 15-min rest on the examination bed. Arterial diameter was measured from high-resolution, two-dimensional ultrasound images obtained using a Vivid 7 ultrasound machine (GE Medical Systems, Horten, Norway) with a 7.5-MHz linear-array transducer. The right brachial artery was scanned over a longitudinal section 3–5 cm above the right elbow. The transmit (focus) zone was set to the depth of the anterior vessel wall, and depth and gain settings were optimized to identify the lumen-to-vessel wall interface. When a reasonable image was obtained, a mark was made on the skin for repeat examination; the arm was kept in the same position throughout the study. Changes in diameter of the right brachial artery were measured at rest, during reactive hyperemia, again at rest and after sublingual nitroglycerin administration. A pneumatic tourniquet placed around the forearm distal to the target artery was inflated to a pressure of 250-mm Hg, and the pressure held for 5 min. After sudden cuff deflation, a second ultrasound scan was performed. Measurements were taken at 30, 60 and 90 s, and their average was calculated. An additional resting scan was recorded 15 min later to confirm the vessel recovery. Sublingual nitroglycerin spray (0.4 mg) was then administered, and the last scans were obtained 3 and 4 min later. Images were stored in digital format to enable off-line analysis. The diameter of the brachial artery was measured from the anterior to the posterior interface between the media and adventitia at a fixed distance. The mean diameter was calculated from four cardiac cycles synchronized with the R-wave peaks on the ECG. The diameter change caused by FMD was expressed as the percentage change relative to that at the initial resting scan (FMD %). The diameter change caused by nitroglycerin administration was also expressed as the percentage change relative to that at the recovery scan (NID %).
Intra-observer variability
Doppler and brachial artery measurements were taken by an experienced cardiologist (T.S.) who was blinded to previously obtained data. In our laboratory, the intra-observer variability was as follows: r = 0.97 for carotid USG measurements and r = 0.98 for brachial artery USG measurements.
Laboratory studies
C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) were obtained from venous blood samples of patients. CRP level was measured using Abbott Architect c16000 device by immunoturbidimetric method and ESH using Alifax test1 device by laser method. Lipid measurements for LDL cholesterol, triglycerides, HDL cholesterol, total cholesterol were taken from approximately 10-h fasting serum samples. Of these parameters, total cholesterol was measured by enzymatic method and triglycerides by glycerol phosphate oxidase method using Abbott Architect c16000 device. LDL was calculated using Friedewald’s equation. For serum ADMA measurement, sera were extracted from blood samples obtained from patient and control groups by centrifugation at 3,500 rpm for 10 min and stored at −80 °C until the time of analysis. Plasma ADMA measurement was taken at the Department of Clinical Biochemistry of Kocaeli University Medical Faculty using Dynex-DSX device (Immundiagnostik AG, Germany) by microELISA method with an immunodiagnostic kit.
Statistical analysis
Statistical analyses of the present study were performed using SPSS for Windows 20.0 (SPSS, Chicago, IL, USA) software package. Results of descriptive analyses were presented as mean ± SD if normal distribution was present and if not, as median [interquartile range (IQR)]. For pair-wise comparisons, independent t test was used for normally distributed numerical variables and Chi-square test or Fisher’s exact test as appropriate was used for categorical variables. Non-normally distributed numerical data were analyzed using Mann–Whitney U test. Pearson’s correlation test was used to investigate the associations between measured variables. All tests were two-sided, and a p value below 0.05 was considered statistically significant.
Results
Clinical findings and treatment of PsA patients
The two groups did not differ in demographic characteristics including age, gender and body mass index. Table 1 lists the characteristics of 20 patients enrolled in the study. The mean age of patients was 41 years, and 75 % were females. None of the patients had clinical CVD or traditional cardiovascular risk factors. During the study, 17 patients (85 %) used disease-modifying antirheumatic drugs (DMARDs). Seventeen patients (85 %) were on methotrexate, and seven (35 %) were using anti-tumor necrosis factor (anti-TNF) alpha therapy. Four (20 %) patients were receiving combination therapy with methotrexate and anti-TNF alpha, and 19 (95 %) patients were on ≤5 mg/day prednisolone.
