Abstract
Kin discrimination in nepotistic as well as in sexual contexts is widespread in animals including humans. However, the underlying mechanisms of kin discrimination are assumed to vary between species and—within species—between contexts. During solitary life stages, kin recognition based on social learning is assumed to be less reliable because kin templates that are not continuously updated may get lost. Therefore, self-referent phenotype matching (“armpit effect”), i.e., the comparison of unknown phenotypes with own characteristics, should be particularly relevant when no social cues are available. However, experimental evidence for this mechanism is scarce. Here, we examine self-referent kin recognition in a mate-choice context in adult male Pelvicachromis taeniatus, a socially monogamous cichlid fish from West Africa with biparental brood care and pronounced kin-mating preferences. Juvenile P. taeniatus live in groups, whereas adult males compete for access to breeding sites which they aggressively defend against rivals. Using computer-animated females as standardized visual stimuli in combination with olfactory cues of related and unrelated females, we show that adult males reared isolated from kin since egg stage were able to discriminate sisters from unrelated females. As males could have learned kin cues only from themselves, our study provides evidence for self-referent kin recognition and indicates that the observed inbreeding preferences are mediated by self-derived olfactory cues. Male preferences for sisters were correlated with male body size. We discuss the implications of quality-related mate choice for the evolution of kin-mating preferences.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nepotistic behaviors as well as optimal outbreeding/inbreeding often rely on cognitive capabilities of individuals to assess their genetic relatedness to others. The proximate mechanisms underlying kin recognition are assumed to vary between species and—within species—between contexts (Mateo 2004). Kin recognition by phenotype matching is widespread in animals (Penn and Frommen 2010). This mechanism is advantageous because it is independent from spatial cues or prior association and enables individuals to recognize even unfamiliar kin (Holmes and Sherman 1982). However, it requires a correlation between phenotypic and genotypic similarity and a reference system for matching the phenotypic cues of an encountered individual. Phenotype matching can be either self-referential or family-referential. Self-referent kin recognition is assumed to be advantageous in species with multiple mating resulting in unequally related siblings who are less appropriate as reliably kin-reference (Hain and Neff 2006). Furthermore, as kin templates need regular updates because they may get lost otherwise (Olsen and Winberg 1996; Mateo 2010), self-reference might be advantageous during solitary phases. While several studies suggest that phenotype matching in shoaling contexts is family-referential rather than self-referential (e.g., Olsen and Winberg 1996; Gerlach et al. 2008, juvenile Pelvicachromis taeniatus Hesse et al. 2012) studies examining the reference of kin recognition in mate choice are scarce. Self-referent kin recognition based on olfactory cues (coined “armpit effect” by Dawkins (1982) might be particularly promising here because self-derived olfactory cues can be continuously used as reference (Hauber and Sherman 2001). However, although the results of several studies are consistent with self-matching (Mateo and Johnston 2000; Hauber and Sherman 2001; Bressan and Zucchi 2009; Schielzeth et al. 2008), definitive experimental evidence for self-reference is still scarce which might be due to the context examined or due to experimental constraints (e.g., difficulties to deprive individuals from becoming familiar with phenotypes from kin, considering for instance in utero familiarization in mammals (Hare et al. 2003).
In this study, we examine kin recognition in the externally fertilizing fish P. taeniatus in a mate-choice context. P. taeniatus is a monogamous cichlid with intense biparental care (Thünken et al. 2010). After some weeks, the young leave their parents and build loose shoals. After becoming sexually mature, solitary males occupy territories and defend them against rivals (Thünken et al. 2011). Previous mate-choice experiments revealed that males and females were able to discriminate between unfamiliar kin and unfamiliar non-kin, suggesting phenotype matching as kin recognition mechanism (Thünken et al. 2007a). Interestingly, kin were preferred as mating partner (Thünken et al. 2007a, b; see also Langen et al. 2011). Further studies suggest that kin recognition is mediated by olfactory cues (Thünken et al. 2011; Hesse et al. 2012). Adult males were shown to be able to recognize their own odor (Thünken et al. 2009) which is an important prerequisite for self-referent kin recognition. In the present study, adult males reared isolated from kin since egg stage were given the choice between the odor of a sister and the odor of an unrelated female. Both female odors were presented in combination with the same computer-animated, brightly colored, and reproductively active female of P. taeniatus in order to simulate a mate-choice situation.
