Abstract
Purpose
Ramucirumab plus docetaxel improved survival in REVEL, a randomized phase 3 trial for patients with Stage IV non-small cell lung cancer after standard platinum-based chemotherapy. This exploratory analysis evaluated the exposure–response relationship of ramucirumab from REVEL.
Methods
Patients received ramucirumab (10 mg/kg) or placebo plus docetaxel (75 mg/m2) every 3 weeks. Pharmacokinetic samples were collected. A population pharmacokinetic analysis predicted ramucirumab minimum concentration after first-dose administration (Cmin,1) and average concentration at steady state (Cave,ss). Predicted Cmin,1 and Cave,ss were used to evaluate the relationship between ramucirumab exposure and efficacy and safety, respectively. Exposure–efficacy was assessed by Kaplan–Meier and Cox regression analyses; exposure–safety was assessed by ordered categorical analyses.
Results
Analyses included 376 patients treated with ramucirumab plus docetaxel and 366 patients treated with placebo plus docetaxel (364 for safety population). After adjusting for corresponding prognostic factors, the association between overall survival (OS) and Cmin,1 was statistically significant (p = 0.0110), although progression-free survival (PFS) showed a marginal association (p = 0.0515). At high ramucirumab exposures (Cmin,1), greater improvements (smaller hazard ratios) were seen for OS and PFS when stratified by Cmin,1 exposure quartiles. A statistically significant correlation was observed between ramucirumab Cave,ss and grade ≥ 3 febrile neutropenia and hypertension.
Conclusions
An association was observed between ramucirumab exposure and efficacy. Higher ramucirumab exposure was associated with improved clinical outcomes and increased toxicity in this analysis. Two exposure–response prospective randomized trials are being conducted to address causation (NCT02443883 and NCT02514551), with encouraging preliminary results (Ajani et al. in Ann Oncol 28:abstr 698P, 2017).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ramucirumab is a recombinant human immunoglobulin G subclass 1 (IgG1) neutralizing monoclonal antibody (mAb) specific for vascular endothelial growth factor receptor-2 (VEGFR-2) [1]. The maximum tolerated dose of ramucirumab as a single agent is 13 mg/kg per week [2]. Safety and efficacy of ramucirumab in patients with previously treated Stage IV non-small cell lung cancer (NSCLC) were evaluated in a randomized, double-blind phase 3 trial (REVEL) comparing ramucirumab or placebo in combination with docetaxel [3]. Overall survival and progression-free survival significantly improved in the ramucirumab arm. The four most common grade ≥ 3 treatment-emergent adverse events (TEAEs) occurring in ≥ 5% of patients treated with ramucirumab plus docetaxel with a > 2% difference in the incidence rate between the ramucirumab arm and placebo arm were neutropenia, febrile neutropenia, hypertension, and fatigue [3]. Based on the outcome of this clinical trial, ramucirumab was approved for use in combination with docetaxel for the treatment of patients with previously treated advanced NSCLC [4].
Exposure–response analyses are at the core of any determination of the safety and effectiveness of drugs [5]. Monoclonal antibodies (mAbs) have been shown to be effective anticancer agents, but assessing the pharmacokinetics (PK) of mAbs is challenging [5, 6]. Often, the maximum tolerated dose is not determined in phase 1 trials, and measuring target occupancy in cancer tissues is not easily accomplished. Indeed, the heterogeneous distribution of systemically administered antibodies in tumor tissue is a well-known concern with the use of mAbs [6]. The variable PK of several mAbs used to treat different tumor types has provided data for potentially improving their individual dosing strategies [7,8,9,10]. The exposure–response analyses of ramucirumab in patients with gastric cancer and in patients with advanced colorectal cancer have shown an association between higher exposure to ramucirumab and longer overall survival and progression-free survival, as well as an increased risk of adverse events [11, 12]. Similarly, a study of four ramucirumab dosing regimens in patients with advanced gastric/gastroesophageal junction adenocarcinoma showed some trends towards improved progression-free survival and overall survival with the higher doses of ramucirumab versus the standard regimen, with safety profiles comparable to the standard regimen [13].
We conducted exposure–efficacy and exposure–safety analyses on data from patients treated with ramucirumab plus docetaxel from the REVEL trial. The objectives of these analyses were to evaluate the relationships between ramucirumab exposure and overall survival, progression-free survival, and commonly reported TEAEs in patients with previously treated Stage IV NSCLC.