Brachial artery reactivity and ADMA level
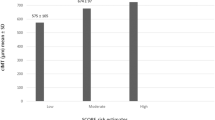
In this cross-sectional study, there was a difference between PsA patients and control group in FMD % which was found to be significantly decreased in PsA patients versus control group [mean %11; median (range) 10.5 % (8–15 %) and mean 13.2 %; median (range) %12.8 (8.1–17.6 %), respectively; p = 0.01] (Fig. 1) (Table 2). The two groups did not differ in NID % [mean 14.2 % median (range) 14.3 % (9.5–21.4 %) and mean 14.3 %; median (range) 13.9 % (9.6–19 %), respectively; p = 0.892] (Table 2) and baseline arterial diameter [mean 3.5 mm; median (range) 3.5 (2.6–4.9 mm) and mean 3.7 mm; median (range) 3.5 (2.9–4.8 mm), respectively; p = 0.158] (Table 2). The FMD % results were not significantly different in males and females. Clinical and laboratory data obtained during the study did not show a significant correlation with FMD % results (Table 3). PsA patients and control groups did not differ in ADMA level either [mean ± SD, 0.538 ± 0.145 and 0.629 ± 0.192, respectively; p > 0.05] (Table 4).
Carotid IMT measurement in PsA patients and control group
When we looked at the mean IMT (mean ± SD, 0.611 ± 0.08 and 0.596 ± 0.08, respectively; p > 0.05) (Table 4) and maximum IMT results, no significant difference was found between PsA patients and control group in these parameters (mean ± SD, 0.753 ± 0.113 and 0.743 ± 0.09, respectively; p > 0.05) (Table 4).
Presence of subclinical atherosclerosis
While atherosclerotic plaques were detected in two (10 %) patients in the PsA group, none of the control subjects had plaques (Table 4).
Discussion
The most prominent changes that occur during the subclinical stage of atherosclerotic disease include increased IMT and endothelial dysfunction of the entire arterial bed [27]. In the present study, endothelial function was demonstrated to be impaired in the PsA patient cohort with no clinical CVD and traditional cardiovascular risk factors.
As in RA, PsA and psoriasis are associated with increased cardiovascular mortality rate [2, 28, 29]. In the literature, impaired endothelial function and increased prevalence of subclinical atherosclerosis have been reported in PsA [30, 31]. In chronic inflammatory disorders such as RA and PsA, markers of inflammation frequently show variations over time, mainly in correlation with therapy. Limited information is available on the effect of control of disease activity by treatment on endothelial dysfunction in PsA patients. Anti-TNF alpha therapy was reported to provide some improvement of endothelial dysfunction in ankylosing spondylitis patients [32]. On the other hand, in studies with RA patients, endothelial dysfunction was reported to ameliorate with anti-TNF alpha therapy, but the effect was transient [33, 34]. Contrastingly, a separate study reported no change in the impaired FMD % response with therapy in RA patients [35]. In the present study, impaired FMD % was demonstrated in PsA patient cohort with MDA who were receiving therapy. Therapy may have affected the change in FMD %. We believe that the difference might be more apparent in PsA patients with increased disease activity who receive inadequate therapy.
No significant difference was found between the two groups in ADMA level. ADMA is an endogenous inhibitor of nitric oxide synthase [36] and also an indicator of endothelial dysfunction; elevations in serum ADMA level have been identified in several disorders associated with increased cardiovascular risk including RA and PsA [7, 37, 38]. Additionally, decreased serum ADMA levels were observed following effective RA treatment with both standard DMARDs and biological agents [37, 38]. This may explain the absence of difference between two groups in ADMA level in the present study.