Material and methods
Experimental fish
The experimental fish were the F2 generation of wild caught fish from the Moliwe, a small river in Cameroon, West Africa. The Moliwe population is highly inbred and spatially (by several waterfalls) as well as genetically structured (Langen et al. 2011). As a consequence, it is very likely that related individuals encounter each other during the reproductive period under natural conditions.
The experimental fish were bred between February and April 2010 in the laboratory at the Institute for Evolutionary Biology and Ecology in Bonn under standardized conditions (see Thünken et al. 2007a for details). Breeding caves were checked for eggs daily. The eggs were then raised isolated from kin in small plastic tanks (16 × 9 × 10 cm) under standardized conditions (see Hesse et al. 2012 for details). In February 2011, subadult males were each transferred individually to larger tanks (30.5 × 20 × 10 cm; water level, 13 cm) in which they remained until they reached sexual maturity. The tanks were equipped with sand, an air stone for air supply, and a breeding cave. Visual isolation was ensured by opaque Styrofoam sheets between the isolation tanks. To habituate the test fish to the background of the computer animation (see below), a hard copy of the background (showing a water plant) was fixed at the broad sides of the isolation tanks. As donor for olfactory stimuli, brightly colored and reproductively active females that were related and unrelated to the focal males were isolated in tanks (25 × 15.5 × 15.5 cm, 1 L water/0.15 g female biomass). Each tank was equipped with a hiding shelter and an air stone for air supply. Scented water was taken from these tanks, serving as odor stimuli during trials. One third of the water was exchanged at the day before scented water for experimental trials was taken. The water temperature was kept 23 ± 1 °C, and the day/night period was 12 L/12D. Experimental fish were fed with frozen Chironomus larvae in the afternoon.
Experimental setup
The experimental tank had the same size as the isolation tanks for the males and was also equipped with a breeding cave. The tank was placed between two monitors of the same model (EIZO Flex Scan F520, 85 Hz, see Thünken et al. 2011). The setup was illuminated by a fluorescent tube (37 W) installed 1 m above the middle of the tank. Additionally, white Styrofoam surrounded the setup. In the experiment, the test males were given the choice between two identical computer-animated brightly colored females of P. taeniatus (4 cm body length on the screen), which were simultaneously presented in combination with different odors. The computer animation was developed based on digital photograph material of five different females that were merged. It moved a horizontal pathway from one side of the monitor to the other for a period of 15 s, including a 2-s stop in the middle. After that, it recurred horizontally and moved back in the same time frame. This sequence was repeated over the total experimental period. For details and preparation of the computer animation, see Baldauf et al. (2008, 2009b, 2013). The applications of computer animation in mate-choice experiments is well established in P. taeniatus (Baldauf et al. 2009a, 2010, 2011, 2013; in combination with olfactory cues Thünken et al. 2011). Water conditioned with the odor of one related female was added centrally in front of one animation and that conditioned with the odor of one unrelated female in front of the other animation (see below). The side on which the related odor was presented was determined randomly. In order to control for potential differences in attractiveness of the individual odors independent from relatedness, we tested the female stimulus pair with two males (paired design, see “Statistical analysis”).
Experimental procedure
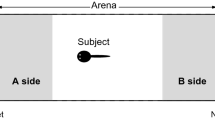
The experiment was conducted in August 2011 with brightly colored, reproductively active males. One day before the experiment, a live, brightly colored female was presented to the males allowing sexual stimulation by visual cues only. The next day, the male was carefully transferred into the test tank; he was given 15 min to habituate to the experimental conditions. Before the two computer animations of the female were simultaneously started on both broad sides of the tank, the olfactory stimuli were introduced for 40 s using a peristaltic pump (flow rate, 5 ml/min). Subsequently, the scented waters were introduced synchronously to every short stop of the female animation for 4.8 s. This was done on the one hand to make sure that the males associate the odor with the visual stimuli and in order to avoid a rapid mixture of the odors. Two hundred milliliters of conditioned water was taken from the isolation tank of the respective females immediately before each trial. Pretest had shown that these settings are appropriate; animated fish with scented water were preferred over animations with blanket water only (TT, TCMB, SAB, unpublished data). After each trial, the tubes of the peristaltic pumps and the tanks containing the stimulus water were thoroughly rinsed with water. Body length of the test male was measured immediately after the experiment. Fish behavior was recorded using a webcam. The video recordings were analyzed with the tracking software BIObserve. Mating preferences were measured as association time near a stimulus of the opposite sex, which is a standard method to estimate mating preferences in animals (Wagner 1998). Association time reliably predicts mating decisions in cichlids (Couldridge and Alexander 2001; Dechaume-Moncharmont et al. 2011) including P. taeniatus (Thünken et al. 2007a, TT, TCMB, N. Henning, H. Kullmann, unpublished data). The time spent in each choice zone (see Fig. 1) was calculated over a period of 10 min after the fish had visited both choice zones (and thus probably had perceived both odors).