Materials and methods
Patients and samples
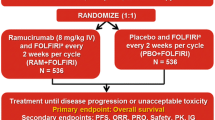
REVEL was a randomized, placebo-controlled, phase 3 clinical trial that included patients with Stage IV NSCLC whose disease had progressed during or after one prior first-line platinum-based chemotherapy regimen (with or without bevacizumab or maintenance therapy) for advanced/metastatic disease. Patients were randomized (1:1) to receive ramucirumab (10 mg/kg) or placebo in combination with docetaxel (75 mg/m2) on day 1 of a 21-day (3-week) cycle, as previously described [3]. The primary endpoint was overall survival, and key secondary endpoints included progression-free survival, analysis of ramucirumab PK parameters, and safety. The blood collection for PK analysis ranged from limited to extensive sampling, and the samples were collected at baseline, pre-infusion, and 1-h post-infusion at cycles 3, 5, 9, and 13, and at the 30-day safety follow-up. The details of the REVEL trial, including informed consent, trial design, and clinical definitions of overall survival and progression-free survival, have been previously described [3].
A population PK model was developed as previously described [14]. Predicted ramucirumab exposure parameters [minimum concentration after first-dose administration (Cmin,1) and average concentration at steady state (Cave,ss)] were determined for ramucirumab-treated patients using a nonlinear mixed-effects modeling approach [NONMEM VI (ICON, Ellicott City, MD)]. Analyses were conducted in accordance with the United States Food and Drug Administration (FDA) Guidance for Industry on Population Pharmacokinetics [5].
The first post-baseline PK sample was collected before the cycle three infusion. Therefore, only patients who received ≥ 3 doses of treatment (both arms) and had evaluable PK data were included in exposure–response analyses. Patients who discontinued before dose 3 of placebo were excluded from the placebo group.
Exposure–efficacy analyses
The exposure–efficacy analyses were performed using Cmin,1 as both continuous and categorical (quartiles) values. Univariate and multivariable Cox regression analyses were performed to evaluate the exposure–efficacy relationship between ramucirumab Cmin,1 and efficacy outcomes (overall survival and progression-free survival). Imbalances in prognostic factors associated with progression-free survival or overall survival were adjusted in a multivariable Cox regression analysis. Factors with potential prognostic significance were identified using a stepwise Cox regression with an entry p value of 0.05 and an exit p value of 0.1 and were Eastern Cooperative Oncology Group performance status (ECOG PS; 0 versus 1); sex (females versus males); prior maintenance therapy (yes versus no); geographic region [region 1: Korea and Taiwan versus region 2: rest of the world (North America, South America, European Union, India, and Australia)]; smoking history (never versus ever); histology (nonsquamous versus squamous); best response to platinum-based chemotherapy [complete response (CR)/partial response (PR)/stable disease (SD) versus progressive disease (PD)]; prior taxane treatment (no versus yes); prior bevacizumab treatment (no versus yes); epithelial growth factor receptor (EGFR) status (wild type versus mutation versus unknown); age (< 65 years versus age ≥ 65 years); race (white versus black versus other); and time since prior therapy (< 9 versus ≥ 9 months).
To evaluate the exposure–response relationship compared with the control group, patients in the exposed population were stratified into defined Cmin,1 exposure quartiles (Q): Q1, Cmin,1 6.26–≤ 15.7 µg/mL (< 25%); Q2, Cmin,1 > 15.7–≤20.7 µg/mL (25–< 50%); Q3, Cmin,1 > 20.7–≤ 27.9 µg/mL (50–< 75%); Q4, Cmin,1 > 27.9–56.4 µg/mL (≥ 75%) (Supplemental Fig. 1). Overall survival and progression-free survival were assessed with the Kaplan–Meier method comparing each ramucirumab Cmin,1 quartile with data from the placebo plus docetaxel patients. The hazard ratios (HRs) for each quartile versus the control arm were estimated using a Cox proportional hazard model adjusted for baseline covariates.
Additional matched case–control analyses for overall survival were explored to adjust for potential imbalances in important prognostic factors between the treatment arms within each exposure quartile group as previously described [15]. In this analysis, the case groups are the four ramucirumab Cmin,1 exposure quartiles. For every patient in each exposure quartile group, a matched control patient was identified from all patients receiving placebo plus docetaxel through a matching scheme based on the significant potential prognostic factors identified in the stepwise Cox regression analysis. The Mahalanobis metric matching technique was employed [16]. The balance of the prognostic factors between the two treatment arms was assessed in each case–control group, before and after matching, using Fisher’s exact test. Missing values in any of the matching factors excluded the patients from the matched case–control study. Kaplan–Meier survival analysis and Cox models were performed to compare the two treatment arms in each of the four matched case–control groups.
