Abstract
Purpose
A curvilinear pathway intervening between the olfactory fossa and nasal vestibule has not been well documented. Therefore, the aim of this study was to examine its structure using magnetic resonance imaging (MRI).
Methods
In total, 84 patients underwent thin-sliced, contrast MRI. Among these patients, 31 underwent additional thin-sliced, sagittal T2-weighted imaging.
Results
A curvilinear pathway intervening between the olfactory fossa and nasal vestibule was delineated on sagittal and coronal imaging in 98% and 82% of patients, respectively. All of these pathways demonstrated communication with the lower limit of the superior sagittal sinus (SSS) or fine venous channels connecting to the SSS in the vicinity of the crista galli. The pathway was identified in the parasagittal regions on both sides with varying lengths, diameters, and curvatures. In 94% of the patients who underwent sagittal T2-weighted imaging, the pathways appeared as linear high-intensity signals. Most pathways were delineated as a single channel coursing extracranially adjacent to the olfactory fossa. In 38% of the patients, post-contrast sagittal images showed variable filling defects between the olfactory bulb and floor of the olfactory fossa, furthermore traversing the venous pathway. Additionally, in 73% of the patients, post-contrast images identified diploic venous channels, variably in the nasal bone and communicating with the venous pathway.
Conclusions
A curvilinear pathway intervening between the olfactory fossa and nasal vestibule is a consistent venous structure and may function as an extracranial route of cerebrospinal fluid drainage.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The cribriform plate has a distinct morphology characterized by numerous bony pores that transmit the olfactory filaments extensively to the nasal cavity [4]. Several experimental investigations have suggested that pathways in the cribriform region may play a distinct role in the drainage of cerebrospinal fluid (CSF) into the extracranial lymphatic vessels [2, 6, 8, 9, 11, 15, 16]. In microscopic examinations using human cadaveric specimens, fine subarachnoid spaces have been identified around the olfactory filaments as they exit the neural foramina of the cribriform plate. The dura mater surrounding the subarachnoid spaces thins out and becomes continuous with the periosteum in the ethmoid bone [1]. Furthermore, a distinct foramen, the ethmoidal foramen, has been identified in the anterior part of the olfactory fossa representing an area of least resistance that is predisposed to CSF leaks and meningocele [10].
Examinations using magnetic resonance imaging (MRI) has encountered a curvilinear venous structure intervening between the olfactory fossa and nasal vestibule. The nasal vestibule has been documented as a distinct region of the nasal cavity that is highly sensitive to the temperature-dependent, tactile stimulation of an airjet, and heavily influences nasal airway resistance [5, 7, 13, 14]. The nasal vestibule is also thought to function as a bacterial reservoir [3]. To our knowledge, the venous pathway has not been well documented [12]. The present retrospective study aimed to characterize the undescribed pathway using contrast and non-contrast MRI.
Materials and methods
The present retrospective study included 84 patients who presented as outpatients to our hospital between April 2010 and April 2016 and underwent MRI examinations. The patients presented with headaches, dizziness, tinnitus, hearing and hemisensory disturbances, and seizures. Patients with a history of traumatic brain injury, hydrocephalus, skull base tumors affecting the anterior and middle cranial fossae, and tumors and inflammatory diseases of the nasal cavity and paranasal sinuses were excluded from the study. The patient population consisted of 37 men and 47 women aged 48.5 ± 16.1 years (median ± SD; range 18–78 years). Initial examinations using axial T1- and T2-weighted imaging, T2 gradient echo, fluid-attenuated inversion recovery and diffusion-weighted sequences confirmed that none of the patients had any signs of the aforementioned pathologies. Then, the patients who were suspected to have tumorous lesions, inflammatory diseases, and vascular pathologies underwent imaging examination with an intravenous gadolinium infusion (0.1 mmol/kg) in the axial, coronal, and sagittal planes involving the whole cranial vault. The following parameters were adopted: repetition time (TR), 4.1 ms; echo time (TE), 1.92 ms; slice thickness, 1 mm; interslice gap, 0 mm; matrix, 320 × 320; field of view (FOV), 250 mm; flip angle (FA), 13°; and scan duration, 7 min 25 s. For 31 patients, an additional thin-sliced, sagittal T2-weighted sequence was performed involving the whole anterior fossa. The following parameters were adopted: TR, 3500.00 ms; TE, 90.00 ms; slice thickness, 2.00 mm; interslice gap, 0 mm; matrix, 300 × 189; FOV, 200 mm; FA, 90°; and scan duration, 2 min 40 s. All imaging sequences were performed using a 3.0-T MRI scanner (Achieva R2.6; Philips Medical Systems, Best, The Netherlands). Imaging data were transferred to a workstation (Virtual Place Lexus 64. 64th edition; AZE, Tokyo, Japan) and independently analyzed by two of the authors (H.I. and H.O.). The post-contrast sagittal and coronal images and sagittal T2-weighted images were used for analysis.
