Abstract
The overall role of the Rcs phosphorelay in Proteus mirabilis is largely unknown. Previous work had demonstrated that the Rcs phosphorelay represses the flhDC operon and activates the minCDE cell division inhibition system. To identify additional cellular functions regulated by the Rcs phosphorelay, an analysis of RNA-seq data was undertaken. In this report, the results of the RNA-sequencing are discussed with an emphasis on the predicted roles of the Rcs phosphorelay in swarmer cell differentiation, motility, biofilm formation, and virulence. RcsB is shown to activate genes important for differentiation and fimbriae formation, while repressing the expression of genes important for motility and virulence. Additionally, to follow up on the RNA-Seq data, we demonstrate that an rcsB mutant is deficient in its ability to form biofilm and exhibits enhanced virulence in a Galleria mellonella waxworm model. Overall, these results indicate the Rcs regulon in P. mirabilis extends beyond flagellar genes to include those involved in biofilm formation and virulence. Furthermore, the information presented in this study may provide clues to additional roles of the Rcs phosphorelay in other members of the Enterobacteriaceae.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The regulator of colonic acid capsule synthesis (Rcs) phosphorelay is a complex signal transduction system present in many members of the Enterobacteriaceae. In the swarming bacterium Proteus mirabilis, the Rcs phosphorelay controls swarmer cell differentiation, represses the flhDC operon and activates the minCDE cell division system (Belas et al. 1998; Liaw et al. 2001; Clemmer and Rather 2008a; Howery et al. 2015). However, our knowledge of the role for the Rcs phosphorelay in other cellular functions in P. mirabilis is limited. Because of the importance of P. mirabilis as a human pathogen, we sought to identify additional functions for the Rcs phosphorelay in the physiology of this bacterium.
The Rcs phosphorelay was first identified during a screen to find genes necessary for capsular polysaccharide (cps) synthesis in Escherichia coli (Gottesman et al. 1985). Further studies revealed that it was not just a simple two-component system, but a multifaceted variation known as a phosphorelay. The Rcs phosphorelay comprises a sensor kinase (RcsC) located at the inner membrane, a response regulator (RcsB) (Brill et al. 1988; Stout and Gottesman 1990), and a phosphotransfer protein (RcsD). RcsD, previously known as YojN, mediates the transfer of the phosphate group from RcsC to RcsB (Takeda et al. 2001). An additional protein, RcsA, is an auxiliary protein that assists RcsB binding at sites denoted as RcsAB boxes (Pristovsek et al. 2003; Stout et al. 1991). Finally, the outer membrane lipoprotein RcsF transmits signals induced by membrane stress or other elements to RcsC thereby activating the phosphorelay (Castanie-Cornet et al. 2006; Majdalani et al. 2005; Gervais and Drapeau 1992).
In addition to cps, other targets for Rcs regulation have been identified in E. coli. The cell division septum protein, FtsZ, responsible for Z-ring formation at the future site of cytokinesis is activated by RcsB (Gervais et al. 1992). A related cell division protein, FtsA, which tethers FtsZ to the cell membrane, was also shown to be Rcs regulated (Carballès et al. 1999). OsmC, a reducer of organic hydroperoxides, and two other osmotically induced proteins, encoded by osmB and osmY, are all Rcs-activated genes (Davalos-Garcia et al. 2001; Hagiwara et al. 2003). RprA, a regulatory RNA also activated by RcsB, affects translation of RpoS, which connects the Rcs phosphorelay to the RpoS regulon (Majdalani et al. 2002). One target in particular, flhDC, encoding the regulator for flagellar synthesis, FlhD4C2, is repressed by the Rcs phosphorelay in E. coli (Francez-Charlot et al. 2003) and Salmonella enterica (Wang et al. 2007). The relationship between the Rcs phosphorelay and flhDC regulation is one that has strong implications for motility and virulence, particularly for organisms that use motility as a means to establish infection.
Proteus mirabilis is a highly motile bacterium noted for its ability to change shape from short vegetative rod cells into elongated swarmer cells during a process known as differentiation. Differentiated swarmer cells are 20- to 50-fold longer than their vegetative counterparts and express thousands of flagella (Hoeniger 1965; Belas and Flaherty 1994). Swarmer cells utilize a flagellum-dependent, multicellular form of motility known as swarming which is different from swimming motility. Swimming occurs when a single cell propels itself through liquid or soft agar. Swarming is a multicellular process where a group of cells producing massive amounts of interweaving flagella collectively align and form rafts which facilitate movement across a solid surface (Jones et al. 2004). Differentiation of rod-shaped cells into swarmer cells occurs upon contact with a solid surface. Elements that contribute to this process include O-antigen-mediated cell surface perturbation (Morgenstein et al. 2010), inhibition of flagellar rotation, and cell-to-cell signaling (Rauprich et al. 1996; Fraser and Hughes 1999; Sturgill and Rather 2004; Belas and Suvanasuthi 2005). After a period of migration, swarmer cells undergo consolidation or de-differentiation, which was once thought to be a resting period. However, during consolidation, cells are more metabolically active than swarming cells (Armitage 1981) and a prominent upregulation of genes involved in cell wall synthesis, nutrient uptake, metabolism, and respiration occurs relative to swarming cells (Pearson et al. 2010), indicating the cells are preparing for another period of energy-expensive swarming. The bulls-eye pattern P. mirabilis forms on agar plates is due to the oscillation between swarming and consolidation.
At the center of swarmer cell differentiation is the flhDC master operon, whose expression is dictated by a myriad of factors (Belas et al. 1998; Dufour et al. 1998; Liaw et al. 2001; Sturgill and Rather 2004; Stevenson and Rather 2006; Clemmer and Rather 2008a; Morgenstein et al. 2010). The product of flhDC, FlhD4C2, is central to the activation of class II flagellar genes (Furness et al. 1997; Claret and Hughes 2000a), and hyper-flagellation is essential for the formation of swarmer cells (Gygi et al. 1995). Expression of flhDC is high during initiation of swarming, but sharp decreases in flhDC expression can be observed shortly after differentiation occurs (Furness et al. 1997; Clemmer and Rather 2008a). In addition to strict transcriptional control, the FlhD and FlhC proteins are tightly regulated post-translationally via proteolytic degradation (Claret and Hughes 2000b; Clemmer and Rather 2008b). Modest changes in flhDC expression can have dramatic effects on the swarm phenotype. Overexpression of flhDC results in a hyperswarming phenotype (Furness et al. 1997), also known as precocious swarming, and flhDC mutants do not swarm.
Other mutations that result in hyperswarming include those in the RppAB two-component system (Wang et al. 2008) and the Rcs phosphorelay. It is common in bacteria to employ more than one two-component system to control differentiation (Kovács 2016). The first rcs mutations resulting in hyperswarming were in rcsC and rcsD (previously rsbA) (Belas et al. 1998; Liaw et al. 2001). An rcsB mutant was later constructed, and it shared the hyperswarming phenotype with other mutants of the Rcs phosphorelay. The hyperswarming phenotype of the rcsB mutant is likely related to increased levels of flhDC observed in this strain (Clemmer and Rather 2008a), and RcsB has been shown to bind to the upstream region of flhDC in E. coli (Francez-Charlot et al. 2003). In addition to hyperswarming, rcs mutants elongate in liquid media, a non-permissive condition for differentiation, suggesting the Rcs phosphorelay influences the expression of genes important for differentiation. Interestingly, flhDC overexpression alone does not induce an elongated phenotype in liquid indicating additional rcs regulated genes control elongation (Clemmer and Rather 2008a).
Recently, RNA sequencing was performed on an rcsB mutant in P. mirabilis to identify Rcs-regulated genes important for differentiation (Howery et al. 2015). This data set was compiled from three independent RNA-Seq analyses between wild type and an RcsB mutant. GEO accession number GSE76341. However, when re-evaluating these data sets, additional RcsB-regulated genes were identified resulting in 221 genes that were differentially regulated in an rcsB mutant when a twofold or greater cutoff is employed (Supplemental Table 1). To aid in determining whether RcsB is acting directly or indirectly on genes within its regulon, a bioinformatic approach was used to identify putative RcsB-binding sites. The results of the RNA-Seq have predicted additional functions that may be controlled by the Rcs regulon in P. mirabilis. Specifically, the role of the Rcs regulon in differentiation, motility, self-recognition, adherence, biofilm formation and virulence are discussed. Lastly, to confirm these predictions, we experimentally verify that the Rcs phosphorelay is important for biofilm formation and influences virulence in P. mirabilis.
