Abstract
Major tributaries such as the renal and adrenal veins have been studied extensively; however, tributaries of the infra-renal segment of the inferior vena cava (IVC) have not been given much attention. Accurate knowledge of the anatomy of these veins is necessary for improved efficacy of surgical interventions in the retroperitoneum. The aim of this study therefore was to provide a comprehensive picture of the internal anatomy of the tributaries of the infra-renal segment of the IVC. Dissection of the posterior abdominal wall was performed on 30 formalin-fixed cadavers. Endoscopic study was carried out followed by a midline venotomy on the anterior wall of the isolated IVC, the location and orientation of its tributaries and their ostia were observed and measurements taken. The results showed that while there was great variation in the drainage pattern of the lumbar veins, most lumbar veins had ostia located between L2 and L3 vertebrae irrespective of the location of renal and gonadal tributaries. Valves were found in 81.81 % of gonadal veins, in 56.60 % of all lumbar veins and discrete ostial valves in 14.81 % of renal veins. The location of the tributaries of the IVC was correlated with the vertebral levels. Empirical data regarding their ostio-valvular complexes were established, which put into question widely accepted concept of valveless tributaries. Our results may implicate surgical procedures in and around the retroperitoneal region.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Despite intensive research in the anatomical sciences for the past two centuries, some structures of the human body still remain controversial or incompletely described. One of these structures is the infra-renal segment of the inferior vena cava (IVC) and in particular its lumbar tributaries. Standard anatomy textbooks describe the infra-renal segment as the longest segment of the IVC arising from the confluence of the right and left common iliac veins at the level of the fifth lumbar vertebra, which extends to about the first lumbar vertebra where the renal veins join it. The tributaries to this portion of the IVC arise from retroperitoneal and pelvic structures and include the lumbar and right gonadal veins [21, 23, 28, 32]. It is widely accepted that the complex embryology of the IVC is responsible for the high incidence of variations among its tributaries [1, 2, 11, 18, 22, 29, 31, 32]. Lumbar veins are thought to drain along the entire length of the IVC accompanying their corresponding arteries. They receive blood from parietal structures of the anterolateral and posterior abdominal wall [9, 21, 23, 28, 32]. Typically, five pairs of lumbar veins are described; however, it has been suggested that the IVC may only receive the third and fourth pairs [9, 21, 25, 28, 29]. Additionally, locations of the first and second lumbar veins have been described as highly variable, rarely emptying into the IVC but frequently join the ascending lumbar, iliolumbar or renal veins to subsequently drain into the azygos–hemiazygos system of veins [12, 21, 23, 29]. There are no data regarding the ostio-valvular complexes of the lumbar tributaries as they enter the IVC. Additionally, there are increasing reports of anatomical variations which may prove important in thoracoabdominal surgical procedures such as IVC thrombectomy, Greenfield filter and IVC stent placement, or in the follow-up of testicular cancer patients where an aberrant vein may be mistaken for a metastatic lymph node [2, 4, 9, 14, 22, 25, 27].
Although the surgical anatomy of the posterior abdominal wall and IVC has been well described, recent reports concerning surgical interventions in the retroperitoneum such as laparoscopic live donor nephrectomy [3, 5, 6, 12, 26, 31], and lymph node dissection as an adjunct to chemotherapy in cancer of the gonads and kidneys [4] are reemphasizing the clinical importance of venous tributaries. Furthermore, these procedures necessitate advanced imaging studies as part of pre-operative screening [12, 14, 22, 25, 27, 31]. Knowledge of the anatomy of the ostio-valvular complexes is essential to avoid erroneous IVC filter placement [18, 31] and inadvertent vessel tearing, which could lead to subsequent hemorrhage into the retroperitoneal space.
Through our experience from many retroperitoneal dissections, we have observed that great variation exists regarding the ostia location of these venous tributaries within the IVC, as well as the presence and distribution of valves. As such, we aimed to explore and describe the range of all the above-mentioned observations, and provide a comprehensive picture of the internal anatomy of the infra-hepatic portion of the IVC across a broad range of specimens.