Several studies have shown increased carotid IMT in PsA patients as an evidence for subclinical atherosclerosis [3, 5, 16, 31, 39]. However, a prospective, observational study conducted by Tam et al. showed that the progression of the atherosclerotic process could be slowed with use of 12 weeks of anti-TNF alpha blocker therapy in PsA patients. The same authors reported that early atherosclerotic lesions may be treated with effective control of inflammation due to regression of maximum IMT over long-term follow-up [40]. In the present study, carotid IMT was greater in the patient group, but the difference versus control group was not statistically significant. This may have resulted from the small sample size. It may be considered that endothelial dysfunction that is an early functional indicator of atherosclerosis and measured by FMD might be a more sensitive marker than IMT which is an early morphological indicator of atherosclerosis.
Currently, inflammation is believed to play an important role in increasing the CVD risk in rheumatic diseases, primarily RA [41]. Inflammation causes endothelial cell activation characterized by the loss of vascular integrity, increased release of leukocyte adhesion molecules, development of hypercoagulable state, cytokine release and upregulation of major histocompatibility complex HLA class II molecules [4]. Chronic inflammation may lead to oxidation, dyslipidemia and insulin resistance, all of which contribute to the development of endothelial dysfunction [4]. Recently, the outstanding role of chronic inflammation in the pathogenesis of accelerated atherogenesis was reported in a number of studies [4, 42, 43]. In one study, endothelial dysfunction in association with impaired FMD was shown in young RA patients with low disease activity. The same study also showed a strong association of chronic inflammation with endothelial dysfunction expressed as mean CRP levels [44]. In the current study, endothelial dysfunction already existing in our PsA patients with low disease activity may be associated with sustained subclinical chronic inflammation.
Limitations of the study include small size of the study population and cross-sectional, observational design which resulted in inadequate power to explore potential causality. Additionally, a single measurement was taken for parameters including inflammatory markers, disease activity and ultrasonographic variables. These data may not accurately reflect changes in the variables over time and the effect of cumulative burden associated with exposure time.
Another potential confounding factor was the use of steroids which is a known risk factor for atherosclerosis and 95 % of patients received steroids, even if at a low dose (≤5 mg/day prednisolone). However, no relationship was reported between cumulative prednisolone dose and endothelial dysfunction in series of PsA patients [15]. Similarly, an association was not observed between a higher cumulative prednisolone dose (mean 15.7 grams) and endothelial dysfunction in a series of patients with long-standing RA with no clinical CVD [45]. A third confounding factor was the inability to exclude the potential effect of hormonal changes on the study results since the female patients were predominant in the PsA group.
In conclusion, there is an increased occurrence of early atherosclerosis in PsA irrespective of CVD risk factors and its underlying mechanism is still unclear. Further prospective studies with larger number of patients are needed to fully elucidate the mechanism of accelerated atherosclerosis in PsA and help identify strategies to protect this group of patients against development of clinical CVD with appropriate therapy similar to those established for RA.