Materials and methods
Bacterial growth conditions
Except where indicated, P. mirabilis strains were grown in modified Lysogeny (LB) broth (10 g tryptone, 5 g yeast extract, 5 g NaCl/L) at 37 °C with shaking at 250 rpm. Agar was used at 30 g/L in plates to prevent swarming. Antibiotic selections included 35 µg/mL streptomycin.
Biofilm assays
Proteus mirabilis strain HI4320 or the isogenic rcsB::Str R were grown overnight in LB with appropriate antibiotics at 37 °C without shaking. Cells were equilibrated to the same optical density (A 600) and a 1:100 dilution of each strain was made in LB only. For the biofilm assays in 96-well polystyrene microtiter plates (Costar®), 150 µL of the diluted cultures were added to 8 wells of the microtiter plate and incubated in a humidified environment at 37 °C for 8 or 24 h. The A 600 was determined for each well to measure growth. Biofilms were stained with by adding 15 µL 0.4 % crystal violet to the well and incubating for 15 min. Afterward, stained biofilms were rinsed with deionized water three times, and biofilms were resuspended in 200 µL 33 % acetic acid. Biofilm was quantified by reading each sample at 580 nm and dividing that value by the A 600 value for the same sample well. For biofilm assays on catheter, tweezers and scissors were pre-sterilized in 100 % ethanol for 20 min. Siliconized latex catheters (Mentor) were cut into 0.25-in. pieces, flamed briefly, and 5 pieces of catheter were fused per well to the bottom of a 6-well plate (Costar®). 9.5 mL of a 1:100 diluted culture was added in duplicate to the plate containing catheter pieces to submerge them. An LB-only control was used in duplicate. After 8 or 24 h of incubation in a humidified environment, 1 mL of the culture was removed to determine the A 600. If any piece of catheter detached from the bottom of the plate, it was discarded prior to staining. Biofilms were stained by adding 0.85 mL of 0.4 % crystal violet to each well and incubated for 15 min. Liquid was removed from the wells, and catheters were rinsed three times by adding 10 mL deionized water to each well to remove planktonic cells. After rinsing, catheter pieces were removed and added to individual 1.5 mL microcentrifuge tubes where they were resuspended in 0.9 mL 33 % acetic acid and vortexed for 2 min. Catheter pieces were removed from tubes and each sample tube was read at 580 nm. A 580 values were adjusted for each strain by subtracting the average A 580 value for catheter incubated in LB only. Biofilm was quantified by dividing the A 580 value by the A 600 value of the well where catheters were incubated.
Identification of potential RcsB-binding sites of RcsB-regulated genes
Based on known RcsB-binding sites in E. coli, the pattern NNNGANNNNCNNN was used as the input for computational microbiology laboratory (CMBL) pattern locator (PATLOC) program (Mrázek and Xie 2006). The search was restricted to intergenic regions within the P. mirabilis HI4320 chromosome (NCBI RefSeq NC_010554.1). Genomic hits returned by PATLOC were compiled using an SAS program to restrict binding sites to RcsB-regulated genes identified using the RNA sequencing data. To confirm location and presence on the direct DNA strand, potential RcsB-binding patterns were input into Nucleotide Basic Local Alignment Search Tool (BLAST) (Altschul et al. 1997) version 2.2.32 adjusted for highly similar (megablast) and short input nucleotide sequences. Potential RcsB-binding sequences located near defined or putative transcriptional regulatory regions were aligned to the consensus E. coli RcsAB box, TAAGAATATTCCTA, using an SAS program. Previously identified RcsB-binding sequences in E. coli were also aligned. The SAS programs written to compile genomic hits and align binding sequences are available from the authors upon request.
Galleria mellonella killing assays
Cells of wild-type PM7002 or the isogenic rcsB::Sm mutant (Clemmer and Rather 2008a) were grown by shaking at 250 rpm in 2.5 mL of LB broth at 37 °C to an optical density (A 600) of 0.45. A 10−6 dilution was prepared in sterile LB and 5 μL representing 10 CFU for each strain was used to inject G. mellonella waxworms (200–250 mg each) in the second proleg. The G. mellonella worms were incubated at 37 °C in a humidified incubator and were checked at 24 h intervals for killing. Worms were considered dead if they did not move when prodded with a pipette tip and were dark brown or black in color. A total of three independent experiments were conducted representing 28 worms for wild type and 29 worms for the rcsB mutant.
Results
RcsB-activated genes identified by RNA-Seq
Cell division and differentiation
Genes involved in cell division and thus septation have long been hypothesized to play a role in swarmer cell elongation, since the prevention of septation in E. coli results in filamentation (de Boer et al. 1989). However, during a previous study to identify genes important for elongation in P. mirabilis, only gidA, involved in glucose-inhibited cell division, was isolated (Belas et al. 1995). Rcs-activated genes involved in cell division include the Min cell division inhibition system, ftsZ, and ftsA (Table 1). The Min system is composed of three proteins, MinC, MinD, and MinE, which oscillate from pole to pole during cell division in order to keep FtsZ-mediated Z-ring formation at the cell center (Ward and Lutkenhaus 1985; de Boer et al. 1990). As previously noted, expression of minC and minD were reduced 3.4- and 6.74-fold, respectively, in the rcsB mutant relative to wild type (Table 1 and Howery et al. 2015). FtsZ is the bacterial homolog of tubulin and mediator of the Z-ring (Bi and Lutkenhaus 1991). FtsA, which is structurally related to actin, is the protein that anchors the Z-ring to the site of septation (Pichoff and Lutkenhaus 2005). ftsZ expression was reduced 3.33-fold and ftsA expression was reduced 2.38-fold in rcsB mutant relative to wild type (Table 1). This is consistent with previous studies demonstrating that ftsZ and ftsA were RcsB activated in E. coli (Gervais et al. 1992; Carballès et al. 1999).
An additional Rcs-activated gene important for differentiation was speB, where expression was reduced 2.69-fold in the rcsB mutant relative to wild type. SpeB functions as an agmatinase required for the conversion of agmatine to putrescine. Mutations in speB result in a delay in the onset of differentiation and shorter migration periods. This swarming defect could be rescued by the addition of extracellular putrescine (Sturgill and Rather 2004).
Fimbriae production
Proteus mirabilis is known in a clinical setting for its ability to cause catheter-associated urinary tract infections (CAUTIs) which can lead to pyelonephritis, urolithiasis, and cystitis. It is not often found in patients undergoing short-term catheterization (Matsukawa et al. 2005), but can be isolated from the urine of approximately 40 % of patients undergoing long-term catheterization (O’Hara et al. 2000; Jacobsen et al. 2008). The first step in establishing a CAUTI is adherence to a catheter, which P. mirabilis readily does (Roberts et al. 1990; Stickler et al. 2006). Fimbriae from other urinary tract pathogens have been implicated in the adherence to catheter material (Mobley et al. 1988; Yakubu et al. 1989). There are 17 putative fimbrial operons in the P. mirabilis HI4320 genome (Pearson et al. 2008). The major fimbrial subunits of the mannose-resistant Proteus-like (MRP) fimbriae, the P. mirabilis fimbriae (PMF), and the uroepithelial cell adhesin fimbriae (UCA) were all identified as Rcs-activated genes. Expression of mrpA, pmfA and ucaA was reduced 68.69-, 63.93-, and 6.97-fold, respectively, in the rcsB mutant relative to wild type (Table 1). Additionally, the expression of an mrpJ paralog (PMI0261) and a fimbrial adhesin, mrpH, paralog (PMI0260) was reduced 28.86- and 12.15-fold (Table 1).