Materials and methods
The institutional review board of St. George’s University School of Medicine approved this study under IRB/06014. Age and patient history were obtained from cadaver records. The anatomy of the IVC was examined in 30 adult cadavers during 2013–2014. The mean age at death was 69 years with a range of 55–86 years; 18 males and 12 females were dissected. All of the cadavers were fixed in a formalin–phenol–alcohol solution. None of the specimens examined revealed any evidence of previous surgical procedures, traumatic lesions, or gross pathologies of the abdomen.
Posterior abdominal wall dissection was done via a surgical retroperitoneal approach as outlined by Loukas et al. [16]. The IVC was then dissected out from the liver and the length of the infra-hepatic portion was measured.
Phase one consisted of endoscopic exploration of the IVC via a superior approach. A rigid 5 mm Stryker endoscope and Stryker unit including a Stryker Quantum 4000 light source, Stryker 3-chip camera system (Stryker Endoscopy, San Jose, CA) and a Wolf 5-mm 30-cm 0° laparoscope was introduced superiorly into the IVC. Images of each ostium with its accompanying valve were recorded with image capture software (Endoscopy Support Services, Inc. Brewster, NY) as previously described [17]. The purpose of endoscopic exploration was to identify and appreciate any morphologic variations in the ostio-valvular complexes without disturbing its normal anatomy.
Subsequently, dissection of the IVC and morphological study of the ostio-valvular complexes were carried out. The IVC was fixed with pins to the vertebral column at the iliocaval junction and superior to the left renal vein to preserve its spatial relationship with other retroperitoneal structures. A midline incision was made in the anterior wall, beginning at the iliocaval confluence and extending superiorly to the left renal vein. Each tributary (renal, gonadal and lumbar) was inspected and the corresponding vertebral level was recorded. The position of the ostia along the wall of the IVC (right, left, anterior or posterior) as well as the orientation (oblique or straight) was documented. The external course of all tributaries flowing into each ostium was followed distally as far as the dissection would allow. These data were later used to construct a composite diagram illustrating the drainage pattern of the lumbar tributaries (Fig. 1). Descriptive statistical analysis was carried out using Microsoft Excel and SPSS software. Tables were plotted using this statistical data.
Frequently observed patterns of drainage of lumbar tributaries into the IVC. a and c Show multiple ipsilateral and contralateral veins uniting to form common trunks before entering into the IVC. b Highlights single lumbar veins draining through individual ostia as is suggested in standard anatomy texts
The tributaries were then inspected for the presence of ostial and parietal valves and correlated to the position and orientation of the vessel. Ostial valves were considered to be any valve attached directly at the point of union to the IVC with the tributary, whereas parietal valves were considered to be any valve within the lumen of the vein as described by Tasch and Brenner [33]. For all observed valves, the number and shape of valve cusps, and the relative distance from the ostia were noted. In addition to morphological observations, the vertical and horizontal diameters of each ostium were measured and recorded. All measurements were taken using a flexible metric ruler calibrated to the nearest millimeter. Photographs were taken of all dissections.
Results
The infra-hepatic portion of the IVC extended to L1 in most cadavers; however, in one specimen a left renal ostium was located at the T12/L1 intervertebral disc (IVD). The confluence of the left and right common iliac veins formed the IVC consistently at L5. The mean length of the IVC measured from the superior border of the highest renal vein ostia to the inferior border of the iliocaval confluence segment was 152.6 mm (±3.23 SD). Endoscopic study revealed a fibrous midline septa at the junction of the iliac veins in 17 cadavers, which in most cases was deviated to the right. Apart from this finding, there was no difference in the observations made with endoscopy and that of the open technique. A total of 180 ostia were observed, of which 27 were right renal, 25 were left renal, 22 were right gonadal and 106 were lumbar veins (Tables 1, 2).