The statistical analyses were conducted with SAS software (Version 9.1.2 or higher; SAS Institute, Cary, NC) or comparable software.
Exposure–safety analyses
The safety endpoints for the exposure–safety analysis were febrile neutropenia, neutropenia, fatigue, and hypertension. These were the four most common grade ≥ 3 TEAEs from the REVEL study occurring in ≥ 5% of patients in the ramucirumab plus docetaxel arm and with a > 2% difference in incidence rate between the ramucirumab arm and placebo arm. All TEAEs were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.0, as previously described [3]. Ordered categorical models were developed to explore the relationship between the predicted average concentration of ramucirumab at steady state (Cave,ss) and TEAEs.
Results
Exposure–response population
Of the 625 patients randomized to the placebo plus docetaxel (placebo) arm in the phase 3 REVEL trial, 366 patients (59%) received at least three cycles of treatment. However, two of these patients received one dose of ramucirumab in error instead of placebo. These two patients were included in the placebo arm for the exposure–efficacy population and excluded from the placebo arm for the exposure–safety population (n = 364; 58%) [3]. Among the 628 patients randomized to the ramucirumab plus docetaxel (ramucirumab) arm, 376 (60%) patients received at least three cycles of ramucirumab and had available ramucirumab concentration data; the rest either discontinued before the third dose of ramucirumab was administered (210 patients), or the PK data were not available (42 patients) [3]. The baseline characteristics of the factors evaluated for potential prognostic significance (ECOG PS, sex, prior maintenance therapy, geographic region, smoking history, histology, best response to platinum therapy, prior taxane, prior bevacizumab, EGFR status, age, race, and time since start of prior therapy) were similar between the treatment and placebo arms in the exposure–response population and consistent with the intent-to-treat (ITT) population, suggesting that the exposure–response population is reflective of the ITT population enrolled in REVEL (Table 1).
Exposure–efficacy
A univariate Cox regression analysis with Cmin,1 as the continuous covariate with data from the treatment arm showed significant association between Cmin,1 and overall survival (p = 0.0004). A stepwise Cox regression identified factors significantly associated with overall survival of ECOG PS, sex, geographic region, histology, best response to platinum-based therapy, and time since prior therapy. After adjusting for these factors, the association between overall survival and Cmin,1 remained statistically significant (p = 0.0110). Cmin,1 was also significantly associated with progression-free survival (p = 0.0057). After adjusting for covariates significantly associated with progression-free survival (ECOG PS, sex, and time since prior therapy), a similar association between Cmin,1 and progression-free survival was observed, although the association was marginal (p = 0.0515).
For comparison with the control group, Cmin,1 was distributed into quartiles to examine the relationship between exposure and efficacy. Kaplan–Meier plots demonstrated separation between the overall survival curves for the Cmin,1 quartiles (Fig. 1a). Median overall survival was 11.1, 14.6, 12.6, 17.1, and 13.3 months for Cmin,1 Q1, Q2, Q3, Q4, and placebo, respectively. Median progression-free survival was 5.6, 7.0, 5.7, 7.0, and 5.5 months for Cmin,1 Q1, Q2, Q3, Q4, and placebo, respectively (Table 2). Longer overall survival favoring ramucirumab was observed in Q2 and Q4 compared to placebo. The Kaplan–Meier plots for progression-free survival also showed a similar trend (Fig. 1b). For progression-free survival, ramucirumab was favored in Q2 and Q4 (HR 0.71 for each) as compared to placebo. By contrast, patients in the lowest exposure quartile had the largest HRs for both overall survival (HR 1.19, 95% CI 0.89, 1.59) and progression-free survival (HR 0.92, 95% CI 0.72, 1.18).