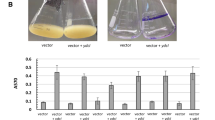
Given the importance of fimbriae to biofilm formation (Jansen et al. 2004; Rocha et al. 2007), the Rcs-mediated activation of certain fimbrial genes in P. mirabilis, and the contribution of Rcs to biofilm formation in other bacteria (Ferrières and Clarke 2003; Hinchliffe et al. 2008) biofilm assays were performed using P. mirabilis strain HI4320 and an isogenic rcsB mutant. After 8 h of incubation in 96-well polystyrene microtiter plates, the rcsB mutant was unable to form biofilm (Fig. 1a). After 24 h of incubation, the rcsB mutant formed 2.9-fold less biofilm than wild-type cells. The reduction in biofilm formation seen in the rcsB mutant at 24 h is unlikely to be the result of increased flhDC expression as a wild-type strain that overexpresses flhDC on a plasmid (pACYC-flhDC) forms the same level of biofilm as cells containing the vector alone (Fig. S1). At 48 h the difference in biofilm formation was not statistically significant, although at this time point the wild-type biofilm had decreased 6.8-fold compared to 24 h. Biofilm reduction after longer periods of incubation is not an uncommon phenomenon and can be seen in other organisms (Jackson et al. 2002; Berne et al. 2010). The reduction over time in wild type is not seen in the rcsB mutant, where only a 0.6-fold reduction in biofilm occurred between 24 and 48 h (Fig. 1a).
An rcsB mutant is deficient in biofilm formation. In Panel a, P. mirabilis strain HI4320 (dark grey bars) or the isogenic rcsB::Str R (light grey bars) was grown overnight at 37 °C without shaking. Cells were equilibrated to the same optical density (A 600), diluted, incubated in polystyrene microtiter plates for 8, 24, and 48 h. Biofilm formation was assessed as described in the “Materials and methods”. In Panel b, biofilms were formed on 0.25-in. latex catheter pieces for 8 and 24 h. For all experiments, the average of three independent experiments is shown. Error bars represent standard deviations. *p value <0.05
To examine the importance of rcsB to establishing CAUTIs, additional biofilm assays were performed using siliconized latex catheters. Recapitulating the results of the previous biofilm assays, the rcsB mutant did not form detectable biofilm on catheter after 8 h of incubation. After 24 h of incubation, the rcsB mutant did form biofilm, but 2.7-fold less than wild type (Fig. 1b). Overall biofilm formation for both strains was higher on catheter material, with the exception of the rcsB mutant after 8 h of incubation. In fact, the wild-type strain formed 6.3-fold more biofilm on catheter (Fig. 1b) compared to polystyrene (Fig. 1a) after 8 h of incubation.
RcsB-repressed genes
Motility
As the Rcs phosphorelay is a well-characterized repressor of swarming, it was not surprising that a large number of flagellar genes were identified as Rcs-repressed (Table 2). There are 15 proteins that comprise the external structural proteins of the flagellum, and 13 of those were recognized as Rcs-repressed. They include the proximal rod (FlgB, FlgC, FlgF), the distal rod (FlgG) and rod cap (FlgJ), the hook (FlgE), the hook cap (FlgD), hook-length control protein (FliK), the hook-flagellar junction proteins (FlgK, FlgL), the major flagellar subunit (FlaA) and flagellar cap (FliD), and the anti-σ28 regulatory protein (FlgM) (Table 2). Secretion of the structural proteins involves seven proteins located within the flagellar basal body, four of which have been identified as Rcs-repressed (FlhB, FliF, FliO, and FliP). All three proteins which comprise the ATPase that drives export of flagellar components (FliI, FliH, and FliJ), the secretion chaperones (FlgN, FliS and FliT), and the alternative sigma factor FliA (σ28) were all negatively regulated by RcsB (Table 2).
Additional flagellar proteins that were identified as Rcs-repressed are within the basal body and the flagellar motor. They include the flagellar basal body protein FliL, the L-ring protein FlgH, the P-ring proteins FlgA and FlgI, the MS-ring protein FliF, and the C ring proteins (FliG, FliM, and FliN). motA, motB, and all genes encoding members of the chemosensory (Che) complex with the exception of cheW, and two putative methyl-accepting chemotaxis proteins, PMI2808 and PMI2809, were also identified as negatively regulated by RcsB (Table 2).
Important regulators of flagellar synthesis, in addition to fliA (σ28) and flgM (anti-σ28), were repressed by RcsB. They include the class I operon for flagellar synthesis, flhDC; umoA and umoD (which were named for the upregulated expression of flhDC), and fliZ. Finally, ccmA, a gene important for cell shape and motility but unrelated to flagella production (Hay et al. 1999) was identified as Rcs-repressed (Table 2).
Virulence genes
RcsB influences the expression of two well-studied virulence genes, the IgA-degrading protease zapA and hemolysin (hpmBA). Both zapA and zapB, encoding a type-1 secretion ATP-binding protein, and zapD, encoding a type I secretion outer membrane protein, were found to be Rcs-repressed. The expression of zapA, zapB, and zapD were upregulated 43.44-, 39.44-, and 13.87-fold, respectively, in the rcsB mutant relative to wild type (Table 2). Expression of the hemolysin encoding genes, hpmA and hpmB, was also upregulated 12.51 and 10.41-fold, respectively, in the rcsB mutant relative to wild type (Table 2).
The RNA-Seq data suggested that the virulence of an rcsB mutant might be enhanced as the hpmBA hemolysin and zapAB protease genes were overexpressed. To examine this, we used an invertebrate G. mellonella waxworm model. In three independent experiments, the average killing at day 1 after infection was 92 % for the rcsB mutant versus 34 % for wild-type cells (Fig. 2). At day 2, the percent killing was 96 % for the rcsB mutant and 53 % for wild type and at day 3, the rcsB mutant killed 96 % of the worms versus 60 % for wild-type cells.
An rcsB mutant exhibits enhanced virulence in a Galleria mellonella waxworm model. Cells of wild-type PM7002 (light grey bars) or the isogenic rcsB::Str R mutant (dark grey bars) were grown to the same optical density. A 10−6 dilution was prepared in sterile LB and was used to inject G. mellonella in the second proleg. The G. mellonella worms were incubated at 37 °C in a humidified incubator and were checked at 24 h intervals for killing. Worms were considered dead if they did not move when prodded with a pipette tip and were dark brown or black in color. A total of three independent experiments were conducted representing 28 worms for wild type and 29 worms for the rcsB mutant. Error bars represent standard deviations
Self-recognition
Proteus mirabilis undergoes an interesting phenomenon during swarming where different strains will form boundaries (Dienes lines) between swarming colonies, but swarming colonies of the same strain will merge (Dienes 1946). The ability to distinguish between self and non-self has been well studied in P. mirabilis strain BB2000. Two systems, ids and idr, are important for self-recognition, and their products are exported by a type VI secretion system encoded by tss (Gibbs et al. 2008; Wenren et al. 2013, Alteri et al. 2013). The BB2000 homologs of idsA (PMI2990), idrA (PMI0750), and idrB (PMI0751) were all negatively regulated by RcsB (Table 2). PMI1117 (hcp) is a paralog of PMI0750, and PMI1118 is a paralog of PMI0751; both were identified as Rcs-repressed. Several homologs of the tss system were identified as Rcs-repressed. They include tssA (PMI0749), tssB (PMI0748), tssC (PMI0747), tssE (PMI0745), tssF (PMI0744), tssH (PMI0742), tssI (PMI0741), tssJ (PMI0740), and tssL (PMI0738) (Table 2). The idr and tss operons neighbor one another, are positioned divergently, and are separated by a large intergenic region (1899 bp) on the BB2000 chromosome. This organization is mimicked in HI4320 with PMI0250 being separated by an intergenic region of 2396 bp from the BB2000 homolog of tss. However, none of the genes downstream of PMI0251 had significant sequence similarity to idrC, idrD, or idrE. Other Rcs-repressed genes, which could be involved in self-recognition include PMI1331, which shares 56 % identity with IdsB, and PMI1332, which shares 44 % identity with IdsF.