The experimental setup. Male Pelvicachromis taeniatus could choose between the odor of a sister and that of an unrelated female. Two identical computer animations of a brightly colored, reproductively active female were simultaneously presented as visual stimuli on both sides. A choice zone of 5 cm was defined in front of each monitor to measure male preferences
Statistical analysis
All calculations were performed with the R. 2.9.1 statistical software package (R Development Core Team 2008). Parametric tests were used because data did not significantly differ from normal distribution according to the Shapiro–Wilk test (paired data: N = 6, W = 0.912, p = 0.449; preference index: N = 12, W = 0.898, p = 0.149). To analyze mating preferences, we conducted two analyses. In order to control for potential differences in attractiveness of the individual stimulus odors (independent from relatedness), we tested a female stimulus pair in two trials. Trial 1: male 1 with female A (a sister) and female B (a non-kin). Trial 2: male 2 with the same female A (a non-kin) and the same female B (a sister). Accordingly, the sister average was (proportion time 1A + proportion time 2B)/2 and the non-kin average was (proportion time 1B + proportion time 2A)/2. The proportions were compared using a paired t test. Furthermore, we conducted a linear mixed effect model (“lme”) with kin preference index (time spent with sister minus time spent with non-sister) of each experiment as response variable. This analysis allowed us on the one hand to examine size-related mating preference (male body size was entered as covariate) and on the other hand to control for the multiple use of some families. Family origin of the males was entered as random factor (random factor “family”: likelihood-ratio tests (LRT), df = 1, χ 2 = 0.144, p = 0.704). The 12 test males and 8 stimulus females originated from five different families (number of males per family: 4, 3, 1, two times: 2). LRT with maximum likelihood assessed whether the removal of a variable caused a significant decrease in the model fit. Reported p values of models refer to the increase in deviance when the respective variable was removed. Hence, degrees of freedom differ by one.
Results
Males significantly preferred the odor of the unfamiliar sister over that of the unfamiliar, unrelated female (paired t test, df = 5, t = −3.05, p = 0.028; Fig. 2). The result remains significant when controlling for the multiple use of families (lme, intercept estimate = 136.458, df = 7, t = 2.46, p = 0.043; random factor: “family” (intercept) SD = 0.148, residual SD = 183.698). Body size of the test males ranged from 4.3 to 5.0 cm (mean ± SD, 4.60 ± 0.16 cm). Male body size which was independent from a male’s age (Pearson correlation, N = 12, r = −0.28, p = 0.41) was positively correlated with kin preference (LRT, slope estimate = 723.477, χ 2 = 5.55, p = 0.018; Fig. 3).
Discussion
Here, we showed that males of P. taeniatus that were reared isolated from kin since egg stage were able to discriminate between unfamiliar sisters and unfamiliar unrelated females based on olfactory cues in the context of mate choice. As expected from previous studies (Thünken et al. 2007a, 2007b, 2011), they preferred sisters. Because males could have learned kin cues exclusively from themselves, our study provides clear evidence for self-referent phenotype matching. Several other studies examining phenotype matching in non-sexual, i.e., grouping contexts reported that individuals reared isolated from kin did not discriminate between kin and non-kin (e.g., Olsen and Winberg 1996; Gerlach et al. 2008). This is also true for juvenile of P. taeniatus which live in shoals (Hesse et al. 2012). Other studies showed that kin templates appear to get lost after a certain period of separation from kin (Olsen and Winberg 1996; Mateo 2010). This suggests that contact to kin is often essential for successful recognition of kin and that phenotype matching is rather based on familial imprinting than self-reference during social stages (Frommen et al. 2007, but see Hain and Neff 2006). When individuals become reproductive, their own odor patterns may change and develop more pronounced and, consequently, own odors may gain more importance. For example, in male sticklebacks, certain MHC signals are only produced in the reproductive state (Milinski et al. 2010). In Atlantic salmon, olfactory receptors of adult fish were shown to be different from those of juveniles (Johnstone et al. 2011). Reproductively active males of P. taeniatus are solitary, highly territorial, and defend breeding sites aggressively against rivals (Thünken et al. 2011). As in other cichlids (Barata et al. 2007), olfactory signals play a role in the territorial behavior of adult males. When given the choice between two caves in which different scents were artificially added, males preferred caves with the own odor over caves with foreign odors; interestingly, they avoided caves with the odor of brothers against caves with the odor of unrelated males (maybe to avoid competition with kin; Thünken et al. 2009)). These results indicate that adult males of P. taeniatus are able to recognize own olfactory cues, and the results of the present study indicate that they use self-derived olfactory cues to recognize kin. In conclusion, in contrast to juvenile kin recognition which is based on familial imprinting in P. taeniatus (Hesse et al. 2012), adult kin recognition seems to rely on self-reference, suggesting that mechanisms of kin recognition are context-dependent.