Kaplan–Meier analysis of overall survival and progression-free survival in REVEL exposure–response population by ramucirumab Cmin,1 quartile. Overall survival (a) and progression-free survival (b) in the REVEL exposure–response population stratified by Cmin,1 quartile and each compared with patients treated with docetaxel plus placebo who had at least three cycles of treatment. C min,1 minimum concentration following the first dose, DOC docetaxel, PBO placebo, Q quartile, Q1, C min,1 6.26–≤ 15.7 µg/mL (< 25%), Q2, C min,1 > 15.7–≤ 20.7 µg/mL (25–< 50%), Q3, C min,1 > 20.7–≤ 27.9 µg/mL (50–< 75%), Q4, C min,1 > 27.9–56.4 µg/mL (≥ 75%), RAM ramucirumab
Several baseline characteristics were slightly different among the quartiles (Supplemental Table 1). Therefore, we adjusted for the potential impact of imbalances in baseline characteristics and important prognostic factors between the treatment arms within each exposure group using a matched case–control analysis for overall survival. Seven matching factors with prognostic significance associated with overall survival were identified using a stepwise Cox regression with entry p value of 0.05 and exit p value of 0.1 and adjusted for sex (female versus male), ECOG PS at baseline (0 versus 1), geographic region (Japan/East Asia versus rest of world), histology (nonsquamous versus squamous), best response to platinum-based chemotherapy (CR/PR/SD versus PD), best response to platinum-based chemotherapy (missing versus PD), and time since prior therapy (< 9 versus ≥ 9 months). The matching was performed separately within each of the four Cmin,1 exposure quartiles in the ramucirumab plus docetaxel arm (Supplemental Table 2). The two treatment arms in each of the four matched case–control groups were compared using Kaplan–Meier methodology for overall survival (Fig. 2). Overall, there was a trend for overall survival HRs to decrease with increasing ramucirumab exposure. The analysis demonstrated clinical benefits in Q2, Q3, and Q4 compared to matched control, although Q1 showed results similar to the control (Fig. 2). Among the four quartile groups, the higher exposure quartile groups (Q2–Q4) were generally associated with longer survival and HR < 1 (Fig. 2). Overall, our findings suggest (i) that there is a significant association between exposure and efficacy when exposure was treated as continuous covariate and (ii) that there may be a greater extent of clinical benefit in patients with higher exposures when compared to the control group. In addition, Kaplan–Meier univariate analysis showed inferior survival for Q1 compared to control (Table 2), likely due to unbalanced prognostic factors, since no adjustments were made for prognostic factors in the Kaplan–Meier univariate analysis. After adjusting for prognostic factors in a multivariable analysis, there was little or no detrimental effect (Table 2) at Q1 exposure versus the control group for both overall survival (HR 1.19, 95% CI 0.89, 1.59) and progression-free survival (HR 0.92, 95% CI 0.72, 1.18). However, the adjusted hazard ratios for progression-free survival in Table 2 are not consistent with a clear, ordered monotonic exposure–response.
Overall survival Kaplan–Meier curves in each matched case–control Cmin,1 quartile. Kaplan–Meier curves of overall survival for each ramucirumab plus docetaxel case–control Cmin,1 quartile compared with patients treated with placebo plus docetaxel who had at least three cycles of treatment. C min,1 minimum concentration following the first dose, DOC docetaxel, PBO placebo, Q quartile, Q1, C min,1 6.26–≤ 15.7 µg/mL (< 25%), Q2, C min,1 > 15.7–≤ 20.7 µg/mL (25–<50%), Q3, C min,1 > 20.7–≤ 27.9 µg/mL (50–< 75%), Q4, C min,1 > 27.9–56.4 µg/mL (≥ 75%), RAM ramucirumab
Exposure–safety
Observed incidences of grade ≥ 3 hypertension, neutropenia, febrile neutropenia, and fatigue for the ramucirumab and placebo arms were similar between the REVEL ITT safety population and the exposure–safety population (Table 3).
The observed incidences of each safety endpoint by Cave,ss quartile are shown in Fig. 3. Ordered categorical analyses were further performed to evaluate the relationship between predicted measures of exposure and the incidences of grade ≥ 3 febrile neutropenia, neutropenia, fatigue, and hypertension. Based on this analysis, a statistically significant relationship (p < 0.001) was identified between Cave,ss and the incidence of grade ≥ 3 febrile neutropenia and hypertension.
Incidence of grade ≥ 3 adverse events by Cave,ss quartile. The observed incidence of select grade ≥ 3 adverse events by ramucirumab plus docetaxel Cave,ss quartile. Predicted Cave,ss exposure quartiles: Q1 = 37.9–≤79.3 µg/mL; Q2 = > 79.3–≤97.4 µg/mL; Q3 = > 97.4–≤118 µg/mL; Q4 = > 118–207 µg/mL. aIncidence of grade 3 fatigue. C ave,ss average concentration at steady state, DOC docetaxel, PBO placebo, Q quartile, RAM ramucirumab
There was no apparent relationship between Cave,ss and incidence of neutropenia or fatigue (Fig. 3).