The renal veins were the largest with the respective mean vertical and horizontal diameter of their ostia being 13.38 mm (±3.22 SD) and 9.76 mm (±2.06 SD) for the right and 15.90 mm (±2.47 SD) and 10.60 mm (±3.12 SD) for the left. A low lying right kidney was observed in one specimen, the ostia of its renal vein being at L3, while all other right renal veins were found between L1 and L2/L3 (IVD). Although both renal veins were found at the same level, the left was often seen slightly higher than the right. In one of our specimen, a retroaortic left renal vein was present. Accessory tributaries to the right renal vein were present in two specimens, one of which had three additional veins; two of these aberrant renal veins had ostial valves (Fig. 2).
A cadaveric picture showing a bicuspid apparently incompetent parietal valve in the right renal vein (RRV). The right kidney (RK) is partially hidden by the reflected vena cava. The left renal vein (LRV) of this specimen had no valves. A indicates a bicuspid ostial valve of a posteriorly located lumbar ostia, other lumbar ostia in the immediate vicinity also have valves
Several variations of the gonadal veins were documented. A right gonadal tributary was present in twenty-two cadavers, twenty had its ostium located on the anterior wall and two entered on the right side of the IVC. In one specimen, the left gonadal vein drained into the IVC and in three cadavers no gonadal tributaries to the IVC were observed. In these last variants, the gonadal veins were tributary to their respective renal veins as seen in Fig. 3. Gonadal vein ostia were found approximately at the L2/3 IVD in most specimens; however, in two, ostia were located as low as L4. The mean vertical and horizontal diameters were 6.53 (±2.98 SD) and 4.53 (±1.81 SD), respectively (Table 2).
Lumbar tributaries were defined as any vein emerging from the medial border of the psoas major and entering into the IVC at any point along, which were not gonadal, hepatic, ureteric or renal. A total of 106 lumbar ostia were found for an average of 4.24 per cadaver and ranged from 4 to 7 veins per cadaver. While the diameters of the renal and gonadal tributaries were relatively constant, there was great variability in the diameters of the lumbar tributaries. The largest ostia were located between L2 and L3 where most lumbar tributaries were found (Table 1). Lumbar veins were frequently found close to the right gonadal vein as seen in Fig. 4. In 73 % of cases, the lumbar tributaries drained obliquely into the infra-renal IVC. More than half (60/106) of all lumbar ostia were located on the posterior aspect of the IVC, while 40 were found on the left and 6 on the right side of the wall. The right sided tributaries were found between the L1 and L2 vertebral levels (Fig. 5).
A cadaveric picture showing the ostial location of the tributaries of the inferior vena cava as being proximal to the renal veins. The ostium of both right renal vein (RRV) and left (LRV) were located at the L2. The right gonadal vein RGV in this specimen was located at L2/3 intervertebral disc (IVD). The arrowheads point to lumbar veins which can be seen draining at the same level or close to the RGV. Only one lumbar vein drained in the lower segment close to the iliocaval confluence
A composite diagram illustrating the location of the lumbar tributaries of the inferior vena cava. Lumbar veins were most frequently observed between the second (L3) and third (L3) lumbar vertebrae. They were infrequent at L1 (1.88 %) and below the L4/5 IVD (9.43 %). RK and LK represent the right and left kidneys, respectively
All gonadal veins examined presented with ostial valves, the majority being bicuspid. It was observed that valves are also present in lumbar and renal veins (Figs. 2, 4, 6). It was noted that 63 % of all obliquely draining lumbar tributaries had ostial valves compared to only in 5 % of straight draining veins. The ostial valves observed were predominately monocuspid with a rudimentary cusp; however, several were bicuspid as illustrated in Fig. 6. Three main renal veins also had valves as demonstrated in Fig. 2.