The mating preferences reported in the present study confirm the inbreeding preferences found in previous laboratory studies in P. taeniatus (Thünken et al 2007a, 2007b, 2011, 2012). These findings received further support by genetic analyses, revealing that the natural Moliwe population shows generally a very low genetic diversity (in terms of microsatellite allelic diversity and heterozygosity) as well as a significant heterozygote deficit (Langen et al. 2011). The latter was present at population level but also within subpopulations and sampling sites, indicating that mating among related individual occurred more often than expected by chance even at relatively small spatial scales.
Mating with kin often results in inbreeding depression (Charlesworth and Charlesworth 1987), but theory also predicts benefits (reviewed in Kokko and Ots 2006). However, as the costs often appear to exceed the benefits, many animals show inbreeding avoidance strategies (Pusey and Wolf 1996). Although there are examples that inbreeding is tolerated in some species, evidence for active inbreeding is rare (Szulkin et al. 2013). Inbreeding might be beneficial in species with biparental brood care because it may reduce the sexual conflict over care in pairs consisting of related individuals (Thünken et al. 2007a). This hypothesis was supported in P. taeniatus in which related individuals were less aggressive during care and spent more time caring. In contrast, in the cichlid Neolamprologus pulcher, highly related pairs showed higher within-pair aggression (Stiver et al. 2008). Furthermore, inbreeding individuals might increase their inclusive fitness by providing opposite sex siblings a mating partner (“altruistic inbreeding”, Kempenaers 2007; Kokko and Ots 2006). This might be particularly important in species with strong intra-sexual competition like P. taeniatus in which females compete among each other for access to males (Baldauf et al. 2011).
In the present—as well as in a previous study (Thünken et al. 2011)—male kin preference was positively correlated with male body size. As male size did not correlate with age, we assume that variation in size reflect variation in genetic quality. Variation in mating preferences has received considerable attention in evolutionary research (reviewed in Jennions and Petrie 1997; Widemo and Saether 1999; Cotton et al. 2006). As large males have advantages in intra- (Thünken et al. 2011) as well as in inter-sexual competition (Baldauf et al. 2009a), they are able to select the preferred partner and gain accordant fitness benefits. In contrast, low-quality males might be generally less choosy or show instantly assortative preferences for low-quality females (see Baldauf et al. 2013). Interestingly, size-related responses do not seem to require any prior social experience and thus might be innate.
In the context of inbreeding, quality-related inbreeding might contribute to “purging”, i.e., selection against deleterious alleles in the long-term due to inbreeding (Crnokrak and Barrett 2002; Glemin 2003). Because of purging, inbreeding individuals might increase individual inclusive fitness across generations (Lehmann and Perrin 2002). Quality-related mate choice in general might contribute to purging by sexual selection because higher choosiness of high-quality individuals in mate choice should result in higher fitness compared to low-quality individuals. Moreover, low-quality individuals might be less attractive as mating partners. Recently, we have shown that small sisters are rejected against large unrelated females by males as mates (Thünken et al. 2012). In particular, quality-related kin-mating preferences might be of importance for the evolution of inbreeding preference. The stronger kin preferences of high-quality individuals should lead to fast spread of “inbreeding alleles”; in contrast, reduced kin preferences of low-quality individuals might prevent the reintroduction of deleterious mutations.