Identification of putative RcsB-binding sites upstream of RcsB-regulated genes
As RcsB modulates the expression of known transcriptional regulators such as flhDC, crp, rpoN, and rpoE, a bioinformatic approach was used to identify potential RcsB-binding sites upstream of genes identified by the RNA sequencing data. After aligning previously identified RcsB-binding sites (Francez-Charlot et al. 2003; Carballès et al. 1999; Davalos-Garcia et al. 2001), it is clear that the guanine at position 4, the adenine at position 5, and the cytosine at position 10 are the most conserved residues within RcsB-binding sites.
In addition, if point mutations are introduced into the adenine at position 5 or the cytosine at position 10, repression of flhDC expression is reduced in E. coli (Francez-Charlot et al. 2003). In an S. enterica mutant where the Rcs system is constitutively active, a point mutation of the adenine, cytosine, or guanine at position 4 was able to restore swimming motility in the previously non-motile strain (Wang et al. 2007). These three sites within the RcsAB box are the only residues conserved across strains and multiple genes, both activated and repressed. Therefore, NNNGANNNNNCNNN was used as the input sequence for the Pattern Locator (PATLOC) program (Mrázek and Xie, 2006) against the P. mirabilis strain HI4320 genome. When used as the input for PATLOC, 4605 genomic hits were returned. 21.47 % were within pseudogenes, 73.05 % were intergenic, and 5.48 % were partial overlaps. These hits were compiled to be specific for genes identified using the RNA sequencing data. After compilation, the number of hits was reduced to intergenic or partially overlapping regions of 126 genes identified using RNA sequencing. Since PATLOC did not take into account whether the putative site was on the top or bottom strand, each potential sequence was input into BLAST to confirm its location and position. Previously proposed or experimentally identified transcriptional start sites and promoter regions were considered when determining a potential binding site. Thirty-four putative RcsB-binding sites were identified for RcsB-activated genes (Table 3), and 31 putative RcsB-binding sites were identified for RcsB-repressed genes (Table 4). These binding sites were aligned to the original consensus RcsAB box TAAGAATATTCCTA (Wehland and Bernhard 2000) based on the number of shared residues. The consensus RcsAB box was chosen for alignment as it is the originally defined binding sequence for RcsB, though there is divergence amongst identified RcsB-binding sequences, and the RcsB-binding site in P. mirabilis is unknown. Shared residues in aligned sequences are shown in red. In addition to putative binding sites, the known RcsA-independent RcsB-binding sites for the promoters of flhD, ftsA, and osmC in E. coli are also aligned (Francez-Charlot et al. 2003; Carballès et al. 1999; Davalos-Garcia et al. 2001). The sequence for the RcsA-dependent RcsB-binding site for the flhD promoter is also included (Francez-Charlot et al. 2003). Their inclusion in the tables illustrates the divergence of identified RcsB-binding sites from the consensus sequence. The fold-change in gene expression based on the RNA sequencing data and the location of binding site relative to important residues are also included in the table. Unfortunately, for many of the identified genes, no transcriptional start sites or promoters have been mapped and the relevance of the putative binding sites is unclear.
Of the Rcs-activated genes, cfa (encoding a cyclopropane fatty acyl phospholipid synthase), eco (encoding the protease inhibitor ecotin), and mrpA shared the most residues with the RcsB consensus sequence, with 11 out of 14 base pairs aligning with the sequence (Table 3). The location of the RcsB-binding site of mrpA is next to the −35 region of a putative σ70 promoter, which was identified when the mrp operon was first sequenced (Bahrani and Mobley 1994). The mrp transcriptional start site and another σ70 promoter were recently identified within the invertible repeat region when the phase variable mrp operon is in ON phase (Bode et al. 2015). This places the putative binding site 100 base pairs downstream of the transcriptional start site. Regulatory elements of cfa or eco have not been identified.
Genes directly regulated by RcsB in other organisms, which were identified in P. mirabilis using RNA sequencing include osmB and ftsA. Their putative binding sites are located 60 and 396 base pairs, respectively, upstream of their start codons. The osmB sequences shares 9 out of 14 identities with the consensus RcsAB box and shares 10 out of 14 residues with the osmCp1 RcsB-binding sequence (Davalos-Garcia et al. 2001). The ftsA binding sequence shares seven residues with the consensus sequence, but nine with the ftsA1p binding site of E. coli. The putative RcsB-binding site and the ftsA1p binding site are found in similar locations: at the center of ftsQ (Carballès et al. 1999).
Of the Rcs-repressed genes, the identified RcsB-binding site of flhD shared 11 out of 14 residues with the RcsAB consensus sequence and the RcsAB binding site of flhD in E. coli (Francez-Charlot et al. 2003). It shared nine residues with the RcsA-independent binding site flhD3p described in the same manuscript (Table 4). The putative flhD binding site is located two base pairs upstream of the mapped transcriptional start site (Furness et al. 1997; Clemmer and Rather 2008a; Bode et al. 2015). The putative RcsB-binding sites of PMI1359, which encodes a putative attachment invasion outer membrane protein, and PMI1075, a hypothetical protein with a domain of unknown function (DUF1795) aligned 10 of 14 residues with the RcsAB box. Many of the motility-associated genes have putative RcsB-binding sites overlapping their promoters (hpmB, flaD, fliF), start codons (flgB, flgM, motA) or transcriptional start sites (flhB). Additionally, the genes encoding self-recognition proteins PMI1117 (hcp/idrA homolog), PMI0750 (idrA paralog), and PMI2990 (idsA homolog) all share the same putative RcsB-binding sequence in the same location (Table 4).
Discussion
The importance of the Rcs phosphorelay in P. mirabilis has long been associated with the control of swarming via the negative regulation of flhDC. Through RNA sequencing, we have shown that the impact of Rcs on gene expression extends far beyond the inhibition of swarming-related genes. RcsB is not just a repressor of flhDC, but a repressor of many genes essential for motility, including flagella, chemotactic proteins, and positive regulators of motility. In addition, it represses the expression of virulence factors associated with swarmer cells, including zapA and hemolysin, and genes important for self-recognition. In contrast, RcsB is an activator of genes important for swarmer cell differentiation, including minCDE and speB, and also fimbrial genes, which are important for adherence and biofilm formation. This work will pave the way for future studies to identify the specific RcsB-regulated genes that carry out the above functions. In addition, the role of downstream transcriptional regulators in the overall RcsB regulon can be assessed.
As expected, RcsB was shown to negatively regulate the expression of flagellar structural genes and known activators of flagellar synthesis. The negative effect of RcsB on motility is seen in a myriad of bacterial species (Takeda et al. 2001; Wang et al. 2007; Hinchliffe et al. 2008). A putative RcsB-binding site was identified for the master operon for flagellar synthesis, flhDC (Table 4); this site shares high sequence homology to the RcsAB consensus binding site and is located in a similar region as the RcsB-binding site of flhD in E. coli. RcsB-mediated regulation of flg, fli, or flhBA operons could be indirect, as FlhD4C2 has been shown to bind directly to these operons in E. coli (Claret and Hughes 2002). FliA, which also has a predicted RcsB-binding motif (Table 4) has also been shown to bind directly upstream of motAB-cheAW, predicted methyl-accepting chemotaxis proteins, and the E. coli homolog of flaA (Fitzgerald et al. 2014).
The RNA sequencing data have revealed new processes controlled by RcsB. First, the expression of self-recognition systems involved in the territoriality of swarmer cells (ids, idr, tss) were repressed by RcsB. Since self-recognition systems are primarily observed under swarming permissive conditions, the increased expression of these genes when swarming has initiated would make biological sense. Second, expression of the speB gene, encoding agmatinase, was reduced 2.69-fold in the RcsB mutant (Table 1). SpeB is required for the production of putrescine and speB mutants exhibit both delay in the onset of differentiation and a crippled swarming phenotype.