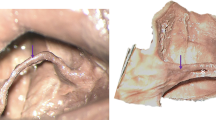
Examples of valves observed at the ostium of the lumbar tributaries of the IVC by endoscopy and by direct observation after venotomy. a A picture of a posterior lumbar vein ostium captured after anterior venotomy of the IVC; a bicuspid valve can be seen at the lumen. b A picture of a bicuspid ostial valve as observed by endoscopy, the cusps of valvular leaflets approximate each other suggesting some degree of competency
Discussion
Surgical interventions into the retroperitoneum such as laparoscopic live donor nephrectomy, lymph node dissection and lumbar disc surgery require accurate knowledge of the anatomy of the retroperitoneal vasculature [3, 4, 12, 13, 15, 22, 25, 30]. While the hepatic, adrenal and renal veins have been studied extensively [6–8, 19, 26], some have proposed that adequate knowledge of lumbar venous vasculature is paramount to prevention of life threatening retroperitoneal bleeding, as a result of damage to aberrant or accessory renal and adrenal veins and lumber tributaries to the renal veins [1, 3, 4, 7, 9, 12, 19, 22, 26, 27]. Our findings of right renal vein ostia at the L3/L4 IVD, multiple accessory renal veins [3, 12, 15, 17, 25], a retroaortic left renal vein [13, 31] and absent gonadal tributaries to the IVC support claims of great variation in the anatomy of the infra-renal IVC.
Our data support the observation of Miles et al. [20] that lumbar vein ostia are more prevalent on the left and posterior aspect of the IVC. We demonstrated that the majority of ostia were concentrated around the L2–L3 vertebral level. Miles et al. [20] studied 100 cadavers and described lumbar ostia position and frequency relative to their distance from the renal, gonadal and iliac tributaries. They reported that lumbar veins were mostly between the iliocaval confluence and right gonadal vein. Likewise, Shchukin et al. [27] proposed the use of the right renal vein as a marker for identifying lumbar veins and reported that most of these were located in the vicinity of the lower border of the right renal vein. Reports of aberrant renal veins [8, 31] and our own findings of absent gonadal tributaries in three cadavers (Fig. 3) and four cases of a low lying renal vein bring into question the reliability of these identification methods during surgery. In contrast, Beveridge et al. [33] used the proximity from the iliocaval confluence to classify lumbar veins. While this is an acceptable landmark in cadaveric studies, the importance of rapid identification and isolation of ruptured vessels during surgery supersedes this. Furthermore, given that laparoscopic surgery has become the gold standard for many retroperitoneal interventions [12, 30], more easily discernible landmarks such as vertebral levels are preferable.
Reports of ostial locations are inconclusive with the same authors often using different descriptions [20]. This furthers the need for descriptions of venous ostia using stable landmarks such as vertebral levels or intervertebral discs. Knowing that most lumbar veins drain between the second and third lumbar vertebrae (Table 1) may allow surgeons to quickly localize a ruptured tributary during retroperitoneal interventions, thus decreasing surgical time.
Our results showed that there was a disparity between the number of tributaries and the number of ostial openings, a feature better observed with direct visualization than endoscopy. Multiple lumbar veins frequently formed a common trunk before draining into a single ostium along the IVC (Fig. 1). These findings contradict information presented in clinical anatomy textbooks which suggest a regular arrangement of lumbar veins [23, 29, 32]. In our observations, lumbar veins had a haphazard arrangement and were not frequently found paired to the contralateral vein as their corresponding arteries.
As expected, our results confirmed the presence of ostial valves in the gonadal veins [3, 10, 28, 32]; however, ostial valves were also found in lumbar veins (Figs. 4, 6). This finding does not support data available in contemporary surgical textbooks which suggest valveless lumbar veins [26, 28], however is in line with Franklin [10] who claimed that most tributaries of the IVC had ostial valves including lumbar. Similarly, in a venographic study using 40 cadavers, Oleaga reported valves along the length of renal veins and at the caval ostium of the right renal, alluding to their importance in selecting a site for splenorenal shunt placement [24]. Emergent research, however, has described the ostial diameter of the tributaries of the IVC [6, 8, 19, 27] or classified them based on their course [5], but has not reported on the presence of valves. It appears that information was either considered unimportant or simply not deemed worthy of mention, leading to respective anatomy texts stating that “except for the gonadal, valves are not present in the tributaries to the inferior vena cava” [28].
