Abstract
Chronic inflammation in ankylosing spondylitis (AS) is associated with vascular endothelial dysfunction. Infliximab improves inflammatory disease activity in AS patients, but its effect on endothelial dysfunction has still not been tested in these patients. Twelve anti-TNF naive AS patients (mean age, 32.6 ± 3.94 years; disease duration, 5.6 ± 0.8 years) with high disease activity [Bath ankylosing spondylitis disease activity index (BASDAI score > 4)] despite treatment with stable doses of conventional disease modifying anti-rheumatic drugs (DMARDs) were investigated. Inflammatory disease activity [BASDAI and Bath ankylosing spondylitis functional index (BASFI) scores, erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP) levels], serum nitrite concentration, and endothelium-dependent and endothelium-independent vasodilation of the brachial artery were measured at baseline and 12 weeks of therapy after single intravenous infusion of infliximab (5 mg/kg). Previous DMARD(s) regimen remained unchanged throughout the study period. After treatment with infliximab, flow-mediated vasodilation improved from 9.81 ± 1.70% to 26.93 ± 2.34% (p < 0.001), whereas there was no significant change in endothelium-independent vasodilation with nitroglycerin and baseline diameter (13.65 ± 2.10% versus 14.59 ± 1.93%, p = 0.08, and 4.45 ± 0.15 versus 4.46 ± 0.15 mm, p = 0.3, respectively). Nitrite concentration reduced from 6.50 ± 0.21 to 2.57 ± 0.18 µmol/l (p < 0.001), ESR from 40.90 ± 6.00 to 11.50 ± 1.38 mm in the first hour (p < 0.001), and CRP level from 29.08 ± 4.11 to 2.69 ± 0.43 mg/dl (p < 0.001). BASDAI and BASFI scores were significantly reduced from 5.40 ± 1.14 to 1.40 ± 0.70 (p < 0.05) and 5.05 ± 1.76 to 0.20 ± 0.63 (p < 0.05), respectively. The study suggests that in ankylosing spondylitis, endothelial dysfunction is a part of the disease process and infliximab improves both endothelial dysfunction and inflammatory disease activity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Ankylosing spondylitis (AS) is a chronic inflammatory rheumatic disease of the sacroiliac joints and spine with a worldwide prevalence ranging up to 0.9% [1, 2]. AS patients have an approximately twofold increase in mortality compared to the general population, which is predominantly contributed by an increased cardiovascular risk [3–5]. Accelerated atherosclerosis probably contributes to the increased cardiovascular mortality in AS [6, 7]. In fact, AS may represent a coronary heart disease risk factor [8]. Accelerated atherosclerotic disease may be due to traditional risk factors (smoking, hypertension, impaired lipid status, and higher BMI), but the inflammation in active AS also contributes to atherosclerosis as in rheumatoid arthritis (RA) and systemic lupus erythematosus [9, 10].
Chronic inflammation in active AS also contributes to endothelial dysfunction [2, 11] and microvascular dysfunction [8]. Endothelial dysfunction is an essential marker and a good predictor of atherosclerosis, thereby acting as a “barometer” of cardiovascular health [12]. Acute inflammation may interfere with endothelial function through the action of pro-inflammatory cytokines which can modify the vascular release of both nitric oxide and endothelium-derived hyperpolarizing factor [13]. The hallmark of endothelial dysfunction is impaired nitric oxide-mediated endothelial-dependent vasodilatation, which may be due to either decreased production or activity of nitric oxide [14]. Tumor necrosis factor alpha (TNF-α) plays a key role in the pathogenesis of AS initiation and amplification of the inflammatory cascade in atherogenesis [15]. TNF-α blocks the activation of endothelial nitric oxide synthase (eNOS) by interfering with the phosphorylation and directly degrading eNOS messenger RNA (mRNA), thus inhibiting nitric oxide release which is essential for flow-dependent relaxation of blood vessels [16].