References
Gladman DD (2004) Psoriatic arthritis. In: Maddison PJ, Isenberg DA, Woo P, Glass DN, Breedveld F (eds) Oxford textbook of rheumatology, 3rd edn. Oxford University Press, Oxford, pp 766–778
Wong D, Gladman DD, Husted J, Long JA, Farewell VT (1997) Mortality studies in psoriatic arthritis: results from a single outpatient clinic. I. Causes and risk of death. Arthritis Rheum 40:1868–1872
Di Minno MN, Iervolino S, Peluso R, Scarpa R, Di Minno G (2011) Carotid intima-media thickness in psoriatic arthritis: differences between tumor necrosis factor-α blockers and traditional disease-modifying antirheumatic drugs. Arterioscler Thromb Vasc Biol 31(3):705–712 CaRRDs study group
Sattar N, McCarey DW, Capell H, McInnes IB (2003) Explaining how “high-grade” systemic inflammation accelerates vascular risk in rheumatoid arthritis. Circulation 108(24):2957–2963
Tam LS, Shang Q, Li EK, Tomlinson B, Chu TT, Li M, Leung YY, Kwok LW, Wong KC, Li TK, Yu T, Zhu TY, Kun EW, Yip GW, Yu CM (2008) Subclinical carotid atherosclerosis in patients with psoriatic arthritis. Arthritis Rheum 59(9):1322–1331
Eder L, Jayakar J, Shanmugarajah S, Thavaneswaran A, Pereira D, Chandran V, Rosen CF, Gladman DD (2013) The burden of carotid artery plaques is higher in patients with psoriatic arthritis compared with those with psoriasis alone. Ann Rheum Dis 72(5):715–720
Turiel M, Atzeni F, Tomasoni L, de Portu S, Delfino L, Bodini BD, Longhi M, Sitia S, Bianchi M, Ferrario P, Doria A, De Gennaro CV, Sarzi-Puttini P (2009) Non-invasive assessment of coronary flow reserve and ADMA levels: a case-control study of early rheumatoid arthritis patients. Rheumatology (Oxford) 48(7):834–839
Miyazaki H, Matsuoka H, Cooke JP, Usui M, Ueda S, Okuda S, Imaizumi T (1999) Endogenous nitric oxide synthase inhibitor: a novel marker of atherosclerosis. Circulation 99(9):1141–1146
Vallance P (2001) Importance of asymmetrical dimethylarginine in cardiovascular risk. Lancet 358(9299):2096–2097
Atzeni F, Sarzi-Puttini P, Sitia S, Tomasoni L, Gianturco L, Battellino M, Boccassini L, De Gennaro CV, Marchesoni A, Turiel M (2011) Coronary flow reserve and asymmetric dimethylarginine levels: new measurements for identifying subclinical atherosclerosis in patients with psoriatic arthritis. J Rheumatol 38(8):1661–1664
Brunner H, Cockcroft JR, Deanfield J, Donald A, Ferrannini E, Halcox J et al (2005) Working Group on Endothelins and Endothelial Factors of the European Society of Hypertension. Endothelial function and dysfunction. Part II: association with cardiovascular risk factors and diseases. A statement by the Working Group on Endothelins and Endothelial Factors of the European Society of Hypertension. J Hypertens 23(2):233–246
Deanfield J, Donald A, Ferri C, Giannattasio C, Halcox J, Halligan S et al (2005) Working Group on Endothelin and Endothelial Factors of the European Society of Hypertension. Endothelial function and dysfunction. Part I: methodological issues for assessment in the different vascular beds: a statement by the Working Group on Endothelin and Endothelial Factors of the European Society of Hypertension. J Hypertens 23(1):7–17
Nadar S, Blann AD, Lip GY (2004) Endothelial dysfunction: methods of assessment and application to hypertension. Curr Pharm Des 10(29):3591–3605
Lorenz MW, Markus HS, Bots ML, Rosvall M, Sitzer M (2007) Prediction of clinical cardiovascular events with carotid intima-media thickness: a systematic review and meta-analysis. Circulation 115(4):459–467