Our findings show no significant morphological difference between the ostia of the tributaries of the IVC using endoscopy or direct observation after venotomy. One advantage of endoscopy is the ability to view valves and their cusps without manipulation of the three-dimensional architecture of the IVC. This allowed for observation regarding the approximation of the leaflets and the possibility of their consequent occlusive ability to the smaller vessel (Fig. 6). The endoscopic method also assisted in discerning if the valve(s) observed in the lumen of the tributaries were located ostially or parietally. Endoscopic examination should therefore be considered a valuable tool for studying large vascular structures such as the IVC, as it provides a more accurate picture of three-dimensional anatomy.
Conclusion
Our study is the first to describe the locational frequency of the tributaries of the IVC based on vertebral levels. This could prove valuable in retroperitoneal surgery. Our findings reiterate the highly variable anatomy of the lumbar tributaries of the post renal IVC and refute previous reports claiming valveless tributaries, adding valuable data to current anatomical knowledge. Further investigation of the lumbar veins is warranted in particular their ostio-valvular complexes.
References
Abassi A, Johnson T, Kleris R, Ying K, Bonner M, Shishir MK, Kooby DA, Marshall FF, Master VA (2012) Posterior lumbar vein of the retro-hepatic inferior vena cava: a novel anatomical variant with surgical implication. J Urol 187:296–301
Ang WC, Doyle T, Stringer MD (2013) Left-sided and duplicate inferior vena cava: a case series and review. Clin Anat 26:990–1001
Baniel J, Foster RS, Donohue JP (1995) Surgical anatomy of the lumbar vessels: implications for retroperitoneal surgery. J Urol 153:1422–1425
Baniel J, Foster RS, Rowland RG, Bihrle R, Donohue JP (1995) Testis cancer: complication of post chemotherapy lymph node dissection. J Urol 153:976–980
Beveridge TS, Power A, Johnson M, Power NE, Allman BL (2015) The lumbar arteries and veins: quantification of variable anatomical positioning with application to retroperitoneal surgery. Clin Anat. doi:10.1002/ca.22504
Camargo AM, Texiera GG, Ortale JR (1995) Anatomy of the ostia venae hepaticae and the retrohepatic segment of the Inferior Vena Cava. J Anat 188:59–64
Cesmebasi A, Du Plessis M, Iannatuono M, Shah S, Tubbs RS, Loukas M (2014) A review of the anatomy and clinical significance of adrenal veins. Clin Anat 27:1253–1263
Chang RW, Shan-Quan S, Yen WWC (1989) An applied anatomical study of the ostia venae hepaticae and the retrohepatic segment of the inferior vena cava. J Anat 164:41–47
Duty B, Daneshmand S (2008) Venous resection in urological surgery. J Urol 180:2338–2342
Franklin KJ (1927) Valves in veins: an historical survey. Proc R Soc Med 21:1–33
Grubb R 3rd (2014) Reply: left-sided and duplicate inferior vena cava. A case series and review. Clin Anat 27:948–949. doi:10.1002/ca.22175
He B, Hamdorf JM (2013) Clinical Importance of anatomical variations of renal vasculature during laparoscopic donor nephrectomy. Open Access Anatomy 1:2. doi:10.13172/2052-7829-3-865
Inzunza HO, Inzunza MA, Salgado GA (2011) Vena renal recurrente retroaortica: reporte de una rara variation. Int J Morphol 29:339–343
Jaskolka JD, Kwok RPW, Gray SH, Mojibian HR (2010) The value of pre-procedure computed tomography for planning insertion of inferior vena cava filters. Can Assoc Radiol J 61:223–229
Li G, Dong J, Lu JS, Quiang Z, Su-Xia Y, Hong-Zhao L, Xin M, Xu Z (2011) Anatomical variation of the posterior lumbar tributaries of the left renal vein in retroperitoneoscopic left living donor nephrectomy. Int J Urol 18:503–509. doi:10.1111/j.1442-2042.2011.02778
Loukas M, Benninger B, Tubbs RS (2012) Gray’s clinical photographic dissector of the human body. Saunders Elsevier, Philadelphia, pp 205–220