The most noteworthy development in the treatment of AS in the last decade has been the emergence of biologic therapies, specifically the anti-TNF-α agents [17, 18]. Chronic anti-inflammatory treatment with infliximab has shown improvement in inflammatory disease activity in AS patients [19] and in endothelial function as assessed by the flow-mediated dilatation (FMD) of the brachial artery in patients with RA [16, 20], systemic vasculitis [21], and Crohn’s disease [22]. However, the effect of infliximab on endothelial dysfunction in AS has not been studied to date. In this study, we tested the hypotheses that chronic inflammation in AS is associated with endothelial dysfunction despite treatment with conventional disease modifying anti-rheumatic drugs (DMARDs), which can be reversed with intravenous (i.v.) infusion of infliximab. We studied the effect of infliximab intravenous infusion on endothelial dysfunction in anti-TNF-naïve AS patients lacking adequate response to conventional DMARDs therapy in the context of systemic inflammation.
Materials and methods
Patients
A prospective, uncontrolled, open-label, pilot study was designed. Twelve anti-TNF-naive AS patients [ten men, two women; mean age, 32.6 ± 3.9 years (range, 18–57 years); mean disease duration, 5.6 ± 0.8 years] meeting the modified New York criteria, for AS, with high disease activity despite treatment with single or combination of conventional DMARDs were enrolled in the study [23]. All the patients included have active AS, defined by the presence of Bath ankylosing spondylitis disease activity index (BASDAI) score ≥ 4, spinal pain of 4 or greater on a 10-cm visual analogue scale, and morning stiffness that lasted for at least 30 min [24]. Only patients on stable dose of DMARDs for at least 6 months were included in the study. These patients were administered infliximab 5 mg/kg as a single i.v. infusion.
All patients were on a combination of non-selective NSAIDs (mostly on diclofenac or indomethacin) and sulfasalazine (1–3 g/day), and none was on steroids. Exclusion criteria were: age ≤18 years, history of acute inflammatory joint disease other than AS, prior use of TNF antagonists, active tuberculosis, unstable ischemic heart disease, pregnant and lactating women, concomitant diseases (diabetes mellitus, hypertension, and hyperlipidemia) and concomitant use of drugs known to affect endothelial function (nitrates, statins, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, and beta-blockers), and history of sensitivity to the study medication.
The study protocol was approved by the regional ethical research committee and was performed in accordance with the declaration of Helsinki and the code of Good Clinical Practice. The investigators (two radiologists and one pharmacologist) involved in estimating the two critical components of endothelial dysfunction assessment—serum nitrite concentration and FMD of brachial artery—were blinded to the treatment protocol. Other clinical investigations and assessment of participants were carried by the rheumatologist. All patients provided written informed consent to participate after a full explanation of the study.
Assessment
In all subjects, blood was drawn in the morning after overnight fasting, and the following variables were determined: high-density lipoprotein (HDL) cholesterol, triglycerides (TG), low-density lipoproteins (LDL), erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), total leukocyte count (TLC), differential leukocyte count (DLC), and hemoglobin by conventional methods using standard commercial kits.
Assessment of endothelial dysfunction
Two measures were taken: serum nitrite concentration and FMD of brachial artery.
-
1.
Serum nitrite was estimated at baseline and 12 weeks after single-dose treatment with infliximab (5 mg/kg i.v. infusion) as described by Sastry et al. [25] to assess indirectly the generation of nitric oxide (NO). Measurement of NO production in vivo is difficult because of its short half-life. Consequently, its metabolite serum nitrite has been used as a surrogate marker for estimating NO production [26]. Serum samples for this estimation were collected after at least 14 h of fasting. This was obtained by fasting after the evening meal at 6:00 p.m. and taking sample the next morning after 8.00 a.m. [27]. All the subjects were teetotalers and nonsmokers and taking a normal regular diet. Samples for serum nitrite were estimated in duplicate. Mean of the two measured value was considered for analysis.