Discussion
REVEL was a positive phase 3 clinical trial that established the efficacy and safety of ramucirumab plus docetaxel for patients with previously treated Stage IV NSCLC [3]. An exploratory analysis identified a relationship between ramucirumab exposure and survival outcomes. The results described here provide evidence for a positive exposure–response relationship between ramucirumab exposure and survival and toxicity in patients with previously treated Stage IV NSCLC. Separation between the Kaplan–Meier overall survival curves was observed among Cmin,1 exposure groups stratified by quartile (Fig. 1a), indicating that the higher exposure groups were generally associated with improved survival over the range of exposures achieved by a 10-mg/kg dose of ramucirumab given once every 3 weeks in combination with docetaxel. After adjusting for prognostic factors in a multivariable analysis (Table 2), the HRs for overall survival in Q2, Q3, and Q4 were below 1, indicating a trend towards treatment benefit in these quartiles. When the imbalances in prognostic factors were controlled for in the matched case–control analysis (Fig. 2), the HRs for overall survival in each quartile were very similar to the adjusted HRs from the multivariable analysis (Table 2). The trend for improvement in progression-free survival in the Cmin,1 exposure quartiles was not as linear as the results for overall survival, indicating that there may still be imbalances in unknown prognostic factors among quartiles.
One limitation of this study is that the first measurable PK samples were taken immediately before cycle 3. Therefore, patients in the ramucirumab plus docetaxel arm who discontinued before this third dose did not have ramucirumab PK data and no exposure–response relationship could be evaluated. To perform an appropriate comparison, patients in the placebo arm who discontinued before cycle 3 were also excluded from the exposure–efficacy analyses. Thus, the analysis was biased in both treatment arms for those patients who remained on treatment for at least three cycles, which may have impacted assessment of the true exposure–response effect. Thus, the observed relationship in the exposure–response population may be different from that in the ITT population. A better study design would be to measure cycle-1 levels of ramucirumab exposure to minimize this potential bias. As a result of only including patients who remained on treatment for at least three cycles, median progression-free survival (5.5 months) and overall survival (13.3 months) in the placebo plus docetaxel arm (N = 366) of the exposure–response population were longer than those reported in the control arm of the REVEL ITT population (median progression-free survival = 3.0 months, median overall survival = 9.1 months) [3]. This report showed an overall survival benefit of 1.4 months and a progression-free survival benefit of 1.5 months for ramucirumab as compared to placebo in the ITT population. In the current analysis, the highest quartile showed 3.8-month overall survival benefit and 1.5-month progression-free survival benefit for ramucirumab as compared to placebo; these outcomes are similar to the 5.2-month overall survival benefit and 2.8-month progression-free survival benefit in the highest ramucirumab exposure quartiles compared to the placebo arm in the RAINBOW exposure–response analysis that included patients with gastric cancer treated with 8 mg/kg ramucirumab plus paclitaxel [11]. These results suggest that a higher exposure to ramucirumab may improve efficacy outcomes.
In the REVEL ITT population, ramucirumab in combination with docetaxel was well tolerated with manageable side effects. The safety profile was consistent with that observed for ramucirumab established in previous studies in gastric cancer, NSCLC, and urothelial carcinoma as well as the established safety profile for docetaxel [3, 17,18,19]. In the REVEL trial, grade ≥ 3 TEAEs occurring in at least 5% of patients and at a > 2% higher incidence in the ramucirumab arm versus the control arm were neutropenia (49 versus 40%), febrile neutropenia (16 versus 10%), fatigue (14 versus 11%), and hypertension (5 versus 2%) [3]. Independent of the treatment arm, neutropenia was the most frequently reported grade ≥ 3 adverse event [3]. Our exploratory exposure–safety analysis demonstrated that the incidence of grade ≥ 3 hypertension and febrile neutropenia correlated with predicted ramucirumab concentration and increased exposure. A statistically significant association with exposure was only found for hypertension and febrile neutropenia. No covariates were found that altered the relationship between ramucirumab exposure and either hypertension or febrile neutropenia. The lack of association between exposure and neutropenia is presumably due to the high incidence of neutropenia (47.8%) in the control arm due to docetaxel.
A population PK meta-analysis of 11 ramucirumab trials that included different indications identified a modest relationship between body weight and ramucirumab disposition, which confirms that a weight-normalized dosing regimen is appropriate for therapy with ramucirumab. No other covariates were shown to have a clinically significant influence on the disposition of ramucirumab [14]. In the current study, we were not able to identify patients who would be more likely to have higher or lower ramucirumab exposure. Thus, therapeutic drug monitoring to measure the ramucirumab concentrations may be one option to help optimize the benefit for patients with advanced NSCLC.