Gonzalez-Juanatey C, Llorca J, Miranda-Filloy JA, Amigo-Diaz E, Testa A, Garcia-Porrua C, Martin J, Gonzalez-Gay MA (2007) Endothelial dysfunction in psoriatic arthritis patients without clinically evident cardiovascular disease or classic atherosclerosis risk factors. Arthritis Rheum 57(2):287–293
Gonzalez-Juanatey C, Llorca J, Amigo-Diaz E, Dierssen T, Martin J, Gonzalez-Gay MA (2007) High prevalence of subclinical atherosclerosis in psoriatic arthritis patients without clinically evident cardiovascular disease or classic atherosclerosis risk factors. Arthritis Rheum 57:1074–1080
Coates LC, Fransen J, Helliwell PS (2010) Defining minimal disease activity in psoriatic arthritis: a proposed objective target for treatment. Ann Rheum Dis 69(1):48–53
Taylor W, Gladman D, Helliwell P, Marchesoni A, Mease P, Mielants H (2006) Classification criteria for psoriatic arthritis: development of new criteria from a large international study. Arthritis Rheum 54(8):2665–2673 CASPAR study group
van der Heijde DM, van’t Hof MA, van Riel PL, van Leeuwen MA, van Rijswijk MH, van de Putte LB (1992) Validity of single variables and composite indices for measuring disease activity in rheumatoid arthritis. Ann Rheum Dis 51(2):177–181
Berth-Jones J, Grotzinger K, Rainville C, Pham B, Huang J, Daly S, Herdman M, Firth P, Hotchkiss K (2006) A study examining inter- and intrarater reliability of three scales for measuring severity of psoriasis: psoriasis area and severity index, Physician’s Global Assessment and Lattice System Physician’s Global Assessment. Br J Dermatol 155:707–713
Fries JF, Spitz P, Kraines RG, Holman HR (1980) Measurement of patient outcome in arthritis. Arthritis Rheum 23(2):137–145
Committee Guidelines (2003) European society of Hypertension-European society of cardiology guidelines for the management of arterial hypertension. J Hypertens 21:1011–1013
Persson J, Stavenow L, Wikstrand J, Israelsson B, Formgren J, Berglund G (1992) Noninvasive quantification of atherosclerotic lesions. Reproducibility of ultrasonographic measurement of arterial wall thickness and plaque size. Arterioscler Thromb 12(2):261–266
Roman MJ, Saba PS, Pini R, Spitzer M, Pickering TG, Rosen S, Alderman MH, Devereux RB (1992) Parallel cardiac and vascular adaptation in hypertension. Circulation 86(6):1909–1918
Joakimsen O, Bønaa KH, Stensland-Bugge E (1997) Reproducibility of ultrasound assessment of carotid plaque occurrence, thickness, and morphology the Tromsø study. Stroke 28(11):2201–2207
Celermajer DS, Sorensen KE, Gooch VM et al (1992) Non-invasive detection of endothelial dysfunction in children and adults at risk of atherosclerosis. Lancet 340:1111–1115
Glagov S, Weisenberg E, Zarins CK, Stankunavicius R, Kolettis GJ (1987) Compensatory enlargement of human atherosclerotic coronary arteries. N Engl J Med 316:1371–1375
Gladman DD, Farewell VT, Wong K, Husted J (1998) Mortality studies in psoriatic arthritis: results from a single outpatient center. II. Prognostic indicators for death. Arthritis Rheum 41(6):1103–1110
Mallbris L, Akre O, Granath F, Yin L, Lindelöf B, Ekbom A, Ståhle-Bäckdahl M (2004) Increased risk for cardiovascular mortality in psoriasis inpatients but not in outpatients. Eur J Epidemiol 19(3):225–230
Tam LS, Tomlinson B, Chu TT, Li M, Leung YY et al (2008) Cardiovascular risk profile of patients with psoriatic arthritis compared to controls–the role of inflammation. Rheumatology (Oxford) 47(5):718–723
Kimhi O, Caspi D, Bornstein NM, Maharshak N, Gur A, Arbel Y, Comaneshter D, Paran D, Wigler I, Levartovsky D, Berliner S, Elkayam O (2007) Prevalence and risk factors of atherosclerosis in patients with psoriatic arthritis. Semin Arthritis Rheum 36(4):203–209
