Abstract
Purpose
To evaluate the relationship between the volume, cross-sectional area, and peak isometric muscle strength of the gluteus maximus and gluteus medius muscles in patients with hip osteoarthritis, and to use this information to identify effective sites for measurement of the cross-sectional area of these muscles.
Methods
Twenty-four patients with hip osteoarthritis were included. The muscle cross-sectional area and volume were calculated from magnetic resonance images. The cross-sectional area was calculated at three levels: the inferior point of the sacroiliac joint, just above the femoral head, and at the greatest muscle diameter. Peak isometric strength was assessed using hand-held dynamometry, using the extension and external rotation for the gluteus maximus and abduction and internal rotation for the gluteus medius. Measured outcomes were compared between the two muscles, and the association between muscle volume, cross-sectional area, and peak isometric muscle strength was evaluated using Pearson’s correlation.
Results
Volume was correlated with the cross-sectional area in the gluteus maximus (r ≥ 0.707) and with the cross-sectional area (r ≥ 0.637) and peak isometric strength (r ≥ 0.477) in the gluteus medius. There was no difference between the cross-sectional area measured at the greatest muscle diameter and just above the femoral head (p = 0.503) for the gluteus maximus and at the inferior point of the sacroiliac joint (p = 0.651) for the gluteus medius.
Conclusion
The cross-sectional area, when used to calculate the muscle volume, should be evaluated just above the femoral head for the gluteus maximus and at the inferior point of the sacroiliac joint for the gluteus medius.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Hip osteoarthritis (OA) is common among individuals over the age of 60 years, causing pain in weight-bearing and impaired mobility [2, 22, 30]. Chronic hip OA generally leads to progressive hip deformity, resulting in hip joint dysfunction, muscle weakness, decreased range of hip joint motion, and restrictions in walking function [4, 13, 14, 18, 27]. The gluteus maximus and the gluteus medius have been implicated in OA-related hip dysfunction, and there are many prior studies investigating their role [1, 9, 10, 12, 29]. Thus, an objective assessment of the strength capacity of these muscles is clinically important.
Magnetic resonance (MR) and computed tomography (CT) imaging can be used to quantify the cross-sectional area (CSA) of the gluteus maximus and the gluteus medius muscles, with the volume calculated by multiplying the measured CSAs by the thickness of the MR image slices. Muscle volume, which provides a measure of the whole muscle rather than across just one area, has a higher correlation to muscle strength than muscle CSA [6, 10], and has previously been used to assess hip OA [9, 16, 17, 25]. However, the CSA may provide a more easily obtained assessment index than muscle volume [12, 15]. Of specific concern is the variation in the CSA according to the site selected for measurement, including smaller CSA values towards the points of muscle attachment. A previous study among healthy individuals reported a significant relationship between the CSA, volume, and muscle strength of the gluteus maximus and medius [12].
As hip OA is associated with muscle atrophy [9, 10, 29] and fatty infiltration [21], it is unclear whether the relationship between muscle mass and CSA identified in healthy subjects also applies to patients with hip OA. Therefore, the purpose of our study was to investigate the relationship between the volume, CSA and peak isometric strength of the gluteus maximus and medius in patients with hip OA. Ultimately, we aim to use this information to identify effective sites for CSA measurement.
Materials and methods
Statement of ethics
The design of this study was approved by the Ethics Committee of Niigata Bandai Hospital (approval no. 54), and participants provided informed consent.
Study group
Over the study period, from June 2017 to December 2018, 65 patients with hip OA were enrolled. All patients included were able to walk independently, without the use of a gait aid, and had no cognitive decline. In addition, all subjects were free of pain at the time of muscle testing. The exclusion criteria were as follows: pain in any joint other than the affected hip (n = 26); advanced or end-stage OA in the contralateral hip joint, according to the criteria of the Japanese Orthopaedic Association (JOA) criteria [26] (n = 5); the history of hip disease other than OA (n = 6); and participants for which all data could not be measured (n = 4). After exclusion, a total of 24 patients (6 men, 18 women) were included in the final analysis. All 24 patients included in the analysis were diagnosed with unilateral secondary hip OA, based on the JOA criteria [26], and were able to maintain the prone and supine position without being affected by the limited hip range of motion. Many of the subjects had advanced and end-stage hip deformity by the JOA criteria. Timed up and go test and leg length difference (spinomalleolar distance) and pain of loading hip joint in gait were also measured as basic information about the subject’s physical function.