-
2.
Flow-mediated vasodilation and nitroglycerin-induced (400 µg sublingual tablet) vasodilation of the brachial artery were assessed at baseline and 12 weeks after single-dose treatment with infliximab (5 mg/kg i.v. infusion) using a high-resolution ultrasound vessel wall tracking device with a 10-MHz linear array transducer (Voluson Vyge, 730 expert model) by two radiologists, who were blinded to the treatment protocol, using the standard method as described in earlier studies [16]. Subjects were asked to fast, with the exception of water, for at least 12 h prior to being assessed. Subjects lay supine for 20 min before the first vascular ultrasound scan was conducted. Flow-mediated dilation of the brachial artery was induced by release of a wrist cuff inflated to suprasystolic pressure for 5 min. Recording of arterial diameter commenced 30 s before cuff deflation and after every 15 s for 3 min. After a rest period of 10 min, when brachial artery diameter had returned to baseline levels, a second baseline scan was recorded. This was followed by sublingual administration of 400 µg of nitroglycerin (NTG) tablet. After nitroglycerin administration, the diameter was recorded every 30 s for 5 min [16].
FMD was calculated in percentage as:
Assessment of disease activity
The following measures of clinical evaluation of inflammation were employed:
-
1.
The BASDAI and the Bath ankylosing spondylitis functional index (BASFI) disease activity scores were calculated using the standard and validated questionnaire as described in the previous published studies [24, 28].
-
2.
ESR was measured by Westergreen method and CRP level was determined using standard commercial kits.
Statistical analysis
Results are presented as mean ± SEM. Komlogorov–Smirnov test was used to determine if continuous quantitative variables were normally distributed. Differences in all continuous variables after treatment were examined by Wilcoxan signed-rank test because of the skewed distribution of data. The relationships between the different variables were analyzed using the Pearson or Spearman’s rank correlation coefficients. For FMD%, mean of the two measures taken by two radiologists were considered for analysis. Intra- and inter-observer variability for estimating FMD% was 1.13% and 1.20%, respectively. Sigma Stat V. 3.1 was used for statistical analysis. A p value of <0.05 was considered as statistically significant.
Results
The demographic and clinical characteristics of the patients at baseline and after treatment are presented below (Table 1).
Infliximab improves endothelial dysfunction
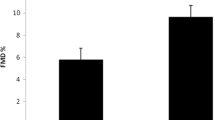
Flow-mediated dilation improved significantly from 9.81 ± 1.70 to 26.93 ± 2.34 (p < 0.001) after 12 weeks of initial treatment (Fig. 1). Endothelial-independent vasodilation-induced by NTG was statistically insignificant (13.65 ± 2.10 to 14.59 ± 1.93, p = 1.00) after 12 weeks (Fig. 2). There was no significant difference found in baseline brachial artery diameter from baseline to end visit (4.45 ± 0.15 to 4.46 ± 0.15 mm, p = 0.3).
Serum nitrite concentration (µmol/l) decreased significantly from 6.50 ± 0.21 to 2.57 ± 0.18 (p < 0.001) after 12 weeks.
Infliximab improves inflammatory disease activity
All patients included in the study had high disease activity, i.e., BASDAI score ≥4. After 12 weeks of single infliximab infusion, disease activity of all the patients had fallen to the level of low disease activity (BASDAI score <3.2). Overall, a composite BASDAI score decreased significantly from 5.40 ± 1.14 to 1.40 ± 0.70 (p = 0.004) and BASFI score from 5.05 ± 1.76 to 0.20 ± 0.63 (p = 0.002).
ESR and CRP levels also decreased significantly from 40.90 ± 6.00 to 11.50 ± 1.38 mm in first hour (p = 0.004) and from 29.08 ± 4.11 to 2.69 ± 0.43 mg/dl (p < 0.001) after 12 weeks, respectively (Table 1).