Syngle A, Vohra K, Sharma A, Kaur L (2010) Endothelial dysfunction in ankylosing spondylitis improves after tumor necrosis factor-alpha blockade. Clin Rheumatol 29(7):763–770
Gonzalez-Juanatey C, Testa A, Garcia-Castelo A, Garcia-Porrua C, Llorca J, Gonzalez-Gay MA (2004) Active but transient improvement of endothelial function in rheumatoid arthritis patients undergoing long-term treatment with anti-tumor necrosis factor alpha antibody. Arthritis Rheum 51(3):447–450
Bosello S, Santoliquido A, Zoli A, Di Campli C, Flore R, Tondi P, Ferraccioli G (2007) TNF-alpha blockade induces a reversible but transient effect on endothelial dysfunction in patients with long-standing severe rheumatoid arthritis. Clin Rheumatol 27(7):833–839
Spinelli FR, Metere A, Barbati C, Pierdominici M, Iannuccelli C et al (2013) Effect of therapeutic inhibition of TNF on circulating endothelial progenitor cells in patients with rheumatoid arthritis. Mediators Inflamm. doi:10.1155/2013/537539
Vallance P, Leone A, Calver A, Collier J (1992) Accumulation of an endogenous inhibitor of nitric oxide synthesis in chronic renal failure. Lancet 339(8793):572–575
Surdacki A, Martens-Lobenhoffer J, Wloch A et al (2007) Elevated plasma asymmetric dimethyl-l-arginine levels are linked to endothelial progenitor cell depletion and carotid atherosclerosis in rheumatoid arthritis. Arth Rheum 56(3):809–819
Di Franco M, Spinelli FR, Metere A et al (2012) Serum levels of asymmetric dimethylarginine and apelin as potential markers of vascular endothelial dysfunction in early rheumatoid arthritis. Mediators Inflamm. doi:10.1155/2012/347268
Eder L, Zisman D, Barzilai M, Laor A, Rahat M, Rozenbaum M, Bitterman H, Feld J, Rimar D, Rosner I (2008) Subclinical atherosclerosis in psoriatic arthritis: a case-control study. J Rheumatol 35(5):877–882
Tam LS, Li EK, Shang Q, Tomlinson B, Li M, Leung YY, Kuan WP, Kwok LW, Li TK, Zhu Y, Kun EW, Yip G, Yu CM (2011) Tumour necrosis factor alpha blockade is associated with sustained regression of carotid intima-media thickness for patients with active psoriatic arthritis: a 2-year pilot study. Ann Rheum Dis 70(4):705–706
Van Doornum S, McColl G, Wicks IP (2002) Accelerated atherosclerosis: an extraarticular feature of rheumatoid arthritis? Arthritis Rheum 46(4):862–873
Gonzalez-Gay MA, Gonzalez-Juanatey C, Martin J (2005) Rheumatoid arthritis: a disease associated with accelerated atherogenesis. Semin Arthritis Rheum 35(1):8–17
Gonzalez-Gay MA, Gonzalez-Juanatey C, Lopez-Diaz MJ, Piñeiro A, Garcia-Porrua C et al (2007) HLA-DRB1 and persistent chronic inflammation contribute to cardiovascular events and cardiovascular mortality in patients with rheumatoid arthritis. Arthritis Rheum. 57(1):125–132
Vaudo G, Marchesi S, Gerli R, Allegrucci R, Giordano A, Siepi D et al (2004) Endothelial dysfunction in young patients with rheumatoid arthritis and low disease activity. Ann Rheum Dis 63(1):31–35
Gonzalez-Juanatey C, Testa A, Garcia-Castelo A, Garcia-Porrua C, Llorca J et al (2003) HLA-DRB1 status affects endothelial function in treated patients with rheumatoid arthritis. Am J Med 114(8):647–652
Conflict of interest
The authors declare no conflict of interest. This study was funded by the Kocaeli University Coordinating Unit for Scientific Research Projects.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yilmazer, B., Sahin, T., Unlu, B.Ö. et al. Investigation of subclinical atherosclerosis in psoriatic arthritis patients with minimal disease activity. Rheumatol Int 35, 1385–1392 (2015). https://doi.org/10.1007/s00296-015-3228-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-015-3228-y