RcsB also regulated expression of the major fimbrial subunits of the uroepithelial cell adhesion (UCA), the P. mirabilis fimbriae (PMF), and the mannose-resistant Proteus-like (M/RP) fimbriae, which were highly down-regulated in the rcsB mutant relative to wild type. Additional genes within each of these fimbrial operons were identified as Rcs-regulated. However, due to low levels of transcript reads, the p values for each gene were too high to be deemed significant, and they were not included. RcsB has been shown to directly bind to the promoter region of the mat fimbriae and activate its expression in the E. coli (Lehti et al. 2012). It also activates the transcription of the fim operon in E. coli by influencing the orientation of the fimAp invertible repeat (Schwan et al. 2007). In addition, fim genes are positively regulated by RcsB in S. enterica serovar typhimurium (Wang et al. 2007). Identification of major fimbrial subunits in P. mirabilis as RcsB regulated is important, as all three systems have been implicated in virulence. Though the contribution of P. mirabilis fimbriae to catheter adherence has not been well-characterized, their importance to the establishment of urinary tract infections has been recognized. Mutations in ucaA lead to impaired adhesion to uroepithelial cells and in the ability to colonize the kidneys of mice (Pellegrino et al. 2013). Mutants of pmfA have been shown to be deficient in bladder colonization in a mouse model of ascending urinary tract infection, but able to colonize the kidneys (Massad et al. 1994). Another study showed that pmfA mutants were unable to colonize the bladder and the kidneys (Zunino et al. 2003). The ability of a pmfA mutant to adhere to uroepithelial cells also differed between these two reports. The differences seen between these two studies could be due to the route of infection, different strains of mice, uroepithelial cells, and P. mirabilis. Mutants of mrpA reduce P. mirabilis colonization of the bladder, kidneys, and urine of mice (Bahrani et al. 1994; Zunino et al. 2001). M/RP fimbriae are also important for adherence to uroepithelial cells (Zunino et al. 2001) and biofilm formation in P. mirabilis (Jansen et al. 2004).
mrpJ has 14 paralogues in P. mirabilis strain HI4320, and many of them repress motility (Pearson and Mobley 2008). MrpJ itself has been shown to directly repress flhDC expression (Li et al. 2001; Bode et al. 2015). One mrpJ paralog, PMI0261, was highly repressed in the rcsB mutant based on our RNA sequencing data. The neighboring mrpH paralog, PMI0260, was also significantly down-regulated in the rcsB relative to wild type. Expression of the mrpA paralog of this operon (PMI0254) was also reduced in the rcsB mutant relative to wild type as well as other members of the paralog operon. Their exclusion is due to a high p value which resulted from low transcript levels. Why the two terminal genes of this operon produced more read transcript relative to the rest of operon members is unknown. An internal promoter could exist or perhaps more levels of mrpH and mrpJ are required to initiate transcription of the operon, as mrpJ binds and activates expression of mrpA (Bode et al. 2015), and assembly of the fimbriae, which mrpH is essential for (Li et al. 1999). However, PMI0256, encoding a fimbrial chaperone mrpD paralog, is a pseudogene due to the presence of a stop codon. This likely renders this mrp paralog non-functional. Overexpression of PMI0261 has been shown to reduce motility in P. mirabilis (Pearson and Mobley 2008) in the same manner as mrpJ, illustrating its function in counteracting motility is intact. This negative impact fimbrial activators have on motility makes biological sense as motile bacteria would not want to express adherence factors and vice versa.
The formation of biofilms was strongly impacted by the rcsB mutation. Two different biofilm assays were performed, one using polystyrene (Fig. 1a) and the other using catheter sections as biofilm substrates (Fig. 1b). With polystyrene as the substrate, the rcsB mutant formed no detectable biofilm after 8 h of incubation, formed 2.9-fold less biofilm than wild type after 24 h, and formed similar amounts of biofilm as wild type after 48 h of incubation. The wild-type strain exhibited a large reduction in biofilm formation between 24 and 48 h of incubation which was not as exaggerated in the rcsB mutant (Fig. 1a). The inability of the rcsB mutant to form biofilm was recapitulated when assays were performed on catheters. Again, it did not form detectable biofilm after 8 h of incubation, and formed 2.7-fold less biofilm than wild type after 24 h of incubation (Fig. 1b). The inability of the rcsB mutant to form biofilm initially may be due to the down-regulation of adherence factors, such as the major fimbrial subunits (ucaA, pmfA, and mrpA). Rcs mutants of Yersinia pseudotuberculosis are deficient in their ability to adhere to HEp-2 cells and form biofilm on abiotic surfaces; fimbrial proteins are a part of its RcsB regulon (Hinchliffe et al. 2008). Our RNA-seq data also showed that outer membrane protein A (ompA) is highly repressed in the rcsB mutant; it has been shown to bind abiotic surfaces, which could contribute to initial adherence (Lower et al. 2005). However, other factors are likely mediating adherence as the rcsB mutant does form biofilm after 24 h of incubation. Like wild-type cells, biofilm production of the rcsB mutant decreases after 48 h. Overall these data, combined with a previous report that a P. mirabilis rcsD mutant is deficient in biofilm production (Liaw et al. 2004), reiterate the contribution of the Rcs phosphorelay to biofilm formation in P. mirabilis. These results also suggest a preference for P. mirabilis to form biofilm on catheter versus polystyrene, a medically relevant finding, as the wild-type strain formed 6.8-fold more biofilm on catheter versus polystyrene after 8 h of incubation.
The virulence genes zapAB and hpmBA were strongly upregulated in the rcsB mutant; both have increased expression in swarming bacteria (Walker et al. 1999; Fraser et al. 2002). ZapA is a metalloprotease, which cleaves immunoglobulins (Senior et al. 1991), host cytoskeletal and matrix proteins, complement (C1q and C3), and antimicrobial peptides (human beta-defensin 1 and LL-37) (Belas et al. 2004). It is important for the colonization and subsequent infection of the urinary tract (Phan et al. 2008). HpmA functions as a hemolysin (Mobley et al. 1991; Chippendale et al. 1994) and induces cytotoxicity (Swihart and Welch 1990). Virulence proteins are RcsB regulated in Y. pseudotuberculosis, and genes within the Salmonella enterica serovar typhimurium SP-2 pathogenicity island are highly repressed by RcsB (Hinchliffe et al. 2008; Wang et al. 2007). Additionally, a mutant of S. enterica with a constitutively activated Rcs system is attenuated in virulence within a mouse (Garcia-Calderon et al. 2005). Based on these studies and our RNA-seq data, it was hypothesized that RcsB would regulate virulence in P. mirabilis. This was tested using a Galleria mellonella waxworm model. It should be noted that this model has not been previously used in P. mirabilis, although it has been used in other bacteria, where a good correlation has been observed with mammalian virulence (Jander et al. 2000; Seed and Dennis 2008; Murherjee et al. 2010; Fedhila et al. 2010; Ramarao et al. 2012). Moreover, the relevance of this model to a mouse model of urinary tract infection is unknown, but this model was only intended to measure general differences in virulence. One day after inoculation, the rcsB mutant had killed 94 % of worms versus 37 % for wild-type HI4320. By day three, the rcsB mutant had killed 96 % of worms versus 60 % of wild type.
A putative RcsB-binding sequence lies upstream of the hpmBA operon, but not upstream of the zap operon. RcsB has been shown to directly bind to the promoter region of the hemolysin homolog (shlBA) in Serratia marcescens (Di Venanzio et al. 2014), and our putative RcsB-binding site does overlap the σ28 promoter identified by Fraser et al. However, a limitation exists in our identification of putative RcsB-binding sites. By using NNNGANNNNCNNN as the input, many genomic hits were returned to due the sequence being nonspecific. The input sequence retaining only 3 of 14 base pairs is due to the variability in known RcsB-binding sites. The original RcsAB Box was defined as TaAGaataTCctA, with capital letters being the most conserved residues (Wehland and Bernhard 2000). This binding site was defined based on the RcsA-dependent RcsB-binding site of 12 promoters that were RcsAB regulated. It does not take into account sequence of RcsA-independent RcsB-binding sites of multiple genes. Presently, the RcsB-binding site in P. mirabilis is unknown.