Measured parameters
The following parameters for both the gluteus maximus and the gluteus medius were included in our analysis; CSA, volume, and peak isometric muscle strength. Using previously described methods [12], CSA values were measured, for both muscles, at three levels. Figure 1 shows a whole image of the measured MRI; Fig. 2 shows the inferior point of the sacroiliac joint; Fig. 3 shows the region just above the femoral head and at the level of greatest muscle diameter. The measurement of CSA at these three points was chosen because of the existence of clear bone indices and the possibility of simple and reproducible measurements; this protocol has already been used in previous studies [12].
The gluteus maximus and gluteus medius are shown on the sagittal plane at each measurement height. Since the entirety of the gluteus maximus and medius cannot be drawn in the image on the sagittal plane, the image with the best positional relationship was selected. The gluteus maximus is larger just above the femoral head than at the lowest point of the sacroiliac joint; the gluteus medius attaches to the great dislocation lower than just above the femoral head, so the gluteus medius is thinner just above the femoral head
The cross-sectional area of the measurement site and each muscle at the level just above the femoral head is shown. As can be seen from Fig. 1, the muscle cross-sectional area of the gluteus medius is depicted to be smaller as it approaches the great inversion. In addition, the gluteus maximus muscle can be clearly identified
Imaging protocol and calculation of muscle volume and CSA
MR images were obtained as previously described [12, 29], using the Signa HDe system (GE Healthcare, Waukesha, WI, USA) system. Prior to imaging, patients were screened for MR contraindications, with safety confirmed by a trained technician. Images were obtained with patients in the supine position, using the following imaging parameters: coronal T1 fast-spin echo; field of view, 400 × 400; 3 mm slice thickness, without a gap between slices; echo time, 10.1 ms; repetition time, 450 ms; and voxel size, 320/400 × 192/400 × 3.0 ms. The position of the pelvis was standardized during imaging by ensuring that the line connecting the anterior superior iliac crest on both sides was perpendicular to the bed [12].
Data from magnetic resonance imaging of the muscles were imported into ZedHip (Lexi Co., Ltd., Tokyo, Japan) and analyzed. The method used to measure each muscle’s cross-sectional area and volume was based on previous studies [12, 29]. To measure the CSA, the gluteus maximus and medius were identified in the images (Fig. 1) and traced around their peripheries. The tracings were then used to calculate the CSA of each muscle. The muscle volume was calculated using the following formula: Volume = sum (all CSAs × slice thickness).
Evaluation of steatosis
The extent of steatosis was evaluated using the Goutallier classification [8], as used in several previous studies on hip OA [1, 5, 18, 29]. The degree of steatosis was evaluated on cross-sectional images and classified into the following four grades [8, 29] (Table 1): grade 0, normal muscle volume; grade 1, muscle contains some fatty streaks; grade 2, fatty infiltration, but still more muscle than fat; grade 3, equal amounts of muscle and fat; and grade 4, more fat than muscle.
Measures of isometric muscle strength
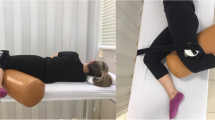
The peak isometric muscle strength of the gluteus maximus and of the gluteus medius was measured on the same day as the MR imaging, as per the methods described by Thorborg et al. [24] and used in another study [12]. Isometric strength was quantified using a hand-held dynamometer (μTas F-1, Anima, Tokyo, Japan), with the patient position and placement site for the dynamometer shown in Fig. 2. Each resisted contraction was held for 5 s, with the highest value recorded for analysis. For the gluteus maximus, peak isometric strength was evaluated for hip extension and external rotation, and hip abduction and internal rotation for gluteus medius.
Statistical analysis
Continuous variables were reported as mean ± standard deviation (SD), with categorical variables reported as count. The normality of the distribution of the data was evaluated using the Shapiro–Wilk test. Pearson’s correlation coefficient was used to evaluate the relationship between CSA, volume, and peak isometric strength. Differences in CSA between the two muscles were evaluated using a one-way analysis of variance (ANOVA), with a Tukey post-hoc test (p < 0.05). The degree of fatty muscle degeneration was not normally distributed, with between-muscle differences evaluated using the Wilcoxon’s signed-rank test. All statistical analyses were performed using SPSS (version 21; IBM Corp., Armonk, NY, USA), with the level of significance set at a two-tailed p value < 0.05.