Association of endothelial dysfunction with inflammatory disease activity
From univariate correlation analysis (Table 2), we could not find any significant correlation of FMD with BASDAI score (r = 0.07, p = 0.83), nitrite concentration (r = 0.11, p = 0.75), BASFI score (r = 0.02, p = 0.93), total cholesterol (r = 0.22, p = 0.53), and ESR (r = 0.05, p = 0.87) at baseline. FMD showed negative correlation with CRP (r = −0.72, p < 0.01) both at baseline (Fig. 3) and after the treatment (r = −0.59, p < 0.05). But serum nitrite was found positively correlated with total cholesterol (0.66, p = 0.03) and BASFI (r = 0.64, p < 0.05) at baseline, which means that a decrease in nitrite concentration in AS patients significantly leads to a decrease in disease activity and improved quality of life.
Adverse effects
Minor adverse effects, which were self-reported by the patients, included skin rashes and nausea in two patients. Serum hematological and biochemical profile was found in the course of treatment (Table 1).
Discussion
The present study has demonstrated the improvement of both endothelial dysfunction and inflammatory disease activity with single dose of a TNF-α inhibitor, infliximab, in anti-TNF-naïve patients with established AS not responding to long-term DMARD therapy. We found that improvement in all the functions remained intensified for at least 12 weeks post-infusion. Improvement in endothelial dysfunction along with the disease activity provides a promising strategy to manage spondylitis and its atherosclerotic complications. It is the first study to date that shows efficacy of a single dose of TNF-alpha blockade with infliximab as an adjunct with sulfasalazine in targeting endothelial dysfunction in ankylosing spondylitis patients.
Systematic histological studies of vascular changes in spondyloarthritides or AS patients are sparse. Data on endothelial dysfunction in AS continue to emerge [2, 11], while the disease creates “favorable” conditions for endothelial lesion and the development of endothelial dysfunction [29]. A study by Caliskan et al. [30] revealed that coronary flow reserve was significantly lower in AS patients, suggesting impaired coronary microvascular function. Mathieu et al. [31] found a trend toward increased subclinical atherosclerosis in AS patients, but reports no improvement in arterial stiffness in ten patients treated with 14 weeks of infliximab therapy. Sari et al. [2] measured carotid artery intima-media thickness (IMT) and FMD of the brachial artery to assess endothelial function in patients with AS. This study revealed that IMT was not different between AS and healthy controls, but FMD was, however, significantly lower in AS patients, suggesting impaired endothelial function. In another study, Van Eijk et al. [8] demonstrated microvascular dysfunction (which is linked to several cardiovascular risk factors) in AS. This study revealed that endothelial-dependent vasodilatation of the skin as measured by laser Doppler fluxmetry technique improves after 1 month of treatment with anti TNF-α etanercept in AS patients with active disease. Moreover, in this study, a significant correlation was also observed between changes in endothelium-dependent vasodilatation and changes in ESR (r = −0.56, p = 0.03) after treatment with etanercept [8]. Improvement in endothelium-dependent vasodilatation with infliximab in the present study and by etanercept (the study by Eijk et al.) suggests that improvement in endothelial dysfunction could be a group effect of anti-TNF targeting therapies.
Elevated levels of CRP may contribute to the development of atherosclerosis as an independent risk factor [5]. In the present study, a significant correlation was observed between FMD and CRP at baseline (p < 0.01) and between changes in endothelium-dependent vasodilatation and changes in CRP (p < 0.05) after the treatment. Interestingly, impaired coronary microvascular function and the improved microvascular function in AS correlated well with CRP, suggesting a detrimental effect of inflammation on vascular function. Our findings support this hypothesis by demonstrating that blocking TNF-α with infliximab results in the improvement of endothelium-dependent vasodilation. In patients with RA, the ability of biologic [16] and non-biologic therapy [32] to improve endothelial function has already been demonstrated by brachial ultrasonography, but this has not been the case in AS so far. TNF-α contributes to microvascular dysfunction [8]. This is possibly because TNF-α blocks the activation of eNOS by interfering with Akt phosphorylation, which is essential for flow-dependent vasodilatation. In addition, TNF-α directly degrades eNOS mRNA [33, 34]. In addition to the specific TNF-α effects on endothelium function, it is likely that in line with the inflammation-driven atherosclerosis, improvement in endothelial dysfunction was the result of a generally decreased inflammatory burden as evidenced by a significant decline in the inflammatory biomarkers—CRP and ESR—and serum nitrite. Hence, it appears that endothelial dysfunction is accelerated by inflammation, and as the inflammation decreases, it improves the endothelial function.