The role of the Rcs phosphorelay in establishing CAUTIs has not been determined, but likely depends on whether the swarmer cell or swimmer cell is more important for colonization of the urinary tract. Conflicting data exist concerning this topic. Studies have shown that differentiated swarmer cells invade uroepithelial cells more efficiently than vegetative cells (Allison et al. 1992), and non-swarming mutants are unable to establish ascending urinary tract infections or kill mice (Allison et al. 1994; Mobley et al. 1996). Other studies have shown swimmer cells, not swarmer cells, are the dominant cell morphology in a mouse model of ascending urinary tract infection (Jansen et al. 2003) and non-flagellated P. mirabilis were isolated from a human infection (Zunino et al. 1994). Corroborating these studies is one that showed flagellar genes were down-regulated during the early stages of ascending urinary tract infection. Fimbrial genes, associated with swimmer cells, were upregulated during early infection (Pearson et al. 2011). Perhaps swarmer cells are needed to establish infection, as P. mirabilis readily moves across catheters (Sabbuba et al. 2002), but the antigenic potential due to flagellar components and cell length leads to immune recognition. This leads to the selection of swimmer cells in the urinary tract. The upregulation of fimbriae during colonization offers another set of antigens subject to immune recognition. To counteract this, P. mirabilis may differentiate into swarmer cells during later stages of infection, consistent with a previous study where fimbrial gene expression decreased while the expression of some flagellar genes increased during the later stages of a mouse ascending urinary tract infection (Pearson et al. 2011). The possibility of cycling differentiation and consolidation during infection reiterates the origin of the name Proteus, named for a god who changed shape to avoid capture and questioning. The interplay of the Rcs phosphorelay, together with additional important genetic factors contributes to this elusive change in cell morphology and affects motility, biofilm formation, and virulence in P. mirabilis.
References
Allison C, Coleman N, Jones PL, Hughes C (1992) Ability of Proteus mirabilis to invade human urothelial cells is coupled to motility and swarming differentiation. Infect Immun 60:4740–4746
Allison C, Emody L, Coleman N, Hughes C (1994) The role of swarm cell differentiation and multicellular migration in the uropathogenicity of Proteus mirabilis. J Infect Dis 169:1155–1158
Alteri CJ, Himpsl SD, Pickens SR, Lindner JR, Zora JS, Miller JE, Arno PD, Straight SW, Mobley HLT (2013) Multicellular bacteria deploy the type VI secretion system to preemptively strike neighboring cells. PLoS Pathog 9(9):e1003608
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Armitage JP (1981) Changes in metabolic activity of Proteus mirabilis during swarming. J Gen Microbiol 125:445–450
Bahrani FK, Mobley HLT (1994) Proteus mirabilis MR/P fimbrial operon: genetic organization, nucleotide sequence, and conditions for expression. J Bacteriol 176(11):3412–3419
Bahrani FK, Massad G, Lockatell CV, Johnson DE, Russell RG, Warren JW, Mobley HLT (1994) Construction of an MR/P fimbrial mutant of Proteus mirabilis role in virulence in a mouse model of ascending urinary tract infection. Infect Immun 176:3412–3419
Belas R, Flaherty D (1994) Sequence and genetic analysis of multiple flagellin-encoding genes from Proteus mirabilis. Gene 128:33–41
Belas R, Suvanasuthi R (2005) The Ability of Proteus mirabilis to sense surfaces and regulate virulence gene expression involves FliL, a flagellar basal body protein. J Bacteriol 187(19):6789–6803
Belas R, Goldman M, Ashliman K (1995) Genetic analysis of Proteus mirabilis mutants defective in swarmer cell elongation. J Bacteriol 177:823–828
Belas R, Schneider R, Melch M (1998) Characterization of Proteus mirabilis precocious swarming mutants: identification of rsbA, encoding a regulator of swarming behavior. J Bacteriol 180(23):6126–6139
Belas R, Manos J, Suvanasuthi R (2004) Proteus mirabilis ZapA metalloprotease degrades a broad spectrum of substrates, including antimicrobial peptides. Infect Immun 72(9):5159–5167
Berne C, Kysela DT, Brun YV (2010) A bacterial extracellular DNA inhibits settling of motile progeny cells within a biofilm. Mol Microbiol 77(4):815–829
Bi EF, Lutkenhaus J (1991) FtsZ ring structure associated with division in Escherichia coli. Nature 354:161–164
Bode NJ, Debnath I, Kuan L, Schulfer A, Ty M, Pearson MM (2015) Transcriptional analysis of the MrpJ network: modulation of diverse virulence-associated genes and direct regulation of mrp fimbrial and flhDC flagellar operons in Proteus mirabilis. Infect Immun 83(6):2542–2556
Brill JA, Quinlan-Walshe C, Gottesman S (1988) Fine-structure mapping and identification of two regulators of capsule synthesis in Escherichia coli K-12. J Bacteriol 170:2599–2611
Carballès F, Bertrand C, Bouche JP, Cam K (1999) Regulation of Escherichia coli cell division genes ftsA and ftsZ by the two-component system rcsC–rcsB. Mol Microbiol 34(3):442–548
Castanie-Cornet MP, Cam K, Jacq A (2006) RcsF is an outer membrane lipoprotein involved in the RcsCDB phosphorelay signaling pathway in Escherichia coli. J Bacteriol 188:4264–4270
Chippendale GR, Warren JW, Trifillis AL, Mobley HLT (1994) Internalization of Proteus mirabilis by human renal epithelial cells. Infect Immun 62(8):3115–3121
Claret L, Hughes C (2000a) Functions of the subunits of FlhD2C2 transcriptional master regulator of bacterial flagellum biogenesis and swarming. J Mol Biol 303:467–478
Claret L, Hughes C (2000b) Rapid turnover of FlhD and FlhC, the flagellar regulon transcriptional activator proteins, during Proteus swarming. J Bacteriol 182(3):833–836
Claret L, Hughes C (2002) Interaction of the atypical prokaryotic transcription activator FlhD2C2 with early promoters of the flagellar gene hierarchy. J Mol Biol 321(2):185–199
Clemmer KM, Rather PN (2008a) Regulation of flhDC expression in Proteus mirabilis. Res Microbiol 158:295–302
Clemmer KM, Rather PN (2008b) The Lon protease regulates swarming motility and virulence gene expression in Proteus mirabilis. J Med Microbiol 57(8):931–937
Davalos-Garcia M, Conter A, Toesca I, Gutierrez C, Cam K (2001) Regulation of osmC gene expression by the two component system rcsB-rcsC in Escherichia coli. J Bacteriol 183:5870–5876
de Boer PA, Crossley RE, Rothfield LI (1989) A division inhibitor and a topological specificity factor coded for by the minicell locus determine proper placement of the division septum in E. coli. Cell 56:641–649
de Boer PA, Crossley RE, Rothfield LI (1990) Central role for the Escherichia coli minC gene product in two different cell division-inhibition systems. Proc Natl Acad Sci USA 87:1129–1133
Di Venanzio G, Stepanenko TM, Garcia Vescovi E (2014) Serratia marcescens ShlA pre-forming toxin is responsible for early induction of autophagy in host cells and is transcriptionally regulated by RcsB. Infect Immun 82(9):3542–3554
Dienes L (1946) Reproductive processes in Proteus cultures. Proc Soc Exp Biol Med 63:265–270
Dufour A, Furness RB, Hughes C (1998) Novel genes that upregulate the Proteus mirabilis flhDC master operon controlling flagellar biogenesis and swarming. Mol Microbiol 29:741–751