Results
The general demographics of our study group were as follows (Table 2): mean age, 62.2 ± 7.5 years; height, 158.3 ± 6.5 cm; body weight, 55.4 ± 10.0 kg; and radiographic stage of hip OA, advanced and end stage. There was no difference in the degree of fatty infiltration between the gluteus maximus and the gluteus medius muscles.
The CSA, volume, and peak isometric strength of the gluteus maximus and the gluteus medius muscles are reported in Table 3. For the gluteus maximus, there was a significant positive correlation between muscle volume and the three CSA measures (peak CSA, r = 0.924, p < 0.001; inferior sacroiliac joint, r = 0.707, p < 0.001; and just above the femoral head, r = 0.849, p < 0.001), with no correlation between muscle volume and peak isometric strength (extension, r = 0.074, p = 0.732; external rotation, r = 0.323, p = 0.123). For the gluteus medius, there was again a significant correlation not only between muscle volume and the three CSA measures (peak CSA, r = 0.871, p < 0.001; inferior sacroiliac joint, r = 0.800, p < 0.001; and just above the femoral head, r = 0.637, p < 0.001), but also between muscle volume and peak isometric strength (abduction, r = 0.591, p = 0.002; internal rotation, r = 0.477, p = 0.019). The CSA values are reported in Table 4. For the gluteus maximus, there was no significant difference between the peak CSA (measured at the greatest muscle diameter) and the CSA measured just above the femoral head (p = 0.503). By contrast, for the gluteus medius, there was no significant difference between the peak CSA and the CSA measured at the inferior point of the sacroiliac joint (p = 0.651).
Discussion
The gluteus maximus and the gluteus medius muscles are important for proper hip function. As such, patients with hip OA would benefit from an objective assessment of the degeneration and fatty infiltration of these two muscles. Of note, the appropriate site for valid measurement of CSA of these muscles has not been previously determined. We investigated the relationship between volume, CSA, and muscle strength in a healthy group [12], but there were no reports of hip OA in this group. In fact, the relationship between CSA and peripelvic muscle contraction and fatty degeneration was investigated in subjects with unilateral hip disorders; however, the relationship between CSA and muscle functions was not clear [20]. Among the patients in our study group, the CSA measured above the femoral head was not different than the CSA measured at the greatest diameter of the muscle and correlated with the muscle volume. In contrast, for the gluteus medius, the CSA measured at the inferior point of the sacroiliac joint was no different than the CSA measured at the greatest diameter of the muscle and correlated with both the muscle volume and peak isometric strength. These findings are novel and provide an anatomical reference for the standard measurement of the CSA of these two muscles, which are involved in the progressive dysfunction in hip OA. Therefore, the CSA measured at these reference levels could provide an evaluative index in this clinical population. However, unlike a previous study with healthy individuals [12], the volume of the gluteus maximus correlated with the CSA but not with the peak isometric strength. The absence of a correlation between the volume and strength of the gluteus maximus might be related to the fact that all patients had advanced or end-stage hip OA, with measurements obtained just before total hip arthroplasty. It is possible that the hip deformity in these patients progressed as a function of increasing muscle weakness, resulting in increasing hip dysfunction, including the development of a flexion contracture, which would inhibit hip extensor strength. Patients with hip OA develop a range of motion limitations in the hip joint [13]. In particular, hip flexion contractures generally develop among patients with hip OA due to the maintained hip flexion position, which is used to decrease joint pain as OA progresses. The resultant restriction in hip extension likely contributes to the decreased CSA of hip extensors [23]. Specifically, development of a hip flexion contracture would place the fibers of the gluteus maximus muscle in a constant position of elongation, which would lead to a loss of muscle strength over time [3]. We do note that the hip flexion contracture was not excessive in our study group, with all patients being able to assume the prone position for measurement of peak isometric hip extensor strength, and all measures were obtained with the hip having 0º hip extension, as per previously published methods [12, 24]. The lack of correlation between the CSA and muscle strength in the gluteus maximus could be attributed to the measured level of CSA. The gluteus maximus has both upper and lower fibers; Grimaldi [10] showed that there is a volume difference in the lower fibers of the gluteus maximus, but no change in the upper fibers of the muscle. Since the lower fibers are active in hip extension and external rotation, it may be important to assess this area when assessing muscle weakness during these movements. Because we did not measure and analyze CSA separately for upper and lower fibers in this study, it is possible that there was no significant correlation between peak CSA and muscle strength.