Another limitation of this study is the loss of the effect of randomization when patients in the ramucirumab arm are stratified by estimated exposure quartiles. We addressed the resulting imbalance among patient factors by two approaches in the exposure–efficacy analysis. First, the same control group consisting of patients in the placebo arm who received ≥ 3 cycles of treatment was used as the comparator for each exposure group and prognostic factors were adjusted by multivariable analysis. Second, we identified a matched control group for each exposure quartile group, so that each case–control group had a similar distribution of patient characteristics. A similar strategy based on exposure subgroups, prognostic factors, and matched placebo controls was utilized by the FDA to analyze the exposure–response data for ramucirumab in advanced gastric cancer from the RAINBOW study [20]. Similar exposure–response relationships for ramucirumab were observed in the analyses used by the FDA to account for unbalanced patient factors in advanced gastric cancer, in the analysis performed on patients with different types of cancer in other studies (REGARD: NCT00917384, RAISE: NCT01183780), and in the current analysis of NSCLC [11, 12]. Very similar exposure–response outcomes have been reported for other mAbs being used to treat various types of cancer [8,9,10].
The similarity between the exposure–response analyses of ramucirumab in patients with gastric cancer (REGARD and RAINBOW trials) and the current REVEL exposure–response findings raises the possibility that higher exposure to ramucirumab may be beneficial with respect to the overall survival and progression-free survival in these patients. In fact, the findings presented here from REVEL have informed the decision to increase the ramucirumab dose in a randomized, phase 3 study of first-line ramucirumab (10 mg/kg given every 2 weeks) plus erlotinib to treat patients with EGFR mutation-positive metastatic NSCLC (RELAY; NCT02411448). In addition, the exposure–response relationship seen in REGARD and RAINBOW led the FDA to request a Post Marketing Commitment trial to test the hypothesis that higher doses of ramucirumab might be beneficial [21]. In this postmarketing phase 2 trial, advanced gastric/GEJ patients were treated with paclitaxel 80 mg/m2 on days 1, 8, and 15 of a 28-day cycle and randomized to the standard dose of ramucirumab of 8 mg/kg every 2 weeks versus 12 mg/kg every 2 weeks (NCT02514551). In Europe, the European Medicines Agency requested a monotherapy study of ramucirumab in a 4-arm phase 2 trial testing different doses and schedules (NCT02443883). The three experimental arms of the trial included ramucirumab regimens of 8 mg/kg every 2 weeks, 12 mg/kg every 2 weeks, and 6 mg/kg weekly versus the standard monotherapy regimen of 8 mg/kg on days 1 and 8 every 3 weeks. Pharmacokinetic analyses showed that the trough concentrations of the experimental regimens were greater than the standard regimen. Trends towards improved progression-free survival and overall survival were observed in the experimental regimens versus the standard regimen, with safety profiles similar to the standard regimen [13].
In conclusion, this exploratory analysis of population PK data in the REVEL study suggests a positive association between efficacy and ramucirumab exposure using a ramucirumab dose of 10 mg/kg every 3 weeks in combination with docetaxel. This regimen is effective and safe and offers a favorable benefit-to-risk profile in patients with previously treated Stage IV NSCLC. Patients may benefit from a ramucirumab dosing regimen that could produce higher ramucirumab exposure. Further prospective validation of these findings is currently underway.