Fedhila S, Buisson C, Dussurgent C, Serror P, Glomsk IJ, Leihl P, Lereclus C, Nielson-LeRoux C (2010) Comparative analysis of the virulence of invertebrate and mammalian pathogenic bacteria in the oral insect infection model Galleria mellonella. J Invertebr Pathol 103:24–29
Ferrières L, Clarke DJ (2003) The RcsC sensor kinase is required for normal biofilm formation in Escherichia coli K-12 and controls the expression of a regulon in response to growth on a solid surface. Mol Microbiol 50:1665–1682
Fitzgerald DM, Bonocora RP, Wade JT (2014) Comprehensive mapping of the Escherichia coli flagellar regulatory network. PLoS Genet 10(10):e1004649
Francez-Charlot A, Laugel B, VanGemert A, Dubarry N, WiorowskiF Castanié-Cornet MP, Gutierrez C, Cam K (2003) RcsCDB His-Asp phosphorelay system negatively regulates the flhDC operon in Escherichia coli. Mol Microbiol 49:823–832
Fraser GM, Hughes C (1999) Swarming motility. Curr Opin Microbiol 2:630–635
Fraser GM, Claret L, Furness R, Gupta S, Hughes C (2002) Swarming-coupled expression of the Proteus mirabilis hpmBA haemolysin operon. Microbiol. 148(7):2191–2201
Furness RB, Fraser GM, Hay NA, Hughes C (1997) Negative feedback from a Proteus Class II flagellum export defect to the flhDC master operon controlling cell division and flagellum assembly. J Bacteriol 179:5585–5588
Garcia-Calderon CB, Garcia-Quintanilla M, Casadesus J, Ramos-Morales F (2005) Virulence attenuation in Salmonella enterica rcsC mutants with constitutive activation of the Rcs system. Microbiol 151(Pt 2):579–588
Gervais FG, Drapeau GR (1992) Identification, cloning, and characterization of rcsF, a new regulator gene for exopolysaccharide synthesis that suppresses the division mutation ftsZ84 in Escherichia coli K-12. J Bacteriol 174:8016–8022
Gervais FG, Phoenix P, Drapeau GR (1992) The rcsB gene, a positive regulator of colonic acid biosynthesis in Escherichia coli, is also an activator of ftsZ expression. J Bacteriol 174:3964–3971
Gibbs KA, Urbanowski ML, Greenberg EP (2008) Genetic determinants of self-identity and social recognition in bacteria. Science 321(5886):256–259
Gottesman S, Trisler P, Torres-Cabassa AS (1985) Regulation of capsular polysaccharide synthesis in Escherichia coli K12: characterization of three regulatory genes. J Bacteriol 162:1111–1119
Gygi D, Bailey MJ, Allison C, Hughes C (1995) Requirement for FlhA in flagella assembly and swarm cell differentiation by Proteus mirabilis. Mol Microbiol 15:761–769
Hagiwara D, Sugiura M, Oshima T, Mori H, Aiba H, Yamashino T, Mizuno T (2003) Genome-wide analyses revealing a signaling network of the RcsC-YojN-RcsB phosphorelay system in Escherichia coli. J Bacteriol 185(19):5735–5746
Hay NA, Tipper DJ, Gygi D, Hughes C (1999) A Novel Membrane Protein Influencing Cell Shape and Multicellular Swarming of Proteus mirabilis. J Bacteriol 181(7):2008–2016
Hinchliffe SJ, Howard SL, Huang YH, Clarke DJ, Wren BW (2008) The importance of the Rcs phosphorelay in the survival and pathogenesis of the enteropathogenic yersiniae. Microbiol 154(4):1117–1131
Hoeniger JFM (1965) Development of flagella by Proteus mirabilis. J Gen Microbiol 40:29–42
Howery KE, Clemmer KM, Simsek E, Kim M, Rather PN (2015) Regulation of the Min cell division inhibition system by the Rcs phosphorelay in Proteus mirabilis. J Bacteriol 197(15):2499–2507
Jackson DW, Suzuki K, Oakford L, Simecka JW, Hart ME, Romeo T (2002) Biofilm formation and dispersal under the influence of the global regulator CsrA of Escherichia coli. J Bacteriol 184(1):290–301
Jacobsen SM, Stickler DJ, Mobley HL, Shirtliff ME (2008) Complicated catheter-associated urinary tract infections due to Escherichia coli and Proteus mirabilis. Clin Microbiol Rev 21(1):26–59
Jander G, Rahme LG, Ausubel FM (2000) Positive correlation between virulence of Pseudomonas aeruginosa mutants in mice and insects. J Bacteriol 182:3843–3845
Jansen AM, Lockatell CV, Johnson DE, Mobley HL (2003) Visualization of Proteus mirabilis morphotypes in the urinary tract: the elongated swarmer cell is rarely observed in ascending urinary tract infection. Infect Immun 71:3607–3950
Jansen AM, Lockatell V, Johnson DE, Mobley HLT (2004) Mannose-resistant proteus-like fimbriae are produced by most Proteus mirabilis strains infecting the urinary tract, dictate the in vivo localization of bacteria, and contribute to biofilm formation. Infect Immun 72:7294–7305
Jones BV, Young R, Mahenthiralingam E, Stickler DJ (2004) Ultrastructure of Proteus mirabilis swarmer cell rafts and the role of swarming in catheter-associated urinary tract infections. Infect Immun 72:3941–3950
Kovács AT (2016) Bacterial differentiation via the gradual activation of global regulators. Curr Genet 62:125–128
Lehti TA, Heikkinen J, Korhonen TK, Westerlund-Wikström B (2012) The response regulator RcsB activates expression of Mat fimbriae in meningitic Escherichia coli. J Bacteriol 194(13):3475–3485
Li X, Johnson DE, Mobley HLT (1999) Requirement of MrpH for mannose-resistant Proteus-like fimbriae-mediated hemagglutination by Proteus mirabilis. Infect Immun 67(6):2822–2833
Li X, Rasko DA, Lockatell CV, Johnson DE, Mobley HLT (2001) Repression of bacterial motility by a novel fimbrial gene product. EMBO J 20:4854–4862
Liaw SJ, Lia HC, Ho SW, Luh KT, Wang WB (2001) Characterisation of p-nitrophenylglycerol-resistant Proteus mirabilis super-swarming mutants. J Med Microbiol 50(12):1039–1048
Liaw SJ, Lai HC, Wang WB (2004) Modulation of swarming and virulence by fatty acids through the RsbA protein in Proteus mirabilis. Infect Immun 72(12):6836–6845
Lower BH, Yongsunthon R, Vellano FP, Lower SK (2005) Simultaneous force and fluorescence measurements of a protein that forms a bond between a living bacterium and a solid surface. J Bacteriol 187(6):2127–2137
Majdalani N, Hernandez D, Gottesman S (2002) Regulation and mode of action of the second small RNA activator of RpoS translation. RprA Mol Microbiol 46:813–826
Majdalani N, Heck M, Stout V, Gottesman S (2005) Role of RcsF in signaling to the Rcs phosphorelay pathway in Escherichia coli. J Bacteriol 187:6770–6778
Massad G, Lockattell CV, Johson DE, Mobley HLT (1994) Proteus mirabilis fimbriae: construction of an isogenic pmfA mutant and analysis of virulence in a CBA mouse model of ascending urinary infection. Infect Immun 62:536–542
Matsukawa M, Kunishima Y, Takahashi S, Takeyama K, Tsukamoto T (2005) Bacterial colonization on intraluminal surface of urethral catheter. Urology 65:440–444
Mobley HL, Chippendale GR, Tenney JH, Mayrer AR, Crisp LJ, Penner JL, Warren JW (1988) MR/K hemagglutination of Providencia stuartii correlates with adherence to catheters and with persistence in catheter-associated bacteriuria. J Infect Dis 157(2):264–271
Mobley HL, Chippendale GR, Swihart KG, Welch RA (1991) Cytotoxicity of the HpmA hemolysin and urease of Proteus mirabilis and Proteus vulgaris against cultured human renal proximal tubular epithelial cells. Infect Immun 59:2036–2042
Mobley HL, Belas R, Lockatell V, Chippendale G, Trifillis AL, Johnson DE, Warren JW (1996) Construction of a flagellum-negative mutant of Proteus mirabilis: effect on internalization by human renal epithelial cells and virulence in a mouse model of ascending urinary tract infection. Infect Immun 64(12):5332–5340