FMD depends on the production of NO, and other vasodilators by the endothelium and inflammation have the potential to impair FMD by reducing the bioavailability of NO [35]. Our findings of apparently low FMD values (9.81 ± 1.70%) at baseline are in accordance with the previous findings of Sari et al. [2] [14.1 ± 6.7% (patients) vs. 17.6 ± 8% (control), p = 0.03] in AS patients. This blunted vasodilator response to FMD implies a decrease in NO bioavailability which may be caused by a decreased expression of eNOS, lack of substrate or cofactors for eNOS, alteration in the signaling pathways activating eNOS, and/or accelerated degradation of NO by reactive oxygen species [36]. Improvement in FMD and decrease in NO concentration after treatment with infliximab suggests that there is decreased inflammation-induced nitric oxide synthase (iNOS) expression and thus NO production through an increase in endothelial (eNOS) expression. Increase in the FMD response but not the NTG response after treatment suggests that there is increased NO bioavailability but not the sensitivity of vascular smooth muscle to NO [37].
As NO levels are influenced by nitrate-rich foods and alcoholic beverages, they were measured when they had reached a steady-state plasma concentration (after at least 14 h of fasting) [38]. This was based on the observations from a Japanese study which proved that overnight fasting (9–10 h) is not sufficient for measuring steady-state plasma concentrations of NO, and randomly collected blood samples show nearly ten times higher serum nitrate and nitrite concentrations. Even an extremely NO-restricted diet needs 13 h to reach a steady-state concentration [27].
There are no characteristic diagnostic laboratory tests for spondyloarthropathies. ESR and CRP relate more to peripheral than to axial arthritis [1]. Serum nitrite has the potential to emerge as a much better biomarker in AS than the currently used biomarkers like ESR and CRP because they show poor correlation with disease activity [39]. Recently, Sari et al. [40] has reported increased asymmetric dimethylarginine (ADMA) levels in relatively young AS patients who did not have classical CV risk factors, suggesting that NO metabolism is impaired in AS. ADMA, an endogenous inhibitor of NO, is associated with reduced nitric oxide synthesis and vascular dysfunction [41]. Elevated ADMA level is recognized as a novel risk marker for cardiovascular disease and mortality [42]. Thus, on one side, inflammatory cascade inhibits eNOS activity due to elevated ADMA levels, leading to a decrease in NO biosynthesis and, on the other side, accelerates iNOS activity, leading to increased nitrite level and endothelial dysfunction.
In the present study, improvement in all the inflammatory variables like BASDAI and BASFI scores, morning stiffness, and pain intensity shows that the protective effect of infliximab infusion was conserved for at least 12 weeks post-infusion. Significant correlation between BASFI and serum nitrite reveals that a decrease in nitrite concentration leads to functional improvement. The effect of infliximab on serum nitrite level in AS has not been reported so far.