References
Baldauf SA, Bakker TCM, Herder F, Kullmann H, Thünken T (2010) Male mate choice scales female ornament allometry in a cichlid fish. BMC Evol Biol 10:301
Baldauf SA, Bakker TCM, Kullmann H, Thünken T (2011) Female nuptial coloration and its adaptive significance in a mutual mate choice system. Behav Ecol 22:478–485
Baldauf SA, Kullmann H, Bakker TCM (2008) Technical restrictions of computer-manipulated visual stimuli and display units for studying animal behaviour. Ethology 114:737–751
Baldauf SA, Kullmann H, Schroth SH, Thünken T, Bakker TCM (2009a) You can't always get what you want: size assortative mating by mutual mate choice as a resolution of sexual conflict. BMC Evol Biol 9:129
Baldauf SA, Kullmann H, Thünken T, Winter S, Bakker TCM (2009b) Computer animation as a tool to study preferences in the cichlid Pelvicachromis taeniatus. J Fish Biol 75:738–746
Baldauf SA, Engqvist L, Ottenheym T, Bakker TCM, Thünken T (2013) Sex-specific conditional mating preferences in a cichlid fish: implications for sexual conflict. Behav Ecol Sociobiol 67:1179–1186
Barata EN, Hubbard PC, Almeida OG, Miranda A, Canario AVM (2007) Male urine signals social rank in the Mozambique tilapia (Oreochromis mossambicus). BMC Biol 5:54
Bressan P, Zucchi G (2009) Human kin recognition is self-rather than family-referential. Biol Lett 5:336–338
Charlesworth D, Charlesworth B (1987) Inbreeding depression and its evolutionary consequences. Annu Rev Ecol Evol S 18:237–268
Couldridge VCK, Alexander GJ (2001) Does the time spent near a male predict female mate choice in a Malawian cichlid? J Fish Biol 59:667–672
Cotton S, Small J, Pomiankowski A (2006) Sexual selection and condition-dependent mate preferences. Curr Biol 16:755–765
Crnokrak P, Barrett SCH (2002) Purging the genetic load: a review of the experimental evidence. Evolution 56:2347–2358
Dawkins R (1982) The extended phenotype. Oxford University Press, Oxford
Dechaume-Moncharmont FX, Cornuau JH, Keddar I, Ihle M, Motreuil S, Cezilly F (2011) Rapid assessment of female preference for male size predicts subsequent choice of spawning partner in a socially monogamous cichlid fish. C R Biol 334:906–910
Frommen JG, Luz C, Bakker TCM (2007) Kin discrimination in sticklebacks is mediated by social learning rather than innate recognition. Ethology 113:276–282
Gerlach G, Hodgins-Davis A, Avolio C, Schunter C (2008) Kin recognition in zebrafish: A 24-hour window for olfactory imprinting. Proc R Soc Lond B 275:2165–2170
Glemin S (2003) How are deleterious mutations purged? Drift versus nonrandom mating. Evolution 57:2678–2687
Hain TJA, Neff BD (2006) Promiscuity drives self-referent kin recognition. Curr Biol 16:1807–1811
Hare JF, Sealy SG, Underwood TJ, Ellison KS, Stewart RLM (2003) Evidence of self-referent phenotype matching revisited: airing out the armpit effect. Anim Cogn 6:65–68
Hauber ME, Sherman PW (2001) Self-referent phenotype matching: theoretical considerations and empirical evidence. Trends Neurosci 24:609–616
Hesse S, Bakker TCM, Baldauf SA, Thünken T (2012) Kin recognition by phenotype matching is rather family- than self-referential in juvenile cichlid fish. Anim Behav 84:451–457
Holmes WG, Sherman PW (1982) The Ontogeny of kin recognition in two species of ground squirrels. Am Zool 22:491–517
Jennions MD, Petrie M (1997) Variation in mate choice and mating preferences: a review of causes and consequences. Biol Rev Camb Philos Soc 72:283–327
Johnstone KA, Lubieniecki KP, Koop BF, Davidson WS (2011) Expression of olfactory receptors in different life stages and life histories of wild Atlantic salmon (Salmo salar). Mol Ecol 20:4059–4069
Kempenaers B (2007) Mate choice and genetic quality: a review of the heterozygosity theory. Adv Stud Behav 37:189–278
Kokko H, Ots I (2006) When not to avoid inbreeding. Evolution 60:467–475
Langen K, Schwarzer J, Kullmann H, Bakker TCM, Thünken T (2011) Microsatellite support for active inbreeding in a cichlid fish. PLoS One 6:e24689