Morgenstein RM, Clemmer KM, Rather PN (2010) Loss of the waaL O-antigen ligase prevents surface activation of the flagellar gene cascade in Proteus mirabilis. J Bacteriol 192(12):3213–3221
Mrázek J, Xie S (2006) Pattern locator: a new tool for finding local sequence patterns in genomic DNA sequences. Bioinformatics 22:3099–3100
Murherjee K, Altincicek B, Hain T, Domann E, Vilcinskas A, Chakraborty T (2010) Galleria mellonella as a model system for studying Listeria pathogenesis. Appl Environ Microbiol 76:310–317
O’Hara CM, Brenner FW, Miller JM (2000) Classification, identification, and clinical significance of Proteus, Providencia, and Morganella. Clin Microbiol Rev 13(4):534–546
Pearson MM, Mobley HLT (2008) Repression of motility during fimbrial gene expression: identification of 14 mrpJ gene paralogues in Proteus mirabilis. Mol Microbiol 69:548–558
Pearson MM, Sebaihia M, Churcher C, Quail MA, Seshasayee AS, Luscombe NM, Abdellah Z, Arrosmith C, Atkin B, Chillingworth T, Hauser H, Jagels K, Moule S, Mungall K, Norbertczak H, Rabbinowitsch E, Walker D, Whithead S, Thomson NR, Rather PN, Parkhill J, Mobley HL (2008) Complete genome sequence of uropathogenic Proteus mirabilis, a master of both adherence and motility. J Bacteriol 190(11):4027–4037
Pearson MM, Rasko DA, Smith SN, Mobley HLT (2010) Transcriptome of swarming Proteus mirabilis. Infect Immun 78:2834–2845
Pearson MM, Yep A, Smith SN, Mobley HL (2011) Transcriptome of Proteus mirabilis in the murine urinary tract: virulence and nitrogen assimilation gene expression. Infect Immun 79(7):2619–2631
Pellegrino R, Scavone P, Umpiérrez A, Maskell DJ, Zunino P (2013) Proteus mirabilis uroepithelial cell adhesin (UCA) fimbria plays a role in the colonization of the urinary tract. Pathog Dis 67(2):104–107
Phan V, Belas R, Gilmore BF, Ceri H (2008) ZapA, a virulence factor in a rat model of Proteus mirabilis-induced acute and chronic prostatitis. Infect Immun 76(11):4859–4864
Pichoff S, Lutkenhaus J (2005) Tethering the Z ring to the membrane through a conserved membrane targeting sequence in FtsA. Mol Microbiol 55(6):1722–1734
Pristovsek P, Sengupta K, Lohr F, Schafer B, von Trebra MW, Ruterjans H, Bernhard F (2003) Structural analysis of the DNA-binding domain of the Erwinia amylovora RcsB protein and its interaction with the RcsAB box. J Biol Chem 278:17752–17759
Ramarao N, Nielsen-Leroux C, Lereclus D (2012) The insect Galleria mellonella as a powerful infection model to investigate bacterial pathogenesis. J Visual Exp 11(70):e4392
Rauprich O, Matsushita M, Weijer CJ, Siegert F, Esipov SE, Shapiro JA (1996) Periodic phenomena in Proteus mirabilis swarm colony development. J Bacteriol 178:6525–6538
Roberts JA, Fussell EN, Kaack MB (1990) Bacterial adherence to urethral catheters. J Urol 144:264–269
Rocha S, Elias W, Cianciarullo A, Menezes M, Nara J, Piazza R, Silva M, Moreira C, Pelayo J (2007) Aggregative adherence of uropathogenic Proteus mirabilis to cultured epithelial cells. FEMS Immun Med Microbiol 51(2):319–326
Sabbuba N, Hughes G, Stickler DJ (2002) The migration of Proteus mirabilis and other urinary tract pathogens over Foley catheters. BJU Int 89(1):55–60
Schwan WR, Shibata S, Aizawa S, Wolfe AJ (2007) The two-component response regulator RcsB regulates type 1 piliation in Escherichia coli. J Bacteriol 189(19):7159–7163
Seed KD, Dennis JJ (2008) Development of Galleria mellonella as an alternative infection model for the Burkholderia cepacia complex. Infect Immun 76:1267–1275
Senior BW, Loomes LM, Kerr MA (1991) The production and activity in vivo of Proteus mirabilis IgA protease in infections of the urinary tract. J Med Microbiol 35(4):203–207
Stevenson LG, Rather PN (2006) A Novel gene involved in regulating the flagellar gene cascade in Proteus mirabilis. J Bacteriol 188:7830–7839
Stickler DJ, Lear JC, Morris NS, Macleod SM, Downer A, Cadd DH, Feast WJ (2006) Observations on the adherence of Proteus mirabilis onto polymer surfaces. J Appl Microbiol 100:1028–1033
Stout V, Gottesman S (1990) RcsB and RcsC: a two-component regulator of capsule synthesis in Escherichia coli. J Bacteriol 172:659–669
Stout V, Torres-Cabassa A, Maurizi MR, Gutnick D, Gottesman S (1991) RcsA, an unstable positive regulator of capsular polysaccharide synthesis. J Bacteriol 173:1738–1747
Sturgill G, Rather PN (2004) Evidence that putrescine acts as an extracellular signal required for swarming in Proteus mirabilis. Mol Micobiol 51:437–446
Swihart KG, Welch RA (1990) Cytotoxic activity of the Proteus hemolysin HpmA. Infect Immun 58(6):1861–1869
Takeda S, Fujisawa Y, Matsubara M, Aiba H, Mizuno T (2001) A novel feature of the multistep phosphorelay in Escherichia coli: a revised model of the RcsC → YojN → RcsB signalling pathway implicated in capsular synthesis and swarming behavior. Mol Microbiol 40:440–450
Walker KE, Moghaddame-Jafari S, Lockatell CV, Johnson D, Belas R (1999) ZapA, the IgA-degrading metalloprotease of Proteus mirabilis, is a virulence factor expressed specifically in swarmer cells. Mol Microbiol 32(4):825–836
Wang Q, Zhao Y, McClelland M, Harshey RM (2007) The RcsCDB signaling system and swarming motility in Salmonella enterica serovar typhimurium: dual regulation of flagellar and SPI-2 virulence genes. J Bacteriol 189(23):8447–8457
Wang WB, Chen IC, Jiang SS, Chen HR, Hsu CY, Hseuh PR, Hsu WB, Liaw SJ (2008) Role of RppA in the regulation of polymyxin B susceptibility, swarming and virulence factor expression in Proteus mirabilis. Infect Immun 76:2051–2062
Ward JE, Lutkenhaus J (1985) Overproduction of FtsZ induces minicell formation in E. coli. Cell 42:941–949
Wehland M, Bernhard F (2000) The RcsAB box. Characterization of a new operator essential for the regulation of exopolysaccharide biosynthesis in enteric bacteria. J Biol Chem 275(10):7013–7020
Wenren LM, Sullivan NL, Cardarelli L, Septer AN, Gibbs KA (2013) Two independent pathways for self-recognition in Proteus mirabilis are linked by type VI-dependent export. Mbio 4(4):e374–e413
Yakubu DE, Old DC, Senior BW (1989) The haemagglutinins and fimbriae of Proteus penneri. J Med Microbiol 30(4):279–284
Zunino P, Piccini C, Legnani-Fajardo C (1994) Flagellate and non-flagellate Proteus mirabilis in the development of experimental urinary tract infection. Microb Pathog 16:379–385
Zunino P, Geymonat L, Allen AG, Preston A, Sosa V, Maskell DJ (2001) New aspects of the role of MR/P fimbriae in Proteus mirabilis urinary tract infection. FEMS Immunol Med Microbiol 31:113–120
Zunino P, Sosa V, Allen AG, Preston A, Schlapp G, Maskell DJ (2003) Proteus mirabilis fimbriae (PMF) are important for both bladder and kidney colonization in mice. Microbiol. 149(11):3231–3237
Acknowledgments
This work was supported by a Merit Review award 1IOIBX001725 and by a Research Career Scientist award to P.N.R., both from the Department of Veterans Affairs. K.H. was supported by the Burroughs Wellcome Fund’s Molecules to Mankind program. The authors thank Lewis Jordan, PhD, for aid in the bioinformatic identification of RcsB-binding sites.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Kupiec.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Howery, K.E., Clemmer, K.M. & Rather, P.N. The Rcs regulon in Proteus mirabilis: implications for motility, biofilm formation, and virulence. Curr Genet 62, 775–789 (2016). https://doi.org/10.1007/s00294-016-0579-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-016-0579-1