In the present study, total cholesterol and total cholesterol/HDL ratio were decreased significantly (p < 0.001), though within normal range. Deterioration of the atherogenic index (the ratio between total cholesterol and HDL cholesterol) is an important cardiovascular risk factor indicator [43]. Infliximab has been shown to have a variable effect on cholesterol level. A study by Popa et al. [44] reports an increase of HDL-cholesterol and a decrease of CRP and IL-6 after 2 weeks of infliximab treatment. A study by Kiortsis et al. [43] revealed an increase in total cholesterol levels, but atherogenic indices (total cholesterol/HDL-cholesterol and triglyceride/HDL-cholesterol ratios) were not altered after 6 months of infliximab treatment. Increase in total cholesterol has also been found to be correlated with increased serum nitrite level. Here, acute arrest of inflammation by single infliximab injection has resulted in a decrease in total cholesterol level in short run by an unknown mechanism as opposed to its effects observed in other studies. Moreover, patients were lacking NSAIDs (non-selective COX inhibitors) linked atherogenic cardiovascular adverse effects, as has been reported in an earlier study by Farkouh et al. [45]. Thus, the present study suggests a safe effect of a single dose of infliximab on the atherogenic index.
Our results suggest that patients who are chronically ill and partly disabled by ankylosing spondylitis could have more than palliative treatment. It is significant that the benefits resulted from a single infliximab dose and were conserved for at least 12 weeks post-administration, which was the study end point. Although this was an open-label, pilot study without a control group or randomization, it is very unlikely that the high percentage of response (90% of the treated patients) was due to a placebo effect of intravenous infusions; such an effect would be expected in no more than 30% of the patients. The evidence that the anti-TNF alpha therapy with infliximab was efficacious is further strengthened by the fact that all the patients had persistently active disease for at least several months. In addition, the fact about the significantly decreased serum levels of the best currently available biomarkers of inflammation, CRP, ESR, and nitrite can hardly be explained by a placebo effect. The most significant limitation of this study was the small sample size and limited statistical power, and that may be the probable reason for not detecting the direct correlation between markers of inflammation and endothelial dysfunction. Despite these limitations, this study demonstrates improvement of endothelial function and inflammatory disease activity. The present results must be explored further in large-sized double-blind clinical trials so that the true potential of efficacy of infliximab in improving endothelial dysfunction in AS could be found.
References
Agarwal V (2010) Spondyloarthropathies. In: Syngle A, Deodhar SD (eds) Rheumatology principles and practice. Jaypee, India, pp 179–189
Sari I, Okan T, Akar S, Cece H, Altay C, Secil M, Birlik M et al (2006) Impaired endothelial function in patients with ankylosing spondylitis. Rheumatology 45:283–286
Lautermann D, Barun J (2002) Ankylosing spondylitis—cardiac manifestations. Clin Exp Rheumatol 20:S11–S15
Lehtinen K (1993) Mortality and causes of death in 398 patients admitted with ankylosing spondylitis. Ann Rheum Dis 52:174–176
Peters MJ, Irene E, Horst-Bruinsma V et al (2004) Cardiovascular risk profile of patients with spondylarthropathies, particularly ankylosing spondylitis and psoriatic arthritis. Semin Arthritis Rheum 34:585–592