Re-evaluation of the association between muscle volume and strength for the gluteus maximus using alternative hip positions, as well as other measures of hip function, including EMG is, therefore, warranted. Of note, despite a presumptive selective weakness of the gluteus maximus due to progressive hip flexion contracture, there was no difference in the degree of fatty degeneration between the gluteus maximus and the gluteus medius. This might indicate that, despite progressive hip dysfunction, the gluteus maximus is sufficiently used in activities of daily living, thus avoiding excessive steatosis. Again, this indicates that there is a need to consider other metrics of muscle hip function than those included in our study.
A previous study reported the gluteus medius volume as 206 ± 57 cm3 among patients with hip OA [17], which is in reasonable agreement with our value of 203.8 ± 47.2 cm3. Moreover, there was no difference in the CSA measured at the greatest muscle diameter and at the inferior point of the sacroiliac joint, with both CSA values being positively correlated with the peak isometric strength. The volume and CSA of the gluteus medius muscle were also correlated with muscle strength. We hypothesized that this correlation may also be influenced by the high daily activity of the gluteus medius muscle. A previous study showed the average muscle activity of the gluteus medius to be 47% over the walking cycle among patients with hip OA [11]. It has been shown that the gluteus medius is more active during the daily gait cycle among patients with hip OA.
Of note, this study also reported an average activation level of 55% for the gluteus maximus over the gait cycle in patients with hip OA, which would further explain the absence of a difference in the degree of steatosis between the gluteus maximus and the gluteus medius.
The gluteus medius, unlike the gluteus maximus, exists at the frontal plane [7]. Patients with hip OA have changes on the sagittal plane, as represented by the hip spine syndrome [19], which is associated with the joint deformity. In particular, many patients with secondary hip arthropathy in this study have anterior lumbar spine bending [28]; the enhancement of lumbar lordosis is considered a factor in hip flexion contracture, and the changes in the sagittal plane are significant. Because of this, the gluteus medius may be less affected by the progressive change in the sagittal plane alignment in hip OA than the gluteus maximus. These factors likely explain the positive correlation between the volume, CSA, and peak isometric strength for the gluteus medius.
From a clinical perspective, our findings indicate that the CSA measured just above the femoral head for the gluteus maximus and the inferior point of the sacroiliac joint for the gluteus medius might provide an effective evaluation index of the function of these muscles in hip OA. These CSA measures could be used, in combination with functional measures, to determine the benefit of prescribing hip strength exercises in patients with hip OA. If the patient with hip OA has muscle atrophy or dysfunction, hip extension exercise for gluteus maximus should be initiated. To improve the function of the gluteus medius, we believe it is necessary to engage in a hip strengthening program to improve abductor muscle strength and pelvic stability when weight is applied.
The soft tissues of the subject were assessed using MRI; although MRI is not a standard assessment, this study may provide a baseline to investigate other hip diseases, such as abductor muscle tears, femoroacetabular impingement, dysplasia, slipped capital femoral epiphysis, and perthes, and should be analyzed in different diseases.
The limitations of our study should be noted. Foremost, patients included in our study group had advanced and end-stage OA, with measurements obtained just before total hip arthroplasty. Therefore, all patients had long-standing hip OA. Validation of our measures is needed as values are likely to be influenced by the degree of OA-associated hip deformity. As previously stated, other measures of the strength capacity of the gluteus maximus might be needed, including electromyography, as well as across multiple hip positions to account for various degrees of hip deformity. Finally, it is important to note that the sample size of our study group is small, which limits the determination of causality. The results of this study may vary by sex and disease stage; therefore, the number of subjects needs to be increased to allow consideration of these other factors.