References
Spratlin JL, Mulder KE, Mackey JR (2010) Ramucirumab (IMC-1121B): a novel attack on angiogenesis. Future Oncol 6:1085–1094. https://doi.org/10.2217/fon.10.75
Spratlin JL, Cohen RB, Eadens M, Gore L, Camidge DR, Diab S, Leong S, O’Bryant C, Chow LQ, Serkova NJ, Meropol NJ, Lewis NL, Chiorean EG, Fox F, Youssoufian H, Rowinsky EK, Eckhardt SG (2010) Phase I pharmacologic and biologic study of ramucirumab (IMC-1121B), a fully human immunoglobulin G1 monoclonal antibody targeting the vascular endothelial growth factor receptor-2. J Clin Oncol 28:780–787. https://doi.org/10.1200/JCO.2009.23.7537
Garon EB, Ciuleanu TE, Arrieta O, Prabhash K, Syrigos KN, Goksel T, Park K, Gorbunova V, Kowalyszyn RD, Pikiel J, Czyzewicz G, Orlov SV, Lewanski CR, Thomas M, Bidoli P, Dakhil S, Gans S, Kim JH, Grigorescu A, Karaseva N, Reck M, Cappuzzo F, Alexandris E, Sashegyi A, Yurasov S, Perol M (2014) Ramucirumab plus docetaxel versus placebo plus docetaxel for second-line treatment of stage IV non-small-cell lung cancer after disease progression on platinum-based therapy (REVEL): a multicentre, double-blind, randomised phase 3 trial. Lancet 384:665–673. https://doi.org/10.1016/S0140-6736(14)60845-X
Larkins E, Scepura B, Blumenthal GM, Bloomquist E, Tang S, Biable M, Kluetz P, Keegan P, Pazdur R (2015) U.S. Food and Drug Administration approval summary: ramucirumab for the treatment of metastatic non-small cell lung cancer following disease progression on or after platinum-based chemotherapy. Oncologist 20:1320–1325. https://doi.org/10.1634/theoncologist.2015-0221
U.S. Department of Health and Human Services (2003) Guidance for industry, exposure–response relationships-study design, data analysis, and regulatory applications. http://www.fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm072109.pdf. Accessed 30 Aug 2015
Thurber GM, Schmidt MM, Wittrup KD (2008) Antibody tumor penetration: transport opposed by systemic and antigen-mediated clearance. Adv Drug Deliv Rev 60:1421–1434. https://doi.org/10.1016/j.addr.2008.04.012
Cosson VF, Ng VW, Lehle M, Lum BL (2014) Population pharmacokinetics and exposure–response analyses of trastuzumab in patients with advanced gastric or gastroesophageal junction cancer. Cancer Chemother Pharmacol 73:737–747. https://doi.org/10.1007/s00280-014-2400-5
Feng Y, Roy A, Masson E, Chen TT, Humphrey R, Weber JS (2013) Exposure–response relationships of the efficacy and safety of ipilimumab in patients with advanced melanoma. Clin Cancer Res 19:3977–3986. https://doi.org/10.1158/1078-0432.CCR-12-3243
Wang J, Song P, Schrieber S, Liu Q, Xu Q, Blumenthal G, Amiri KL, Cortazar P, Ibrahim A, Justice R, Wang Y, Tang S, Booth B, Mehrotra N, Rahman A (2014) Exposure–response relationship of T-DM1: insight into dose optimization for patients with HER2-positive metastatic breast cancer. Clin Pharmacol Ther 95:558–564. https://doi.org/10.1038/clpt.2014.24
Zhu M, Tang R, Doshi S, Oliner KS, Dubey S, Jiang Y, Donehower RC, Iveson T, Loh EY, Zhang Y (2015) Exposure–response analysis of rilotumumab in gastric cancer: the role of tumour MET expression. Br J Cancer 112:429–437. https://doi.org/10.1038/bjc.2014.649
Tabernero J, Ohtsu A, Muro K, Van Cutsem E, Oh SC, Bodoky G et al (2017) Exposure–response analyses of ramucirumab from two randomized, phase III trials of second-line treatment for advanced gastric or gastroesophageal junction cancer. Mol Cancer Ther 16:2215–2222. https://doi.org/10.1158/1535-7163.MCT-16-0895
Cohn AL, Yoshino T, Heinemann V, Obermannova R, Bodoky G, Prausová J, Garcia-Carbonero R, Ciuleanu T, Garcia-Alfonso P, Portnoy DC, Van Cutsem E, Yamazaki K, Clingan PR, Polikoff J, Lonardi S, O’Brien LM, Gao L, Yang L, Ferry D, Nasroulah F, Tabernero J (2017) Exposure–response relationship of ramucirumab in patients with advanced second-line colorectal cancer: exploratory analysis of the RAISE trial. Cancer Chemother Pharmacol 80:599–608. https://doi.org/10.1007/s00280-017-3380-z
Ajani JA, Udrea A, Sarosiek T, Shenker M, Morgan C, Pikiel J, Wojcik E, Swinson D, Joseph M, Luft A, Salek T, Tournigand C, Ferry D, Zhang Y, Long A, Kuo W-L, Gao L, Kauh J, Mansoor W (2017) A dose-response study of ramucirumab treatment in patients with gastric cancer/gastroesophageal junction adenocarcinoma: Primary results of 4 dosing regimens in the phase 2 trial I4T-MC-JVDB. Ann Oncol 28(suppl 5):abstr 698P