Divecha H, Sattar N, Rumley A, Cherry L, Lowe GDO, Sturrock R (2005) Cardiovascular risk parameters in men with ankylosing spondylitis in comparison with non-inflammatory control subjects: relevance of systemic inflammation. Clin Sci 09:171–176
Heeneman S, Daemen MJ (2007) Cardiovascular risks in spondyloarthritides. Curr Opin Rheumatol 19:358–362
Van Ejick IC, Peters MJL, Seme EH, van der Horst-bruinsma IE, Dijkmans BAC, Smulders YM, Nurmohamed MT (2009) Microvascular function is impaired in ankylosing spondylitis and improves after tumour necrosis factor α blockade. Ann Rheum Dis 66:362–366
Gonzalez-Gay MA, Gonzalez Juananey C, Martin J (2005) Rheumatoid arthritis; a disease associated with accelerated atherogenesis. Semin Arthritis Rheum 35:8–17
Roman MJ, Shanker BA, Davis A, Lockshin MD, Sammantano L, Simantov R et al (2003) Prevalence and correlates of accelerated atherosclerosis in systemic lupus erythematosus. N Engl J Med 349:2399–2406
Poddubnyĭ DA, Rebrov AP (2007) Endothelial dysfunction in patients with Bechterew’s disease (ankylosing spondylitis). Klin Med (Mosk) 85:66–69
Bilsborough W, Keen H, Taylor A, O’Driscoll GJ, Arnolda L, Green DJ (2006) Anti tumour necrosis factor-alpha therapy over conventional therapy improves endothelial function in adults with rheumatoid arthritis. Rheumatol Int 26:1125–1131
Vaudo G, Marchesi S, Gerli R, Allegrucci R, Giordano A, Siepi D et al (2004) Endothelial dysfunction in young patients with rheumatoid arthritis and low disease activity. Ann Rheum Dis 63:31–35
Davignon J, Ganz P (2004) Role of endothelial dysfunction in atherosclerosis. Circulation 09:III27–32
Blake GJ, Ridker PM (2001) Novel clinical markers of vascular wall inflammation. Circ Res 89:763–771
Hurlimann D, Forster A, Noll G, Enseleit F, Chenevard R, Distler O et al (2002) Anti-tumour necrosis factor-alpha treatment improves endothelial function in patients with rheumatoid arthritis. Circulation 106:2184–2187
Zochling J (2008) Assessment and treatment of ankylosing spondylitis: current status and future directions. Current Opinion in Rheumatology 20:398–403
Syngle A (2006) Arthritis and its treatment. In: Rattan S, Kassem M (eds) Prevention and treatment of age-related diseases. Springer, the Netherlands, pp 105–32
Braun J, Brandt J, Listing A, Zink R, Alten G, Burmester W, Golder E et al (2003) Long-term efficacy and safety of infliximab in the treatment of ankylosing spondylitis. Arthritis & Rheumatism 48:2224–2233
Cardillo C, Schinzari F, Mores N et al (2006) Intravascular tumor necrosis factor α blockade reverses endothelial dysfunction in rheumatoid arthritis. Clin Pharmacol Ther 80:275–281
Booth AD, Jayne DR, Kharbanda RK et al (2004) Infliximab improves endothelial dysfunction in systemic vasculitis: a model of vascular inflammation. Circulation 109:1718–1723
Schinzari F, Armuzzi A, De Pascalis B et al (2008) Tumor necrosis factor-α antagonism improves endothelial dysfunction in patients with Crohn’s disease. Clin Pharmacol Ther 83:70–76
Vander LS, Valkerburg HA, Cats A (1984) Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheum 27:361–368
Zochling J, Braun J, van der Heijde D (2007) Assessments in ankylosing spondylitis. Best Practice & Research Clinical Rheumatology 21:699–712
Sastry KV, Moudgal RP, Mohan J, Tyagi JS, Rao GS (2002) Spectrophotometric determination of serum nitrite and nitrate by copper–cadmium alloy. Anal Biochem 306:79–82
Wanchu A, Khullar M, Sud A, Bambrey P (1999) Nitric oxide production is increased in patients with inflammatory myositis. Nitric oxide 3:454–458
Himento M, Ishibashi T, Nakano S, Furuya K, Kigoshi T, Uchida K et al (2003) A practical procedure for achieving a steady state of NO x concentration in plasma: with special reference to NO x content of Japanese daily food. Tohoku J Exp Med 199:95–110