Conclusion
The volume of the gluteus maximus was found to correlate with all CSAs, but not with muscle strength, and may be affected by the site of measurement. CSA at the inferior point of the sacroiliac joint for the gluteus medius can provide an index of the power of these muscles among patients with hip OA.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ahedi H, Aitken D, Scott D, Blizzard L, Cicuttini F, Jones G (2014) The association between hip muscle cross-sectional area, muscle strength, and bone mineral density. Calcif Tissue Int 95:64–72. https://doi.org/10.1007/s00223-014-9863-6
Allen KD, Golightly YM (2015) State of the evidence. Curr Opin Rheumatol 27:276–283. https://doi.org/10.1097/BOR.0000000000000161
Bamman MM, Clarke MS, Feeback DL, Talmadge RJ, Stevens BR, Lieberman SA, Greenisen MC (1998) Impact of resistance exercise during bed rest on skeletal muscle sarcopenia and myosin isoform distribution. J Appl Physiol 84:157–163. https://doi.org/10.1152/jappl.1998.84.1.157
Bejek Z, Paróczai R, Illyés A, Kiss RM (2006) The influence of walking speed on gait parameters in healthy people and in patients with osteoarthritis. Knee Surg Sports Traumatol Arthrosc 14:612–622. https://doi.org/10.1007/s00167-005-0005-6
Bogunovic L, Lee SX, Haro MS, Frank JM, Mather RC 3rd, Bush-Joseph CA, Nho SJ (2015) Application of the Goutallier/Fuchs rotator cuff classification to the evaluation of hip abductor tendon tears and the clinical correlation with outcome after repair. Arthroscopy 31:2145–2151. https://doi.org/10.1016/j.arthro.2015.04.101
Fukunaga T, Miyatani M, Tachi M, Kouzaki M, Kawakami Y, Kanehisa H (2001) Muscle volume is a major determinant of joint torque in humans. Acta Physiol Scand 172:249–255. https://doi.org/10.1046/j.1365-201x.2001.00867.x
Gottschalk F, Kourosh S, Leveau B (1989) The functional anatomy of tensor fasciae latae and gluteus medius and minimus. J Anat 166:179–189
Goutallier D, Postel JM, Bernageau J, Lavau L, Voisin MC (1994) Fatty muscle degeneration in cuff ruptures: pre-and postoperative evaluation by CT scan. Clin Orthop Relat Res 304:78–83
Grimaldi A, Richardson C, Durbridge G, Donnelly W, Darnell R, Hides J (2009) The association between degenerative hip joint pathology and size of the gluteus maximus and tensor fascia lata muscles. Man Ther 14:611–617. https://doi.org/10.1016/j.math.2008.11.002
Grimaldi A, Richardson C, Stanton W, Durbridge G, Donnelly W, Hides J (2009) The association between degenerative hip joint pathology and size of the gluteus medius, gluteus minimus and piriformis muscles. Man Ther 14:605–610. https://doi.org/10.1016/j.math.2009.07.004
Homma D, Jigami H, Sato N (2016) Effects of Nordic walking on pelvis motion and muscle activities around the hip joints of adults with hip osteoarthritis. J Phys Ther Sci 28:1213–1218. https://doi.org/10.1589/jpts.28.1213
Homma D, Minato I, Imai N, Miyasaka D, Sakai Y, Horigome Y, Suzuki H, Dohmae Y, Endo N (2019) Investigation on the measurement sites of the cross-sectional areas of the gluteus maximus and gluteus medius. Surg Radiol Anat 41:109–115. https://doi.org/10.1007/s00276-018-2099-9
Hurwitz DE, Hulet CH, Andriacchi TP, Rosenberg AG, Galante JO (1997) Gait compensations in patients with osteoarthritis of the hip and their relationship to pain and passive hip motion. J Orthop Res 15:629–635. https://doi.org/10.1002/jor.1100150421
Kubota M, Shimada S, Kobayashi S, Sasaki S, Kitade I, Matsumura M, Kamei K, Kitano M, Takeno K, Baba H (2007) Quantitative gait analysis of patients with bilateral hip osteoarthritis excluding the influence of walking speed. J Orthop Sci 12:451–457. https://doi.org/10.1007/s00776-007-1160-z
Malloy P, Stone AV, Kunze KN, Neal WH, Beck EC, Nho SJ (2019) Patients with unilateral femoroacetabular impingement syndrome have asymmetrical hip muscle cross-sectional area and compensatory muscle changes associated with preoperative pain level. Arthroscopy 35:1445–1453. https://doi.org/10.1016/j.arthro.2018.11.053
Mastenbrook M, Commean PK, Hillen TJ, Salsich GB, Meyer GA, Mueller MJ, Clohisy JC, Harris-Hayes M (2017) Hip abductor muscle volume and strength differences between women with chronic hip joint pain and asymptomatic controls. J Orthop Sports Phys Ther 47:923–930. https://doi.org/10.2519/jospt.2017.7380