Lehmann L, Perrin N (2002) Altruism, dispersal, and phenotype-matching kin recognition. Am Nat 159:451–468
Mateo JM (2004) Recognition systems and biological organization: the perception component of social recognition. Ann Zool Fenn 41:729–745
Mateo JM (2010) Self-referent phenotype matching and long-term maintenance of kin recognition. Anim Behav 80:929–935
Mateo JM, Johnston RE (2000) Kin recognition and the 'armpit effect': evidence of self-referent phenotype matching. Proc R Soc Lond B 267:695–700
Milinski M, Griffiths SW, Reusch TBH, Boehm T (2010) Costly major histocompatibility complex signals produced only by reproductively active males, but not females, must be validated by a 'maleness signal' in three-spined sticklebacks. Proc R Soc Lond B 277:391–398
Olsen KH, Winberg S (1996) Learning and sibling odor preference in juvenile arctic char, Salvelinus alpinus (L.). J Chem Ecol 22:773–786
Penn DJ, Frommen JG (2010) Kin recognition: an overview of conceptional issues, mechanisms and evolutionary theory. In: Kappeler P (ed) Animal behavior: evolution and mechanisms. Springer, Heidelberg, pp 55–85
Pusey A, Wolf M (1996) Inbreeding avoidance in animals. Trends Ecol Evol 11:201–206
Development Core Team R (2008) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, http://www.R-project.org
Schielzeth H, Burger C, Bolund E, Forstmeier W (2008) Assortative versus disassortative mating preferences of female zebra finches based on self-referent phenotype matching. Anim Behav 76:1927–1934
Stiver KA, Fitzpatrick JL, Desjardins JK, Neff BD, Quinn JS, Balshine S (2008) The role of genetic relatedness among social mates in a cooperative breeder. Behav Ecol 19:816–823
Szulkin M, Stopher KV, Pemberton JM, Reid JM (2013) Inbreeding avoidance, tolerance, or preference in animals? Trends Ecol Evol 28:205–211
Thünken T, Bakker TCM, Baldauf SA, Kullmann H (2007a) Active inbreeding in a cichlid fish and its adaptive significance. Curr Biol 17:225–229
Thünken T, Bakker TCM, Baldauf SA, Kullmann H (2007b) Direct familiarity does not alter mating preference for sisters in male Pelvicachromis taeniatus (Cichlidae). Ethology 113:1107–1112
Thünken T, Baldauf SA, Kullmann H, Schuld J, Hesse S, Bakker TCM (2011) Size-related inbreeding preference and competitiveness in male Pelvicachromis taeniatus (Cichlidae). Behav Ecol 22:358–362
Thünken T, Meuthen D, Bakker TCM, Baldauf SA (2012) A sex-specific trade-off between mating preferences for genetic compatibility and body size in a cichlid fish with mutual mate choice. Proc R Soc Lond B 279:2959–2964
Thünken T, Meuthen D, Bakker TCM, Kullmann H (2010) Parental investment in relation to offspring quality in the biparental cichlid fish Pelvicachromis taeniatus. Anim Behav 80:69–74
Thünken T, Waltschyk N, Bakker TCM, Kullmann H (2009) Olfactory self-recognition in a cichlid fish. Anim Cogn 12:717–724
Wagner WE (1998) Measuring female mating preferences. Anim Behav 55:1029–1042
Widemo F, Saether SA (1999) Beauty is in the eye of the beholder: causes and consequences of variation in mating preferences. Trends Ecol Evol 14:26–31
Acknowledgments
We thank Saskia Hesse for her help to breed the fish, the Bakker research group for discussion, and Corinna Seibt and anonymous referees for useful comments on the manuscript. This research was funded by the Deutsche Forschungsgemeinschaft (DFG) (BA 2885/2-3 and TH 1516/1-1).
Ethical standards
The experiments comply with the current laws of the country in which they were performed.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. Krause
Rights and permissions
About this article
Cite this article
Thünken, T., Bakker, T.C.M. & Baldauf, S.A. “Armpit effect” in an African cichlid fish: self-referent kin recognition in mating decisions of male Pelvicachromis taeniatus . Behav Ecol Sociobiol 68, 99–104 (2014). https://doi.org/10.1007/s00265-013-1626-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-013-1626-2