The study was conducted in accordance with the guidelines of our institution regarding human research. Written informed consent was obtained from all patients prior to their participation in the study.
Results
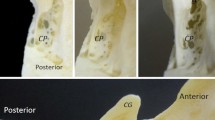
The morphology of the curvilinear pathway intervening between the olfactory fossa and nasal vestibule was well delineated in 82 (98%) and 69 (82%) of the 84 patients on post-contrast sagittal and coronal imaging, respectively. Because all of the pathways demonstrated communication with the lower limit of the superior sagittal sinus (SSS) or fine venous channels connecting to the SSS in the vicinity of the crista galli, they were considered as venous structures. The pathways were consistently identified in the parasagittal regions on both sides with segmental dilation at the nasal vestibule (Fig. 1). On sagittal sections, the pathways were delineated as linear structures with varying lengths, diameters, and curvatures (Fig. 2a–h, upper rows). On coronal sections, the pathways were consistently identified as lying below the olfactory bulbs (Fig. 2a–h, lower rows). In 29 of the 31 patients (94%) who underwent thin-sliced, sagittal T2-weighted imaging, the pathways appeared as linear high-intensity signals in accordance with that delineated on post-contrast sagittal images (Fig. 3). Most of the pathways were identified as a single channel coursing extracranially adjacent to the olfactory fossa, whereas in two patients, the courses divided into many branches (Fig. 4). Furthermore, in 32 patients (38%), post-contrast sagittal images showed variable filling defects of gadolinium between the olfactory bulb and the floor of the olfactory fossa, furthermore traversing the venous pathway (Fig. 5). There were no differences found in these patients compared to the remaining patients. Additionally, in 61 patients (73%), post-contrast sagittal images showed diploic venous channels variably distributed in the adjacent nasal bone and communicating with the venous pathway (Fig. 6).
a–d Seral images of post-contrast sagittal magnetic resonance imaging of a 71-year-old female patient showing a curvilinear venous pathway (arrows) in the left parasagittal region intervening between the olfactory fossa and nasal vestibule. e Post-contrast coronal magnetic resonance image at the level of the posterior edge of the bulb showing venous pathways on both parasagittal regions lying below the olfactory bulbs (arrows). The venous pathway shows segmental dilation at the nasal vestibule (c). IRM inferior rectus muscle, MRM medial rectus muscle, OB olfactory bulb, SObM superior oblique muscle, SRM superior rectus muscle
Post-contrast sagittal (a–f, upper rows) and coronal (a–f, lower rows) magnetic resonance images of different patients showing variable morphologies of the curvilinear venous pathway (a–f, arrow in upper rows) intervening between the olfactory fossa and nasal vestibule. The venous pathway courses below the olfactory bulb (a–f, arrows in lower rows). *Olfactory bulb
Post-contrast sagittal (a) and sagittal T2-weighted (b) magnetic resonance images at the same level in the same patient showing a curvilinear venous pathway in the left parasagittal region (arrows). The venous pathway appears as a high-intensity signal on T2-weighted imaging in accordance with that delineated on post-contrast sagittal imaging (b, arrows)
Discussion
In this study, the curvilinear venous pathway intervening between the olfactory fossa and nasal vestibule was consistently delineated both on sagittal and coronal images. It was identified parasagittal on both sides with varying lengths, diameters, and curvatures. In 38% of the 84 patients, post-contrast sagittal images showed variable filling defects between the olfactory bulb and the floor of the olfactory fossa, furthermore traversing the venous pathway. Because the olfactory filaments arising from the olfactory bulb, coursing downward, and exiting the neural foramina of the cribriform plate are accompanied by subarachnoid spaces at the exits of the neural foramina [1], the filling defects, probably containing the olfactory filaments and their surrounding CSF-filled spaces, could join the venous pathway lying underneath. Furthermore, in most of the patients who underwent sagittal T2-weighted imaging, the venous pathway appeared as a high-intensity signal in accordance with that delineated on post-contrast sagittal images. Based on these findings, we assumed that the venous pathway intervening between the olfactory fossa and nasal vestibule could function as an extracranial route of CSF drainage into the lymphatic vessels around the nasal vestibule. The reasons for the venous pathway consistently reaching the nasal vestibule and exhibiting segmental dilation at the nasal vestibule remain unknown.
In this study, diploic venous channels variably distributed in the nasal bone and communication with the venous pathway were identified in more than 70% of the 84 examined patients. This finding may reflect a unique characteristic of the venous pathway and warrants further verification in a sufficiently large population.