Momose T, Inaba Y, Choe H, Kobayashi N, Tezuka T, Saito T (2017) CT-based analysis of muscle volume and degeneration of gluteus medius in patients with unilateral hip osteoarthritis. BMC Musculoskelet Disord 18:457. https://doi.org/10.1186/s12891-017-1828-2
Murray MP, Gore DR, Clarkson BH (1971) Walking patterns of patients with unilateral hip pain due to osteo-arthritis and avascular necrosis. J Bone Jt Surg Am 53:259–274
Offierski CM, MacNab I (1983) Hip-spine syndrome. Spine 8:316–321. https://doi.org/10.1097/00007632-198304000-00014
Ogawa T, Takao M, Otake Y, Yokota F, Hamada H, Sakai T, Sato Y, Sugano N (2020) Validation study of the CT-based cross-sectional evaluation of muscular atrophy and fatty degeneration around the pelvis and the femur. J Orthop Sci 25:139–144. https://doi.org/10.1016/j.jos.2019.02.004
Pfirrmann CW, Notzli HP, Dora C, Hodler J, Zanetti M (2005) Abductor tendons and muscles assessed at MR imaging after total hip arthroplasty in asymptomatic and symptomatic patients. Radiology 235:969–976. https://doi.org/10.1148/radiol.2353040403
Quintana JM, Arostegui I, Escobar A, Azkarate J, Goenaga JI, Lafuente I (2008) Prevalence of knee and hip osteoarthritis and the appropriateness of joint replacement in an older population. Arch Intern Med 168:1576–1584. https://doi.org/10.1001/archinte.168.14.1576
Semciw AI, Pizzari T, Woodley S, Zacharias A, Kingsley M, Green RA (2018) Targeted gluteal exercise versus sham exercise on self-reported physical function for people with hip osteoarthritis (the GHOst trial–gluteal exercise for hip osteoarthritis): a protocol for a randomised clinical trial. Trials 19:511. https://doi.org/10.1186/s13063-018-2873-3
Thorborg K, Petersen J, Magnusson SP, Hölmich P (2010) Clinical assessment of hip strength using a hand-held dynamometer is reliable. Scand J Med Sci Sports 20:493–501. https://doi.org/10.1111/j.1600-0838.2009.00958.x
Uemura K, Takao M, Sakai T, Nishii T, Sugano N (2016) Volume increases of the gluteus maximus, gluteus medius, and thigh muscles after hip arthroplasty. J Arthroplasty 31:906–912.e.1. https://doi.org/10.1016/j.arth.2015.10.036
Ueno R (1971) Staging of osteoarthritis of the hip joint according to the roentgenographic findings. J Jpn Orthop Assoc 45:826–828
Watelain E, Dujardin F, Babier F, Dubois D, Allard P (2001) Pelvic and lower limb compensatory actions of subjects in an early stage of hip osteoarthritis. Arch Phys Med Rehabil 82:1705–1711
Yoshimoto H, Sato S, Masuda T, Kanno T, Shundo M, Hyakumachi T, Yanagibashi Y (2005) Spinopelvic alignment in patients with osteoarthrosis of the hip: a radiographic comparison to patients with low back pain. Spine 30:1650–1657. https://doi.org/10.1097/01.brs.0000169446.69758.fa
Zacharias A, Pizzari T, English DJ, Kapakoulakis T, Green RA (2016) Hip abductor muscle volume in hip osteoarthritis and matched controls. Osteoarthr Cartil 24:1727–1735. https://doi.org/10.1016/j.joca.2016.05.002
Zhang Y, Jordan JM (2010) Epidemiology of osteoarthritis. Clin Geriatr Med 26:355–369. https://doi.org/10.1016/j.cger.2010.03.001
Acknowledgements
DH is grateful to the collaborators and staff at Niigata Bandai Hospital for their work. We would like to thank Editage (https://www.editage.com) for English language editing.
Funding
This study did not receive any funding or financial support.
Author information
Authors and Affiliations
Contributions
DH: data collection or management, analysis, and manuscript writing/editing. IM: protocol/project development. NI: manuscript writing/editing. DM: protocol/project development. YS: data collection or management. YH: data analysis. HS: data analysis. HS: data analysis. YD: data collection or management. NE: protocol/project development.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The design of this study was approved by the Ethics Committee of Niigata Bandai Hospital (approval no. 54), and participants provided informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Homma, D., Minato, I., Imai, N. et al. Appropriate sites for the measurement of the cross-sectional area of the gluteus maximus and the gluteus medius muscles in patients with hip osteoarthritis. Surg Radiol Anat 43, 45–52 (2021). https://doi.org/10.1007/s00276-020-02535-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-020-02535-2