Zochling J, Braun J, van der Heijde D (2006) Assessments in ankylosing spondylitis. Best Practice & Research Clinical Rheumatology 20:521–537
Karoli NA, Rebrov AP (2007) Vascular wall lesion and disorder of endothelial vasoregulating function in patients with ankylosing spondylitis. Klin Med (Mosk) 85:54–57
Caliskan M, Erdogan D, Gullu H et al (2008) Impaired coronary microvascular and left ventricular diastolic functions in patients with ankylosing spondylitis. Atherosclerosis 196:306–312
Mathieu S, Joly H, Baron G, Tournadre G, Dubost J-J, Ristori JM (2008) Trend towards increased arterial stiffness or intima-media thickness in ankylosing spondylitis patients without clinically evident cardiovascular disease. Rheumatology (Oxford) 47:1203–7
Syngle A, Vohra K, Kaur L, Sharma S (2009) Effect of spironolactone on endothelial dysfunction in rheumatoid arthritis. Scand J Rheumatol 38:15–22
Cardaropoli S, Silvango F, Morra E, Pescarmona GP, Tordos T (2003) Infectious and inflammatory stimuli decrease endothelial nitric oxide synthase activity in vitro. J Hypertens 21:2103–2110
Yoshizumi M, Perrella MA, Burnett JC Jr, Lee ME (1993) Tumor necrosis factor down regulates an endothelial nitric oxide synthase mRNA by shortening its half life. Circ Res 73:205–209
Vita JA, Keaney JF Jr, Larson MG, Keyes MJ, Massaro JM, Lipinska I et al (2004) Brachial artery vasodilator function and systemic inflammation in the Framingham Offspring Study. Circulation 110:3604–3609
Bergholm R, Leirisalo-Repo M, Vehkavaara S, Mäkimattila S, Taskinen MR, Yki-Järvinen H (2002) Impaired responsiveness to NO in newly diagnosed patients with rheumatoid arthritis. Arterioscler Thromb Vasc Biol 22:1637–1641
Koppenol WH et al (1992) Peroxynitrite, a cloaked oxidant formed by nitric oxide and superoxide. Chem Res Toxicol 5:834–842
Sierksma A, van der Gaag MS, Grobbee DE, Hendriks HF (2003) Acute and chronic effects of dinner with alcoholic beverages on nitric oxide metabolites in healthy men. Clin Exp Pharmacol Physiol 30:504–506
Kumar A, Falodia SK, Shankar S, Grover R, Marwaha V, Aneja R, Srivastava K, Das N (2009) Assessment of serum nitrite as biomarker of disease activity in ankylosing spondylitis. Indian J Rheumatology 4:47–50
Sari I, Kebapcilar L, Alacacioglu A, Bilgir O, Yildiz Y, Taylan A (2009) Increased levels of asymmetric dimethylarginine (ADMA)in patients with ankylosing spondylitis. Inter Med 48:1363–1368
Boger RH (2006) Asymmetric dimethylarginine (ADMA): a novel risk marker in cardiovascular medicine and beyond. Ann Med 38:126–136
Boger RH, Maas R, Schulze F, Schwedhelm E (2005) Elevated levels of asymmetric dimethylarginine (ADMA) as a marker of cardiovascular disease and mortality. Clin Chem Lab Med 43:1124–1129
Kiortsis DN, Mavridis AK, Filippatos TD, Vasakos S, Nikas SN, Drosos AA (2006) Effects of infliximab treatment on lipoprotein profile in patients with rheumatoid arthritis and ankylosing spondylitis. J Rheumatol 33:921–933
Popa C, Netea MG, Radstake T et al (2005) Influence of anti-tumour necrosis factor therapy on cardiovascular risk factors in patients with active rheumatoid arthritis. Ann Rheum Dis 64:303–5
Farkouh ME, Greenberg JD, Jeger RV, Ramanathan K, Verheugt FW, Chesebro JH et al (2007) Cardiovascular outcomes in high risk patients with osteoarthritis treated with ibuprofen, naproxen or lumiracoxib. Ann Rheum Dis 66:764–70
Acknowledgments
Support and acknowledgment: None.
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Syngle, A., Vohra, K., Sharma, A. et al. Endothelial dysfunction in ankylosing spondylitis improves after tumor necrosis factor-α blockade. Clin Rheumatol 29, 763–770 (2010). https://doi.org/10.1007/s10067-010-1402-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-010-1402-x