Loukas M, Kinsella CR, Louis RG, Gandhi GS, Curry B (2006) Surgical anatomy of the accessory phrenic nerve. Ann Thorac Surg 82:1870–1875
Loukas M, Strike DRG, Tubbs RS (2009) An interrupted inferior vena cava in Situs Inversus: a case report and review of literature. Folia Morph (Warsz) 68:184–187
Matusz P, Pusztai AM (2010) Regarding the course of the retrohepatic portion of the inferior vena cava. Clin Anat 23:467–470
Miles RM, Flowers BF, Parsons HL, Benitone JD (1973) Some surgical implications of the Cava-iliofemoral system. Ann Surg 177:740–747
Moore KL, Dalley AF, Agur AMR (eds) (2014) Clinically oriented anatomy, 7th edn. Lippincott Williams & Wilkins, Philadelphia, pp 315–320
Netter Frank H (2014) Netter atlas of human anatomy, 6th edn. Saunders Elsevier, Philadelphia, pp 260–379
Nayak SB, Ravindra SS, Madhav VN, Sirasanagandla SR, Guru A, Kumar N (2013) A common lumbar vein formed by union of left second lumbar and bilateral third lumbar veins: a case report. OA Anatomy 1:18. doi:10.13172/2052-7829-2-751
Oleaga JA, Ring EJ, Freiman D, Husted JW (1978) Renal vein valves. Am J Roentgenol 130:927–928
Perez JA, Torres FG, Toribio AM, Fernandez KL, Hayoun C, Naranjo ID (2013) Angio-CT assessment of anatomical variants in renal vasculature: its importance in the living donor. Insights Imaging 4:199–211
Ross IL, Louw GJ (2015) Embryological and molecular development of the adrenal glands. Clin Anat 28:235–242. doi:10.1002/ca.22422
Shchukin DV, Altukhov OO, Lisova GV, Ilyukhin YA (2013) Identification of bleeding sources during removal of inferior vena cava tumor thrombi: multidetector computed tomography study. UroToday Int J 6(6):art 75. doi:10.3834/uij.1944-5784.2013.12.10
Sinnatamby CS (ed) (2006) Lasts anatomy: regional and applied, 11th edn. Churchill Livingstone Elsevier, China, pp 286–288
Skandalakis JE, (2004) Skandalakis’ surgical anatomy. The embryonic and anatomic basis of modern Skandalakis surgical anatomy, vol 1. Paschalidis Medical Publications, Greece, pp 598–608
Skrekas G, Vassilios EP, Mitsis M, Naday HS (2003) Laporoscopic live donor nephrectomy: a step forward in kidney transplantation? JSLS 7:197–206
Spentzouris G, Zandian A, Cesmebasi A, Kinsella CR, Muhleman M, Mirzayan N, Shirak M, Tubbs RS, Shaffer K, Loukas M (2014) The clinical anatomy of the inferior vena cava: a review of common congenital anomalies and considerations for clinicians. Clin Anat 27:1234–1243
Standring S (2008) Chap 13: Early embryonic circulation and Section 8: Abdomen and pelvis. In: Standring S (ed) Gray’s anatomy, 40th edn. Churchill-Livingstone Elsevier, China, pp 206–209, 1075–1076, 1247–1278
Tasch C, Brenner E (2012) The ostial valve at the great saphenous vein. Phlebology 27:1790–1830
Acknowledgments
The authors wish to thank Jessica Holland, MS, medical illustrator at St. George’s University, Grenada, West Indies, for the creation of her illustrations used in this publication. The authors also wish to acknowledge the individuals who donated their bodies without whom this project would not have been possible.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical considerations
All specimens were handled in accordance to the laws and regulations of the country in which the study was performed. The specimens used for this research are protected under IRB approval (06014) as issued by the School of Medicine at St Georges University Grenada.
Rights and permissions
About this article
Cite this article
Bubb, K., du Plessis, M., Hage, R. et al. The internal anatomy of the inferior vena cava with specific emphasis on the entrance of the renal, gonadal and lumbar veins. Surg Radiol Anat 38, 107–114 (2016). https://doi.org/10.1007/s00276-015-1527-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-015-1527-3