O’Brien L, Westwood P, Gao L, Heathman M (2017) Population pharmacokinetic meta-analysis of ramucirumab in cancer patients. Br J Clin Pharmacol 83:2741–2751. https://doi.org/10.1111/bcp.13403
Yang J, Zhao H, Garnett C, Rahman A, Gobburu JV, Pierce W, Schechter G, Summers J, Keegan P, Booth B, Wang Y (2013) The combination of exposure–response and case–control analyses in regulatory decision making. J Clin Pharmacol 53:160–166. https://doi.org/10.1177/0091270012445206
Rubin D (1980) Bias reduction using Mahalanobis metric matching. Biometrics 36:293–298
Fuchs CS, Tomasek J, Yong CJ, Dumitru F, Passalacqua R, Goswami C, Safran H, dos Santos LV, Aprile G, Ferry DR, Melichar B, Tehfe M, Topuzov E, Zalcberg JR, Chau I, Campbell W, Sivanandan C, Pikiel J, Koshiji M, Hsu Y, Liepa AM, Gao L, Schwartz JD, Tabernero J (2014) Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet 383:31–39. https://doi.org/10.1016/S0140-6736(13)61719-5
Wilke H, Muro K, Van Cutsem E, Oh SC, Bodoky G, Shimada Y, Hironaka S, Sugimoto N, Lipatov O, Kim TY, Cunningham D, Rougier P, Komatsu Y, Ajani J, Emig M, Carlesi R, Ferry D, Chandrawansa K, Schwartz JD, Ohtsu A (2014) Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol 15:1224–1235. https://doi.org/10.1016/S1470-2045(14)70420-6
Herbst RS, Sun Y, Eberhardt WE, Germonpre P, Saijo N, Zhou C, Wang J, Li L, Kabbinavar F, Ichinose Y, Qin S, Zhang L, Biesma B, Heymach JV, Langmuir P, Kennedy SJ, Tada H, Johnson BE (2010) Vandetanib plus docetaxel versus docetaxel as second-line treatment for patients with advanced non-small-cell lung cancer (ZODIAC): a double-blind, randomised, phase 3 trial. Lancet Oncol 11:619–626. https://doi.org/10.1016/S1470-2045(10)70132-7
Jin R, Li H, Zhang LH, Zhao H, Fashoyin-Aje L, Lemery S, Keegan P, Booth B, Atiqur Rahman N, Wang Y, Sinha V, Zhao L (2015) Exposure–response (E–R) and case–control analyses of ramucirumab leading to recommendation for dosing optimization in patients with gastric cancer. J Clin Oncol 33(suppl):abstr 2578. https://doi.org/10.1200/jco.2015.33.15_suppl.2578
Casak SJ, Fashoyin-Aje I, Lemery SJ, Zhang L, Jin R, Li H, Zhao L, Zhao H, Zhang H, Chen H, He K, Dougherty M, Novak R, Kennett S, Khasar S, Helms W, Keegan P, Pazdur R (2015) FDA approval summary: ramucirumab for gastric cancer. Clin Cancer Res 21:3372–3376. https://doi.org/10.1158/1078-0432.CCR-15-0600
Acknowledgements
We thank the patients, their families, the study sites, and the study personnel who participated in this clinical trial. Eli Lilly and Company contracted with Syneos Health for writing support provided by Andrea D. Humphries, PhD, and Ira Ayene, PhD, and editing support provided by Angela C. Lorio, ELS.
Funding
Funded by: Eli Lilly and Company.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Ling Gao, Lisa M. O’Brien, Pablo Lee, Annamaria Zimmermann, David R. Ferry, and Allen S. Melemed are full-time employees and stockholders of Eli Lilly and Company. Egbert F. Smit has received honoraria from Eli Lilly and Company. Edward B. Garon received research funding from AstraZeneca, Bristol-Myers Squibb, Genentech, Merck, Mirati, Pfizer, Novartis, Boehringer Ingelheim, and Eli Lilly and Company. Martin Reck has received honoraria for lectures and consultancy from AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Eli Lilly and Company, Merck, Merck Sharp & Dohme, Novartis, Pfizer, and Roche/Genentech. Federico Cappuzzo has received consultancy fees from Eli Lilly and Company. Maurice Pérol has received honoraria as a consultant/advisory board member for Eli Lilly and Company. Paolo Bidoli is on the advisory board for Boehringer Ingelheim, Bristol-Myers Squibb, and Eli Lilly and Company. Roger B. Cohen declares grant support to his institution from ImClone. This study is funded by Eli Lilly and Company.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Smit, E.F., Garon, E.B., Reck, M. et al. Exposure–response relationship for ramucirumab from the randomized, double-blind, phase 3 REVEL trial (docetaxel versus docetaxel plus ramucirumab) in second-line treatment of metastatic non-small cell lung cancer. Cancer Chemother Pharmacol 82, 77–86 (2018). https://doi.org/10.1007/s00280-018-3560-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-018-3560-5