The present study has limitations and weaknesses. The study population consisted of patients with inhomogeneous age distribution and uneven sex ratio. They were retrospectively evaluated and not randomly assigned to the contrast and non-contrast T2-weighted MRI examinations. Furthermore, the hypothetical function of the venous pathway as an extracranial route of CSF drainage was derived only from MRI findings rather than quantitative analysis. Despite these limitations, we believe that the results of this study can provide a clearer understanding of the distinct venous pathway intervening between the olfactory fossa and nasal vestibule.
Conclusions
A curvilinear pathway intervening between the olfactory fossa and nasal vestibule is a consistent venous structure and may function as an extracranial route of CSF drainage.
References
Dare AO, Balos LL, Grand W (2003) Neural–dural transition at the medial anterior cranial base: an anatomical and histological study with clinical applications. J Neurosurg 99:362–365
Erlich SS, McComb JG, Hyman S, Weiss MH (1986) Ultrastructural morphology of the olfactory pathway for cerebrospinal fluid drainage in the rabbit. J Neurosurg 64:466–473
Glück U, Gebbers JO (2000) The nose as bacterial reservoir: important differences between the vestibule and cavity. Laryngoscope 110:426–428
Gomez Galarce M, Yanez-Siller JC, Carrau RL, Montaser A, Lima LR, Servian D, Otto BA, Prevedello DM, Naudy CA (2018) Endonasal anatomy of the olfactory neural network: surgical implications. Laryngoscope 128:2473–2477
Jones AS, Crosher R, Wight RG, Lancer JM, Beckingham E (1987) The effect of local anaesthesia of the nasal vestibule on nasal sensation of airflow and nasal resistance. Clin Otolaryngol Allied Sci 12:461–464
Kida S, Pantazis A, Weller RO (1993) CSF drains directly from the subarachnoid space into nasal lymphatics in the rat. Anatomy, histology and immunological significance. Neuropathol Appl Neurobiol 19:480–488
Li C, Jiang J, Kim K, Otto BA, Farag AA, Cowart BJ, Pribitkin EA, Dalton P, Zhao K (2018) Nasal structural and aerodynamic features that may benefit normal olfactory sensitivity. Chem Senses 43:229–237
Murtha LA, Yang Q, Parsons MW, Levi CR, Beard DJ, Spratt NJ, McLeod DD (2014) Cerebrospinal fluid is drained primarily via the spinal canal and olfactory route in young and aged spontaneously hypertensive rats. Fluids Barriers CNS 11:12
Nagra G, Koh L, Zakharov A, Armstrong D, Johnston M (2006) Quantification of cerebrospinal fluid transport across the cribriform plate into lymphatics in rats. Am J Physiol Regul Integr Comp Physiol 291:R1383–R1389
Patron V, Berkaoui J, Jankowski R, Lechapt-Zalcman E, Moreau S, Hitier M (2015) The forgotten foramina: a study of the anterior cribriform plate. Surg Radiol Anat 37:835–840
Silver I, Kim C, Mollanji R, Johnston M (2002) Cerebrospinal fluid outflow resistance in sheep: impact of blocking cerebrospinal fluid transport through the cribriform plate. Neuopathol Appl Neurobiol 28:67–74
Tsutsumi S, Ono H, Yasumoto Y (2016) A possible venous connection between the cranial and nasal cavity. Surg Radiol Anat 38:911–916
Wong AS, Thian Y-L, Kapur J, Leong C-N, Kee P, Lee C-T, Lee MB (2018) Pushing the limits of immune-related response: a case of “extreme pseudoprogression”. Cancer Immunol Immunother 67(7):1105–1111. https://doi.org/10.1007/s00262-018-2167-3
Yu S, Liu Y, Sun X, Li S (2008) Influence of nasal structure on the distribution of airflow in nasal cavity. Rhinology 46:137–143
Zakharov A, Papaiconomou C, Johnston M (2004) Lymphatic vessels gain access to cerebrospinal fluid through unique association with olfactory nerves. Lymphat Res Biol 2:139–146
Zhang ET, Richards HK, Kida S, Weller RO (1992) Directional and compartmentalised drainage of interstitial fluid and cerebrospinal fluid from the rat brain. Acta Neuopathol 83:233–239
Funding
None.
Author information
Authors and Affiliations
Contributions
ST conceived the study. HI and YY collected the imaging data. HO and HI analyzed the imaging data. ST wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare regarding the materials or methods used in this study or the findings presented in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tsutsumi, S., Ono, H., Ishii, H. et al. An undescribed venous pathway intervening between the olfactory fossa and nasal vestibule. Surg Radiol Anat 41, 485–490 (2019). https://doi.org/10.1007/s00276-019-02208-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-019-02208-9